Abstract
Soil organic carbon (C) and nitrogen (N) play a critical role in plant nutrient dynamics and crop yield performance under different soils. In this study, the amount of carbon (C), N and phosphorus (P) mineralization as influenced by type of organic residue and soil contact variation was studied for an incubation period of 75 days under controlled laboratory conditions. Different types of residues, viz. subabul residue (Leucaena leucocephala) (T1), biochar of subabul residue (T2) and FYM (T3), along with unamended soil with no residue constituted the three modes of application, viz. buried (t1), incorporated (t2) and surface applied (t3) treatments. Among all the treatments, subabul residue applied treatments showed highest rate of C mineralization in which surface applied (SA) treatments varied from 19.54 at 24 h to 239.55 mg CO2–C/100 g soil at 75 days followed by subabul incorporated (I) (17.04–237.8 mg CO2–C/100 g soil) and subabul buried (B) (15.91–225.5 mg CO2–C/100 g soil) treatments. Subabul incorporated (I) treatment showed lowest rate of mineralization of NO3–N, which varies from 0.010 and 0.613 mg kg−1 at 7 days to 4.640 and 10.03 mg kg−1 at 75 days after incubation followed by subabul buried (B) and SA treatments. These results will be useful in the selection of agriculture crop inputs and their proper placement in soils with respect to nutrient mineralization.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Soil organic matter (SOM), one of the crucial indicators for the soil fertility and crop productivity, has received global attention recently. An appropriate package and practices of SOM management not only improve crop productivity [18, 26], but also reduce the atmospheric concentration of carbon dioxide (CO2) [20, 24, 51]. The use of more stable substances such as carbonized materials from incomplete combustion of organic materials such as black C, pyrogenic feedstocks and charcoal could provide a long-term stability for maintaining high levels of SOM and available nutrients in the soil [3, 45, 50]. Biochar as defined by Lehmann and Joseph [41] is a fine-grained, porous, stable C product remaining after plant biomass subjected to thermochemical conversion process at low temperature (350–600 °C) in a little or no oxygen environment known as pyrolysis [54, 60, 63, 64]. In recent years, it has gained attention as a potential amendment to boost soil fertility and crop productivity across the globe with multidimensional opportunity to reduce the C emission from soils by storing C in long-lived C pools [45, 48]. As a source of C-negative recalcitrant soil C pool, biochar serve as a sink of atmospheric CO2 stored in highly recalcitrant soil C stocks [9, 59]. The biochar provides a pool of C that undergoes minimal microbial degradation in soil [28, 46]. Accumulation of this persistent pool of C within the soil can improve soil structure, water holding capacity, cation exchange capacity, surface sorption capacity, base saturation and nutrient cycling [19, 21, 22, 34]. In addition, the biochar can have a protective effect for other sources of C within the soil and has been reported to decrease the mineralization rate of both native SOC and fresh inputs of C, i.e., raw residues [7, 35, 43, 58]. Estimation of net C mineralized or converted into CO2 from biochars decomposition was needed to improve understanding on the efficiency of biochars in enhancing soil quality, C sequestration and biochar stability in soils [55]. Biochar could offset 12% of the current anthropogenic CO2–C equivalent emissions from soils [47]. Positive effects of biochar on the soil ecosystem and functions were proposed to derive either directly from the nutrients within biochar itself, or indirectly from its ability to sorb and retain nutrients [33, 42]. Besides sequestering C, biochar has been observed for potential improvement in N and P cycling in the soil– plant system [44, 58]. A major portion of N exists in a complex organic form that must be ammonified to NH4+–N and then nitrified to NO3−–N prior to plant uptake. Biochar improved N and phosphorus (P) mineralization kinetics in soils [13, 31]. It plays an important role in the biochemical cycling of N and P through its large surface area, pH and nutrient content [23], which varied among different types of biochar depending on their source of raw material [6, 31]. Recent studies have demonstrated that the addition of biochar to surface mineral soils may directly influence N transformations [67]. Gaskin et al. [29] reported that N from biochar could not be available to plants at the initial stage of application in the soil due to lower rate of decomposition. The addition of biochar to soils resulted slower mineralization rate compared uncharred material [40] and decreased net C and N mineralization in soil [11, 17]. Many researchers revealed that biochar application directly involved in the biochemical process related to mostly for N and P utilization [2], but reduced the activities of C cycle enzymes [42, 53, 57]. Asai et al. [1] showed that biochar application improves P availability and crop yield under low-P status soils. Although most soil properties could be improved through the application of crop residues or pyrolyzed material, there is limited research work on the effect of subabul biochar with respect to C, N and P dynamics under different soil contact variations. Keeping this in mind, the hypothesis of this study was to evaluate the effect of organic residues and soil contact variation on C, N and P mineralization in Vertisol of central India.
Materials and Methods
Experimental Design and Treatment Details
An incubation experiment was conducted at ICAR-Indian Institute of Soil Science, Bhopal. For this, the bulk surface soil (0–15 cm) sample was collected, air-dried and ground to pass through a 2-mm sieve. The physicochemical properties of the soil were analyzed as per the procedure described in Singh et al. [56] and are presented in Table 1. The different residues, i.e., subabul residue, biochar of subabul and FYM were collected locally and analyzed for total organic carbon content (TOC); properties are described in Table 2. Subabul (Leucaena leucocephala) stem and twigs were collected locally term as subabul residue and dried at 80 °C for 12 h. Further these materials were used for biochar preparation by the indigenous technique (drum method). The experiment was conducted for 75 days with treatments based on the type of organic residues and their method of placement. Three different types of residues, viz. subabul residue (T1), biochar of subabul (T2) and FYM (T3), were formulated by mixing 100 g soil in 250-mL conical flasks. These residues were applied as buried (B) (t1), incorporated (I) (t2) and surface applied (SA) (t3) treatments. The highest TOC content was observed in biochar (74.0%) followed by subabul residue (47.1%) and FYM (24.2%).
Incubation Experiment for C Mineralization
For this, 100 g soil was taken into a 250-mL conical flask and was subjected to different treatments based on the C equivalent basis equal to biochar C content with three replications. Throughout the experiment, moisture content of each sample was maintained at the field capacity level. For C mineralization, vial containing 10 mL of 2 M NaOH was kept inside the conical flask in hanging position and airtight with parafilms. Simultaneously blank sample run to compute the exact C mineralization rate. Soil C mineralization was measured under controlled conditions in the laboratory at an interval of 1, 7, 15, 30, 45, 60 and 75 days after incubation. Produced CO2–C was determined by titration of the NaOH solution with 0.5 M HCl in an excess of BaCl2, using phenolphthalein as an indicator [70].
Incubation Experiment for Mineralization of N and P
The 100 g soil was transferred to moisture box, and all the treatments were formulated as in case of the above-mentioned soil incubation experiment. These moisture boxes were kept in an incubator at 25 °C temperature, and a destructive sampling was done at different time interval of 7, 15, 30, 45, 60 and 75 days after incubation. Ammoniacal nitrogen (NH4+–N) and nitrate nitrogen (NO3−–N) were determined by transferring 10 g soil to a 100-mL conical flask and 50 mL of 2 M KCl. NH4+–N and NO3−–N were measured calorimetrically using continuous-flow auto-analyzer (Sanþþ System, Skalar, the Netherlands). Phosphorus content in the samples at same interval was measured by Olsen et al. [52].
Statistical Analysis
The experiment was conducted in complete randomized design (CRD) with three replications as per the procedure described in Gomez and Gomez [30]. To assess the statistical differences among different treatments, C, N and P mineralization was analyzed via repeated measures ANOVA at the p < 0.05 significance level. All statistical analyses were performed using SPSS 16.0 software (SPSS Inc., Chicago, IL, USA).
Results and Discussion
Effect of Treatments on Carbon Mineralization
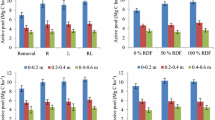
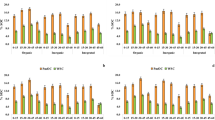
The cumulative amount of CO2–C was higher in soils amended with different residues as compared to unamended soil during the incubation period. The measured ANOVA results showed that cumulative C mineralization rate was significantly affected by residue type and placement method over unamended. Surface applied treatment showed the highest rate of C mineralization as compared to other treatments followed by incorporated and buried treatments. On average, C mineralization rate in different treatments with their modes of application ranged between 14.77 and 225.5 mg CO2–C/100 g soil after one day and 75 days of incubation, respectively, for buried; 9.20 and 237.8 mg CO2–C/100 g soil, respectively, for incorporated; and 8.50 and 239.5 mg CO2–C/100 g soil for surface applied treatments. Maximum C mineralization was observed under subabul residue applied treatments in which surface applied treatment varied from 19.54 at 24 h to 239.55 mg CO2–C/100 g soil at 75 days followed by subabul incorporated (I) (17.04–237.8 mg CO2–C/100 g soil) and subabul buried (B) (15.91–225.5 mg CO2–C/100 g soil) treatment. On the contrary, all the biochar applied treatments remained on a par with control throughout the incubation period (Fig. 1).
Soil treated with FYM remained on a par with subabul residue applied treatments up to 15 days after incubation, but after 30 days subabul residue-treated soils showed the highest level of C mineralization over all other treatments. The response differences in total CO2–C emission in the study were clear among all the applied treatments as compared to control soil, which is regulated by higher microbial activity and availability of C substrates [25, 44]. The cumulative C mineralization was higher for residues placed on the soil surface than for residues incorporated and buried into the soils [14]. Previous studies showed that the C mineralization rate was larger when the residues were subjected to surface applied treatment as compared to that of incorporated and buried treatments [44]. Differences in C mineralization between residues incorporated into soils and residues placed on the soil surface have been found to relate to moisture conditions and temperature [65], while moisture conditions and temperature were controlled in our incubation experiment. The incorporation of residues into soils reduced gas diffusivity and might further decrease residue decomposition rate [68]. The incorporation of organic residue in soil enhances the soil microbial population and diversity which affect the plant nutrient dynamics in soil. The C-to-N ratio of residues and microbial population affect the kinetics of organic residue. Although C-use efficiency could be enhanced under anaerobic conditions compared to aerobic, CO2 emissions were higher for residues placed on the soil surface than for residues incorporated into the soils. These results indicated that the incorporation of residues into the soils inhibited residue decomposition, most likely by modifying the availability of oxygen to decomposer microorganisms [68]. Experimental results were in line with many studies conducted by other researchers [10, 44, 58], which revealed that uncharred subabul residue showed higher rates of C mineralization as compared to same material was converted to biochar. When uncharred feedstocks went through pyrolysis process, some of the elements in biochar were concentrated due to loss volatile materials, while some elements experienced concentration declined [49, 55]. Due to significant increase in C content, it increased the C/N ratio and reduced microbial activity in biochar applied treatments which ultimately affected C mineralization [8]. Conducted incubation experiments indicated that in case of biochar applied treatments only a small fraction usually less than 5% is degraded within the time fraction of laboratory incubations, depending both on the material from which it has been produced and on the processing conditions [69]. Jien et al. [39] observed that SOM stabilization by biochar application in the soil and found decreased C mineralization due to the formation of macro-aggregates which wrapped the biochar and compost and prevent from rapid decomposition by microbes [36, 38].
Effect of Treatments on NH4+–N and NO3−–N Mineralization rate
During the period of investigation, within the organic treatments no significant difference (p = 0.05) was found regarding net NH4+–N mineralization. However, different types of placement methods found significant differences between subabul and biochar and FYM (Table 3). Among the treatments, lower rate of ammoniacal nitrogen (NH4+–N) mineralization was observed at up to 45 days after incubation, while slight increment was observed after 60 and 75 days of incubation. Impacts of types of organic residues and their modes of application on NO3−–N mineralization are given in Table 4. Results revealed that subabul residue applied treatments showed the lowest rate of NO3−–N mineralization as compared to other treatments. Among all the treatments, subabul incorporated (I) treatment observed lowest rate of mineralization of nitrate nitrogen (NO3−–N) as compared to unamended soil, which varies from 0.010 mg kg−1 at 7 days to 4.640 mg kg−1 at 75 days after incubation, respectively, followed by subabul buried (B) and surface applied (SA) treatments (Table 4). Even at 30 days after incubation, no content of NO3−–N was observed in all the subabul residue applied treatments. NO3−–N mineralization rate was highest in FYM applied treatments followed by biochar and subabul residue applied treatments. Experimental results indicated that the application of subabul residue in the form of biochar mineralization was found on a par with unamended soil up to 45 and increased two- to threefold up to 75 days after incubation as compared to unamended soil. Incubated results were in accordance with studies conducted by many researchers which revealed that when fresh biochar material is applied to soil because of its higher surface area and cation and anion exchange capacity, NH4+–N gets immobilized on the anion exchange sites of biochar at initial stages of application [8, 12, 13, 37, 66]. The define rate of NH4+–N was at 15 and 45 days was due to lower amount of labile N in the organic matter and promotes more microbial immobilization in soil. Shortage of labile N and more amount of recalcitrant organic N in soil lead to the imbalance in the C-to-N ratio and reduce the mineralization rate of organic residue. Such findings were also reported by Li et al. [44]. Results also indicated that biochar application stabilized SOM through sorption that rendered SOM inaccessible to microbial decomposition may have caused a reduction in NH4+–N mineralization [42]. Black cotton soil, relatively rich in clay, further enhanced with the application of biochar, which increased the surface acidic functional groups and adsorbed NH4+–N on surface [35, 43]. The primary and most consistent effects of biochar observed in this study were the increased NO3−–N concentration with days of incubation. Several studies have indeed demonstrated that the application of biochar in soil stimulates the nitrifying community in soil due to its porous structure, increased CEC and adsorption of NH4+–N and dissolved organic C on its surface [13, 42, 61]. Additionally biochar adsorbs the allelopathic molecules from soil solution that may otherwise inhibit nitrification onto the biochar surface and likewise influences nitrifying bacteria [27]. Studies conducted by DeLuca et al. [15, 16] shows that the application of biochar in soil enhanced adsorption of compound with a high C-to-N ratio onto the surface of biochar that otherwise would increase N immobilization upon decomposition is likely to contribute to the stimulation of ammonification and nitrification [32]. Soil N availability may be important factor for residue decomposition [14]. The soils amended with subabul residue had a lower cumulative N mineralization than unamended soil. Furthermore, the addition of biochar resulted in a higher mineral N compared to unamended soil; the cumulative mineral N for residues placed on the soil surface was higher than for residues incorporated and buried into the soils.
Effect of Treatments on P Mineralization
Results revealed that the addition of biochar adversely mediated P mineralization rate in all the treatments along with unamended soil was observed after 60 days of incubation (Fig. 2). All the methods of subabul residue application (23.25 and 6.91 mg kg−1 at 15 and 60 days after incubation, respectively) were on a par with unamended soil (23.96 mg kg−1 and 4.49 mg kg−1 at 15 and 60 days after incubation, respectively) for P mineralization. Biochar-treated soil showed 1 to twofold increase in P mineralization rate, while the same was four- to eightfold in case of FYM-treated soil. Among the treatments, highest rate of P mineralization was observed when the residues were subjected to surface applied treatment followed by incorporated and buried treatments. The influence of different organic residues and their method of placement on P mineralization was generally of a smaller consistency across different treatments and days of incubation. The decrease in available P with days after incubation at the application of biochar could be a result of P sorption to biochar surfaces. A few research observations are available on P adsorption to biochar surfaces were conducted in forest soils and found small decrease in P availability after incubation [4]. Beaton et al. [5] demonstrated adsorption of P on biochar surfaces as a result of hydrogen bonding between orthophosphate and biochar surfaces. Takaya et al. [62] revealed that incomplete digestion and P adsorption by the residual biochar might have resulted in lower P mineralization. Phosphorus mineralization is much affected by the soil as well as organic residue properties. Lower concentration of P in residue slows down the mineralization process due to wider difference in C-to-P ratio in soil for proper mineralization. The soil properties much affected the P mineralization dynamics in soil and also decide the plant nutrient availability in soil solution. Phosphorus is an immobile nutrient in soil and its mineralization process is mediated by microbial biomass and their diversity in soil, which is directly or indirectly affected the phosphorus-use efficiency in soil–plant system.
Conclusions
Incubation results indicated that C mineralization was highest in subabul application than biochar and FYM application over control. Soil surface applied residues showed higher C mineralization as compared to incorporated and buried treatments. Further, results showed that lower rate of NO3−–N mineralization in subabul residue applied treatments as compared to control, but when the same material was applied in the form of biochar it showed a higher rate of nitrate mineralization even 2 to 3 times higher after 45 days of incubation. Phosphorus mineralization showed a nonsignificant change with days after incubation among all the treatments. This type of study could be used in the rational use of organic residues for soil fertility management and sustainable crop production without compromising environmental health.
References
Asai H, Samson BK, Stephan HM, Songyikhangsuthor K, Homma K, Kiyono Y, Inoue Y, Shiraiwa T, Horie T (2009) Biochar amendment techniques for upland rice production in Northern Laos 1. Soil physical properties, leaf SPAD and grain yield. Field Crops Res 111:81–84
Bailey VL, Fansler SJ, Smith JL, Bolton H (2011) Reconciling apparent variability in effects of biochar amendment on soil enzyme activities by assay optimization. Soil Biol Biochem 43:296–301
Baldi E, Toselli M (2014) Mineralization dynamics of different commercial organic fertilizers from agro-industry organic waste recycling: an incubation experiment. Plant Soil Environ 60:93–99
Beaton JD (1959) The influence of burning on the soil in the timber range area of Lac le Jeune BC II. Chemical properties. Canad J Soil Sci 39:6–11
Beaton JD, Peterson HB, Bauer N (1960) Some aspects of phosphate adsorption by charcoal. Soil Sci Soc Am J 24:340–346
Biederman LA, Harpole WS (2012) Biochar and its effects on plant productivity and nutrient cycling: a meta-analysis. Glob Change Biol Bioener 5(2):202–214
Borchard N, Ladd B, Eschemann S, Hegenberg D, Moseler BM, Amelung W (2014) Black carbon and soil properties at historical charcoal production sites in Germany. Geoderma 232–234:236–242
Bruun EW, Ambus P, Egsgaard H, Hauggaard-Nielsen H (2012) Effects of slow and fast pyrolysis biochar on soil C and N turnover dynamics. Soil Biol Biochem 46:73–79
Bruu EWMD, Müller-Stöver D, Ambus P, Hauggaard-Nielsen H (2011) Application of biochar to soil and N2O emissions: potential effects of blending fast pyrolysis biochar with anaerobically digested slurry. Eur J Soil Sci 62:581–589
Cantrell KB, Martin JH (2012) Stochastic state-space temperature regulation of biochar production. J Sci Food Agric 92:481–489
Castaldi S, Riondino M, Baronti S, Esposito FR, Marzaioli R, Rutigliano FA (2012) Impact of biochar application to a Mediterranean wheat crop on soil microbial activity and greenhouse gas fluxes. Chemosphere 85:1464–1471
Clough T, Condron L, Kammann C, Müller C (2013) A review of biochar and soil nitrogen dynamics. Agron 3:275–293
Clough TJ, Condron LM (2010) Biochar and the nitrogen cycle. J Environ Qual 39:1218–1223
Coppens F, Garnier P, De-Gryze S, Merckx R, Recous S (2006) Soil moisture, carbon and nitrogen dynamics following incorporation and surface application of labelled crop residues in soil columns. Euro J Soil Sci 57:894–905
DeLuca TH, Aplet GH (2007) Charcoal and carbon storage in forest soils of the Rocky Mountain West. Front Ecol Environ 6:1–7
DeLuca TH, MacKenzie MD, Gundale MJ (2009) Biochar effects on soil nutrient transformations. In: Lehmann J, Joseph S (eds) Biochar for environmental management. Earthscan, London, pp 251–270
Dempster DN, Gleeson DB, Solaiman ZM, Jones DL, Murphy DV (2012) Decreased soil microbial biomass and nitrogen mineralization with eucalyptus biochar addition to a course textured soil. Plant Soil 354:311–324
Dotaniya ML (2013) Impact of various crop residue management practices on nutrient uptake by rice-wheat cropping system. Curr Adv Agric Sci 5(2):269–271
Dotaniya ML, Datta SC (2014) Impact of bagasse and press mud on availability and fixation capacity of phosphorus in an Inceptisol of north India. Sugar Tech 16(1):109–112
Dotaniya ML, Datta SC, Biswas DR, Dotaniya CK, Meena BL, Rajendiran S, Regar KL, Lata M (2016) Use of sugarcane industrial byproducts for improving sugarcane productivity and soil health—a review. Intl J Recyc Org Waste Agric 5(3):185–194
Dotaniya ML, Datta SC, Biswas DR, Kumar K (2014) Effect of organic sources on phosphorus fractions and available phosphorus in Typic Haplustept. J Ind Soc Soil Sci 62(1):80–83
Dotaniya ML, Datta SC, Biswas DR, Meena HM, Kumar K (2014) Production of oxalic acid as influenced by the application of organic residue and its effect on phosphorus uptake by wheat (Triticum aestivum L.) in an Inceptisol of north India. Natl Acad Sci Lett 37(5):401–405
Dotaniya ML, Datta SC, Biswas DR, Meena HM, Rajendiran S, Meena AL (2015) Phosphorus dynamics mediated by bagasse, press mud and rice straw in inceptisol of north India. Agrochimica 59(4):358–369
Dotaniya ML, Rajendiran S, Meena BP, Meena AL, Meena BL, Jat RL, Saha JK (2016) Elevated carbon dioxide (CO2) and temperature vis- a-vis carbon sequestration potential of global terrestrial ecosystem. In: Bisht JK, Meena VS, Mishra PK, Pattanayak A (eds) Conservation agriculture: an approach to combat climate change in Indian Himalaya. Springer, New York, pp 225–256
Dotaniya ML, Rajendiran S, Meena VD, Saha JK, Coumar MV, Kundu S, Patra AK (2017) Influence of chromium contamination on carbon mineralization and enzymatic activities in Vertisol. Agric Res 6(1):91–96
Dotaniya ML, Sharma MM, Kumar K, Singh PP (2013) Impact of crop residue management on nutrient balance in rice-wheat cropping system in an Aquic hapludoll. The J Rural Agric Res 13(1):122–123
Elmer WH, Pignatello JJ (2011) Effect of biochar amendments on mycorrhizal associations and Fusarium crown and root rot of Asparagus in replant soils. Plant Dis 95:960–966
Farrell MKTK, Macdonald LM, Maddern TM, Murphy DV, Hall PA, Singh BP, Baumann K, Krull ES, Baldock JA (2013) Microbial utilisation of biochar-derived carbon. Sci Total Environ 465:288–297
Gaskin JW, Speir RA, Harris K, Das C, Lee DR, Morris LA, Fisher DS (2010) Effect of peanut hull and pine chip biochar on soil nutrients, corn nutrient status and yield. Agron J 102:1096–1106
Gomez KA, Gomez A (1983) Statistical procedures for agricultural research, 2nd edn. Wiley, New York
Gul S, Whalen JK (2016) Biochemical cycling of nitrogen and phosphorus in biochar amended soils. Soil Biol Biochem 103:1–15
Gundale MJ, DeLuca TH (2006) Temperature and source material influence ecological attributes of ponderosa pine and Douglas-fir charcoal. Forest Ecol Manage 231(1–3):86–93
Hammes K, Schmidt M (2009) Changes in biochar in soil. In: Lehmann J, Joseph S (eds) Biochar for environmental management. Earthscan, London, pp 169–182
Harvey OR, Li-Jung K, Andrew RZ, Patrick L, James EA, Bruce EH (2012) An index-based approach to assessing recalcitrance and soil carbon sequestration potential of engineered black carbons (biochars). Environ Sci Technol 46:1415–1421
Hernandez-Soriano MC, Kerre B, Goos P, Hardy B, Dufey J, Smolders E (2016) Long-term effect of biochar on the stabilization of recent carbon: soils with historical inputs of charcoal. Glob Change Biol Bioener 8:371–381
Hseu ZY, Jien SH, Chien WS, Liou RC (2014) Impacts of biochar on physical properties and erosion potential of a mudstone slope land soil. Sci World J. https://doi.org/10.1155/2014/602197
Huang M, Yang L, Qin H, Jiang L, Zou Y (2014) Fertilizer nitrogen uptake by rice increased by biochar application. Biol Fert Soils 50:997–1000
Jien SH, Wang CS (2013) Effects of biochar on soil properties and erosion potential in a highly weathered soil. CATENA 110:225–233
Jien SH, Wang CC, Lee CH, Lee TY (2015) Stabilization of organic matter by biochar application in compost amended soils with contrasting pH values and textures. Sustainability 7:13317–13333
Knoblauch C, Maarifat AA, Pfeiffer EM, Haefele SM (2012) Degradability of black carbon and its impact on trace gas flux and carbon turnover in paddy soils. Soil Biol Biochem 43:1768–1778
Lehmann J, Joseph S (2009) Biochar for environmental management: an introduction. In: Lehmann J, Joseph S (eds) Biochar for environmental management. Science and technology. Earthscan, London, pp 1–12
Lehmann J, Rillig MC, Thies J, Masiello CA, Hockaday WC, Crowley D (2011) Biochar effects on soil biota—a review. Soil Biol Biochem 43:1812–1836
Liang B, Lehmann J, Sohi SP, Thies JE, Neill BO, Trujillo L, Gaunt J, Soloman D, Grosmann J, Neves EJ, Liuzao FJ (2010) Black carbon affects the cycling of non-black carbon in soil. Org Geochem 41:206–213
Li L, Xiao-Zeng H, You MY, Yuan YR, Ding XL, Qiao YF (2013) Carbon and nitrogen mineralization patterns of two contrasting crop residues in a Mollisol: effects of residue type and placement in soils. Euro J Soil Biol 54:1–6
Manya JJ (2012) Pyrolysis for biochar purposes: a review to establish current knowledge gaps and research needs. Environ Sci Technol- 46:7939–7954
Maria CHS, Bart K, Peter MK, Benjamin H, Erik S (2016) Biochar affects carbon composition and stability in soil: a combined spectroscopy-microscopy study. Nat Sci Rep 6:1–13
Mukherjee A, Zimmerman AR (2013) Organic carbon and nutrient release from a range of laboratory-produced biochars and biochar–soil mixtures. Geoderma 193–194:122–130
Novak JM, Busscher WJ, Laird DL, Ahmenda M, Watts DW, Niandou MAS (2009) Impact of biochar amendment on soil fertility of a southeastern Coastal Plain soil. Soil Sci 174:105–112
Novak JM, Cantrell KB, Watts DW, Busscher WJ, Johnson MG (2014) Designing relevant biochars as soil amendments using lignocellulosic-based and manure-based feedstocks. J Soils Sed 14:330–343
Novara A, Gristina L, Rühl J, Pasta S, D’Angelo G, La Mantia T, Pereira P (2013) Grassland fire effect on soil organic carbon reservoirs in a semiarid environment. Solid Earth 4:381–385
Oliveira SP, Lacerda NB, Blum SC, Escobar MEO, Oliveira TS (2014) Organic carbon and nitrogen stocks in soils of northeastern Brazil converted to irrigated agriculture. Land Degrad Dev 26:9–21
Olsen SR, Cole CV, Watanabe FS, Dean LA (1954) Estimation of available phosphorus in soils by extraction with sodium bicarbonate. US Depart Agric Circ No 939
Paz-Ferreiro J, Fu S, Mendez A, Gasco G (2014) Interactive effects of biochar and the earthworm Pontoscolex corethrurus on plant productivity and soil enzyme activities. J Soils Sed 14:483–494
Sigua GC, Novak JM, Watts DW, Cantrell KB, Shumaker PD, Szogi AA, Johnson MG (2014) Carbon mineralization in two ultisols amended with different sources and particle sizes of pyrolyzed biochar. Chemosphere 103:313–321
Sigua GC, Novak JM, Watts DW, Szogi AA, Shumaker PD (2016) Impact of switch grass biochars with supplemental nitrogen on carbon–nitrogen mineralization in highly weathered Coastal Plain Ultisols. Chemosphere 145:135–141
Singh D, Chhonkar PK, Pandey RN (1999) Soil plant water analysis: a methods manual. Westville Publishing House, New Delhi
Song D, Xi X, Huang S, Liang G, Sun J, Zhou W (2016) Short-term responses of soil respiration and C-cycle enzyme activities to additions of biochar and urea in a calcareous soil. PLoSOne 11(9):e0161694
Spokas KA, Cantrell KB, Novak JM, Archer DA, Ippolito JA, Collins HP, Boateng AA, Lima IM, Lamb MC, Mc-Aloon AJ (2012) Biochar: a synthesis of its agronomic impact beyond carbon sequestration. J Environ Qual 41:973–989
Sui Y, Gao J, Liu C, Zhang W, Lan Y, Li S (2016) Interactive effects of straw-derived biochar and N fertilization on soil C storage and rice productivity in rice paddies of Northeast China. Sci Total Environ 544:203–210
Sun JN, He FH, Zhang ZH, Shao HB, Xu G (2016) Temperature and moisture responses to carbon mineralization in the biochar-amended saline soil. Sci Total Environ 569:390–394
Taghizadeh-Toosi A, Clough TJ, Condron LM, Sherlock RR, Anderson CR, Craigie RA (2011) Biochar incorporation into pasture soil suppresses in situ nitrous oxide emissions from ruminant urine patches. J Environ Qual 40:468–476
Takaya CA, Fletcher LA, Singh S, Anyikude KU, Ross AB (2016) Phosphate and ammonium sorption capacity of biochar and hydrochar from different wastes. Chemosphere 145:518–527
Tejada M, Benitez C (2014) Effects of crushed maize straw residues on soil biological properties and soil restoration. Land Degrad Dev 25:501–509
Vasu D (2015) Effect of biochar addition on soil carbon emission and nitrogen mineralization in some typical Indian soils. Intl J Emerg Res Manage Technol 4(11):17–22
Wang J, Sainju UM (2014) Soil carbon and nitrogen fractions and crop yields affected by residue placement and crop types. PLoSONE 9(8):1–11
Yao Y, Gao B, Zhang M, Inyang M, Zimmerman AR (2012) Effect of biochar amendment on sorption and leaching of nitrate, ammonium, and phosphate in a sandy soil. Chemosphere 89:1467–1471
Zheng H, Wang Z, Deng X, Herbert S, Xing B (2013) Impacts of adding biochar on nitrogen retention and bioavailability in agricultural soil. Geoderma 206:32–39
Zibilske LM, Bradford JM (2007) Oxygen effects on carbon, polyphenols, and nitrogen mineralization potential in soil. Soil Sci Soc Am J 71:133–139
Zimmerman AR (2010) Abiotic and microbial oxidation of laboratory-produced black carbon (Biochar). Environ Sci Technol 44:1295-130
Zibilski LM (1994) Carbon mineralization. In: Bingham JM, Mickelson SH (eds) Method of soil analysis, part 2. Microbiological and biochemical properties. SSSA book series no. 5. ASA, SSSA, Madison
Acknowledgements
The authors are highly thankful to the Director, HOD and technical staff of the Division of Soil Chemistry and Fertility, ICAR-IISS, Bhopal, as well as Director, ICAR-IIFSR, Modipuram, for necessary help during the course of the investigation. The authors are equally thankful to Dr B L Lakaria, Division of Soil Chemistry and Fertility, ICAR-IISS, Bhopal, for valuable help during the course of study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Meena, A.L., Jha, P., Dotaniya, M.L. et al. Carbon, Nitrogen and Phosphorus Mineralization as Influenced by Type of Organic Residues and Soil Contact Variation in Vertisol of Central India. Agric Res 9, 232–240 (2020). https://doi.org/10.1007/s40003-019-00425-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40003-019-00425-7