Abstract
Micro(nano)plastic (MNP) pollutants have not only impacted human health directly, but are also associated with numerous chemical contaminants that increase toxicity in the natural environment. Most recent research about increasing plastic pollutants in natural environments have focused on the toxic effects of MNPs in water, the atmosphere, and soil. The methodologies of MNP identification have been extensively developed for actual applications, but they still require further study, including on-site detection. This review article provides a comprehensive update on the facile detection of MNPs by Raman spectroscopy, which aims at early diagnosis of potential risks and human health impacts. In particular, Raman imaging and nanostructure-enhanced Raman scattering have emerged as effective analytical technologies for identifying MNPs in an environment. Here, the authors give an update on the latest advances in plasmonic nanostructured materials-assisted SERS substrates utilized for the detection of MNP particles present in environmental samples. Moreover, this work describes different plasmonic materials—including pure noble metal nanostructured materials and hybrid nanomaterials—that have been used to fabricate and develop SERS platforms to obtain the identifying MNP particles at low concentrations. Plasmonic nanostructure-enhanced materials consisting of pure noble metals and hybrid nanomaterials can significantly enhance the surface-enhanced Raman scattering (SERS) spectra signals of pollutant analytes due to their localized hot spots. This concise topical review also provides updates on recent developments and trends in MNP detection by means of SERS using a variety of unique materials, along with three-dimensional (3D) SERS substrates, nanopipettes, and microfluidic chips. A novel material-assisted spectral Raman technique and its effective application are also introduced for selective monitoring and trace detection of MNPs in indoor and outdoor environments.
Graphical abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Plastics of polystyrene (PS), polypropylene (PP), polyethylene (PE), polyethylene terephthalate (PET), and so on are today not only extremely versatile, but also the most-produced polymer materials worldwide [1, 2]. These polymers have been introduced as significant components for specific manufacturing processes due to their unique properties of cost-effectiveness, flexible shaping, and long-lasting robustness. Plastic materials are currently used in universal applications for the industrial manufacturing of numerous commercial products, including automotive parts (e.g., instrument panels, knobs, and trim), electronic components (interior and exterior materials for displays, calculators, and computers), food processing (packaging and labeling), temperature control apparatuses (heaters and refrigerators), medical devices (diagnostic components, test tubes, disposable products), and storage (foams and compact discs) [3, 4]. Although plastic materials have provided many economic benefits and conveniences in modern life, they have also caused serious risks, inducing harmful impacts on global public health and ecological influences over the natural environments of soil, plants, and oceans [5, 6]. The implications of the COVID-19 pandemic on plastic pollution have become one of the most pressing environmental issues [7, 8]. For example, the harmful effects of either polycarbonate microplastics [9] or PS MNPs may critically impact the health of plastic factory workers and general consumers who use these products due to their uptake via various routes, including inhalation [10, 11], dermal contact [12, 13], and ingestion of PS-contaminated foods, water, and marine products [14, 15].
The severe impacts of MNPs comprise various aspects of degradation and their fate in natural environments. Polluted plastic fragments (nano- or micro-sized) are a crucial issue for aquatic organisms, such as zebrafish [16], in the case of either ingestion or uptake, resulting in negative metabolic responses, such as neurotoxicity, pulmonary toxicity, organ accumulation, oxidative stress, and organ damage [17, 18]. In addition, MNPs can influence the natural environment not only by themselves, but also in combination with other toxic contaminants, including benzo(a)pyrene [19], cadmium [20], arsenic [21], and bisphenol A [22], through adsorption and desorption processes. In particular, MNPs with different surface charges [23], shapes (e.g., microplastic fibers and granules) [24], and sizes [25, 26] cause the risk of chronic exposure in living organisms. Furthermore, these plastic products typically have numerous additives aimed at improving their properties, such as phthalates [27] and polybrominated diphenyl ethers [28]. These toxic additives can be released into the natural environment from plastic products, resulting in toxic effects in aquatic ecosystems. In this case, plastic products have been an indirect source of pollutants [29]. For this reason, the waste disposal of plastic products [1, 3] is a significant issue because of the threat from MNP pollutants in global ecological systems [30, 31].
Consequently, a series of studies on the chronic toxicity of MNP exposure has reported on potential risks that may be linked to various human health issues, including endocrine disturbances, reproductive toxicity [32], neurotoxicity, gastrointestinal toxicity, liver toxicity [14], cardiac fibrosis [33], pulmonary fibrosis, and even carcinogenesis [10]. Additionally, a line of proof has emerged of negative effects from exposure to PS MNPs and additives for blood plasma [34, 35] and human health [36]. Based on scientific evidence, the serious risks of PS MNPs have been fully justified as a cause of cellular toxicity in mammals [37]. Many researchers have studied the chemical and physical toxicities of micro-sized (or nano-sized) PS fragments by in vitro experiments using [13], for example, human lung cells [38], human HepG2 cells [39], human-derived cells [40], RAW 264.7 macrophages [41], human gastric adenocarcinoma cells [42], and L02 cells [43]. Furthermore, in vivo experiments were conducted using mice [26, 44], Daphnia magna [45], zebra fish [17], red tilapia [25], and fathead minnow [46].
In addition to these investigations of toxicity, to provide an early warning about the potential risks of MNPs, many researchers have evaluated the current research on microplastics by means of the systematic assessment [47] of quality assurance/quality control and data quality [48]. Recently, there has been a large number of scientific reviews promising to elucidate not only the effects of MNP pollution on risks to human health [49] and the environment [50], but also many methodologies for the trace detection of MNPs in aquatic [51, 52], sedimentary [53, 54], freshwater, and coastal ecosystems [55], as well as other environments [56, 57], in wastewater [58], and on beaches [59]. Furthermore, several researchers have investigated a variety of analytical technologies for the trace detection of MNPs, including fluorescence microscopy [18, 60,61,62], impedance spectroscopy [63], microwave-based techniques [64], hyperspectral imaging [65, 66], Fourier transform infrared spectroscopy (FTIR) [67, 68], near-infrared (NIR) hyperspectral imaging and chemometrics [69], semi-automated analysis [70], a thermo analytical method [71], and mass- or particle-based analysis [72]. However, these thermal analysis methodologies often require additional procedures to separate analytes in the sample preparation, leading to time-consuming and toxic organic solvents, as well as further adverse effects on the environment. The detection of MNPs require more consideration and a focus on novel detection methods, such as plasmonic resonance-based Raman spectroscopy sensors. Interestingly, spectroscopic methods that have been well investigated and shown to provide highly sensitive trace detection of MNPs include Raman scattering microscopy methods, such as Raman imaging, normal Raman spectrum [73, 74], deep learning for reconstructing low-quality FTIR and Raman spectra [75], or the combination of Raman imaging and matrix-assisted laser desorption/ionization mass spectrometry [76], thermal gravimetric analysis, FTIR spectroscopy, or gas chromatography mass spectrometry [77]. Recently, metal plasmonic nanostructures have emerged as potential materials utilized in various fields such as energy application [78, 79], catalytic reduction [80], and efficient dye removal [81]. Especially, since plasmonic nanomaterials exhibit a powerful ability to amplify signal intensities in sensor technologies [82,83,84,85], there is a series of studies on the development of novel and unique nanostructures used for technique detection such as 3D nanocomposite-assisted tartrazine determination [86], sensitive guanine-based DNA biosensor [82], cyanazine herbicide monitoring [83], and Ponceau dyes sensing [87]. Furthermore, due to their excellent enhancement of resonance Raman scattering spectroscopy, many researchers have been interested in developing the plasmonic nanomaterials-based SERS sensing method. In particular, also successfully developed for MNP detection are the plasmonic nanostructure-based SERS technique [88,89,90,91] with supporting silver nanoparticles (AgNPs) [92], gold nanoparticle (AuNP) substrates [93], gold nanostars [94], commercialized Klarite substrates [95], AuNP-decorated sponges [96], silver-coated gold nanostars [97], a single gold nanopore fabricated at the tip of a glass nanopipette [98], SiO2 self-assembly sputtered with Ag films [99], Ag nanowire arrays on regenerated cellulose (RC) films [100], and microfluidic chips [101].
Considering that having environmental significance, MNP pollutants have impacted not only ecological systems but also human health [102, 103], this mini-review provides a critical updated and comprehensive overview of published works regarding the on-site detection of MNPs by means of Raman spectroscopy, which is attracting scientific attention. There have been numerous review studies on the effects of microplastics on freshwater environments [104], drinking water [105], the atmosphere [106, 107], seawater and sediment [108], terrestrial plants [109, 110], soil [111], groundwater [112], aquatic environments [113], various organisms [16, 114], cellular toxicity in mammals [37], potential health repercussions [115, 116], and much more. However, further study is still urgently needed regarding trace detection of MNPs that are aimed at early warning of harsh effects on human health based on accumulated evidence, which must be done before it is too late due to the tremendous manufacturing utility of plastics. Although there have been several reports on MNP detection using thermogravimetric analysis [117], Raman spectroscopy [118], FTIR and Raman [119], NIR and nuclear magnetic resonance (NMR) [120], spectroscopy and thermal analysis [121], and many methods [122, 123], there are few reviews focusing solely on surface-enhanced Raman spectroscopy, despite its universal application. This review evaluates and discusses recent developments and trends in designing uniquely structured materials that aid in platforms used for the efficient detection of MNPs that pose a risk to human health to understand the correlations between the Raman technique and plasmonic resonance phenomena (Fig. 1). The main content used for on-site detection of MNPs by Raman spectroscopy can be divided into three parts: (1) Raman detection of MNPs, including Raman imaging and normal Raman spectra; (2) novel plasmonic nanostructure-based SERS detection of MNPs, including pure noble metals, hybrid nanomaterials, and 3D SERS substrates; and (3) other materials that introduce SERS detection of MNPs, including nanopipettes and microfluidic chips. This literature provides sufficient evidence supporting the analysis of MNPs in the natural environment with different levels of dose, sizes, shapes, and surface charges that have adverse impacts on human health. In addition, this article includes recently updated developments on convenient SERS technology and trends in the functionalization of unique nanostructure-introduced monitoring of MNPs. The current challenges for and perspectives on novel nanomaterial templates used for SERS are discussed in terms of the detection of MNPs.

Copyright 2020, American Chemistry Society. Adapted with permission from Ref. [146]. Copyright 2021 under the terms and conditions provided by Elsevier and Copyright Clearance Center
Pathways of MNP particle exposures to human
MNP particle exposure can occur directly to workers in industrial manufacturing or people using plastic household products. There are three main pathways for human exposure to MNPs: inhalation, dermal contact, and digestion [114]. After entering the human body, MNP particles show basic effects in the physical, chemical, microbiological, and immune barriers of the body, leading to dispersal throughout the body and inducing toxic effects [116]. Also, the toxic risks of MNPs could depend on various factors, including the dosage, length of exposure, and the specific plastic particle-associated contaminants involved.
Inhalation
It is recognized that evidence has emerged that airborne MNP particle pollutants damage health due to their production processes and uses [10, 106]. The inhalation of MNP particles is a critical issue associated with health risks [24]. The potential risks from respiratory exposure are significant due to the small sizes (micro or nano) of plastic particles, which could cause serious damage directly to tissues of the nose, throat, and lungs [38, 124]. A study of the atmosphere in the metropolitan area of Hamburg, Germany, indicated that the atmospheric deposition of microplastics was at an estimated level of 275 microplastic particles/m2/day [125]. In addition, a group of researchers has found that there are many risks for respiratory threats to using products containing expanded PS, because not only styrene monomers but also other chemicals (volatile organic compounds) have been emitted from commercial products containing expanded PS beads. Accordingly, the emission fluxes of styrene monomer (µg/(m2h)) were measured at different temperatures to illustrate typical product use. At 25 °C, this value was estimated from 25.3 to 8.73 × 103 for the samples. In the case of increased temperature from 36 °C (human body’s temperature) to 50 °C (inside an automobile’s temperature in summer), this value significantly increased from range = 124–2.44 × 104 to range = 474–4.59 × 104, respectively. In a bedroom and automobile at 36 °C, due to styrene emissions from expanded PS beads, the maximum estimated styrene concentrations were higher than the relevant guidelines in air [11].
Dermal contact
Contact with MNP particles in water or air causes exposure [126,127,128], and due to daily use of commercial products containing MNP particles, dermal contact is one of the most effective and common pathways of human intake [12]. The level of dermal exposure to MNP particles may depend not only on the sizes of the particles, but also on their concentration. The size of MNP particles may be one of the main factors leading to skin pore penetration, and this has been investigated by many scientists. For example, numerous researchers have studied a systematic comparison of neurotoxic hazard risk by not only different types, shapes, and sizes of plastic particles, but also by exposure to different concentrations and for different durations [129]. High concentrations may also be a critical feature in exposure absorption that results in the bioaccumulation of large quantities of MNP particles in the human body. Additionally, the duration of contact or the chemical contaminants in the MNP particles may also increase skin absorption.
Ingestion
MNP pollutants can enter the food chain, leading to potential harsh impacts on human health [15, 108, 130]. There is one report that MNPs were generated when opening plastic packaging [4]. Many scientists have indicated that oral ingestion of MNP particles may cause the most extreme poisoning, resulting in bioaccumulation in the intestines and liver [131]. For example, researchers have reported on the toxicity of nano-sized polymethylmethacrylate (PMMA) plastic particles dependent on the exposure route and food items [132]. Oral exposure to MNP particles through the frequent use of bottles or food containers means that the MNPs enter the human body through the food chain [102], primarily in drinking water [105] and seafood [133]. Many scientific investigations have illustrated the negative effects on health caused by drinking MNP particles from soft drink bottles or after drinking water stored in bottles contaminated with MNP particles. For example, many researchers have underestimated the health risks of PS MNP particles that have jointly induced intestinal barrier dysfunction by reactive oxygen species-mediated epithelial cell apoptosis and lipid storage distribution [44, 134]. Workers handling plastics or equipment for the application of plastics may also ingest plastics if they do not wash their hands prior to eating or smoking. Consequently, those handling such applications should be carefully instructed on the handling of nano-sized plastics, including warnings not to clear a spray line or nozzle by blowing through it with their mouth.
MNP detection by Raman spectroscopy
Analytical technologies based on spectroscopic Raman—including Raman imaging, normal Raman spectroscopy, and SERS spectra—have been widely adopted in many fields. Raman platforms used for SERS methods may be divided into pure noble metal nanostructured materials, hybrid nanomaterials, 3D Raman substrates, or other materials.
Raman imaging
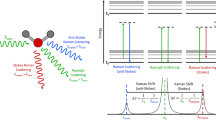
Based on different spectroscopy methods, Raman imaging— separating the species of molecules present in the sample—is reflected in two-dimensional maps [135, 136]. Due to its potential applications, this method has been universally employed in many fields, including the environment, materials, and biology [119, 137]. Based on data from multiple individual studies, Fig. 2 shows the combined summary result of the detection of MNPs by Raman imaging. For example, Fig. 2A illustrates a quick process for the detection of four ubiquitous types of MNPs—PE, PS, PP, and PET—that exist in environmental matrices worldwide. Although many efficient techniques for MNP detection have been successfully investigated after special sample preparation, it remains a significant necessity for developing a novel method allowing for the direct identification and visualization of MNP particles in real samples as well as environmental and organismal matrices. In this article, the authors first introduce a novel method of detecting MNPs in a complex background by Raman hyperspectral imaging with multivariate analysis data of curve resolution-alternating least squares, which aimed to directly identify and visualize either individual analytes or mixed analytes without a pre-treatment sample, washing, or removal of organic matter. Interestingly, multivariate curve resolution-alternating least squares method-based Raman imaging has been successfully applied to in situ MNP identification in not only environmental sand samples, but also fish feces biological samples. Moreover, this method indicated that Raman imaging with multivariate analysis of curve resolution-alternating least squares has allowed for the successful detection of MNP particles in samples that have complex backgrounds while eliminating fluorescence interference and overlapping Raman bands [135].

Copyright 2021 under the terms and conditions provided by Elsevier and Copyright Clearance Center. Adapted with permission from Ref. [138]. Copyright 2020, American Chemistry Society. Adapted with permission from Ref. [139]. Copyright 2021 under the terms and conditions provided by Elsevier and Copyright Clearance Center. Adapted with permission from Ref. [136]. Copyright 2020 under the terms and conditions provided by Elsevier and Copyright Clearance Center
Schematic diagram of spectroscopic Raman detection of MNP particles contaminating in A real-world samples by means of Raman imaging [135], B in ambient particulate matter using Raman spectral imaging [138]. C Schematic diagram of visualization and identification of MNPs via principal component analysis (PCA) to decode Raman spectral imaging [139]. D Schematic diagram of visualization and identification of MNP particles by Raman imaging [136]. Adapted with permission from Ref. [135].
In addition, Fig. 2B illustrates an optimized method using different chemometric techniques of spectral Raman imaging for microplastic particle identification in indoor and outdoor air. This study’s results demonstrate that this analysis resulted in appropriate identification of spectral Raman imaging using chemometric statistical approaches for not only virgin, but also environmental microplastic particles by size. For sensitivity, in spectral data sets, the developed analysis of an agglomerative hierarchical cluster and Pearson’s correlation are recommended for MNP identification. The results also show a potential application in the identification of airborne microplastic particles outdoors, using data obtained in an urban part of London, UK. Airborne microplastics have been raised as an area of concern for human exposure, and this method can detect exposure concentrations of airborne microplastics, providing early warnings of potential human health impacts before it becomes too late [138].
Figure 2C shows a comparison of two types of decoding approaches to visualizing MNPs by means of Raman imaging. The first approach—a logic-based algorithm—is used to merge images of samples containing several standard microplastics for which the spectra are known. Based on a selected characteristic band, the Raman intensity can be mapped as one selected image, or several images at characteristic bands can be merged into one image. However, this approach means a low signal-to-noise ratio because signals that are not selected peaks are ignored. The second approach—a principal component analysis-based algorithm—is used to decode the spectrum matrix for complex samples containing several microplastics for which spectral standards are unknown. In this study, the authors successfully justified and assigned unclear samples to MNPs and other materials by selecting and generating principal component score curves to mimic Raman spectra. In this way, the MNPs and other materials were simultaneously visualized by mapping the principal component loadings as images. Overall, the principal component analysis-based algorithm that decoded the spectrum matrix for an accurate map showed some advantages over visualizing the MNPs via Raman imaging [139]. Other authors have investigated various factors influencing the quality of Raman imaging in visualizing and identifying MNPs < 100 nm, such as a distinguishing laser spot, pixel size or image resolution, size or position of MNPs within a laser spot, signal intensity of Raman spectra, steps of pre-treatment of samples, and so on (Fig. 2D). In the present study, the authors successfully estimated billions to trillions of MNPs ranging in size from ~ 7 mm down to ~ 200 nm [136].
Normal Raman spectroscopy
In normal Raman spectra-based MNP detection, a laser source is used as an incident ray directly on MNP samples [118, 140]. The laser light is scattered by the bindings inside the molecular analyte, and the inelastically scattered light is collected and processed into a Raman spectroscopic image [141]. The Raman spectroscopy-assisted sensor technique is a simple method that has been widely used in the selective detection of analytes based on their distinguishing spectra in the sample background. This method can be widely applied in the environment [142], food, and biology [143,144,145]. For example, as shown in Fig. 3A, this study successfully combined micro-Raman spectroscopy and Nile red staining for a novel analytical method of MNP detection. Micro-Raman spectroscopy with 442 nm laser-induced Nile red luminescence could target specific MNP identification via an orange filter, which leads to reducing the number of MNP particles for improving sample throughput and selective detection. Besides effective quantified properties, Nile red staining has been advantageously applied with little interference as an objective criterion for the pre-selection detection of MNP particles by micro-Raman spectroscopy. This method has therefore successfully detected targeted analysis of MNP particles after the Nile red staining dye induced a map of suspected MNPs [146].

Copyright 2021 under the terms and conditions provided by Elsevier and Copyright Clearance Center. Adapted with permission from Ref. [147]. Copyright 2021 under the terms and conditions provided by Elsevier and Copyright Clearance Center
A Schematic diagram of Raman spectroscopy-based selective detection of MNPs using Nile red staining [146]. B Schematic diagram of Raman spectroscopic detection of MNP particles in water samples via a particle capturing medium as well as perfluorocarbon [147]. Adapted with permission from ref. [146].
Figure 3B illustrates a simple scheme for online MNP detection in water using Raman spectroscopy. Here, perfluorohexane (PFH) is used as an MNP-capturing medium rather than a conventional physical filter for MNP separation. Due to its high density and hydrophobicity, PFH is induced as a firm droplet at the bottom of the 90° curve of a water-filled L-shaped tube. Subsequently, a sample of tap water containing PE particles was added and flowed through the L-shaped tube, causing PE particles to be captured in the PFH droplet. For reliable quantitative analysis, a 6 mm diameter laser beam was used to measure the Raman spectral acquisition of the whole droplet instead of only partial spectroscopic sampling. In the collected spectral Raman, the quantities of dispersed PE particles in the water samples were clearly elevated based on the increased intensity ratios of Raman signal bands for PE and PFH. Due to the shapes of PFH droplets, the captured PE particle did not change with greater ionic strength, and this method successfully detected PE particles in seawater samples. The results demonstrate that this analytical scheme is feasible for the accurate field analysis of MNPs [147].
Nanomaterial-enhanced detection of MNPs
Pure noble metal-based nanostructure-enhanced detection
Due to the best enhanced ability of Raman signal, plasmonic nanomaterials have been adopted as one of the most widely used substrates for sensor technologies by means of Raman spectrum. Among numerous nanostructured materials, the pure noble metals—gold (Au) and silver (Ag)—have emerged as potential nanomaterials contributing to spectral Raman-active platform-based detection [88]. Furthermore, the unique shapes and sizes of pure noble metals have helped generate highly dense plasmonic hot spots to enhance the selective and sensitive method by means of Raman spectra. Studies have developed more unique nanostructures made from pure noble metals that aimed to maximize the enhancements for establishing a high density of plasmonic hot spots, leading to not only enhanced electromagnetic fields but also amplified Raman signals [148]. In addition to the synthesis of new nanostructures, modification of the surface of nanostructured materials has been investigated using specific probes for Raman sensing with sensitive and selective abilities. For example, Fig. 4A shows the great effort made to detect MNPs in aquatic environments by means of in situ SERS spectroscopy. AgNPs based MNP detection in environmental samples has been evaluated in comparisons of the potential SERS spectroscopy with normal Raman spectra for the analysis of MNPs. In the present study, the authors tested various conditions—including the ratio of the sample volume to AgNPs, NaCl concentrations, and sample concentrations—to assess the detection of MNPs, such as PS, PE, and PP, in either pure water or seawater samples. Due to the Raman-active substrate showing good, enhanced efficiency, the AgNP-assisted SERS method was successfully developed for the qualitative detection of MNPs of various sizes as well as 100 nm, 500 nm, and 10 µm. The optimal enhancement factor was estimated to be 104, and it was also demonstrated that the AgNP-based SERS detection method could overcome the limitations to the detection of MNPs in liquids or the detectability of small-sized plastics ranging from 100 nm down to 40 µg/mL. The results indicate that this potential method could be applied to the rapid detection of MNPs in aquatic environments in the future [92].

Copyright 2020 under the terms and conditions provided by Elsevier and Copyright Clearance Center. Adapted with permission from Ref. [100]. Copyright 2021 under the terms and conditions provided by Elsevier and Copyright Clearance Center
A Schematic diagram of in situ SERS spectroscopy-introduced MNPs detection in aquatic environments using AgNPs [92]. B FE-SEM images of PS nano-sized particles on the plasmonic nanoparticles/RC film: B-a 630 nm PS nanoparticles on an AuNRs/RC film (surface density of AuNRs on RC: 0.063 mg/cm2); B-b 630 nm PS nanoparticles on an AgNWs/RC film; and B-c 84 nm PS nanoparticles on an AgNWs/RC film (surface density of AgNWs on RC: 0.99 mg/cm2). SERS spectra of B-d 84 nm PS nanoparticles (0.1–10 mg/mL), and B-e 630 nm PS nanoparticles (0.1–10 mg/mL) on AgNWs/RC films [100]. Adapted with permission from Ref. [92].
Recently, MNP pollution has steadily become a crucial global issue due to the universal manufacture of MNPs and their use in plastic products [54, 149]. Figure 4B illustrates rapid MNP detection using a novel substrate that was synthesized from plasmonic nanostructured materials—Au nanorods (AuNRs) and Ag nanowires (AgNWs)—applied to RC hydrogel films. The authors reported that the enhanced SERS signal of the crystal violet AgNWs/RC film was estimated to be approximately six times greater than that from AuNRs/RC film, which exhibited the SERS active array with a high enhanced factor of 107. In this way, AgNWs/RC film substrates have provided great potential for nano-sized PS detection with a limited concentration down to 0.1 mg/mL. Due to many advantages, including good reproducibility, flexibility, low cost, and ultra-sensitive characteristics of the nano-sized PS-SERS signal, this AgNWs/RC film could provide a potential application in SERS-based rapid detection of nano-plastics in an environment [100].
Hybrid nanomaterial-assisted nanostructure-enhanced detection
In addition to the pure noble metals, hybrid nanomaterials of either bimetallic nanostructures or nanocomposites have been adapted by using effective plasmonic materials for Raman-active substrates, which are applied in a variety of fields of biosensors and environments [150]. Because MNP pollutants are a serious ecological threat [52, 66], researchers have investigated the label-free identification of several MNPs using fluorescence lifetime imaging microscopy [60] and the weathering of MNP interactions with coexisting constituents [20,21,22, 151]. In addition, Fig. 5 shows a unique NIR SERS-labeled MNP model that aimed to obtain not only signal stability, but also completely prevent false positive problems in both complex biological environments and in vivo. The authors provided an ideal analytical tool for a reliable bio-MNP interaction investigation of various organisms. In vivo behavior imaging has been successfully studied in living adult zebrafish and zebrafish embryos, as well as in Brassica rapa green vegetables. Due to the many advantages of MNP particles, including ultra-sensitive single MNP particles, anti-interference, multiplex labeling ability, and detection of deep tissue, the applicability of MNP particles from the aspect of optical properties has been successfully verified by means of plasmonic gold nanostar-based SERS spectra [94].
A-a Schematic diagram of synthesis procedure of SERS labeled nano-plastic models (SERS@PS) for reliable bio-nano interaction investigations. TEM image of A-b Au nanostar, A-c SERS@PS. A-d In situ Raman detection based on SERS@PS of a living zebrafish. B To investigate in the uptake of developmental SERS@PS nano-plastics, their exposures were checked at several different periods: B-a SERS@PS nano-plastics exposure at the embryonic stage (6 hpf), B-b Pictures of the dechorionated embryo and stripped chorion (36 hpf), and B-c the correlated Raman intensities; B-d SERS@PS nano-plastics exposure at the first larvae stage (48 hpf), B-e Pictures of larvae (72 hpf) containing of SERS@PS nano-plastics, and B-f the organs of larva at 48, 72 and 96 hpf-correlated Raman intensities; B-g SERS@PS nano-plastics exposure time was 0, 24 and 48 h, respectively, at the second larvae stage (120 hpf), B-h pictures of larvae (120 hpf) containing of SERS@PS nano-plastics, and the digestive system of zebrafish larvae at 0, 12, 24 and 48 h-correlated Raman intensities. The errors were checked from the measured result of either 6 embryos or larvae [94]. Adapted with permission from Ref. [94].
3D nanostructure-enhanced substrate-introduced MNP detection
Recently, the authors studied a combination of Raman microscopy and scanning electron microscopy with the goal of identifying 100 nm plastic particles in a variety of environmental samples [152]. Other authors have successfully combined 3D SERS active substrates and Raman microscopy. As shown in Fig. 6A, MNP pollutants have been detected in various environmental samples, including aquatic life and mammalian (including human) health, by Klarite substrate-based SERS spectra. Klarite is known to be an exceptional 3D SERS substrate that has a dense grid structure fabricated from Au shapes with inverted pyramidal cavities. The Klarite substrate exhibited the potential to detect a single MNP particle at sizes down to 360 nm in atmospheric/aquatic samples [95]. Figure 6B shows an MNP detection scheme using SERS spectra and a unique 3D platform. In the present study, a novel nano-well-enhanced Raman-active template was successfully developed, which was composed of self-assembled SiO2 and sputtering silver films on the surface of the SiO2 (SiO2@Ag). Due to the coffee ring effect, numerous robust nano-wells in a 3D substrate exhibited the potential to trap MNPs, leading to a specific method for testing of environmental samples. Interestingly, a single PS MNP particle was directly visualized on this substrate, and the limited detection of PS concentrations in bottled water, tap water, and river water was estimated at 5 ppm. This novel 3D template provides a method for ultra-sensitive detection of MNPs by means of SERS spectra [99]. As shown in Fig. 6C, the ultra-sensitive detection of MNPs based on SERS spectra used a novel 3D SERS active substrate, as well as a sponge supported by the AuNP layer. The AuNP-decorated sponge pores exhibited the effective ability to capture MNP particles out of water samples. This template allows for the sponge size to be adjusted, leading to control of distance and the generation of many hot spots on the surface of the 3D substrate. In turn, the many hot spots mean that the Raman signal intensity for MNPs in water is greatly enhanced on the substrate [96].

Copyright 2020, American Chemistry Society. Adapted with permission from Ref. [99]. Copyright 2021, Royal Society of Chemistry. Adapted with permission from ref. [96]. Copyright 2021 under the terms and conditions provided by Elsevier and Copyright Clearance Center. Adapted with permission from Ref. [97]. Copyright 2020 under the terms and conditions provided by Elsevier and Copyright Clearance Center
A Schematic diagram of Klarite substrates-induced SERS detection of < 1 μm MNPs in the environmental samples [95]. B Schematic diagram of the nano-well-enhanced SERS visualization and quantification of MNPs in the environmental samples [99]. C Schematic diagram of rapid and sensitive detection of trace concentrated MNP particles based on Au-nanoparticle-decorated sponge by means of SERS spectra [96]. D Self-organized Ag-coated Au nanostar dimers in nano-porous anodic aluminum oxide leading to generate Raman active substrates (AuNSs@Ag@AAO) for the sensitive detection of PS MNP pollutants in water samples. E-a 0.4 μm PS concentration-correlated SERS spectra with a range of 0–0.1% in distilled water, E-b PS concentration-corresponded calibration curve of the 1000 cm−1 SERS signal as a function of PS concentration using AuNSs@Ag@AAO platforms. Three measured SERS spectra estimate error bars indicate standard deviations [97]. Adapted with permission from Ref. [95].
To address the toxic effects of PS nano-plastics [153], the present study presents a novel 3D Raman template that sensitively detects PS MNP particles in a variety of water samples, as shown in Fig. 6D, E. The Ag-coated Au nanostar (AuNSs@Ag) dimers were well aligned as a vertical dimension array into a template containing anodized aluminum oxide (AAO) nanopores. This uniformity of AuNSs@Ag dimers illustrates a great potential application as a 3D Raman-active substrate used for trace detection of MNP pollutants, including those with diameters of 0.8–4.8 µm, in water samples. The vertical dimensions of the AuNSs@Ag dimers were well designed by the controlled diameter of the AAO pores and the diameter of the AuNSs@Ag. This unique nanostructure exhibits not only a small gap but also a unique anisotropic nanostar dimer, providing many electromagnetic field enhancements at the tips of nanomaterials. This, in turn, leads to highly dense hot spots that induce an enhanced Raman signal of PS MNPs. This study found that AuNSs@Ag dimer-based 3D SERS active substrates yielded stronger Raman signals at the same weight ratios for PS MNP particles with diameters as small as 0.4 µm, whereas such behaviors could not be observed for larger PS MNP particles. The limited detection of 0.4 µm PS MNP particles by means of SERS spectra was estimated to be ∼ 0.05 mg/g and was accompanied by not only a fast detection time, but also no sample pretreatments. This 3D SERS template composed of nano-sized AuNSs@Ag dimer vertical dimensional matching platforms may prove to be a useful analytical tool for detecting MNP pollutants in water samples [97].
Other material-supported MNP detection
Nanopipettes
Over the past two decades, MNPs have become an increasingly crucial issue, and a series of studies have focused on analyzing MNPs in environmental samples [8, 55, 70, 122, 154]. This topic still attracts the most scientists, who have widely investigated and performed a variety of novel analytical methodologies for MNP detection [103, 117, 155]. Recently, nanopipettes, which are fabricated by applying heat during the pull-out process of a glass capillary, have been described as exhibiting many potential applications in the analytical community [156]. Nanopipettes emerged not only as a route for nanoscale studies of ion transport, but also the development of chemical and biochemical sensors [157, 158]. Figure 7A shows a single Au nanopore with a highly SERS active template fabricated on the tip of a glass nanopipette. PS nanoparticles can be identified by means of the SERS spectrum while passing through a single nanopore, as shown in Fig. 7B [98].

Copyright 2019, Royal Society of Chemistry. Adapted with permission from Ref. [101]. Copyright 2021, Springer Nature
A-a Schematic diagram of preparation a glass nanopipette which has fabricated a single Au nano-pore at the tip. SEM images of A-b the bare glass nanopipette, A-c the Au nanopores-coated glass nanopipette, A-d and Au nanopores formed by one high voltage electric pulse in 0.1 mM KCl. B The waterfall plot of 0.5% PS concentration-correlated SERS spectra in 100 mM KCl with– 1 V potential [98]. C-a A microscope picture of microfluidic chip reservoir containing trapped PS particles (diameter 80 µm), corresponding with Raman spectrum. C-b A microscope picture of microfluidic chip reservoir containing a mixture of 20 µm PS particles and 6 µm PMMA particles, corresponding with Raman spectra. C-c A microscope picture of microfluidic chip reservoir containing 10 µm PMMA particles, corresponding with Raman spectroscopy of a single 10 µm PMMA particle [101]. Adapted with permission from Ref. [98].
Microfluidic chips
There is still a need to develop and improve a novel analytical method for detecting MNP particles in environmental samples within a short time [51, 53, 56, 57, 67, 121, 159]. Figure 7C displays a novel micro‑optofluidic platform used for fast detection of MNP particles by means of Raman spectra. The micro-optofluidic platform was fabricated with micro‑reservoirs leading to micro‑filters that are designed to trap all MNP particles in an ultra‑compact area. The researchers successfully enabled fast optical spectroscopy and imaging to detect the MNP nature and type contained in this ultra‑compact area. Furthermore, according to the size ranges in different reservoirs, passive size sorting was implemented for sorting the MNP particles. A reference method with flow cytometry was used to retrieve the distributed sizes of the MNP samples, but data on the chemical nature was therefore lost. To validate the potential application of this micro‑optofluidic platform, model samples containing standard MNP particles of different sizes and chemical natures were mixed and successfully checked [101].
According to several previous reports on MNP identification, instead of using Raman imaging and normal Raman spectroscopy, plasmonic nanomaterial substrates-assisted SERS detection has been successfully developed to detect MNP pollution in environments with lower concentrations than without nanostructures. Depending on the ingredient of real samples, the authors have been many great efforts in developing novel technologies for appropriate MNP monitoring, respectively. To develop unique SERS plasmonic nano-substrates, researchers have fabricated SERS platforms used for detected MNP methods including pure noble metal nanostructured materials, hybrid nanomaterials, 3D Raman substrates, or other materials (nanopipettes and microfluidic chips). Those platforms efficiently monitored Raman signals of MNP particles from practice samples. MNP could be successfully identified by distinct signals, even though there is no specific separated processing. However, there are several existing literature reviews on the topic of plasmonic platform-based SERS detection for MNP particles, this work is still different in comparison with previous reports as shown in Table 1.
SERS is a powerfully analytical technique that has been widely adopted to monitor for the direct detection of analyte molecules in a mixture without separated processing, in spite of their low normal Raman intensity. Interestingly, the nano-plasmonic substrates-assisted SERS method is known as an extreme sensitivity and selectivity detection. In addition, since the COVID-19 pandemic has appeared in the world, disposable plastic products have been over-manufacturing leading to the most pressing issues as well as plastic pollution in the environment. Although the present pollutants of MNP may be low level, it seems difficult to avoid its increment input in the natural environment. Therefore, the nanocomposite substrates-introduced SERS method offers many not only potential applications but also promising features for the detection of MNP particle pollution in environmental nature. Thus, research efforts in the combination of nanostructured materials and Raman spectra are still necessary to continuously develop the novel SERS substrates including different compositions and unique morphologies, which aimed to improve the sensitive, selective, and reliable methods that could be efficiently used in practices. Furthermore, MNP pollution is one of the crucial issues that influence direct human health, alongside a harmful impact on environmental nature. New research provides many efficient technologies protecting human health and avoiding harmful MNP pollution. As summarized in Table 2, based on Raman spectra, numerous novel optical sensor technologies were successfully developed for the trace detection of MNPs, with improvements in aspects such as selectivity and sensitivity, as well as economic and reproducible benefits. Specific aspects were studied and evaluated on the current spectroscopic and optical sensor methods, but there are several perspectives to be addressed in future studies: (1) improving optical sensor methods based on pure, noble metal nanomaterials and hybrid nanostructured materials; (2) developing more 3D SERS substrates that exhibit highly accurate target sensors; (3) exhibiting the ability of multiple detection of similar analytes under various conditions; (4) developing a dual functional sensing platform used for both Raman spectroscopy and fluorescence detection or both colorimetric and fluorescence; and (5) preparing a new nanomaterial such as nanopipette or microfluidic chip that exhibits the ability to detect trace MNP contaminants.
Conclusions
Due to universal applications in various areas, the manufacturing and application of plastic materials around the globe have continuously increased in recent years. The subsequent use of plastic products has led to MNP waste accretion in natural environments, which has emerged as a ubiquitous issue. Here, the goal of this work updates on the latest advances in plasmonic nanomaterials-assisted SERS platforms which aimed to detect the trace MNP particles existing in environmental nature. Therefore, the authors introduce various novel SERS substrates which have been fabricated from either pure noble metal or unique hybrid nanomaterials. The findings revealed that great efforts could be created the new and efficient technologies for the MNP identification, which expose not only high selectivity, but also ultra-sensitive monitoring of MNP with a low limited detection. Furthermore, to protect human health and the environment by avoiding the harsh impact of MNP particles pollution, the combination of plasmonic nanomaterials-assisted SERS substrates and Raman spectroscopy can be a critical issue attracting many researchers in the future for portable and fast analysis of MNP particles in environmental nature. According to the validated previous studies, this concise topical review provided an update on recent developments and trends in MNP detection by means of enhanced Raman scattering using a variety of unique materials, as well as 3D SERS substrates, nanopipettes, and microfluidic chips. A novel materials-assisted spectral Raman technique and its practical application were introduced for the selective monitoring of trace detection of MNPs in indoor and outdoor environments.
References
Maafa, I.M.: Pyrolysis of polystyrene waste: a review. Polymers (Basel) 13, 225 (2021)
Zettler, E.R., Mincer, T.J., Amaral-Zettler, L.A.: Life in the “plastisphere”: microbial communities on plastic marine debris. Environ. Sci. Technol 47, 7137–7146 (2013)
Okunola, A.A., Kehinde, I.O., Oluwaseun, A., Olufiropo, E.A.: Public and environmental health effects of plastic wastes disposal: a review. J. Toxicol. Risk Assess. 5, 021 (2019)
Sobhani, Z., Lei, Y., Tang, Y., Wu, L., Zhang, X., Naidu, R., Megharaj, M., Fang, C.: Microplastics generated when opening plastic packaging. Sci. Rep. 10, 4841 (2020)
Allouzi, M.M.A., Tang, D.Y.Y., Chew, K.W., Rinklebe, J., Bolan, N., Allouzi, S.M.A., Show, P.L.: Micro (nano) plastic pollution: the ecological influence on soil-plant system and human health. Sci. Total Environ. 788, 147815 (2021)
Amelia, T.S.M., Khalik, W.M.A.W.M., Ong, M.C., Shao, Y.T., Pan, H.-J., Bhubalan, K.: Marine microplastics as vectors of major ocean pollutants and its hazards to the marine ecosystem and humans. Prog. Earth Planet. Sci. 8, 12 (2021)
Patrício Silva, A.L., Tubić, A., Vujić, M., Soares, A.M.V.M., Duarte, A.C., Barcelò, D., Rocha-Santos, T.: Implications of COVID-19 pandemic on environmental compartments: is plastic pollution a major issue? J. Hazard. Mater. Adv. 5, 100041 (2022)
Chen, X., Chen, X., Liu, Q., Zhao, Q., Xiong, X., Wu, C.: Used disposable face masks are significant sources of microplastics to environment. Environ. Pollut. 285, 117485 (2021)
Shi, Y., Liu, P., Wu, X., Shi, H., Huang, H., Wang, H., Gao, S.: Insight into chain scission and release profiles from photodegradation of polycarbonate microplastics. Water Res. 195, 116980 (2021)
Facciola, A., Visalli, G., Pruiti Ciarello, M., Di Pietro, A.: Newly emerging airborne pollutants: current knowledge of health impact of micro and nanoplastics. Int. J. Environ. Res. Public Health 18, 2997 (2021)
Iizuka, A., Mizukoshi, A., Noguchi, M., Yamasaki, A.: Emission fluxes of styrene monomers and other chemicals for products containing expanded polystyrene beads. PLoS ONE 15, e0239458 (2020)
Hernandez, L.M., Yousefi, N., Tufenkji, N.: Are there nanoplastics in your personal care products? Environ. Sci. Technol. Lett. 4, 280–285 (2017)
Hwang, J., Choi, D., Han, S., Jung, S.Y., Choi, J., Hong, J.: Potential toxicity of polystyrene microplastic particles. Sci. Rep. 10, 7391 (2020)
Chang, X., Xue, Y., Li, J., Zou, L., Tang, M.: Potential health impact of environmental micro- and nanoplastics pollution. J. Appl. Toxicol. 40(1), 4–15 (2020)
Jaafar, N., Azfaralariff, A., Musa, S.M., Mohamed, M., Yusoff, A.H., Lazim, A.M.: Occurrence, distribution and characteristics of microplastics in gastrointestinal tract and gills of commercial marine fish from Malaysia. Sci. Total. Environ. 799, 149457 (2021)
Agathokleous, E., Iavicoli, I., Barcelo, D., Calabrese, E.J.: Micro/nanoplastics effects on organisms: a review focusing on “dose.” J. Hazard. Mater. 417, 126084 (2021)
Bhagat, J., Zang, L., Nishimura, N., Shimada, Y.: Zebrafish: an emerging model to study microplastic and nanoplastic toxicity. Sci. Total Environ. 728, 138707 (2020)
Li, Z., Feng, C., Wu, Y., Guo, X.: Impacts of nanoplastics on bivalve: fluorescence tracing of organ accumulation, oxidative stress and damage. J. Hazard. Mater. 392, 122418 (2020)
Batel, A., Borchert, F., Reinwald, H., Erdinger, L., Braunbeck, T.: Microplastic accumulation patterns and transfer of benzo[a]pyrene to adult zebrafish (Danio rerio) gills and zebrafish embryos. Environ. Pollut. 235, 918–930 (2018)
Davranche, M., Veclin, C., Pierson-Wickmann, A.C., El Hadri, H., Grassl, B., Rowenczyk, L., Dia, A., Ter Halle, A., Blancho, F., Reynaud, S., Gigault, J.: Are nanoplastics able to bind significant amount of metals? The lead example. Environ. Pollut. 249, 940–948 (2019)
Bhagat, J., Nishimura, N., Shimada, Y.: Toxicological interactions of microplastics/nanoplastics and environmental contaminants: Current knowledge and future perspectives. J. Hazard. Mater. 405, 123913 (2021)
Duan, J., Bolan, N., Li, Y., Ding, S., Atugoda, T., Vithanage, M., Sarkar, B., Tsang, D.C.W., Kirkham, M.B.: Weathering of microplastics and interaction with other coexisting constituents in terrestrial and aquatic environments. Water Res. 196, 117011 (2021)
Canesi, L., Ciacci, C., Fabbri, R., Balbi, T., Salis, A., Damonte, G., Cortese, K., Caratto, V., Monopoli, M.P., Dawson, K., Bergami, E., Corsi, I.: Interactions of cationic polystyrene nanoparticles with marine bivalve hemocytes in a physiological environment: role of soluble hemolymph proteins. Environ. Res. 150, 73–81 (2016)
Gasperi, J., Wright, S.L., Dris, R., Collard, F., Mandin, C., Guerrouache, M., Langlois, V., Kelly, F.J., Tassin, B.: Microplastics in air: are we breathing it in? Curr. Opin. Environ. Sci. Health 1, 1–5 (2018)
Ding, J., Huang, Y., Liu, S., Zhang, S., Zou, H., Wang, Z., Zhu, W., Geng, J.: Toxicological effects of nano- and micro-polystyrene plastics on red tilapia: are larger plastic particles more harmless? J. Hazard. Mater. 396, 122693 (2020)
Luo, T., Zhang, Y., Wang, C., Wang, X., Zhou, J., Shen, M., Zhao, Y., Fu, Z., Jin, Y.: Maternal exposure to different sizes of polystyrene microplastics during gestation causes metabolic disorders in their offspring. Environ. Pollut. 255, 113122 (2019)
Deng, Y., Yan, Z., Shen, R., Huang, Y., Ren, H., Zhang, Y.: Enhanced reproductive toxicities induced by phthalates contaminated microplastics in male mice (Mus musculus). J. Hazard. Mater. 406, 124644 (2021)
Butryn, D.M., Chi, L.H., Gross, M.S., McGarrigle, B., Schecter, A., Olson, J.R., Aga, D.S.: Retention of polybrominated diphenyl ethers and hydroxylated metabolites in paired human serum and milk in relation to CYP2B6 genotype. J. Hazard. Mater. 386, 121904 (2020)
Heinlaan, M., Kasemets, K., Aruoja, V., Blinova, I., Bondarenko, O., Lukjanova, A., Khosrovyan, A., Kurvet, I., Pullerits, M., Sihtmae, M., Vasiliev, G., Vija, H., Kahru, A.: Hazard evaluation of polystyrene nanoplastic with nine bioassays did not show particle-specific acute toxicity. Sci. Total Environ. 707, 136073 (2020)
Velusamy, K., Chellam, P., Kumar, P.S., Venkatachalam, J., Periyasamy, S., Saravanan, R.: Functionalization of MXene-based nanomaterials for the treatment of micropollutants in aquatic system: a review. Environ. Pollut. 301, 119034 (2022)
Priya, A.K., Jalil, A.A., Dutta, K., Rajendran, S., Vasseghian, Y., Qin, J., Soto-Moscoso, M.: Microplastics in the environment: recent developments in characteristic, occurrence, identification and ecological risk. Chemosphere 298, 134161 (2022)
Hojjati-Najafabadi, A., Mansoorianfar, M., Liang, T., Shahin, K., Karimi-Maleh, H.: A review on magnetic sensors for monitoring of hazardous pollutants in water resources. Sci. Total Environ. 824, 153844 (2022)
Li, Z., Zhu, S., Liu, Q., Wei, J., Jin, Y., Wang, X., Zhang, L.: Polystyrene microplastics cause cardiac fibrosis by activating Wnt/beta-catenin signaling pathway and promoting cardiomyocyte apoptosis in rats. Environ. Pollut. 265, 115025 (2020)
Gruner, M.S., Kauscher, U., Linder, M.B., Monopoli, M.P.: An environmental route of exposure affects the formation of nanoparticle coronas in blood plasma. J. Proteom. 137, 52–58 (2016)
Kik, K., Bukowska, B., Krokosz, A., Sicinska, P.: Oxidative properties of polystyrene nanoparticles with different diameters in human peripheral blood mononuclear cells (in vitro study). Int. J. Mol. Sci. 22, 4406 (2021)
Campanale, C., Massarelli, C., Savino, I., Locaputo, V., Uricchio, V.F.: A detailed review study on potential effects of microplastics and additives of concern on human health. Int. J. Environ. Res. Public Health 17, 1212 (2020)
Banerjee, A., Shelver, W.L.: Micro- and nanoplastic induced cellular toxicity in mammals: a review. Sci. Total Environ. 755(Pt 2), 142518 (2021)
Goodman, K.E., Hare, J.T., Khamis, Z.I., Hua, T., Sang, Q.A.: Exposure of human lung cells to polystyrene microplastics significantly retards cell proliferation and triggers morphological changes. Chem. Res. Toxicol. 34(4), 1069–1081 (2021)
He, Y., Li, J., Chen, J., Miao, X., Li, G., He, Q., Xu, H., Li, H., Wei, Y.: Cytotoxic effects of polystyrene nanoplastics with different surface functionalization on human HepG2 cells. Sci. Total Environ. 723, 138180 (2020)
Choi, D., Bang, J., Kim, T., Oh, Y., Hwang, Y., Hong, J.: In vitro chemical and physical toxicities of polystyrene microfragments in human-derived cells. J. Hazard. Mater. 400, 123308 (2020)
Florance, I., Ramasubbu, S., Mukherjee, A., Chandrasekaran, N.: Polystyrene nanoplastics dysregulate lipid metabolism in murine macrophages in vitro. Toxicology 458, 152850 (2021)
Forte, M., Iachetta, G., Tussellino, M., Carotenuto, R., Prisco, M., De Falco, M., Laforgia, V., Valiante, S.: Polystyrene nanoparticles internalization in human gastric adenocarcinoma cells. Toxicol. In Vitro 31, 126–136 (2016)
Li, S., Ma, Y., Ye, S., Tang, S., Liang, N., Liang, Y., Xiao, F.: Polystyrene microplastics trigger hepatocyte apoptosis and abnormal glycolytic flux via ROS-driven calcium overload. J. Hazard. Mater. 417, 126025 (2021)
Liang, B., Zhong, Y., Huang, Y., Lin, X., Liu, J., Lin, L., Hu, M., Jiang, J., Dai, M., Wang, B., Zhang, B., Meng, H., Lelaka, J.J.J., Sui, H., Yang, X., Huang, Z.: Underestimated health risks: polystyrene micro- and nanoplastics jointly induce intestinal barrier dysfunction by ROS-mediated epithelial cell apoptosis. Part Fibre Toxicol. 18, 20 (2021)
Colomer, J., Muller, M.F., Barcelona, A., Serra, T.: Mediated food and hydrodynamics on the ingestion of microplastics by Daphnia magna. Environ. Pollut. 251, 434–441 (2019)
Elizalde-Velazquez, A., Crago, J., Zhao, X., Green, M.J., Canas-Carrell, J.E.: In vivo effects on the immune function of fathead minnow (Pimephales promelas) following ingestion and intraperitoneal injection of polystyrene nanoplastics. Sci. Total Environ. 735, 139461 (2020)
Xu, L., Cao, L., Huang, W., Liu, J., Dou, S.: Assessment of plastic pollution in the Bohai Sea: abundance, distribution, morphological characteristics and chemical components. Environ. Pollut. 278, 116874 (2021)
Ziajahromi, S., Leusch, F.D.L.: Systematic assessment of data quality and quality assurance/quality control (QA/QC) of current research on microplastics in biosolids and agricultural soils. Environ. Pollut. 294, 118629 (2022)
Shi, Q., Tang, J., Liu, R., Wang, L.: Toxicity in vitro reveals potential impacts of microplastics and nanoplastics on human health: a review. Crit. Rev. Environ. Sci. Technol. 5, 1–33 (2021)
Xu, B., Liu, F., Cryder, Z., Huang, D., Lu, Z., He, Y., Wang, H., Lu, Z., Brookes, P.C., Tang, C., Gan, J., Xu, J.: Microplastics in the soil environment: occurrence, risks, interactions and fate—a review. Crit. Rev. Environ. Sci. Technol. 50, 2175–2222 (2019)
Rist, S., Hartmann, N.B., Welden, N.A.C.: How fast, how far: diversification and adoption of novel methods in aquatic microplastic monitoring. Environ. Pollut. 291, 118174 (2021)
Fu, Z., Chen, G., Wang, W., Wang, J.: Microplastic pollution research methodologies, abundance, characteristics and risk assessments for aquatic biota in China. Environ. Pollut. 266, 115098 (2020)
Balestra, V., Bellopede, R.: Microplastic pollution in show cave sediments: first evidence and detection technique. Environ. Pollut. 292, 118261 (2022)
Abel, S.M., Primpke, S., Int-Veen, I., Brandt, A., Gerdts, G.: Systematic identification of microplastics in abyssal and hadal sediments of the Kuril Kamchatka trench. Environ. Pollut. 269, 116095 (2021)
Kumar, R., Sharma, P., Bandyopadhyay, S.: Evidence of microplastics in wetlands: extraction and quantification in Freshwater and coastal ecosystems. J. Water Process. Eng. 40, 101966 (2021)
Kutralam-Muniasamy, G., Perez-Guevara, F., Elizalde-Martinez, I., Shruti, V.C.: Review of current trends, advances and analytical challenges for microplastics contamination in Latin America. Environ. Pollut. 267, 115463 (2020)
Cai, H., Xu, E.G., Du, F., Li, R., Liu, J., Shi, H.: Analysis of environmental nanoplastics: progress and challenges. Chem. Eng. J. 410, 128208 (2021)
Dey, T.K., Uddin, M.E., Jamal, M.: Detection and removal of microplastics in wastewater: evolution and impact. Environ. Sci. Pollut. Res. Int. 28, 16925–16947 (2021)
Zhang, T., Lin, L., Li, D., Wu, S., Kong, L., Wang, J., Shi, H.: The microplastic pollution in beaches that served as historical nesting grounds for green turtles on Hainan Island, China. Mar. Pollut. Bull. 173, 113069 (2021)
Monteleone, A., Schary, W., Wenzel, F., Langhals, H., Dietrich, D.R.: Label-free identification and differentiation of different microplastics using phasor analysis of fluorescence lifetime imaging microscopy (FLIM)-generated data. Chem. Biol. Interact. 342, 109466 (2021)
Maes, T., Jessop, R., Wellner, N., Haupt, K., Mayes, A.G.: A rapid-screening approach to detect and quantify microplastics based on fluorescent tagging with Nile Red. Sci. Rep. 7, 44501 (2017)
Annenkov, V.V., Danilovtseva, E.N., Zelinskiy, S.N., Pal’shin, V.A.: Submicro- and nanoplastics: how much can be expected in water bodies? Environ. Pollut. 278, 116910 (2021)
Colson, B.C., Michel, A.P.M.: Flow-through quantification of microplastics using impedance spectroscopy. ACS Sens. 6, 238–244 (2021)
VishnuRadhan, R., Lonappan, A., Eldho, T.I.: A microwave-based technique as a feasible method to detect plastic pollutants in experimental samples. J. Hazard. Mater. 428, 128224 (2022)
Zhu, C., Kanaya, Y., Nakajima, R., Tsuchiya, M., Nomaki, H., Kitahashi, T., Fujikura, K.: Characterization of microplastics on filter substrates based on hyperspectral imaging: Laboratory assessments. Environ. Pollut. 263, 114296 (2020)
Nigamatzyanova, L., Fakhrullin, R.: Dark-field hyperspectral microscopy for label-free microplastics and nanoplastics detection and identification in vivo: a Caenorhabditis elegans study. Environ. Pollut. 271, 116337 (2021)
Boni, W., Arbuckle-Keil, G., Fahrenfeld, N.L.: Inter-storm variation in microplastic concentration and polymer type at stormwater outfalls and a bioretention basin. Sci. Total Environ. 809, 151104 (2021)
Veerasingam, S., Ranjani, M., Venkatachalapathy, R., Bagaev, A., Mukhanov, V., Litvinyuk, D., Mugilarasan, M., Gurumoorthi, K., Guganathan, L., Aboobacker, V.M., Vethamony, P.: Contributions of Fourier transform infrared spectroscopy in microplastic pollution research: a review. Crit. Rev. Environ. Sci. Technol. 51, 2681–2743 (2020)
Vidal, C., Pasquini, C.: A comprehensive and fast microplastics identification based on near-infrared hyperspectral imaging (HSI-NIR) and chemometrics. Environ. Pollut. 285, 117251 (2021)
Horton, A.A., Cross, R.K., Read, D.S., Jurgens, M.D., Ball, H.L., Svendsen, C., Vollertsen, J., Johnson, A.C.: Semi-automated analysis of microplastics in complex wastewater samples. Environ. Pollut. 268, 115841 (2021)
Lin, J., Xu, X.P., Yue, B.Y., Li, Y., Zhou, Q.Z., Xu, X.M., Liu, J.Z., Wang, Q.Q., Wang, J.H.: A novel thermoanalytical method for quantifying microplastics in marine sediments. Sci. Total Environ. 760, 144316 (2021)
Ivleva, N.P.: Chemical analysis of microplastics and nanoplastics: challenges, advanced methods, and perspectives. Chem. Rev. 121, 11886–11936 (2021)
Valsesia, A., Parot, J., Ponti, J., Mehn, D., Marino, R., Melillo, D., Muramoto, S., Verkouteren, M., Hackley, V.A., Colpo, P.: Detection, counting and characterization of nanoplastics in marine bioindicators: a proof of principle study. Microplast. Nanoplast. 1, 5 (2021)
Fang, C., Sobhani, Z., Zhang, D., Zhang, X., Gibson, C.T., Tang, Y., Luo, Y., Megharaj, M., Naidu, R.: Capture and characterisation of microplastics printed on paper via laser printer’s toners. Chemosphere 281, 130864 (2021)
Brandt, J., Mattsson, K., Hassellov, M.: Deep learning for reconstructing low-quality FTIR and Raman spectra horizontal line a case study in microplastic analyses. Anal. Chem. 93, 16360–16368 (2021)
Luo, Y., Sobhani, Z., Zhang, Z., Zhang, X., Gibson, C.T., Naidu, R., Fang, C.: Raman imaging and MALDI-MS towards identification of microplastics generated when using stationery markers. J. Hazard. Mater. 424, 127478 (2022)
Liu, Y., Li, R., Yu, J., Ni, F., Sheng, Y., Scircle, A., Cizdziel, J.V., Zhou, Y.: Separation and identification of microplastics in marine organisms by TGA-FTIR-GC/MS: a case study of mussels from coastal China. Environ. Pollut. 272, 115946 (2021)
Karaman, C.: Orange peel derived-nitrogen and sulfur Co-doped carbon dots: a nano-booster for enhancing ORR electrocatalytic performance of 3D graphene networks. Electroanalysis 33, 1356–1369 (2021)
Buledi, J.A., Mahar, N., Mallah, A., Solangi, A.R., Palabiyik, I.M., Qambrani, N., Karimi, F., Vasseghian, Y., Karimi-Maleh, H.: Electrochemical quantification of mancozeb through tungsten oxide/reduced graphene oxide nanocomposite: a potential method for environmental remediation. Food Chem. Toxicol. 161, 112843 (2022)
Akça, A., Karaman, O., Karaman, C.: Mechanistic insights into catalytic reduction of N2O by CO over Cu-embedded graphene: a density functional theory perspective. ECS J. Solid State Sci. Technol. 10, 041003 (2021)
Karaman, C., Karaman, O., Show, P.L., Karimi-Maleh, H., Zare, N.: Congo red dye removal from aqueous environment by cationic surfactant modified-biomass derived carbon: equilibrium, kinetic, and thermodynamic modeling, and forecasting via artificial neural network approach. Chemosphere 290, 133346 (2022)
Karimi-Maleh, H., Khataee, A., Karimi, F., Baghayeri, M., Fu, L., Rouhi, J., Karaman, C., Karaman, O., Boukherroub, R.: A green and sensitive guanine-based DNA biosensor for idarubicin anticancer monitoring in biological samples: a simple and fast strategy for control of health quality in chemotherapy procedure confirmed by docking investigation. Chemosphere 291, 132928 (2022)
Karimi-Maleh, H., Karimi, F., Fu, L., Sanati, A.L., Alizadeh, M., Karaman, C., Orooji, Y.: Cyanazine herbicide monitoring as a hazardous substance by a DNA nanostructure biosensor. J. Hazard. Mater. 423, 127058 (2022)
Cheraghi, S., Taher, M.A., Karimi-Maleh, H., Karimi, F., Shabani-Nooshabadi, M., Alizadeh, M., Al-Othman, A., Erk, N., Yegya Raman, P.K., Karaman, C.: Novel enzymatic graphene oxide based biosensor for the detection of glutathione in biological body fluids. Chemosphere 287, 132187 (2022)
Lakhdari, D., Guittoum, A., Benbrahim, N., Belgherbi, O., Berkani, M., Vasseghian, Y., Lakhdari, N.: A novel non-enzymatic glucose sensor based on NiFe (NPs)–polyaniline hybrid materials. Food Chem. Toxicol. 151, 112099 (2021)
Mehmandoust, M., Erk, N., Karaman, O., Karimi, F., Bijad, M., Karaman, C.: Three-dimensional porous reduced graphene oxide decorated with carbon quantum dots and platinum nanoparticles for highly selective determination of azo dye compound tartrazine. Food Chem. Toxicol. 158, 112698 (2021)
Ghalkhani, M., Zare, N., Karimi, F., Karaman, C., Alizadeh, M., Vasseghian, Y.: Recent advances in Ponceau dyes monitoring as food colorant substances by electrochemical sensors and developed procedures for their removal from real samples. Food Chem. Toxicol. 161, 112830 (2022)
Sarfo, D.K., Izake, E.L., O’Mullane, A.P., Ayoko, G.A.: Fabrication of nanostructured SERS substrates on conductive solid platforms for environmental application. Crit. Rev. Environ. Sci. Technol. 49, 1294–1329 (2019)
Terry, L.R., Sanders, S., Potoff, R.H., Kruel, J.W., Jain, M., Guo, H.: Applications of surface-enhanced Raman spectroscopy in environmental detection. Anal. Sci. Adv. 3, 113–145 (2022)
Yilmaz, D., Gunaydin, B.N., Yuce, M.: Nanotechnology in food and water security: on-site detection of agricultural pollutants through surface-enhanced Raman spectroscopy. Emergent Mater. 5, 105–132 (2022)
Dey, T.: Microplastic pollutant detection by surface enhanced Raman spectroscopy (SERS): a mini-review. Nanotechnol. Environ. Eng. (2022). https://doi.org/10.1007/s41204-022-00223-7
Lv, L., He, L., Jiang, S., Chen, J., Zhou, C., Qu, J., Lu, Y., Hong, P., Sun, S., Li, C.: In situ surface-enhanced Raman spectroscopy for detecting microplastics and nanoplastics in aquatic environments. Sci. Total Environ. 728, 138449 (2020)
Caldwell, J., Taladriz-Blanco, P., Rothen-Rutishauser, B., Petri-Fink, A.: Detection of sub-micro- and nanoplastic particles on gold nanoparticle-based substrates through surface-enhanced Raman scattering (SERS) spectroscopy. Nanomaterials (Basel) 11, 1149 (2021)
Zhang, P., Wang, Y., Zhao, X., Ji, Y., Mei, R., Fu, L., Man, M., Ma, J., Wang, X., Chen, L.: Surface-enhanced Raman scattering labeled nanoplastic models for reliable bio-nano interaction investigations. J. Hazard. Mater. 425, 127959 (2021)
Xu, G., Cheng, H., Jones, R., Feng, Y., Gong, K., Li, K., Fang, X., Tahir, M.A., Valev, V.K., Zhang, L.: Surface-enhanced Raman spectroscopy facilitates the detection of microplastics <1 μm in the environment. Environ. Sci. Technol. 54, 15594–15603 (2020)
Yin, R., Ge, H., Chen, H., Du, J., Sun, Z., Tan, H., Wang, S.: Sensitive and rapid detection of trace microplastics concentrated through Au-nanoparticle-decorated sponge on the basis of surface-enhanced Raman spectroscopy. Environ. Adv. 5, 100096 (2021)
Lê, Q.T., Ly, N.H., Kim, M.-K., Lim, S.H., Son, S.J., Zoh, K.-D., Joo, S.-W.: Nanostructured Raman substrates for the sensitive detection of submicrometer-sized plastic pollutants in water. J. Hazard. Mater. 402, 123499 (2021)
Nie, X.L., Liu, H.L., Pan, Z.Q., Ahmed, S.A., Shen, Q., Yang, J.M., Pan, J.B., Pang, J., Li, C.Y., Xia, X.H., Wang, K.: Recognition of plastic nanoparticles using a single gold nanopore fabricated at the tip of a glass nanopipette. Chem. Commun. (Camb) 55, 6397–6400 (2019)
Zhang, W., Chang, L., Jiang, S., Luo, J., Zhang, J., Liu, X., Lee, C.-Y.: Nanowell-enhanced Raman spectroscopy enables the visualization and quantification of nanoplastics in the environment. Environ. Sci. Nano 9, 542–553 (2022)
Jeon, Y., Kim, D., Kwon, G., Lee, K., Oh, C.S., Kim, U.J., You, J.: Detection of nanoplastics based on surface-enhanced Raman scattering with silver nanowire arrays on regenerated cellulose films. Carbohydr. Polym. 272, 118470 (2021)
Elsayed, A.A., Erfan, M., Sabry, Y.M., Dris, R., Gasperi, J., Barbier, J.S., Marty, F., Bouanis, F., Luo, S., Nguyen, B.T.T., Liu, A.Q., Tassin, B., Bourouina, T.: A microfluidic chip enables fast analysis of water microplastics by optical spectroscopy. Sci. Rep. 11, 10533 (2021)
Lin, P., Lu, Z., Zhang, Y., Liao, X., He, L., Guo, Y., Zhou, C., Qian, Z.-J., Hong, P., Liang, Y.-Q., Ren, L., Sun, S., Li, C.: Do polystyrene nanoplastics aggravate the toxicity of single contaminants (okadaic acid)? Using AGS cells as a biological model. Environ. Sci. Nano 8, 3186–3201 (2021)
Rai, P.K., Kumar, V., Sonne, C., Lee, S.S., Brown, R.J.C., Kim, K.-H.: Progress, prospects, and challenges in standardization of sampling and analysis of micro- and nano-plastics in the environment. J. Clean. Prod. 325, 129321 (2021)
Szymańska, M., Obolewski, K.: Microplastics as contaminants in freshwater environments: a multidisciplinary review. Ecohydrol. Hydrobiol. 20, 333–345 (2020)
Shruti, V.C., Perez-Guevara, F., Kutralam-Muniasamy, G.: Metro station free drinking water fountain—a potential “microplastics hotspot” for human consumption. Environ. Pollut. 261, 114227 (2020)
Dos Santos Galvao, L., Fernandes, E.M.S., Ferreira, R.R., Dos Santos Rosa, D., Wiebeck, H.: Critical steps for microplastics characterization from the atmosphere. J. Hazard. Mater. 424, 127668 (2022)
Abbasi, S.: Microplastics washout from the atmosphere during a monsoon rain event. J. Hazard. Mater. Adv. 4, 100035 (2021)
Simon-Sanchez, L., Grelaud, M., Franci, M., Ziveri, P.: Are research methods shaping our understanding of microplastic pollution? A literature review on the seawater and sediment bodies of the Mediterranean Sea. Environ. Pollut. 292, 118275 (2022)
Ge, J., Li, H., Liu, P., Zhang, Z., Ouyang, Z., Guo, X.: Review of the toxic effect of microplastics on terrestrial and aquatic plants. Sci. Total Environ. 791, 148333 (2021)
Horton, A.A., Walton, A., Spurgeon, D.J., Lahive, E., Svendsen, C.: Microplastics in freshwater and terrestrial environments: evaluating the current understanding to identify the knowledge gaps and future research priorities. Sci. Total Environ. 586, 127–141 (2017)
Kim, S.K., Kim, J.S., Lee, H., Lee, H.J.: Abundance and characteristics of microplastics in soils with different agricultural practices: Importance of sources with internal origin and environmental fate. J. Hazard. Mater. 403, 123997 (2021)
Chia, R.W., Lee, J.-Y., Kim, H., Jang, J.: Microplastic pollution in soil and groundwater: a review. Environ. Chem. Lett. 19, 4211–4224 (2021)
Gaylarde, C.C., Baptista Neto, J.A., da Fonseca, E.M.: Nanoplastics in aquatic systems—are they more hazardous than microplastics? Environ. Pollut. 272, 115950 (2021)
Kik, K., Bukowska, B., Sicinska, P.: Polystyrene nanoparticles: sources, occurrence in the environment, distribution in tissues, accumulation and toxicity to various organisms. Environ. Pollut. 262, 114297 (2020)
Gonzalez-Acedo, A., Garcia-Recio, E., Illescas-Montes, R., Ramos-Torrecillas, J., Melguizo-Rodriguez, L., Costela-Ruiz, V.J.: Evidence from in vitro and in vivo studies on the potential health repercussions of micro- and nanoplastics. Chemosphere 280, 130826 (2021)
Huang, Z., Weng, Y., Shen, Q., Zhao, Y., Jin, Y.: Microplastic: a potential threat to human and animal health by interfering with the intestinal barrier function and changing the intestinal microenvironment. Sci. Total Environ. 785, 147365 (2021)
Mansa, R., Zou, S.: Thermogravimetric analysis of microplastics: a mini review. Environ. Adv. 5, 100117 (2021)
Dabrowska, A.: Raman spectroscopy of marine microplastics—a short comprehensive compendium for the environmental scientists. Mar. Environ. Res. 168, 105313 (2021)
Song, Y.K., Hong, S.H., Eo, S., Shim, W.J.: A comparison of spectroscopic analysis methods for microplastics: manual, semi-automated, and automated Fourier transform infrared and Raman techniques. Mar. Pollut. Bull. 173, 113101 (2021)
Tirkey, A., Upadhyay, L.S.B.: Microplastics: an overview on separation, identification and characterization of microplastics. Mar. Pollut. Bull. 170, 112604 (2021)
Shim, W.J., Hong, S.H., Eo, S.E.: Identification methods in microplastic analysis: a review. Anal. Methods 9, 1384–1391 (2017)
Renner, G., Schmidt, T.C., Schram, J.: Analytical methodologies for monitoring micro(nano)plastics: which are fit for purpose? Curr. Opin. Environ. Sci. Health 1, 55–61 (2018)
Caldwell, J., Taladriz-Blanco, P., Lehner, R., Lubskyy, A., Ortuso, R.D., Rothen-Rutishauser, B., Petri-Fink, A.: The micro-, submicron-, and nanoplastic hunt: a review of detection methods for plastic particles. Chemosphere 293, 133514 (2022)
Lim, D., Jeong, J., Song, K.S., Sung, J.H., Oh, S.M., Choi, J.: Inhalation toxicity of polystyrene micro(nano)plastics using modified OECD TG 412. Chemosphere 262, 128330 (2021)
Klein, M., Fischer, E.K.: Microplastic abundance in atmospheric deposition within the Metropolitan area of Hamburg, Germany. Sci. Total Environ. 685, 96–103 (2019)
Ita-Nagy, D., Vázquez-Rowe, I., Kahhat, R.: Prevalence of microplastics in the ocean in Latin America and the Caribbean. J. Hazard. Mater. Adv. 5, 100037 (2022)
Imhof, H.K., Laforsch, C., Wiesheu, A.C., Schmid, J., Anger, P.M., Niessner, R., Ivleva, N.P.: Pigments and plastic in limnetic ecosystems: a qualitative and quantitative study on microparticles of different size classes. Water Res. 98, 64–74 (2016)
Huang, D., Tao, J., Cheng, M., Deng, R., Chen, S., Yin, L., Li, R.: Microplastics and nanoplastics in the environment: macroscopic transport and effects on creatures. J. Hazard. Mater. 407, 124399 (2021)
Prust, M., Meijer, J., Westerink, R.H.S.: The plastic brain: neurotoxicity of micro- and nanoplastics. Part Fibre Toxicol. 17, 24 (2020)
Singh, S., Kumar Naik, T.S.S., Anil, A.G., Dhiman, J., Kumar, V., Dhanjal, D.S., Aguilar-Marcelino, L., Singh, J., Ramamurthy, P.C.: Micro (nano) plastics in wastewater: a critical review on toxicity risk assessment, behaviour, environmental impact and challenges. Chemosphere 290, 133169 (2021)
Paul, M.B., Stock, V., Cara-Carmona, J., Lisicki, E., Shopova, S., Fessard, V., Braeuning, A., Sieg, H., Böhmert, L.: Micro- and nanoplastics—current state of knowledge with the focus on oral uptake and toxicity. Nanoscale Adv. 2, 4350–4367 (2020)
Venancio, C., Ciubotariu, A., Lopes, I., Martins, M.A., Oliveira, M.: Is the toxicity of nanosized polymethylmethacrylate particles dependent on the exposure route and food items? J. Hazard. Mater. 413, 125443 (2021)
Dehaut, A., Cassone, A.L., Frere, L., Hermabessiere, L., Himber, C., Rinnert, E., Riviere, G., Lambert, C., Soudant, P., Huvet, A., Duflos, G., Paul-Pont, I.: Microplastics in seafood: benchmark protocol for their extraction and characterization. Environ. Pollut. 215, 223–233 (2016)
Fueser, H., Pilger, C., Kong, C., Huser, T., Traunspurger, W.: Polystyrene microbeads influence lipid storage distribution in C. elegans as revealed by coherent anti-Stokes Raman scattering (CARS) microscopy. Environ. Pollut. 294, 118662 (2022)
Tian, M., Morais, C.L.M., Shen, H., Pang, W., Xu, L., Huang, Q., Martin, F.L.: Direct identification and visualisation of real-world contaminating microplastics using Raman spectral mapping with multivariate curve resolution-alternating least squares. J. Hazard. Mater. 422, 126892 (2022)
Sobhani, Z., Zhang, X., Gibson, C., Naidu, R., Megharaj, M., Fang, C.: Identification and visualisation of microplastics/nanoplastics by Raman imaging (i): down to 100 nm. Water Res. 174, 115658 (2020)
Takahashi, T., Herdzik, K.P., Bourdakos, K.N., Read, J.A., Mahajan, S.: Selective imaging of microplastic and organic particles in flow by multimodal coherent anti-stokes Raman scattering and two-photon excited autofluorescence analysis. Anal. Chem. 93, 5234–5240 (2021)
Levermore, J.M., Smith, T.E.L., Kelly, F.J., Wright, S.L.: Detection of microplastics in ambient particulate matter using Raman spectral imaging and chemometric analysis. Anal. Chem. 92, 8732–8740 (2020)
Fang, C., Luo, Y., Zhang, X., Zhang, H., Nolan, A., Naidu, R.: Identification and visualisation of microplastics via PCA to decode Raman spectrum matrix towards imaging. Chemosphere 286, 131736 (2022)
Zada, L., Leslie, H.A., Vethaak, A.D., Tinnevelt, G.H., Jansen, J.J., de Boer, J.F., Ariese, F.: Fast microplastics identification with stimulated Raman scattering microscopy. J. Raman Spectrosc. 49, 1136–1144 (2018)
Liu, J., Zhang, X., Du, Z., Luan, Z., Li, L., Xi, S., Wang, B., Cao, L., Yan, J.: Application of confocal laser Raman spectroscopy on marine sediment microplastics. J. Oceanol. Limnol. 38, 1502–1516 (2020)
Laptenok, S.P., Martin, C., Genchi, L., Duarte, C.M., Liberale, C.: Stimulated Raman microspectroscopy as a new method to classify microfibers from environmental samples. Environ. Pollut. 267, 115640 (2020)
Wolff, S., Kerpen, J., Prediger, J., Barkmann, L., Muller, L.: Determination of the microplastics emission in the effluent of a municipal waste water treatment plant using Raman microspectroscopy. Water Res. X 2, 100014 (2019)
Valsesia, A., Quarato, M., Ponti, J., Fumagalli, F., Gilliland, D., Colpo, P.: Combining microcavity size selection with Raman microscopy for the characterization of nanoplastics in complex matrices. Sci. Rep. 11, 362 (2021)
Karimi-Maleh, H., Darabi, R., Shabani-Nooshabadi, M., Baghayeri, M., Karimi, F., Rouhi, J., Alizadeh, M., Karaman, O., Vasseghian, Y., Karaman, C.: Determination of D&C Red 33 and Patent Blue V Azo dyes using an impressive electrochemical sensor based on carbon paste electrode modified with ZIF-8/g-C3N4/Co and ionic liquid in mouthwash and toothpaste as real samples. Food Chem. Toxicol. 162, 112907 (2022)
Prata, J.C., da Costa, J.P., Fernandes, A.J.S., da Costa, F.M., Duarte, A.C., Rocha-Santos, T.: Selection of microplastics by Nile Red staining increases environmental sample throughput by micro-Raman spectroscopy. Sci. Total Environ. 783, 146979 (2021)
Cho, S., Kim, Y., Chung, H.: Feasibility study for simple on-line Raman spectroscopic detection of microplastic particles in water using perfluorocarbon as a particle-capturing medium. Anal. Chim. Acta 1165, 338518 (2021)
Liu, Y., Yue, S., Wang, Y.-N., Wang, Y., Xu, Z.-R.: A multicolor-SERS dual-mode pH sensor based on smart nano-in-micro particles. Sens. Actuators B Chem 310, 127889 (2020)
Amorim, M.J.B., Scott-Fordsmand, J.J.: Plastic pollution—a case study with Enchytraeus crypticus—from micro-to nanoplastics. Environ. Pollut. 271, 116363 (2021)
Ly, N.H., Son, S.J., Jang, S., Lee, C., Lee, J.I., Joo, S.W.: Surface-enhanced Raman sensing of semi-volatile organic compounds by plasmonic nanostructures. Nanomaterials (Basel) 11, 2619 (2021)
Das, R.K., Sanyal, D., Kumar, P., Pulicharla, R., Brar, S.K.: Science-society-policy interface for microplastic and nanoplastic: environmental and biomedical aspects. Environ. Pollut. 290, 117985 (2021)
Schmidt, R., Nachtnebel, M., Dienstleder, M., Mertschnigg, S., Schroettner, H., Zankel, A., Poteser, M., Hutter, H.P., Eppel, W., Fitzek, H.: Correlative SEM-Raman microscopy to reveal nanoplastics in complex environments. Micron 144, 103034 (2021)
Pang, M., Wang, Y., Tang, Y., Dai, J., Tong, J., Jin, G.: Transcriptome sequencing and metabolite analysis reveal the toxic effects of nanoplastics on tilapia after exposure to polystyrene. Environ. Pollut. 277, 116860 (2021)
Chaudhry, A.K., Sachdeva, P.: Microplastics’ origin, distribution, and rising hazard to aquatic organisms and human health: socio-economic insinuations and management solutions. Reg. Stud. Mar. Sci. 48, 102018 (2021)
Pérez-Guevara, F., Roy, P.D., Kutralam-Muniasamy, G., Shruti, V.C.: A central role for fecal matter in the transport of microplastics: an updated analysis of new findings and persisting questions. J. Hazard. Mater. Adv. 4, 100021 (2021)
Morris, C.A., Friedman, A.K., Baker, L.A.: Applications of nanopipettes in the analytical sciences. Analyst 135, 2190–2202 (2010)
Ho, V.T.T.X., Park, H., An, S., Kim, G., Ly, N.H., Lee, S.Y., Choo, J., Jung, H.S., Joo, S.-W.: Coumarin–lipoic acid conjugates on silver nanoparticle-supported nanopipettes for in situ dual-mode monitoring of intracellular Cu(II) and potential chemodynamic therapy applications. Sens. Actuators B Chem 344, 130271 (2021)
Nguyen, T.D., Song, M.S., Ly, N.H., Lee, S.Y., Joo, S.-W.: Nanostars on nanopipette tips: a Raman probe for quantifying oxygen levels in hypoxic single cells and tumours. Angew. Chem. Int. Ed. 58, 2710–2714 (2019)
Schmid, C., Cozzarini, L., Zambello, E.: Microplastic’s story. Mar. Pollut. Bull. 162, 111820 (2021)
Vélez-Escamilla, L.Y., Contreras-Torres, F.F.: Latest advances and developments to detection of micro- and nanoplastics using surface-enhanced Raman spectroscopy. Part Part Syst. Charact. 39, 2100217 (2022)
Fang, C., Sobhani, Z., Zhang, X., McCourt, L., Routley, B., Gibson, C.T., Naidu, R.: Identification and visualisation of microplastics/nanoplastics by Raman imaging (iii): algorithm to cross-check multi-images. Water Res. 194, 116913 (2021)
Gwinnett, C.M.B., Osborne, A.O., Jackson, A.R.W.: The application of tape lifting for microplastic pollution monitoring. Environ. Adv. 5, 100066 (2021)
Jiao, M., Cao, S., Ren, L., Li, R.: Analysis of composite microplastics in sediment using 3D Raman spectroscopy and imaging method. J. Hazard Mater. Adv. 3, 100016 (2021)
Luo, Y., Gibson, C.T., Chuah, C., Tang, Y., Naidu, R., Fang, C.: Applying Raman imaging to capture and identify microplastics and nanoplastics in the garden. J. Hazard. Mater. 426, 127788 (2022)
Luo, Y., Gibson, C.T., Tang, Y., Naidu, R., Fang, C.: Characterising microplastics in shower wastewater with Raman imaging. Sci. Total Environ. 811, 152409 (2021)
Acknowledgements
We acknowledge the financial support from the Korea Environment Industry & Technology Institute (KEITI) through Technology Development Project for Safety Management of Household Chemical Product Program, funded by Korea Ministry of Environment (MOE) (2020002970005, 1485017845).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ly, N.H., Kim, MK., Lee, H. et al. Advanced microplastic monitoring using Raman spectroscopy with a combination of nanostructure-based substrates. J Nanostruct Chem 12, 865–888 (2022). https://doi.org/10.1007/s40097-022-00506-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40097-022-00506-0