Abstract
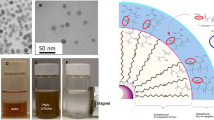
A magnetic nanoparticles-loaded polymeric nanocarrier was developed. Amphiphilic copolymer, methoxy polyethylene glycol-poly(D,L-lactide-co-glycolide)(MPEG-PLGA) could self-assemble to form nanomicelle with the help of emulsion-solvent evaporation technique. This nanocarrier with core-shell structure was loaded with magnetic iron oxide nanoparticles(IONPs) and anticancer drug paclitaxel(PTX). The hydrodynamic diameter of IONPs-PTX-loaded nanocarrier showed an average size of 110 nm with a polydispersity index(PDI) of 0.136, and its zeta potential was (–4.76±0.36) mV. The drug-loading content and encapsulation efficiency were 4.47% and 31.28%, respectively. In vitro drug release experiment was performed and a sustained release profile was observed. 3-(4,5-Dimethyl-2-thiazolyl)-2,5-diphenyl-2-H-tetrazolium bromide(MTT) assay indicated that IONPs-PTX-loaded nanocarrier showed comparable cytotoxicity with free paclitaxel. When an external magnetic field was applied, the nanocarrier significantly localized at the target area, demonstrating that the nanocarrier could be used for potential magnetic targeted drug delivery.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Siegel R. L., Miller K. D., Jemal A., CA-Cancer J. Clin., 2015, 65(1), 5
Wang H., Zhao Y., Wu Y., Hu Y. L., Nan K. H., Nie G. J., Chen H., Biomaterials, 2011, 32(32), 8281
Iyer A. K., Khaled G., Fang J., Maeda H., Drug Discov. Today, 2006, 11(17/18), 812
Nie S. M., Xing Y., Kim G. J., Simons J. W., Annu. Rev. Biomed. Eng., 2007, 9, 257
Obara K., Ishihara M., Ozeki Y., Ishizuka T., Hayashi T., Nakamura S., Saito Y., Yura H., Matsui T., Hattori H., Takase B., Ishihara M., Kikuchi M., Maehara T., J. Control. Release, 2005, 110(1), 79
Ruel-Gariepy E., Shive M., Bichara A., Berrada M., Garrec D. L., Chenite A., Leroux J. C., Eur. J. Pharm. Biopharm., 2004, 57(1), 53
Dordunoo S. K., Oktaba A. M. C., Hunter W., Min W., Cruz T., Burt H. M., J. Control. Release, 1997, 44(1), 87
Brito D. A., Yang Z. Y., Rieder C. L., J. Cell Biol., 2008, 182(4), 623
Elkharraz K., Faisant N., Guse C., Siepmann F., Arica-Yegin B., Oger J. M., Gust R., Goepferich A., Benoit J. P., Siepmann J., Int. J. Pharm., 2006, 314(2), 127
Ta H. T., Dass C. R., Dunstan D. E., J. Control. Release, 2008, 126(3), 205
Fujioka T., Taketani S., Nagasaki T., Matsumoto A., Bioconjugate Chem., 2009, 20(10), 1879
Haag R., Angew. Chem. Int. Ed., 2004, 43(3), 278
Mi Y., Liu Y. T., Feng S. S., Biomaterials, 2011, 32(16), 4058
Maeda H., Nakamura H., Fang J., Adv. Drug Deliv. Rev., 2013, 65(1), 71
Mi Y., Zhao J., Feng S. S., Nanomedicine, 2013, 8(10), 1559
Schroeder A., Heller D. A., Winslow M. M., Dahlman J. E., Pratt G. W., Langer R., Jacks T., Anderson D. G., Nat. Rev. Cancer, 2012, 12(1), 39
Mi Y., Guo Y. J., Feng S. S., Nanomedicine, 2012, 7(12), 1791
Lü Y. Y., Ding G. B., Zhai J. H., Guo Y., Nie G. J., Xu L., Colloid Surf. B, Biointerfaces, 2013, 110, 411
Ding G. B., Liu H. Y., Wang Y., Lv Y. Y., Wu Y., Guo Y., Xu L., Chem. Res. Chinese Universities, 2013, 29(1), 103
Wang Y., Wang X. Y., Lü Y. Y., Xu W. B., Guo Y., Xu L., Chem. J. Chinese Universities, 2013, 34(12), 2866
Chen F., Dong D., Fu Y., Zheng Y. H., Liu S., Chang M. X., Jing X. B., Chem. Res. Chinese Universities, 2012, 28(4), 656
Ding G. B., Wang Y., Guo Y., Xu L., ACS Appl. Mater. Interfaces, 2014, 6(19), 16643
Ding G. B., Liu H. Y., Lü Y. Y., Liu X. F., Guo Y., Sun C. K., Xu L., Chem., Eur. J., 2012, 18(44), 14037
Liu P., Situ J. Q., Li W. S., Shan C. L., You J., Yuan H., Hu F. Q., Du Y. Z., Nanomedicine: Nanotechnology, Biology and Medicine, 2015, 11(4), 855
Ahmed F., Discher D. E., J. Control. Release, 2004, 96(1), 37
Sobczak M., Korzeniowska A., Gos P., Kolodziejski W. L., Eur. J. Med. Chem., 2011, 46(7), 3047
Song Z. M., Feng R. L., Sun M., Guo C. Y., Gao Y., Li L. B., Zhai G. X., J. Colloid Interface Sci., 2011, 354(1), 116
Kim S., Shi Y. Z., Kim J. Y., Park K., Cheng J. X., Expert Opin. Drug Deliv., 2010, 7(1), 49
Xiong X. B., Mahmud A., Uludag H., Lavasanifar A., Pharm. Res., 2008, 25(11), 2555
Soppimath K. S., Liu L. H., Seow W. Y., Liu S. Q., Powell R., Chan P., Yang Y. Y., Adv. Funct. Mater., 2007, 17(3), 355
Banerjee S. S., Chen D. H., J. Nanopart. Res., 2009, 11(8), 2071
Tian J., Stella V. J., J. Pharm. Sci., 2010, 99(3), 1288
Morgan M. T., Nakanishi Y., Kroll D. J., Griset A. P., Carnahan M. A., Wathier M., Oberlies N. H., Manikumar G., Wani M. C., Grinstaff M. W., Cancer Res., 2006, 66(24), 11913
Yang X. Q., Grailer J. J., Pilla S., Steeber D. A., Gong S. Q., Shuai X. T., J. Biofabrication, 2010, 2(2), 025004
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supported by the National Natural Science Foundation of China(Nos.81271697, 81571791, 31571017), the Specialized Research Fund for the Doctoral Program of Higher Education of China(Nos.20100061120077, 20120061110021), the Social Development Project of Science and Technology Department of Jilin Province, China(Nos.20120967, YYZX201264, 20130206069GX) and the “Significant New Drug Creation” Science and Technology Major Program of China (No.2012ZX09503001-003).
Rights and permissions
About this article
Cite this article
Zhang, S., Qian, X., Zhang, D. et al. In vitro anticancer efficacy by magnetic targeted nanocarrier with local delivery of paclitaxel. Chem. Res. Chin. Univ. 32, 149–154 (2016). https://doi.org/10.1007/s40242-015-5115-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-015-5115-1