Abstract
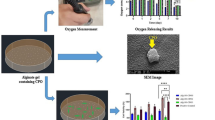
Coagulative necrosis often occurs under hypoxic conditions, causing major limitation in the field of tissue engineering especially those dealing with larger tissues and organs. In this study, a comprehensive work has been performed in developing a tailor made design of a dual layered matrix that can produce oxygen to be utilized in tissue engineering application. Optimizations of protocol, ingredient and condition of the system were carried out specifically based on the responses observed from in vitro studies using L6 rat skeletal muscle cell as a candidate. Oxygen was generated from decomposition of encapsulated hydrogen peroxide. Poly (D,L)-lactide-co-glycolide (PLGA) with molecular weights of 90,000 and 110,000 gmol−1 managed to secure good encapsulation of hydrogen peroxide for this application, while the best stirring time during the encapsulation was found to be 8 hours. The PLGA microspheres were coated with a secondary layer of alginate that was pre-grafted with calatase to form the dual layer system. This dual layered architecture has successfully controlled the release rate of oxygen at an optimum level for the survival of muscle cells under hypoxia condition. It was found that muscle cells have low tolerance limit towards the direct contact with hydrogen peroxide, however the cells maintained high viability within encapsulated hydrogen peroxide in the matrix system. It was observed that 4% of encapsulated hydrogen peroxide in the matrix system can produce efficient amount of oxygen at a controlled release manner to sustain the survival of muscle cells under hypoxic condition.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
LE Freed, G Vunjak-Novakovic, RJ Biron, et al., Biodegradable polymer scaffolds for tissue engineering, Nature Biotechnology, 12, 689 (1994).
MJ Lysaght, J Reyes, The growth of tissue engineering, Tissue Eng, 7, 485 (2001).
AH Reddi, Morphogenesis and tissue engineering of bone and cartilage: inductive signals, stem cells, and biomimetic biomaterials, Tissue Eng, 6, 351 (2000).
A Anthony, BB Stuart, S Shay, et al., Tissue-engineered autologous bladders for patients needing cystoplasty, The Lancet, 367, 1241 (2006).
J Malda, TJ Klein, Z Upton, The roles of hypoxia in the in vitro engineering of tissues, Tissue Eng, 13, 2153 (2007).
P Vaupel, F Kallinowski, P Okunieff, Blood Flow, oxygen and nutrient supply, and metabolic microenvironment of human tumors: a review, Cancer Res, 49, 6449 (1989).
JE Valentin, DO Freytes, JM Grasman, et al., Oxygen diffusivity of biologic and synthetic scaffold materials for tissue engineering, J Biomed Mater Res Part A, 91A, 1010 (2009).
K Kellner, G Liebsch, I Klimant, et al., Determination of oxygen gradients in engineered tissue using a fluorescent sensor, Biotechnol Bioeng, 80, 73 (2002).
J Malda, J Rouwkema, DE Martens, et al., Oxygen gradients in tissue-engineered Pegt/Pbt cartilaginous constructs: measurement and modeling, Biotechnol Bioeng, 86, 9 (2004).
A Khademhosseini, R Langer, J Borenstein, et al., Microscale technologies for tissue engineering and biology, Proceedings of the National Academy of Sciences of the United States of America, 103, 2480 (2006).
BS Harrison, D Eberli, SJ Lee, et al., Oxygen producing biomaterials for tissue regeneration, Biomaterials, 28, 4628 (2007).
SH Oh, CL Ward, A Atala, Oxygen generating scaffolds for enhancing engineered tissue survival, Biomaterials, 30, 757 (2009).
FK Sarwat, C Kyuong-sik, RB Surita, et al., Enhancing oxygen tension and cellular function in alginate cell encapsulation devices through the use of perfluorocarbons, Biotechnol Bioeng, 96, 156 (2007).
HC Kim, TK Kwon, JY Lee, et al., The effect of intake of calcium and vitamin d on the expression of vascular endothelial growth factor receptors (VEGFR) in bone tissue around implant in osteoporosis induced-rats, Tissue Eng Regen Med, 8(S2), 73 (2011).
HR Wang, Y Zhang, B Ozdamar, et al., Regulation of cell polarity and protrusion formation by targeting RhoA for degradation, Science, 302, 1775 (2003).
GG Giordano, MF Refojo, MH Arroyo, Sustained delivery of retinoic acid from microspheres of biodegradable polymer in PVR, Invest, Ophthalmol, Vis, Sci, 34, 2743 (1993).
R Langer, New methods of drug delivery, Science, 249, 1527 (1990).
SM Ng, JY Choi, HS Han, et al., Novel microencapsulation of potential drugs with low molecular weight and high hydrophilicity: hydrogen peroxide as a candidate compound, Inte J Pharma, 384, 120 (2010).
SIH Abdi, SM Ng, JO Lim, An enzyme-modulated oxygenproducing micro-system for regenerative therapeutics, Inte J Pharma, 409, 203 (2011).
A Barbetta, E Barigelli, M Dentini, Porous alginate hydrogels: synthetic methods for tailoring the porous texture, Biomacromolecules, 10, 2328 (2009).
CS Visconti, K Kavalkovich, JJ Wu, et al., Biochemical analysis of collagens at the ligament-bone interface reveals presence of cartilage-specific collagens, Arch Biochem Bioph, 328, 135 (1996).
G Fundueanu, C Nastruzzi, A Carpov, et al., Physico-chemical characterization of Ca-alginate microparticles produced with different methods, Biomaterials, 20, 1427 (1999).
GH Choi, YH Youn, DY Kim, et al., Alginate gel decreases chondroitin sulfate proteoglycan immunoreactivityfollowing spinal cord injury: Preliminary Study, Tissue Eng Regene Med, 3, 472 (2006).
JH Woo, DY Kim, SY Jo, et al., Modification and evaluation of porous poly (lactide-co-glycolide) scaffolds by ion beam with high energy, Biomat Res, 13, 152 (2009).
JW Jang, KS Park, SH Kim, et al., Tissue engineered bone regeneration using DBP-loaded PLGA scaffoldin rabbit model, Tissue Eng Regen Med, 2, 34 (2005).
H Johno, S Takahashi, M Kitamura, Influences of acidic conditions on formazan assay: A Cautionary note, Applied Biochem Biotechnol, 162, 1529 (2010).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abdi, S.I.H., Choi, J.Y., Lau, H.C. et al. Controlled release of oxygen from PLGA-alginate layered matrix and its in vitro characterization on the viability of muscle cells under hypoxic environment. Tissue Eng Regen Med 10, 131–138 (2013). https://doi.org/10.1007/s13770-013-0391-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13770-013-0391-7