Abstract
Purpose
Heavy metals, as significant toxic environmental contaminants, can cause serious adverse health outcomes on the human body even in trace concentrations. There is limited evidence on heavy metal concentrations existing in the body fluids of pregnant women. This study aims to evaluate the urinary levels of arsenic (As) and lead (Pb), as two main toxic heavy metals, among pregnant women and their lifestyle determinants.
Methods
The study was performed in 2019–2020 in Isfahan, Iran. A number of 140 urine samples of pregnant women who were in their first pregnancy trimester were examined. Inductively coupled plasma optical emission spectrometry (ICP-OES) was applied to analyze the urinary concentrations of As and Pb. Socio-demographic data including age, pre-pregnancy body mass index (BMI), education status, and family income, as well as the use of cleaning products (cosmetic and household cleaning products), and lifestyle habits (food intake, smoking, and physical activity) were collected using a validated questionnaire.
Results
The mean concentration of As and Pb were 8.14 ± 10.8 and 9.6 ± 7.1 μg/g creatinine, respectively. The mean urinary concentration of Pb indicated significant differences in the levels of cosmetic usage, second-hand smoking exposure, and the use of Copper, Aluminum, Teflon, Steel, and Enameled utensils for cooking (p-value < 0.05). Furthermore, the mean of urinary Pb concentrations at high levels of physical activity and scratched utensils using was significantly different from the other categories (p-value = 0.02). No significant differences were found between As and Pb concentration with other socio-demographic factors.
Conclusion
The lifestyle determinants and cosmetic products use are important predictors of urinary heavy metals in pregnant women, rather than sociodemographic characteristics. Additional research is necessary to determine long-term adverse birth outcomes of exposure to these heavy metals.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Heavy metals as high-density metals in comparison with organic pollutants are somewhat stable and resistant in the environment and the body tissues at trace concentration [1]. Individuals may be exposed to heavy metals via different routes such as inhalation, ingestion, smoking, and use of cleaning and cosmetic products [2, 3].
Exposure to heavy metal damage nervous system, change the oxidative cell stress, interference with glucose metabolism, and disrupt endocrine [4, 5].
Arsenic and lead are known as two toxic trace elements which cause some serious adverse health outcome on human health especially for vulnerable individuals and pregnant women [6]. Dietary intake is generally considered the most significant way of exposure to heavy metals in human beings. Arsenic in the diet originates from contaminated well water in regions nearby the waste disposal sites, incinerators, electronic manufacturers, mines, and agricultural areas that utilize fertilizer containing arsenic [7, 8]. Previous documents demonstrated that exposure to arsenic can disrupt sugar metabolism and cause gestational diabetes in pregnant women [7]. In addition, chronic arsenic exposure can elevate the risk of developmental disability, mental retardation, and newborn death [9].
Lead also enters the human body through inhalation, ingestion, or absorption through the skin and mucous membrane [10]. It can cause some serious health risks such as disrupting learning ability, growing, and development [10, 11].
Prenatal exposure to environmental toxicants such as heavy metals is a serious public health concern regarding the vulnerability of pregnant women and their fetuses [12]. Heavy metals have been measured in cord blood and amniotic fluid, demonstrating that these metals cross easily through the placenta [13]. So, maternal exposure to these toxic elements may affect fetus growth by altering the homeostatic and metabolic mechanisms [14]. Previous studies have shown increased adverse pregnancy effects when mothers are exposed to heavy metals [15]. Some epidemiological research demonstrated an association between maternal heavy metals exposure and the consequent risks such as abortion, and preterm delivery [16,17,18].
Numerous factors such as lifestyle and socio-demographic status can influence the exposure rate and subsequently the extent of risk on individuals [19,20,21,22,23]. For example, persons with low socioeconomic situations experience a greater rate of exposure [22, 24, 25]. To the best of our knowledge, no previous publication has evaluated the urinary levels of Arsenic and Lead in Iranian pregnant women with regard to their life-style and socio-demographic variables. Therefore, due to the sensitivity of pregnant women and the direct effect of exposure on birth outcomes, this study was conducted to track the urinary concentrations of Arsenic and Lead in a sample of Iranian pregnant women at their first trimester, as an indicator of exposure to heavy metal, and examine associations between prenatal heavy metals exposures with various factors. The findings of this study may also provide an insight into common sources of exposure to heavy metals among Iranian pregnant women which can help decision-makers to take the appropriate measures.
Materials and methods
Study population
This cross-sectional study was performed in 2019–2020 on pregnant women who lived in Isfahan city, Iran. Exclusion criteria included refusal to participate, history of chronic diseases [including liver and renal diseases, diabetes, malignancy, sickle cell anemia, or seropositive to Human Immunodeficiency Virus (HIV)], sleep deprivation and insomnia, depression/depressive symptomatology, stress, abortion and taking regular medications. A number of 140 women in their first trimester were selected randomly. All parents signed informed consent forms. The distribution of participants’ living is demonstrated in Fig. 1. The participants were completely informed about the purposes, procedure, and also voluntary nature of the study. It is noteworthy that the ethical issues related to this study were approved by the ethics committees of Isfahan University of medical sciences. Suggested formula for estimating correlation coefficients was used to compute the sample size. Based on α = 0.05, β = 0.15, and a correlation coefficient of r = 0.23 based on the previous study [26], we reached to 140 subjects. The urine samples were gathered in borosilicate containers and then transferred to the laboratory to be preserved at − 20 °C. The validated questionnaires (PERSIAN Birth Cohort questionnaires) were utilized to collect information about socio-demographic variables (maternal age, family income, education status, maternal occupation), lifestyle determinants (pre-pregnancy BMI, physical activity, smoking habit, and food habits (seafood and canned foods consumption, kind of cookware for cooking (zinc, copper, aluminum, enameled, Teflon, cast iron, steel, and glassware), and household cleaning and cosmetic product use [27].
FFQ (Food frequency questionnaire) was applied to gather data about the food intake habits of participants.
IPAQ (International physical activity questionnaire) was used to compute total physical activity (MET-minutes/week) score [28]. A validated food frequency questionnaire (FFQ) was applied to obtain information about food intake habits [29].
Measuring urinary heavy metals
A 3.0 ml of the urine was poured into a polypropylene tube containing 15 μl of 65% (v/v) HNO3 and then kept in a refrigerator at 5 °C. Two hours before sample preparation, the urine samples were taken to room temperature. One ml of the sample was poured into a 10 ml polypropylene tube and filled up to 5.0 ml with 1.2% (v/v) HNO3 [30]. Then, the prepared solution was injected into the ICP-OES (Varian 720/730-ES) to analyses heavy metals concentration.
Concentrations of heavy metals were adjusted using creatinine level to reduce the bias of the dilution difference between urine samples.
Quality assurance and quality control (QA/QC)
The quality assurance and quality control (QA/QC) assessments were performed to confirm the reliability of the analytical data and to increase confidence in the relevance of obtained responses. Accordingly, the linear regression gave a good fit (R2 ≥ 0.98) with high precision (≤ 13.2% RSD). The limit of detection (LOD) and limit of quantification (LOQ) were based on the signal-to-noise ratio of 3 and 10, respectively. Furthermore, to minimize the bias of the dilution difference between the samples, the heavy metal concentrations were adjusted using creatinine levels. The R2, precision (% RSD), LOD, LOQ, and mean recovery are presented in Table 1.
Data analysis
Continuous determinants have been reported as mean ± SD, minimum, and maximum whereas categorical variables as frequencies (percent). The Kolmogorov–Smirnov test and Q-Q plot were utilized to specify the normality of continuous data. Appropriate transformation approaches were applied for those heavy metal concentrations with the abnormal distribution. Independent samples t-test or analysis of variance (ANOVA) was utilized to compare the mean of heavy metal concentrations across categories of possible lifestyle and demographic variables. Bonferroni post hoc test was used to determine differences between categories after rejecting the null hypothesis in the analysis of variance. All statistical analyses were conducted by SPSS software (IBM SPSS Inc., Chicago, IL, version 23). A p-value less than 0.05 was considered statistically significant.
Results and discussion
The characteristics of the studied population are demonstrated in Table 2. According to the results, 45.7% (n = 64) were less than 30 years old, 54.3% (n = 76) were ≥ 30-years old, 57.1% of women (n = 80) had overweight, while 38.6% of them (n = 54) were classified as normal-weight group, and only 4.3% of women (n = 6) were underweight. A great number of the participants had an academic education (n = 116, 82.8%). Eighty percent of subjects were householder (n = 112) and 63.6% of the total population (n = 89) categorized into the middle- income group.
Based on the obtained results, greater than 50% of pregnant women were the low user of seafood (n = 74) and canned food (n = 81). In addition, 50% of individuals used Teflon utensils for cooking whereas glassware (n = 7, 5%) and enameled (n = 12, 8.6%) containers had minimum consumers for cooking. After using Teflon, steel, zinc, cast iron, copper, and aluminum had respectively the highest consumers for cooking food. Furthermore, more than half of the participants (n = 79, 56.4%) stated that they used scratched containers for cooking.
Moreover, the results showed that household cleaning and cosmetic products were respectively used by 100% (140) and 62.85% (n = 88) of individuals. However, none of the individuals were smokers, about 50.75% (n = 71) were passive smokers.
The majority of the individuals were categorized into the middle (n = 49, 35%) and low (n = 66, 47.1%) physical activity group, though 17.9% of women (n = 25) were classified into the high physical activity group.
The mean, quartile, maximum, and minimum of creatinine-adjusted heavy metals concentration are given in Table 3. Accordingly, As and Pb were respectively detected in 86% and 100% of urine samples. The highest and lowest concentration values were observed with 103 μg/g creatinine and 1.1 μg/g creatinine for As and 41.5 μg/g creatinine and 0.9 μg/g creatinine for Pb.
In the present study, the mean concentration of As and Pb were found to be 8.14 ± 10.8 μg/g creatinine and 9.6 ± 7.1 μg/g creatinine which As was lower than those concentrations reported by other studies conducted in Brazil (As = 11.34 μg/g) [31], and Tokyo (As = 393 μg/g) [32], but Pb was higher than in results reported in Suadi Arabia (Pb = 5.156 ± 0.28 μg/g) [33], Spain (Pb = 4.8 ± 4.3 μg/g) [34], and Tokyo (Pb = 0.483 μg/g) [32]. The inconsistency in results can be explained by the various characteristics of the studied area, lifestyle, and socio-demographic [35]. Furthermore, some researchers announced that the levels of heavy metals detected in the blood population of many low and middle-income developing countries are much higher amount than in developed countries [36, 37].
The distribution of As and Pb concentrations in relation with some influencing determinants including education level, family income, occupation status, seafood, and canned foods consumption, use of the different type of utensils (zinc, copper, aluminum, enameled, Teflon, cast iron, steel, and glassware) and scratched container for cooking, cosmetic and household cleaning products use, smoking and passive smoking habit, and physical activity is given in Table 4.
Socio-demographic- predictors in urinary heavy metals in pregnant women
According to Table 4, the mean of urinary As and Pb concentrations did not show significant differences in the levels of different variables including maternal education level, family income, BMI, maternal age, and maternal occupation.
Similar results were reported by Zeng [38] and Zaw [39]. However, some studies indicated that family income is directly related to the amount of urinary heavy metals such as Cr, As, Hg, and Cd among pregnant women [40]. Education level, family income, and other sociocultural factors are somehow interdependent which can have different results in developed societies compared with developing countries [41]. A research performed among pregnant women in Myanmar demonstrated no significant difference between maternal education status and heavy metals concentration [42]. In the contrast, a study conducted in China showed that women who had lower education levels showed a higher amount of urinary Cd, Ni, Pb concentration [43]. Various results of maternal education may be rooted in that the influence of education is intensely linked with socio-cultural factors [44].
The results of the current study revealed that the mean concentration of urinary As and Pb indicated no significant differences with occupation status and maternal age (p-value > 0.05).Occupational exposure is often considered a risk factor for women fertility, pre-term delivery, and also spontaneous abortion [45]. However, there are some documents that reported a direct association between heavy metal occupational exposure and the increased risk of birth outcome [46]. A recent study carried out in Nigeria revealed that the miscarriage incidence increased by 1.6% in women exposed to some heavy metals [45].
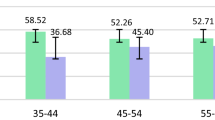
Some studies reported positive associations between heavy metals and matenal age, attributable in part to bioaccumulation [47, 48]. Kasim et al. found that Pb level increased with age, which may be correlated with the release of Pb previously accumulated in tissues [48]. However, numerous studies have been showed inverse results. Esquinas et al. indicated that the older women tended to display lower geometric mean heavy metal levels [49]. This result could be indirectly affected by other co-factors such as lifestyle, genetics, and the metabolism, and sample size which can cause different results. Further intervention studies are needed to elucidate these relationships.
Lifestyle variables- predictors in urinary heavy metals in pregnant women
The mean urinary of Pb concentrations showed significant differences in the levels of the used utensils i.e. Copper, Aluminum, Teflon, Steel, and Enameled (p-value < 0.05). However, the mean concentration of urinary As did not indicate a significant difference with the used utensils by women.
Some studies have revealed that the nature of cookware, cooking process, processing, and storage methods may raise the amount of trace metals in foods [50]. Cabrera et al. [51] reported that some cooking utensils such as Steel, Aluminum, and Teflon can release some substantial trace metals level into foods due to cooking temperatures, heating and acidic condition, damaging and corrosion of container which might consequently lead to disrupting endocrine in the human body [52].
It is must be noted that the scratched cookware increases the risk of leaching heavy metals [53]. According to the statistical results in Table 4, the mean concentrations of Pb indicated a significant difference in the level of the scratched containers for cooking (p-value = 0.02). In developing countries, the majority of people still utilize traditional containers and pots made of Copper, Enameled, and Aluminum for cooking. Unlike modern utensils, these containers do not have any protective layer of inert material to prevent contamination of food [53]. Weidenhameretal et al. reported that Pb can migrate from stainless steels and Aluminum utensils during cooking and then enter food at concentrations beyond the recommended public health guidelines [36]. Said revealed high migration of heavy metals with mean values of 30.3 ± 0.56 mg/l, 21.56 ± 0.12 mg/l, 28.98 ± 0.09 mg/l, 14.54 ± 0.15 mg/l, respectively when scratched fold Iron, Aluminum, stainless steel, and Teflon cookware were used [53].
Daily exposure to heavy metals via cosmetic products has been regarded as a negligible source for individuals compared to the main sources such as food, water, and the air. However, due to the cumulative property of trace metal in the human body, cosmetics can be considered as a significant source of heavy metals [54].
According to the obtained results, the mean concentration Pb show a significant difference in the level of cosmetic usage (p-value < 0.001).
Health Canada revealed that nearly ninety percent of cosmetic products were positively confirmed in terms of the existence of some toxic heavy metals such as Pb, Cd, Ni, and Hg [55]. These heavy metals are extremely toxic and forbidden in cosmetic products as ingredients in the US and EU [56].
A significant amount of Pb was measured by Ajaezi et.al, in skin bleaching agents [57]. Furthermore, the result of a study conducted in Isfahan, Iran indicated 0.08–5.2 µg/l concentration of Pb in lipsticks [58]. However, the Pb range was in the permissible range of FDA, it can be accumulated when used continuously.
The statistical result showed a significant difference between the mean concentrations of Pb and the women who were passively exposed to smoke (p-value = 0.01).
Chiba et al. also detected Cd, Cr, Ni, Pb in smoke/cigarettes and reported also in body fluids of individuals who were smokers. They also announced that individuals who were passively exposed to smoke can absorb more amounts of heavy metals [59]. Some researchers have announced that elevated heavy metals due to smoking and also second-hand smoking may increase spontaneous abortion risk [60], stillbirth, preterm delivery, and smaller infants weight [61].
Bonferroni post hoc test for canned food and seafood consumption showed that the mean urinary of As and Pb concentrations were not significantly different from the other two categories. However, the majority of food-based dietary guidelines insist to consume more seafood during the pregnancy period to provide sufficient iodine and DHA (Docosahexaenoic acid)- a key role in the central nervous system development of fetus [62]. There are some documents detected heavy metals in canned food and seafood. Lin et al. reported that the urinary of Hg and Cd were correlated with canned consumption (r = 0.124, p-value < 0.05) [40]. Furthermore, a study conducted in Iran indicated that the mean contents of As and Pb in canned foods were higher than the standard levels [63]. Additionally, Yu et al. showed the rate of fish consumption during pregnancy was correlated with a greater level of some heavy metals such as Hg and Cd in cord blood [64]. The different results may be due to the frequency of canned food and seafood consumption by individuals.
Our results indicated that the mean of urinary Pb concentrations at high levels of physical activity was significantly different from the other two categories (p-value = 0.02). In other words, higher Pb concentrations were detected in women who had greater physical activities.
There are only limited publications regards the effect of physical exercise on heavy metal excretion from the body fluids and tissues. In parallel with our findings, Milnerowicz et al. have indicated that a higher level of physical activity is positively associated with a higher level of urinary heavy metals and after 24 h they decrease lower than the initial value. It can be supposed that with greater physical exercise, a higher value of heavy metals permeates from blood to urine [65]. It is probable that heavy metals are additionally released from tissues to blood and urine [66]. It can be concluded that heavy physical activity changes heavy metals reserves in tissues [67]. This may be considered a positive occurrence, especially in terms of the long half-life and the range of its toxicity [65, 68].
Physical activity increases oxygen consumption, and accordingly, the generation of reactive oxygen species (ROS) raises [69]. Regular and moderate physical exercise releases oxidative stress, increases cellular adaptation, and also decreases the quantity of lipid peroxidation products developing within activity performance [68, 70]. In this condition, cells in response to the stress generate various proteins. Metallothionein (MT) is one of them. This protein arrest heavy metals therefore lessening their poisonousness. MT is known as a protein of acute-phase serve as an active free radicals scavenger [68, 71].
Strength and limitation
To the best of our knowledge, it is one of the first documents to determine the probable relation between lifestyle and socio-demographic characteristics of Iranian pregnant women with prenatal heavy metals exposure. However, we found some limitations in this investigation that can affect the findings. The medium or small size of the studied samples with typically similar lifestyle and socio-demographic profiles might affect the statistical results. However, we were able to found a significant correlation between socio-demographic and life style determinants with heavy metals concentrations. In addition, in this study, we collected spot urine samples. We were not able to collect representative urine samples (24-h urine) during the day, although this may not severely affect the total distribution of heavy metals among the studied participants. We attempted to address this by creatinine adjusting of urinary samples.
Conclusion
In this study, As and Pb were detected in 86% and 100% of urine samples of pregnant mothers. The detected mean concentration of Pb had higher values in the Iranian pregnant women's urine compared to other societies. Results demonstrated that the mean concentration of urinary Pb was higher in participants who were second-hand smokers, applied Copper, Aluminum, Teflon, Steel, Enameled, and also damaged cookware for cooking, as well as those who had higher physical activity. The lifestyle characteristics and cosmetic product usage were important predictors for urinary heavy metals in pregnant women than socio-demographic profiles.
References
Tchounwou PB, Yedjou CG, Patlolla AK, Sutton DJ. Heavy metal toxicity and the environment. In: Luch A, editor. Molecule, clinic environ toxicol. Basel: Springer; 2012. p. 133–64.
Mohammadi H, Alinejad A, Khajeh M, Darvishmotevalli M, Moradnia M, Mazaheri Tehrani A, Hosseindost G, Zare MR, Mengelizadeh N. Optimization of the 3D electro-Fenton process in removal of acid orange 10 from aqueous solutions by response surface methodology. J Chem Technol Biotechnol. 2019;94(10):3158–71.
Moradnia M, et al. The relation of cancer risk with nitrate exposure in drinking water in Iran. Iran J Public Health. 2019;48(2):362–4.
Gollenberg AL, Hediger ML, Lee PA, Himes JH, Buck Louis GM. Association between lead and cadmium and reproductive hormones in peripubertal US girls. Environ Health Perspect. 2010;118(12):1782–7.
Gundacker C, Fröhlich S, Graf-Rohrmeister K, Eibenberger B, Jessenig V, Gicic D, et al. Perinatal lead and mercury exposure in Austria. Sci Total Environ. 2010;408(23):5744–9.
Egorova KS, Valentine PA. Toxicity of metal compounds: knowledge and myths. Organometallics. 2017;36(21):4071–90.
Milton AH, Hussain S, Akter S, Rahman M, Mouly TA, Mitchell K. A review of the effects of chronic arsenic exposure on adverse pregnancy outcomes. Int J Environ Res Public Health. 2017;14(6):556.
Khlifi R, Olmedo P, Gil F, Feki-Tounsi M, Hammami B, Rebai A, et al. Biomonitoring of cadmium, chromium, nickel and arsenic in general population living near mining and active industrial areas in Southern Tunisia. Environ Monit Assess. 2014;186(2):761–79.
Vahter M. Health effects of early life exposure to arsenic. Basic Clin Pharmacol Toxicol. 2008;102(2):204–11.
Fadaei S, Noorisepehr M, Pourzamani HR, Salari M, Moradnia M, Darvishmotevalli M, Mengelizadeh N. Heterogeneous activation of peroxymonosulfate with Fe3O4 magnetic nanoparticles for degradation of Reactive Black 5: Batch and column study. J Environ Chem Eng. 2021;9(4):105414.
Neeti K, Prakash T. Effects of heavy metal poisoning during pregnancy. Int Res J Environ Sci. 2013;2(1):88–92.
Spielmann H. The toxicity of heavy metals in the pregnant woman, fetus and newborn infant. In: Health evaluation of heavy metals in infant formula and junior food. Berlin: Springer; 1983. p. 57–68.
Huang Y, Li J, Garcia JM, Lin H, Wang Y, Yan P, et al. Phthalate levels in cord blood are associated with preterm delivery and fetal growth parameters in Chinese women. PloS One. 2014;9(2):e87430.
Magnusson C, Jugdaohsingh R, Hulthen L, Westerlund A, Powell JJ, Ransjö M. Urinary excretion of silicon in men, non-pregnant women, and pregnant women: a cross-sectional study. Biol Trace Elem Res. 2019;194:1–7.
Quansah R, Armah FA, Essumang DK, Luginaah I, Clarke E, Marfoh K, et al. Association of arsenic with adverse pregnancy outcomes/infant mortality: a systematic review and meta-analysis. Environ Health Perspect. 2015;123(5):412–21.
Taherkhani S, Darvishmotevalli M, Karimyan K, Bina B, Fallahi A, Karimi H. Dataset on photodegradation of tetracycline antibiotic with zinc stannate nanoflower in aqueous solution–Application of response surface methodology. Data Br. 2018;19:1997–2007.
Moradnia M, Emamjomeh MM. An environmental-friendly study on sanitary wastewater treatment for small community. Desalin Water Treat. 2017;94:25–30.
Darvishmotevalli M, Moradnia M, Asgari A, Noorisepehr M, Mohammadi H. Reduction of pathogenic microorganisms in an Imhoff tank–constructed wetland system. Desalin Water Treat. 2019;154:283–8.
Moradnia M, Movahedian Attar H, Heidari Z, Mohammadi F, Kelishadi R. Prenatal exposure to chromium (Cr) and nickel (Ni) in a sample of Iranian pregnant women: urinary levels and associated socio-demographic and lifestyle factors. Environ Sci Pollut Res. 2021. https://doi.org/10.1007/s11356-021-15201-w.
Asgari G, Mohammad Gholi Mezerji N, Salari M, Asgharnia HA, Darvishmotevalli M, Faraji H, Moradnia M. Forecasting nitrate concentration in babol groundwater resources using the grey model. Int J Env Health Eng. 2020;9(1):16.
Hajizadeh Y, Moradnia M, Feizabadi GK, Rafiei N, Tahmasbizadeh M, Darvishmotevalli M, et al. The sex-specific association between maternal urinary paraben levels and offspring size at birth. Environ Sci Pollut Res. 2021;28(27):36029–38.
Darvishmotevalli M, Bina B, Feizi A, Ebrahimpour K, Pourzamani H, Kelishadi R. Monitoring of urinary phthalate metabolites among pregnant women in Isfahan, Iran: the PERSIAN birth cohort. J Environ Health Sci Eng. 2019;17(2):969–78.
Darvishmotevalli M, Moradnia M, Noorisepehr M, Fatehizadeh A, Fadaei S, Mohammadi H, et al. Evaluation of carcinogenic risks related to nitrate exposure in drinking water in Iran. MethodsX. 2019;6:1716–27.
Sepehr MN, Allani F, Zarrabi M, Darvishmotevalli M, Vasseghian Y, Fadaei S, Fazli MM. Dataset for adsorptive removal of tetracycline (TC) from aqueous solution via natural light weight expanded clay aggregate (LECA) and LECA coated with manganese oxide nanoparticles in the presence of H2O2. Data Br. 2019;22:676–86.
Hajizadeh Y, Ebrahimpour K, Shoshtari-Yeganeh B, Fadaei S, Darvishmotevalli M, Karimi H. Urinary paraben concentrations and their implications for human exposure in Iranian pregnant women. Environ Sci Pollut Res. 2020;27(13):14723–34.
Lewis RC, Meeker JD, Basu N, Gauthier AM, Cantoral A, Mercado-García A, et al. Urinary metal concentrations among mothers and children in a Mexico City birth cohort study. Int J Hyg Envir Heal. 2018;221(4):609–15.
Darvishmotevalli M, Moradnia M, Hosseini R, Bina B, Feizi A, Ebrahimpour K, et al. Association between prenatal phthalate exposure and anthropometric measures of newborns in a sample of Iranian population. Environ Sci Pollut Res. 2021;28:1–11.
Cleland C, Ferguson S, Ellis G, Hunter RF. Validity of the international physical activity questionnaire (IPAQ) for assessing moderate-to-vigorous physical activity and sedentary behaviour of older adults in the United Kingdom. BMC Med Res Methodol. 2018;18(1):176.
Poustchi H, Eghtesad S, Kamangar F, Etemadi A, Keshtkar A-A, Hekmatdoost A, et al. Prospective epidemiological research studies in Iran (the PERSIAN Cohort Study): rationale, objectives, and design. Am J Epidemiol. 2018;187(4):647–55.
Feng W, Cui X, Liu B, Liu C, Xiao Y, Lu W, et al. Association of urinary metal profiles with altered glucose levels and diabetes risk: a population-based study in China. PloS One. 2015;10(4):e0123742.
Rocha G, Steinbach C, Munhoz J, Madia M, Faria J, Hoeltgebaum D, et al. Trace metal levels in serum and urine of a population in southern Brazil. J Trace Elem Med Biol. 2016;35:61–5.
Shirai S, Suzuki Y, Yoshinaga J, Mizumoto Y. Maternal exposure to low-level heavy metals during pregnancy and birth size. Environ Sci Pollut Res Int Part A. 2010;45(11):1468–74.
Al-Saleh I, Shinwari N, Mashhour A, Mohamed GED, Rabah A. Heavy metals (lead, cadmium and mercury) in maternal, cord blood and placenta of healthy women. Int J Hyg Environ Health. 2011;214(2):79–101.
Fort M, Cosín-Tomás M, Grimalt JO, Querol X, Casas M, Sunyer J. Assessment of exposure to trace metals in a cohort of pregnant women from an urban center by urine analysis in the first and third trimesters of pregnancy. Environ Sci Pollut Res. 2014;21(15):9234–41.
Baron R, Manniën J, de Jonge A, Heymans MW, Klomp T, Hutton EK, et al. Socio-demographic and lifestyle-related characteristics associated with self-reported any, daily and occasional smoking during pregnancy. PLoS One. 2013;8(9):e74197.
Weidenhamer JD, Kobunski PA, Kuepouo G, Corbin RW, Gottesfeld P. Lead exposure from aluminum cookware in Cameroon. Sci Total Environ. 2014;496:339–47.
Orisakwe OE, Nduka JK, Amadi CN, Dike DO, Bede O. Heavy metals health risk assessment for population via consumption of food crops and fruits in Owerri, South Eastern, Nigeria. Chem Central J. 2012;6(1):77.
Zeng X, Xu X, Qin Q, Ye K, Wu W, Huo X. Heavy metal exposure has adverse effects on the growth and development of preschool children. Environ Geochem Health. 2019;41(1):309–21.
Zaw YH, Taneepanichskul N. Blood heavy metals and brain-derived neurotrophic factor in the first trimester of pregnancy among migrant workers. PloS One. 2019;14(6):e0218409.
Lin Z, Chen X, Xi Z, Lin S, Sun X, Jiang X, et al. Individual heavy metal exposure and birth outcomes in Shenqiu county along the Huai River Basin in China. Toxicol Res. 2018;7(3):444–53.
Wenzel AG, Brock JW, Cruze L, Newman RB, Unal ER, Wolf BJ, et al. Prevalence and predictors of phthalate exposure in pregnant women in Charleston, SC. Chemosphere. 2018;193:394–402.
Wai KM, Mar O, Kosaka S, Umemura M, Watanabe C. Prenatal heavy metal exposure and adverse birth outcomes in Myanmar: a birth-cohort study. Int J Environ Res Public Health. 2017;14(11):1339.
Chang CH, Liu CS, Liu HJ, Huang CP, Huang CY, Hsu HT, et al. Association between levels of urinary heavy metals and increased risk of urothelial carcinoma. Int J Urol. 2016;23(3):233–9.
DiBenedetto MK, Schunk DH. Self-efficacy in education revisited through a sociocultural lens. Big Theor Revisit. 2018;2(1):117.
Amadi CN, Igweze ZN, Orisakwe OE. Heavy metals in miscarriages and stillbirths in developing nations. Middle East Fertil Soc J. 2017;22(2):91–100.
Yang Y, Liu H, Xiang X-h, Liu F-y. Outline of occupational chromium poisoning in China. Bull Environ Contam Toxicol. 2013;90(6):742–9.
Chien L-C, Han B-C, Hsu C-S, Jiang C-B, You H-J, Shieh M-J, et al. Analysis of the health risk of exposure to breast milk mercury in infants in Taiwan. Chemosphere. 2006;64(1):79–85.
Turan K, Arslan A, Uçkan K, Demir H, Demir C. Change of the levels of trace elements and heavy metals in threatened abortion. Chin Med J. 2019;82(7):554–7.
García-Esquinas E, Pérez-Gómez B, Fernández MA, Pérez-Meixeira AM, Gil E, de Paz C, et al. Mercury, lead and cadmium in human milk in relation to diet, lifestyle habits and sociodemographic variables in Madrid (Spain). Chemosphere. 2011;85(2):268–76.
Dan EU, Ebong GA. Impact of cooking utensils on trace metal levels of processed food items. Ann Food Sci Technol. 2013;14(2):350–5.
Cabrera C, Lloris F, Gimenez R, Olalla M, Lopez MC. Mineral content in legumes and nuts: contribution to the Spanish dietary intake. Sci Total Environ. 2003;308(1–3):1–14.
Kuligowski J, Halperin KM. Stainless steel cookware as a significant source of nickel, chromium, and iron. Arch Environ Contam Toxicol. 1992;23(2):211–5.
Said SA. The impact of using the scratched utensil on food contamination with heavy metals. IOSR J Environ Sci. 2015;9(5):2319–99.
Castro-González M, Méndez-Armenta M. Heavy metals: Implications associated to fish consumption. Environ Toxicol Pharmaco. 2008;26(3):263–71.
Ajaezi GC, Amadi CN, Ekhator OC, Igbiri S, Orisakwe OE. Cosmetic use in Nigeria may be safe: a human health risk assessment of metals and metalloids in some common brands. J Cosmet Sci. 2018;69(6):429–45.
Kohli NJV. Here are other celebs who said no to endorsements on ethical grounds. SCOOPWHOOP. 2017;14:2017.
Mohammed T, Mohammed E, Bascombe S. The evaluation of total mercury and arsenic in skin bleaching creams commonly used in Trinidad and Tobago and their potential risk to the people of the Caribbean. Public Health Res. 2017. https://doi.org/10.4081/jphr.2017.1097.
Nourmoradi H, Foroghi M, Farhadkhani M, Vahid Dastjerdi M. Assessment of lead and cadmium levels in frequently used cosmetic products in Iran. J Environ Public Health. 2013;2013:1–5.
Chiba M, Masironi R. Toxic and trace elements in tobacco and tobacco smoke. Bull World Health Organ. 1992;70(2):269.
Rzymski P, Tomczyk K, Poniedzialek B, Opala T, Wilczak M. Impact of heavy metals on the female reproductive system. Ann Agric Environ Med. 2015;22(2):259–64.
Mittal S. Smoking and tobacco use: Ill effects on reproductive, maternal, newborn, child health, and adolescent (RMNCHA) program—a review. Ann Natl Acad Med Sci. 2019;55(02):065–73.
Tetens I, Sjödin AM. Scientific opinion on health benefits of seafood (fish and shellfish) consumption in relation to health risks associated with exposure to methylmercury. EFSA J. 2014;12(7):3761.
Alidadi H, Dehghan A, Zamand S, Mohammadi A, Taghavimanesh V, Akbari SN. Evaluation the amount of lead, cadmium, mercury, nickel and tin in canned tuna fish available in Neyshabur markets in 2017. J Neyshabur Univ Med Sci. 2019;7(2):48–57.
Yu X-D, Yan C-H, Shen X-M, Tian Y, Cao L-L, Yu X-G, et al. Prenatal exposure to multiple toxic heavy metals and neonatal neurobehavioral development in Shanghai. China Neurotoxicol Teratol. 2011;33(4):437–43.
Blaurock-Busch E, Amin OR, Rabah T. Heavy metals and trace elements in hair and urine of a sample of Arab children with autistic spectrum disorder. Maedica. 2011;6(4):247–57.
Jaishankar M, Tseten T, Anbalagan N, Mathew BB, Beeregowda KN. Toxicity, mechanism and health effects of some heavy metals. Interdiscip Toxicol. 2014;7(2):60.
Jan AT, Azam M, Siddiqui K, Ali A, Choi I, Haq QMR. Heavy metals and human health: mechanistic insight into toxicity and counter defense system of antioxidants. Int J Mol Sci. 2015;16(12):29592–630.
Milnerowicz H, Nowak P, Wielogorska D, Wochynski Z, Sobiech K. Effects of moderate physical exercise on blood and urine concentrations of cadium and metallothionein in runners. Biol Sport. 2004;21(1):81–92.
Kawamura T, Muraoka I. Exercise-induced oxidative stress and the effects of antioxidant intake from a physiological viewpoint. Antioxidants. 2018;7(9):119.
Giimiistas MK. Lipid peroxidation, erythrocyte superoxide-dismutase activity and trace metals in young male footballers. Yonsei Med J. 2003;44(6):979–86.
Lauwerys RR, Bernard A. Early detection of the nephrotoxic effects of industrial chemicals: state of the art and future prospects. Am J Ind Med. 1987;11(3):275–85.
Acknowledgements
The authors are grateful to the team of the Department of Environmental Health Engineering, Isfahan University of Medical Sciences (IUMS)
Funding
This study was financially supported by Isfahan University of Medical Sciences. This paper was extracted from a PhD thesis funded by Isfahan University of Medical Sciences (IR.MUI.RESEARCH.REC.1399.065), and Project Number # 399025.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors announce that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Moradnia, M., Movahedian Attar, H., Heidari, Z. et al. Monitoring of urinary arsenic (As) and lead (Pb) among a sample of pregnant Iranian women. J Environ Health Sci Engineer 19, 1901–1909 (2021). https://doi.org/10.1007/s40201-021-00743-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40201-021-00743-5