Abstract
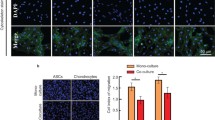
Due to lack of optimal inductive protocols, how to effectively improve chondrogenesis of adiposederived stem cells (ASCs) is still a great challenge. Our previous studies demonstrated that the culture media derived from chondrocyte-scaffold constructs (conditional media) contained various soluble chondrogenic factors and were effective for directing chondrogenic differentiation of bone marrow stem cells. Nevertheless, it remains unclear whether the conditional media can induce ASCs towards chondrogenic differentiation, especially for three-dimensional (3D) cartilage formation in a preshaped scaffold. In this study, it demonstrated that the conditional media derived from chondrocyte-scaffold constructs could promote ASCs to differentiate into chondrocyte-like cells, with similar expression of type II collagen to those induced by chondrogenic growth factors. Moreover, the expression level of chondrocyte-specific genes, such as SOX9, type II collagen, and COMP, was even higher in conditional medium group (CM) than that in optimized chondrogenic growth factor group (GF), indicating that the conditional media can serve as an effective inducer for chondrogenic differentiation of ASCs. Most importantly, the conditional media could also induce ASC-scaffold constructs to form 3D cartilage-like tissue with typical lacunae structures and positive expression of cartilage specific matrices, even higher contents of GAG and type II collagen were achieved in CM group compared to GF group. The current study establishes a simple, but stable, efficient, and economical method for directing 3D cartilage formation of ASCs, a strategy that may be more closely applicable for repairing cartilage defects.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
PA Zuk, M Zhu, H Mizuno, et al., Multilineage cells from human adipose tissue: implications for cell-based therapies, Tissue Eng, 7, 211 (2001).
PA Zuk, M Zhu, P Ashjian, et al., Human adipose tissue is a source of multipotent stem cells, Mol Biol Cell, 13, 4279 (2002).
L Aust, B Devlin, SJ Foster, et al., Yield of human adiposederived adult stem cells from liposuction aspirates, Cytotherapy, 6, 7 (2004).
MJ Varma, RG Breuls, TE Schouten, et al., Phenotypical and functional characterization of freshly isolated adipose tissuederived stem cells, Stem Cells Dev, 16, 91 (2007).
T Hennig, H Lorenz, A Thiel, et al., Reduced chondrogenic potential of adipose tissue derived stromal cells correlates with an altered TGFbeta receptor and BMP profile and is overcome by BMP-6, J Cell Physiol, 211, 682 (2007).
AT Mehlhorn, P Niemeyer, K Kaschte, et al., Differential effects of BMP-2 and beta1 on chondrogenic differentiation of adipose derived stem cells, Cell Prolif, 40, 809 (2007).
K Pelttari, A Winter, E Steck, et al., Premature induction of hypertrophy during in vitro chondrogenesis of human mesenchymal stem cells correlates with calcification and vascular invasion after ectopic transplantation in SCID mice, Arthritis Rheum, 54, 3254 (2006).
E Steck, J Fischer, H Lorenz, et al., Mesenchymal stem cell differentiation in an experimental cartilage defect: restriction of hypertrophy to bone-close neocartilage, Stem Cells Dev, 18, 969 (2009).
A Jikko, Y Kato, H Hiranuma, et al., Inhibition of chondrocyte terminal differentiation and matrix calcification by soluble factors released by articular chondrocytes, Calcif Tissue Int, 65, 276 (1999).
N Ahmed, R Dreier, A Gopferich, et al., Soluble signalling factors derived from differentiated cartilage tissue affect chondrogenic differentiation of rat adult marrow stromal cells, Cell Physiol Biochem, 20, 665 (2007).
X Liu, H Sun, D Yan, et al., In vivo ectopic chondrogenesis of BMSCs directed by mature chondrocytes, Biomaterials, 31, 9406 (2010).
JC Leijten, N Georgi, L Wu, et al., Cell Sources for Articular Cartilage Repair Strategies: Shifting from Monocultures to Cocultures, Tissue Eng Part B Rev, 19, 31 (2013).
VV Meretoja, RL Dahlin, FK Kasper, et al., Enhanced chondrogenesis in co-cultures with articular chondrocytes and mesenchymal stem cells, Biomaterials, 33, 6362 (2012).
JS Lee, GI Im, Influence of chondrocytes on the chondrogenic differentiation of adipose stem cells, Tissue Eng Part A, 16, 3569 (2010).
M Schnabel, S Marlovits, G Eckhoff, et al., Dedifferentiationassociated changes in morphology and gene expression in primary human articular chondrocytes in cell culture, Osteoarthritis Cartilage, 10, 62 (2002).
C Cournil-Henrionnet, C Huselstein, Y Wang, et al., Phenotypic analysis of cell surface markers and gene expression of human mesenchymal stem cells and chondrocytes during monolayer expansion, Biorheology, 45, 513 (2008).
Z Lin, JB Fitzgerald, J Xu, et al., Gene expression profiles of human chondrocytes during passaged monolayer cultivation, J Orthop Res, 26, 1230 (2008).
K Liu, GD Zhou, W Liu, et al., The dependence of in vivo stable ectopic chondrogenesis by human mesenchymal stem cells on chondrogenic differentiation in vitro, Biomaterials, 29, 2183 (2008).
D Yan, G Zhou, X Zhou, et al., The impact of low levels of collagen IX and pyridinoline on the mechanical properties of in vitro engineered cartilage, Biomaterials, 30, 814 (2009).
MF Pittenger, AM Mackay, SC Beck, et al., Multilineage potential of adult human mesenchymal stem cells, Science, 284, 143 (1999).
N Indrawattana, G Chen, M Tadokoro, et al., Growth factor combination for chondrogenic induction from human mesenchymal stem cell, Biochem Biophys Res Commun, 320, 914 (2004).
K Kawamura, CR Chu, S Sobajima, et al., Adenoviral-mediated transfer of beta1 but not IGF-1 induces chondrogenic differentiation of human mesenchymal stem cells in pellet cultures, Exp Hematol, 33, 865 (2005).
JR Matyas, D Huang, M Chung, et al., Regional quantification of cartilage type II collagen and aggrecan messenger RNA in joints with early experimental osteoarthritis, Arthritis Rheum, 46, 1536 (2002).
G Zhou, W Liu, L Cui, et al., Repair of porcine articular osteochondral defects in non-weightbearing areas with autologous bone marrow stromal cells, Tissue Eng, 12, 3209 (2006).
L Vijgen, E Keyaerts, E Moes, et al., Development of one-step, real-time, quantitative reverse transcriptase PCR assays for absolute quantitation of human coronaviruses OC43 and 229E, J Clin Microbiol, 43, 5452 (2005).
GK Reddy, CS Enwemeka, A simplified method for the analysis of hydroxyproline in biological tissues, Clin Biochem, 29, 225 (1996).
S Bjornsson, Simultaneous preparation and quantitation of proteoglycans by precipitation with alcian blue, Anal Biochem, 210, 282 (1993).
GR Srinivas, HJ Barrach, CO Chichester et al., Quantitative immunoassays for type II collagen and its cyanogen bromide peptides, J Immunol Methods, 159, 53 (1993).
H Afizah, Z Yang, JH Hui, et al., A comparison between the chondrogenic potential of human bone marrow stem cells (BMSCs) and adipose-derived stem cells (ADSCs) taken from the same donors, Tissue Eng, 13, 659 (2007).
JD Kisiday, PW Kopesky, CH Evans, et al., Evaluation of adult equine bone marrow- and adipose-derived progenitor cell chondrogenesis in hydrogel cultures, J Orthop Res, 26, 322 (2008).
GI Im, YW Shin, KB Lee, et al., Do adipose tissue-derived mesenchymal stem cells have the same osteogenic and chondrogenic potential as bone marrow-derived cells?, Osteoarthritis Cartilage, 13, 845 (2005).
YJ Kim, HJ Kim, GI Im, et al., PTHrP promotes chondrogenesis and suppresses hypertrophy from both bone marrow-derived and adipose tissue-derived MSCs, Biochem Biophys Res Commun, 373, 104 (2008).
J Fischer, A Dickhut, M Rickert, et al., Human articular chondrocytes secrete parathyroid hormone-related protein and inhibit hypertrophy of mesenchymal stem cells in coculture during chondrogenesis, Arthritis Rheu, 62, 2696 (2010).
Author information
Authors and Affiliations
Corresponding authors
Additional information
These authors contributed equally to this work.
Rights and permissions
About this article
Cite this article
Sun, H., Liu, Y., Jiang, T. et al. Chondrogenic differentiation and three dimensional chondrogenesis of human adipose-derived stem cells induced by engineered cartilage-derived conditional media. Tissue Eng Regen Med 11, 59–66 (2014). https://doi.org/10.1007/s13770-013-1120-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13770-013-1120-y