Abstract
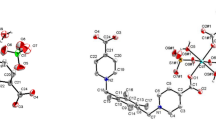
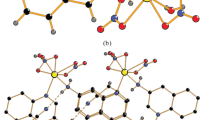
A novel one dimension(1D) cadmium coordination polymer {[Cd(mpda)3]·2(NO3)} n (1) was synthesized via refluxing a mixture of tetradentate Schiff base ligand N,N′-bis(2-pyridinylethylidene)phenylene-1,3-diamine(L) and Cd(NO3)2 in acetonitrile, whose structure was characterized by means of single crystal X-ray diffraction, FTIR spectroscopy, elemental analysis and proton nuclear magnetic resonance(1H NMR). Center metal Cd(II) ion is six-coordinated by six nitrogen atoms from six different m-phenylenediamine(mpda), giving rise to a [CdN6] octahedral coordination environment. The two adjacent cadmium centers are linked by three mpda molecules leading to the construction of 1D chain structure. The crystal structure is stabilized by N—H…O hydrogen bonds to form three-dimension supramolecule. Compound 1 exhibits intense yellow luminescence in solid state at 298 K(λ em=554 nm), which shows a blue shift at 77 K(ca. 147 nm). Additionally, fluorescence characteristics of compound 1 were investigated in different solvents(polarity: DMSO>CH3CN>CH3OH>CHCl3>toluene) at 298 and 77 K. The results show that the emission peak of compound 1 in solvent exhibits a slight bathochromic shift. However, the emission peaks of compound 1 in CH3OH and CHCl3 are red shift compared with that in CH3CN. It is revealed that the luminescence behavior of compound 1 depends on not only the polarity of solvent but also the hydrogen bonding properties between solvent and solute. In addition, the emission peak of compound 1 in solution shows a red shift obviously at 77 K than that at 298 K(ca. 144–159 nm), with the fluorescence lifetime increased at 77 K. The lifetime in DMSO at 77 K(τ=12.470 μs) was the longest one. The quantum yield of compoud 1 increases with increasing the polarity of solvent within a range of 1.8%–8.3 %.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Murray L. J., Dinca M., Long J. R., Chem. Soc. Rev., 2009, 38, 1294
Wang Y. Y., Zhang Z., Sun Y. J., Chem. J. Chinese Universities, 2014, 35(3), 449
Huang Q. D., Li C. L., Zhang Y., Chem. J. Chinese Universities, 2014, 35(3), 524
Li J. R., Kuppler R. J., Zhou H. C., Chem. Soc. Rev., 2009, 38, 1477
Shi W. B., Kou H. Z., Chem. J. Chinese Universities, 2014, 35(1), 12
Lu W. G., Liu H. W., Yin X. G., Chem. J. Chinese Universities, 2013, 34(12), 2691
Plabst M., McCusker L. B., Bein T., J. Am. Chem. Soc., 2009, 131, 18112
Liu Y., Kravtsov V. C., Eddaoudi M., Angew. Chem., Int. Ed., 2008, 47, 8446
Monika P., McCusker L. B., Bein T., J. Am. Chem. Soc., 2009, 131, 18112
Liu H. W., Chen X. F., Zhang J., Acta Polymeric Sinica, 2014, 1, 115
Lee J. Y., Farha O. K., Roberts J., Scheidt K. A., Nguyen S. T., Hupp J. T., Chem. Soc. Rev., 2009, 38, 1450
Ma L., Abney C., Lin W., Chem. Soc. Rev., 2009, 38, 1248
Allendorf M. D., Bauer C. A., Bhakta R. K., Houk R. J. T., Chem. Soc. Rev., 2009, 38, 1330
Wu D. Q., Meng W., Zhang L., Liu L., Hou H. W., Fan Y. T., Inorg. Chim. Acta, 2013, 405, 318
Friedrichs O. D., O’Keeffe M., Yaghi O. M., Acta Crystallogr., 2003, A59, 22
Friedrichs O. D., O’Keeffe M., Yaghi O. M., Acta Crystallogr., 2003, A59, 515
Qi Y., Luo F., Che Y. X., Zheng J. M., Cryst. Growth Des., 2008, 8, 606
Tong M. L., Ye B. H., Cai J. W., Chen X. M., Ng S. W., Inorg. Chem., 1998, 37, 2645
Hennigar T. L., MacQuarrie D. C., Losier P., Rogers R. D., Zaworotko M. J., Angew. Chem., Int. Ed. Engl., 1997, 36, 972
Zhang W. L., Liu Y. Y., Ma J. F., Jiang H., Yang J., Ping G. J., Cryst. Growth Des., 2008, 8, 1250
Sheldrick G. M., Software for Data Extraction and Reduction, Version 6.02, Bruker Axs Inc., Madison, WI, 2002
Sheldrick G. M., SHElXTL NT Crystal Structure Analysis Package, Version 5.10, Bruker Axs Inc., Madison, WI, 1999
Duliban J., J. Appl. Polym. Sci., 2012, 125, 3708
Mbuli B. S., Dlamini D. S., Nxumalo E. N., Krause R. W., Pillay V. L., Oren Y., Linder C., Mamba B. B., J. Appl. Polym. Sci., 2013, 129, 549
Idris A., Kormin F., Noordin M. Y., Separation and Purification Technology, 2006, 49, 271
Lin J. D., Cheng J. W., Du S. W., Cryst. Growth Des., 2008, 8, 3345
Zhang L. P., Yang J., Ma J. F., Jia Z. F., Xie Y. P., Wei G. H., CrystEngComm, 2008, 10, 1395
Habib H. A., Sanchiz J., Janiak C., Dalton Trans., 2008, 1734
Ren P., Liu M. L., Zhang J., Shi W., Cheng P., Liao D. Z., Yan S. P., Dalton Trans., 2008, 4711
Hsu Y. F., Hu H. L., Wu C. J., Yeh C. W., Proserpioc D. M., Chen J. D., CrystEngComm, 2009, 11, 122
Shi X. J., Wang X., Li L., Hou H. W., Fan Y. T., Cryst. Growth Des., 2010, 10, 2490
Kosar B., Albayrak C., Ersanli C. C., Odabasoglu M., Buyukgungor O., Spectrochim. Acta A, 2012, 93, 1
Kasumov V. T., Spectrochim. Acta A, 2003, 57, 1649
Ceyhan G., Köse M., Tümer M., Demirtaş İ., Yağlioğlu A. Ş., McKee V., J. Luminescence, 2013, 143, 623
Lu J., Li Y., Zhao K., Xu J. Q., Yu J. H., Li G. H., Zhang X., Bie H. Y., Wang T. G., Inorg. Chem. Commun., 2004, 7, 1154
Zhang C. M., Lin J., Chem. Soc. Rev., 2009, 8, 1330
Yao X. Q., Zhang M. D., Hu J. S., Li Y. Z., Guo Z. J., Zheng H. G., Cryst. Growth Des., 2011, 11, 3039
Lan A. J., Li K. H., Wu H. H., Olson D. H., Emge T. J., Ki W., Hong M. C., Li J., Angew. Chem., Int. Ed., 2009, 48, 2334
Zang S. Q., Su Y., Li Y. Z., Lin J. G., Duan X. Y., Meng Q. J., Gao S., CrystEngComm, 2009, 11, 122
Habib H. A., Hoffmann A., Höppe H. A., Steinfeld G., Janiak C., Inorg. Chem., 2009, 48, 2166
Liu W. L., Ye L. H., Liu X. F., Yuan L. M., Jiang J. X., Yan C. G., CrystEngComm, 2008, 10, 1395
Wen L. L., Dang D. B., Duan C. Y., Li Y. Z., Tian Z. F., Meng Q. J., Inorg. Chem., 2005, 44, 7161
Hu T. L., Zou R. Q., Li J. R., Bu X. H., Dalton Trans., 2008, 1302
Zencirci N., Gelbrich T., Apperley D. C., Harris R. K., Kahlenberg V., Griesser U. J., Cryst. Growth Des., 2010, 10, 303
Wang X. M., Qiang L. S., Fan R. Q., Wang P., Yang Y. L., Supramolecular Chem., 2013, 25, 416
Dijken A. V., Meulenkamp E. A., Vanmaekelbergh D., Meijerink A., J, Phys. Chem. B, 2000, 104, 1715
Constable E. C., Neuburger M., Rösel P., Schneider G. E., Zampese J. A., Housecroft C. E., Monti F., Armaroli N., Costa R. D., Ortí E., Inorg. Chem., 2013, 52, 885
Ivanov P., Stanimirov S., Kaloyanova S., Petkov I., J. Fluoresc., 2012, 22, 1501
Dobek K., Karolczak J., J. Fluoresc., 2012, 22, 1647
Kubinyi M., Varga O., Baranyai P., Kállay M., Mizsei R., Tárkányi G., Vidóczy T., J. Mol. Struct., 2011, 1000, 77
Rodríguez-Diéguez A., Salinas-Castillo A., Sironi A., CrystEngComm, 2010, 12, 1876
Fan R. Q., Zhang Y. J., Yin Y. B., Su Q., Yang Y. L., Hasi W., Syn. Met., 2009, 159, 1106
Murugan K. D., Natarajan P., Eur. Polym. J., 2011, 47, 16
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supported by the National Natural Science Foundation of China(Nos.21371040, 21171044), the National Key Basic Research Program of China(No.2013CB632900), the Fundamental Research Funds for the Central Universities of China(No.201409) and the Program for Innovation Research of Science in Harbin Institute of Technology, China(Nos.A201416, B201414).
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Xu, M., Liu, Z., Fan, R. et al. Synthesis and crystal structure of 1D Cd-amine coordination polymer and its luminescent properties. Chem. Res. Chin. Univ. 30, 720–725 (2014). https://doi.org/10.1007/s40242-014-4032-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-014-4032-z