Abstract
Microalgae have been highlighted as one of the promising feedstocks for renewable fuel production owing to their high growth rate, ability to grow on wastewater, and to convert CO2 into lipids, the main source of biodiesel. However, lipid extraction from microalgal cells is cumbersome due to its thick cell wall which precludes commercial biodiesel production from microalgae, and to improve the lipid extraction efficiency, optimization of the operational parameters is crucial. One of the well-known challenges is to develop an economical technique to obtain a high lipid extraction efficiency. Hence, this study aimed to investigate the optimized conditions of lipid extraction from marine species of Nannochloropsis sp. PTCC 6016 isolated from the Persian Gulf using Soxhlet, Bligh & Dyer, and ultrasonication methods. Initially, Nannochloropsis sp. was cultivated in a 1000L open pond for the purpose of lipid evaluation and process optimization, to be transferred to a 2000 L and subsequently, a 30000L open pond. Effects of various solvent ratios, mixing time and mixing frequency, pre-treatment, and cell age have been evaluated, and the optimal conditions have been determined. Lipid extraction prediction has been investigated by artificial neural network (ANN) and support vector regression (SVR) methods. Experimental results show that the maximum extraction yield of 0.56 gr extracted lipid/gr dry biomass was obtained using Soxhlet method after 90 days of cultivation using chloroform–methanol (1:1) as extraction solvents. However, considering 10 times higher solvent used in Soxhlet method compared to Bligh and Dyer and ultrasonication methods, the ultrasonication method was the most desired option for lipid extraction efficiency of Nannochloropsis sp. in large-scale operations. In this regard, cost-effective biofuel production should be considered as the focus of future studies to achieve sustainability, energy security and addressing the climate change due to high greenhouse gas emissions.
Graphic abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The rapid growth of population, as well as industrialization, has increased the global energy demand in the past century (Fallahi et al. 2021a). Coal and other fossil fuels have been mainly used to meet the global need for energy (Adholeya and Dadhich 2008). Therefore, the excessive use of such energy sources results in the depletion of these non-renewable sources, and hence, finding an alternative energy source is necessary (Rabbani et al. 2020, 2021; Asgharnejad et al. 2021).
Biofuels such as bioethanol derived from corn starch and biodiesel from soybean have been proposed as a suitable alternative for fossil fuels as they are sustainable and help the environment by reducing emissions (Hajinajaf et al. 2021). However, feedstocks that are used to produce biofuels need a large arable land, usage of a huge amount of fresh water for irrigation, fertilizers, and most importantly can serve as human food and thus might solve famine (Demirbas 2009; Hill et al. 2006).
Oil extracted from microalgae can be regarded as a promising alternative source as it has high photosynthetic efficiency and biomass productivity. Besides, microalgae have the ability to fix carbon dioxide (1.83 kg of CO2 per 1 kg of biomass) and can be cultured in wastewater (Hajinajaf et al. 2021; Yadav et al. 2019); thus, can treat the wastewater by the usage of nitrogen and phosphorus as nutrients and therefore obviates the need for a vast area of land (Hajinajaf et al. 2021; Fallahi et al. 2021b). Notably, the lipid content of microalgae is reported to be approximately ten times higher than that of oil crops (Chisti May 2007; Maggio and Cacciola 2012; Ahmad et al. 2011), and therefore, third-generation biofuel from microalgae looks promising (Anto et al. 2020; Chowdhury and Loganathan 2019). Even though microalgae offer numerous advantages over typical oil crops, many technological bottlenecks including cultivation, lipid maximization, harvesting, lipid extraction, etc., preclude biofuel production from microalgae to become economically practical because of high cost and energy consumption (Hajinajaf et al. 2021; Kumar et al. 2010; Scott, et al. 2010).
Generally, energy production from microalgal cells (specifically biofuel) includes five main stages: species selection, cultivation, biomass harvesting, lipid extraction and fuel production. High biomass productivity and lipid content are the two most important features that should be considered for biodiesel production (Hajinajaf et al. 2021; Shanmugam et al. 2020) in which out of hundred thousand of microalgal strains, only a few of them have the capability to produce high lipid contents (Sharma et al. 2008). Fallahi et al. (2020) investigated the lipid productivity in three species of microalgae, Chlorella vulgaris, Scenedesmus obliquus, and Nannochloropsis sp. and obtained the highest lipid content and productivity of 39% and 66 mg. L−1 d−1, respectively, for Nannochloropsis sp. Therefore, in this study, Nannochloropsis sp., a well-known marine strain of microalgae which has found to be the best potential strain for the purpose of biofuel production, has been selected (Fallahi et al. 2020; Rodolfi et al. 2009; Xiao et al. 2013).
Although selection of a high lipid value strain of microalgae is inevitable, cell lipid content and productivity can be further increased by changing the cultivation or nutritional conditions such as exposure to different CO2 levels, high light intensity, exerting temperature, salt and heavy metal stresses (Hajinajaf et al. 2021). Besides, for the aim of biofuel production from microalgae, lipid extraction stage considered as the most effective stage in an economical point of view (Ranjith Kumar et al. 2015; Corrêa et al. 2021; Kwon and Yeom 2015; Cancela et al. 2017). Commonly used lipid extraction strategies are Soxhlet, Bligh and Dyer, and Folch (Stanley et al. 1957; Bligh and Dyer 1959; Ryckebosch et al. 2012; Khoo et al. 2020). As Bligh and Dyer developed their method for rapid lipid content in fish muscle, Jensen (Jensen 2008) tried to improve their method by adding HCL to methanol–chloroform solvent and shown that adding HCL can increase the lipid extraction and total fatty acids from pig feces by 10–15% and 30–50%, respectively. Also, several studies investigated strategies to increase the lipid content in the microalgal cells. In the research by Abou-Shanab et al. (2014), a new strain of microalga Micractinium reisseri was cultured in an industrial wastewater and its growth, nutrient uptake and lipid content was measured by Bligh and Dyer method. Wahidin et al. (2013) assessed the effect of light photoperiod and intensity on the lipid content of microalgae Nannochloropsis sp. Also, effects of different lipid extraction methods from Nannochloropsis oceanica on the contents of omega-3 fatty acids were also investigated by Figueiredo et al. (2019). Furthermore, lipid content of the microalgae Nannochloropsis sp. was measured by Thawechai et al. (2016) using Bligh and Dyer method under various light intensity and CO2 feeding strategies. Although Bligh and Dyer method was used extensively for lipid content determination from microalgae, a few studies focused on optimizing the lipid extraction efficiency and assessing operational and cultivation parameters on microalgal lipid extraction efficiency. Figure 1 shows the schematic process of lipid extraction from microalgae.
Hence, in this work, lipid extraction from microalgae Nannochloropsis sp. PTCC 6016 was accomplished using Bligh and Dyer, Soxhlet, and ultrasonication methods. The aim of this study was to accomplish a comprehensive study on the evaluation of various operational parameters on oil extraction to obtain the optimal conditions and hence maximize the lipid extraction efficiency. Effect of cell age and optimization of Bligh and Dyer method has been never studied to the best of our knowledge, and therefore, parameters such as solvent ratios, mixing time, cell age, mixing frequency, pre-treatment methods were assessed. Artificial neural network (ANN) and support vector regression (SVR) methods were used to model the lipid extraction efficiency. Observations achieved in this work were used for the cultivation of Nannochloropsis sp. in a 2000 L and then a 30,000 L industrial-scale open raceway ponds.
Material and methods
Microalgae culture and medium
The strain used in this work is Nannochloropsis sp. PTCC 6016 which was isolated from Persian Gulf (26° 32′ N, 53° 56′ E) (Iran) (Moazami et al. 2011) and provided by Iranian Biological Resource Center (IBRC), Tehran, Iran. The F/2 medium was used for the maintenance of the inoculum and cultivation in which its composition is as follows: NaNO3 (75.0 g/l), NaH2HPO4.H2O (5.0 g/l), Na2SiO3.9H2O (30.0 g/l), FeCl3.6H2O (1.17 × 10–5 g/l), Na2EDTA.2H2O (1.17 × 10–5 g/l), CuSO4.5H2O (3.93 × 10–8 g/l), Na2MoO4.2H2O (2.60 × 10–8 g/l), ZnSO4.7H2O (7.56 × 10–8 g/l), CoCl2.6H2O (4.20 × 10–8 g/l), MnCl2.4H2O (9.10 × 10–7 g/l), Thiamine HCL (2.96 × 10–7 g/l), Biotin (2.05 × 10–9 g/l), Cyanocobalamin (3.69 × 10–10 g/l). All the chemicals and media used in this study were purchased from Merck (Merck Chemicals (Shanghai) Co. Ltd.) and CIBA (BASF Schweiz AG, Basel, Switzerland). All reagents were laboratory grade, and deionized distilled water was used. Polarity index and solubility coefficients of the solvents are important factors in lipid extraction from microalgae which are illustrated in Table 1.
Microalgal cultivation and experimental system
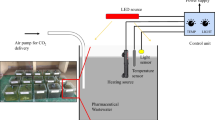
The Nannochloropsis sp. PTCC 6016 microalgae strain cultivated in an open raceway pond with 1000 L working volume as shown in Fig. 2a. This open raceway contains sodium lamp to provide light energy, paddle for flowing and homogenizing medium and CO2 cylinder for pH control. The culture conditions including the light/dark cycles of 12/12, the temperature of 25 °C and pH maintained at 8. The air (carbon dioxide) and nutrient concentration were kept constant during all experiment runs. For sample one, after the medium reached the optical density (OD550) of 0.8 (indicator for stationary phase) the paddle of open raceway turned off and 20 L of biomass medium was separated in order to sediment the biomass by the gravimetric force. The process continued with the rest of the medium. This process was repeated after 1.5 months, and the last sample was separated after 3 months. The sedimented biomass washed two times by distilled water to remove residual cultivation medium and was dried at 105 °C for 2 h. The dried biomass was milled, and the powdered biomass was used for lipid extraction. Results obtained from cultivation of Nannochloropsis sp. in the 1000 L open pond were used for its cultivation in the 2000 L and eventually 30,000 L open ponds, which have shown in Fig. 2b and c, respectively.
Lipid extraction methods
Soxhlet method
Soxhlet methods have been suggested for lipid extraction for its fast extraction, high yield and high-quality lipid samples (Boni et al. 2018; Heinrich et al. 2012). In this regard, lipid extraction through the Soxhlet method was performed using 5.00 ± 0.50 g of dry biomass. As the solubility of polar lipids which are mostly located at the cell wall of the microalgae is limited in hexane, a mixture of chloroform–methanol and hexane–acetone were used as the solvents in the ratios of 1:1, 1:2 and 2:1 as well as 1:0, 1:2 and 1:1, respectively. Primarily, 5 gr of dried cell was added to a 200 ml of solvent mixtures and transferred to the extraction chamber. The final mixture was heated at 65 degrees Centigrade for 24 h. Subsequently, the solvents were evaporated by a rotary evaporator, and the weight of the extracted lipids was measured by gravimetric method.
Bligh and Dyer method
Bligh and Dyer method is one of the most widely used lipid extraction methods from biological tissues such as microalgae (Bligh and Dyer 1959; Breil et al. 2017). Primarily, 1 ± 0.10 g of dried biomass was added to 6 ml chloroform–methanol mixture (1:2) and mixed well for 2 min with 2400 rpm. Then, 2 ml of chloroform was added and mixed for 30 s and subsequently stirred with 2 ml of water. Vacuum pump was then used for the filtration of samples, and then, the system containing three phases was filtered and centrifuged for 15 min at 3500 rpm. As shown in Fig. 3, the upper phase contains water and methanol, the middle phase contains non-lipid components, polar lipids and chloroform, and the lower phase contains chloroform and extracted lipids. Finally, the lipid-rich phase was separated by a funnel and the upper phase (polar phase) was separated by Pasteur pipette and the lower phase (non-polar phase) was transferred to Petri dishes. The weight of lipids was measured after the chloroform was evaporated at 30 °C under high vacuum.
Ultrasonication method
Ultrasonication method was used as a comparative method with Bligh and Dyer and Soxhlet methods (Li et al. 2014). One gram of the dried biomass was added to chloroform–methanol (1:2) mixture in a probe ultrasound (Sonopuls HD 200, 200 W, 20 kHz, Germany) and sonicated for 20 min in the maximum power (200 W) and 100% cycles. Subsequently, 10 ml of chloroform was added to the resulting homogeneous mixture and was mixed by a mixer for 30 s, which was followed by the addition of 10 ml of water and a 30-s mixing. Following that, the sample was centrifuged at 3500 rpm for 15 min, and the supernatant was removed. The lower phase consisting of lipids and chloroform was transferred to a Petri dish, and solvent was evaporated in the ambient temperature. Finally, the weight of the lipids was measured. To optimize this process, various parameters including the biomass/solvent ratio, simultaneous effect of solvent ratios and cavitation time, simultaneous effect of power and cycles frequency were assessed.
Qualitative and quantitative analysis of the extracted lipids
Nile red staining
To identify the amount and the quality of the lipid inside the cell, Nile red staining method was used using fluorescence microscopy (Rumin et al. 2015). Initially, a Nile red solution was prepared, and 5 µL of the solution was added to 1 ml of the cell culture. Samples were kept for 15 min, and images of the samples under the fluorescence microscope have been taken.
Gas chromatography (GC) analysis
GC analysis (GC-TCD-Varian 3400 and Teyfgostar-Compact) with FID detector was performed to quantify the fatty acids. Iso-octane was added to 1 g of lipid. The mixture was added to KOH and mixed well followed by the addition of NaHSO4 after 5 min. The upper transparent liquid was injected to the GC, equipped with FID and capillary column.
Optimization and prediction method
In this study, lipid extraction prediction by Bligh and Dyer method has been investigated by artificial neural network (ANN) and support vector regression (SVR) methods.
Neural network optimization
The artificial neural network is created from simple operating elements, and they process the input information to the network in a parallel manner. According to Eq. (1), inputs multiplied by the adjusting weights after being added to the bias are first summed up and then passed through a function to produce the output for that neuron (Hoskins and Himmelblau 1988; Lee and Chen 1993).
where f is the operating function, Ni is the number of inputs, w is the connection weights, b is the bias, and u and Z are the ith input and jth output of an ANN, respectively. The multi-layer perceptron (MLP) is a feed-forward artificial neural network that can perform a non-linear mapping with arbitrary precision by selecting the number of layers and neurons (Engelbrecht 2007).
The Levenberg–Marquardt algorithm as a learning algorithm is used for training the MLP model. This algorithm is effective and has a desirable accuracy on the learning speed and results (Hagan and Menhaj 1994; Rabbani et al. 2017). In this study, various MLP structures, hidden layers, and neurons’ number were considered, and then, the proper network with the minimum error was selected.
Support vector regression
Support vector regression (SVR) is one of the learning methods with monitoring which is used for arrangement and regression purposes. Support vector regression model is being utilized to solve the forecasting problems and optimization. In SVR, a function related to the dependent variable yi which is a function of several independent variables xi is being estimated. Similar to other regression methods, it is assumed that the relationship between independent and dependent variables is obtained by an algebraic function such as y = f (x), which is similar to the following equation:
where w is the weight vector, b is the bias factor, and ϕ is a kernel function, while the goal is to find a functional form of f (x). This can be achieved by training a SVR model using a set of data which the model operation basis has been previously discussed in the previous study (Bligh and Dyer 1959). Briefly, the characteristics of w and b in the SVR model are calculated using the Karush–Kuhn–Tucker theory conditions where the w is as shown in Eq. (3):
where \(\alpha\) is the Lagrange coefficient, and \(\phi (x)\) is the core function. Calculation of \(\phi (x)\) is very complicated, and thus, to solve this, a kernel function is defined in Eq. (4):
Different kernels are used for a backup vector regression model, which can be linear, quadratic, Gaussian, logistic, or polynomial. To determine an efficient model for the SVR method, the model parameters along with the kernel parameter must be carefully calculated.
3-3-Network evaluation criteria
To design an appropriate neural network and the creation of an appropriate model, the correlation coefficient and root mean square error (RMSE) were used for the evaluation of the results.
The square of the correlation coefficient “R2” and root mean square error is defined as:
where YEXP and YM are the values of experimental and model predictions, respectively, and N is the number of data.
Results and discussion
Microalgae lipids are divided into two groups including non-polar lipids (such as triglycerides, diglycerides, monoglycerides, and sterols) which are cytoplasmic storage lipids and polar lipids (such as free fatty acids, phospholipids, and sphingolipids) which are mainly in the cell membranes (Patil et al. 2007). The type of lipids and extraction yield of lipids from microalgal biomass depends on various parameters. Some of these parameters are temperature, nutrients, the age of microalgae, amount of permeability and selectivity of solvent, type of cell (cell wall composition and thickness), pre-treatment, and mechanical methods of cell disruption (Mercer and Armenta 2011; Pernet and Tremblay 2003; Lee et al. 2010). Proper penetration of extracting solvent into the biomass can guarantee an efficient lipid extraction (Nagappan et al. 2019). The solvents used in this study are chloroform–methanol, hexane–acetone, and hexane as explained in the material and methods section.
Lipid extraction from Nannochloropsis sp. by Soxhlet method
Lipid was extracted using Soxhlet method by the solvent mixtures hexane–acetone and chloroform–methanol in different ratios. Figure 4 shows the amount of lipid extracted from Nannochloropsis sp. by Soxhlet method. From the graph, the lowest and highest extraction efficiency achieved for hexane and chloroform–methanol (1:2), respectively. The extraction efficiency for the hexane was not desired due to the fact that the algal cell wall is made from phospholipids (polar lipids) which is not soluble in hexane in 24 h. In this regard, acetone was added to hexane to increase the lipid extraction efficiency as is observable in Fig. 4.
Chloroform–methanol mixture was also used as solvent mixture to extract lipids from Nannochloropsis sp. cells. Various ratios of 2:1, 1:1 and 1:2 (chloroform–methanol) were selected. As can be seen in Fig. 4, increasing methanol/chloroform ratio results in higher lipid extraction efficiency which can be attributed to the high polarity of methanol and its capability to dissolve phospholipids. Hence, the higher amount of methanol, the higher cell wall disruption and easier penetration of chloroform inside the cell which in turn results in more non-polar lipid solubility in chloroform.
Comparison of the lipid extraction data in Fig. 4 demonstrated that higher efficiency achieved by chloroform–methanol solvent than hexane–acetone. The polarity index (PI) and solubility parameter (δ) can be considered to compare the ability of solvents for lipid extraction. A comparison of the polarity index and solubility parameter of these two solvent mixtures is shown in Table 2. The higher the polarity, the stronger effect on cell wall. As hexane is a non-polar solvent it only has the capability of dissolving non-polar lipids in itself which in turn results in less lipid extraction. On the other hands, the polarity index of chloroform–methanol is higher than that of hexane–acetone which culminates in higher cell wall disruption and hence, a higher lipid extraction efficiency.
Figure 5 shows the algal cell in the various solvents using Soxhlet method, four days after reaching the stationary phase. The addition of more acetone to hexane results in a higher extraction efficiency while adding more acetone reduces the efficiency due to reduction in non-polar extracted lipid by hexane. The effects of solvent impact on cell wall disruption are shown in Fig. 5a–d in hexane (a), acetone–hexane in 1:2 (b), acetone–hexane in 1:1 (c), and chloroform–methanol in 1:1 (d) which validates the best cell wall disruption for the chloroform–methanol mixture. In the study, by Kumar et al. (Figueiredo et al. 2019), ratio of 2: 1 for chloroform/ methanol was selected for the lipid extraction by Soxhlet method, and lipid extraction of 18.6% was achieved.
Lipid extraction from Nannochloropsis sp. by Bligh & Dyer method
The Bligh and Dyer method (Bligh and Dyer 1959) is a three-step lipid extraction method which was formerly used for the extraction of lipids from fish tissue. This well-established method allows an easy and efficient lipid extraction that no pre-drying is needed for analysis. In this method, methanol and water with the polarity indexes of 5.1 and 10.2, and chloroform with 4.1 polarity index as a non-polar solvent were used (Ramluckan et al. 2014). Using this solvent mixture of polar and non-polar solvents allows a high lipid recovery compared to single extraction solvent (Fallahi et al. 2020). However, this method is considered onerous, and chloroform has adverse effects on the environment (Kumar et al. 2019).
In the research conducted by Ryckebosch et al. (Fallahi et al. 2020), chloroform/methanol with 1:1 ratio has been used and recommended for lipid extraction from Chlorella vulgaris. However, Lee et al. (1998) reached the maximum lipid extraction of 28.6% from Botryococcus braunii UTEX 572 using chloroform/methanol with 2:1 (v/v) ratio. An optimization on lipid extraction efficiency of Nannochloropsis salina strain was accomplished by Chatsungnoen and Chisti (Chatsungnoen and Chisti 2016) and suggested the ratio of 5.7:3:1 for chloroform, methanol, and water as the best solvent mixture ratio. A comparison between the Bligh and Dyer method and hexane extraction from a Chlorella vulgaris/cyanobacteria co-culture was carried out recently by Ellison et al. (2019), and the obtained results showed that Bligh and Dyer had the lipid extraction efficiency as twice as the hexane.
To best of our knowledge, there is no evidence on the optimization of operational parameters in Bligh and Dyer method. Hence, the effects of biomass/solvent and solvent/solvent ratios, mixing time, mixing frequency, and pre-treatment method were investigated on Nannochloropsis sp. which are as follows:
Effect of biomass/solvent and solvent/solvent ratios on extraction efficiency
Figure 6 shows the effect of chloroform–methanol solvent mixture and its ratios on lipid extraction efficiency of Nannochloropsis sp., 4 days after reaching the stationary phase. It is observable that the addition of solvent mixture increased the amount of extracted lipid which is due to the fact that more solvent is available to each cell. A twofold increase in lipid extraction from 3 to 6 ml of solvent is represented which is important for scale-up as it can save a large amount of solvent and therefore reduce the operational costs. Another observation from Fig. 6 demonstrates that changing the ratio of solvents from 2:1 (chloroform–methanol) to 1:2 (chloroform–methanol) increases the lipid extraction by 64% when 15 ml of solvent was used. Higher amounts of methanol would result in a single phase that is not desired.
Effect of mixing time on extraction efficiency
In Bligh and Dyer method, typically, the mixing time is considered 2 min. However, the effect of various mixing times on lipid extraction has been investigated in this research. Figure 7 shows the effect of mixing time on the grams of lipid extracted per grams of dry biomass and a decreasing trend for gram lipid extracted by increasing mixing time is observable. Generally, in Bligh and Dyer method, microalgal lipid extraction efficiency is influenced by three main stages: (1) evaporation of part of solvent, (2) absorption of some solvent containing lipid by biomass and (3) formation of bonds between solvent and cell components. Increasing the mixing time is accomplished at the maximum frequency (2400 rpm) and the evaporation of part of solvent due to higher mixing time can be considered as the reason that resulted in the reduction of extraction efficiency at higher mixing times.
Effect of mixing frequency on extraction efficiency
The effect mixing frequency was evaluated on lipid extraction efficiency in Bligh and Dyer method. Seven frequencies of 1200, 1400, 1600, 1800, 2000, 2200, and 2400 were considered and depicted in Fig. 8. It can be seen that increasing the mixing frequency enhances the extraction efficiency at a fixed mixing time due to the fact that higher tension is exerted on the cell wall, and therefore, more cell fracture and lipid extraction efficiency is the result.
Effect of pre-treatment method on extraction efficiency
Generally, extraction using solvent is not feasible in industrial scale due to their low efficiency, high solvent recovery cost, and toxicity issues. Thus, a different strategy should be implemented to raise the efficiency. The pre-treatment method was used in this work as a strategy to boost the lipid extraction efficiency. Figure 9 shows the effect of blending and ultrasonication pre-treatment methods on Nannochloropsis sp. in stationary phase.
As can be seen in Fig. 9, implementing pre-treatment method enhances the lipid extraction efficiency in which highest amount of 0.108 gr lipid was achieved using ultrasound (chloroform). Ultrasound method gained higher amount compared to blending as cell wall gets disrupted individually, and thus, more cell fracture occurs. Also, in ultrasound method, using chloroform will gain more efficiency because of its ability to dissolve a wide range of polar and non-polar lipids. Figure 10 shows the cell samples after using pre-treatment in Bligh and Dyer extraction and justifies the effectiveness of ultrasound pre-treatment on cell wall disruption.
Lipid extraction from Nannochloropsis sp. by ultrasonication method
Ultrasonication considers to be one of the most typical methods for cell membrane disruption which facilitate the accessibility of solvents for a better lipid extraction.
In ultrasonication method, microbubbles are formed by sound waves which become larger by the absorption of ultrasonic energy. As microbubbles reach to their critical size, they start to collapse that is called ultrasonic cavitation (Mason and Lorimer 1989) which in turn propagate waves that disrupt the cell wall (Sivaramakrishnan and Incharoensakdi 2018). Figure 11 shows the ultrasonic cavitation and generation of microbubbles in their implosion. Besides, it helps the lipids to diffuse in the solvents by the turbulence made by breaking the microbubbles (Ranjan et al. 2010; Wang and Weller 2006). To enhance the lipid extraction by ultrasonication method, effect of some parameters has been investigated which are as follows:
Effect of biomass to solvent ratio
As the amount of solid phase in the ultrasonication method is limited, one, two and three grams of dried biomass powder were added to 15 ml of chloroform–methanol (1:2). Table 3 shows the effect of biomass weight on lipid extraction of Nannochloropsis sp. It is observable that increasing the amount of biomass results in the reduction of lipid extraction. This might be due to the decrease in the amount of solvent available and the energy exerted to each cell which lowers the cell disruption, and therefore, less lipid accessible to be extracted.
Effect of solvents ratio and cavitation time
The simultaneous effects of cavitation time and solvents’ ratio on the lipid extraction efficiency of Nannochloropsis sp. are represented in Fig. 12. The maximum efficiency of 20 min is observable for all solvents’ ratio. This can be attributed to the fact that less cell wall disruption occurs for the experiments accomplished in the time less than optimum value (20 min). Besides, the higher cavitation time, the higher cell wall breakage; however, some of the solvents vaporize due to the high temperature and some other absorbs to the biomass which results in a less lipid dissolution.
Another observation from this graph is that the best condition was achieved for chloroform–methanol with the ratio of 1:2. Availability of methanol helps the cell wall disruption happen faster with the help of cavitation bubbles. Higher amount of methanol, on the other hands, limits the dissolution of neutral lipids in it as methanol is a polar solvent.
Effect of power and cycle frequency
Power and cycle frequency of ultrasound are two crucial operating parameters that can affect the lipid extraction. It has found that pigment extracted from microalgae was enhanced by changing the ultrasound frequency (Engelbrecht 2007). Therefore, it was expected that lipid extraction efficiency improves by increasing the power and cycle frequency as shown in Fig. 13. The sudden increase in the lipid extraction at 76 (KE/D) power is notable by changing the cycle frequency from 60 to 80%. This observation is important for the experiments in the industrial scale to save the operational cost by reaching the target at the lowest power and cycle frequency.
Effect of cell age on lipid extraction efficiency of Nannochloropsis sp.
Lipid productivity by microalgae has extensively studied and mostly, researches illustrated that environmental stress could increase the lipid content in microalgal cells. Nitrogen depletion, sudden increase in light intensity, temperature and pH, CO2 addition and cell age are the factors that have suggested to increase the lipid content of microalgal cells, in which the effect of cell age has rarely been studied. Therefore, in this study, the effect of cell age on the lipid type was considered using Soxhlet, Bligh and Dyer and ultrasonication methods.
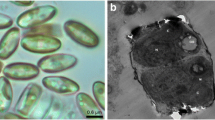
As stated previously, the microalgal cells were transferred to two 20 L flasks after reaching the stationary phase and kept for 1.5 and 3 months for further experiments. Figure 14 shows the effect of cell age on lipid extraction efficiency by Soxhlet, Bligh and Dyer and ultrasonication methods, and Fig. 15 displays the microscopic images of the microalgal cells. Based on the graph, Soxhlet method resulted a higher lipid extraction efficiency at the days 15, 45 and 90. Besides, increasing the cell age resulted in a higher lipid extraction due to the less phospholipids production as well as neutral lipids. Thus, a less phospholipid generation available means a weak cell wall to be disrupted and higher lipid extraction efficiency.
Qualitative and quantitative analysis of the extracted lipids
Nile red staining was used for the further analysis of the intracellular lipid droplets (Greenspan et al. 1985) which is shown in Fig. 16. Red dots in Fig. 16 represent the availability of lipids in the microalgal cell. As the Nannochloropsis sp., cell wall is composed of phospholipids, the red areas seem like spheres or 100% lipid which shows that the Nile red staining is not suitable for the qualitative analysis of the strains with phospholipid cell wall.
Fluorescence microscopy images of Nile red staining, a biomass before extraction (after 3 months), b biomass before extraction (4 days after reaching stationary phase), c before extraction using Bligh and Dyer method 4 days after reaching stationary phase (lower phase), d before extraction using Bligh and Dyer method (upper phase), e after extraction
Therefore, a quantitative analysis of the lipid was assessed for four samples which is shown in Table 4. As the purpose of this work is lipid extraction for the aim of biodiesel production, fatty acids (FAs) such as palmitic (C16:0), stearic (C18:0), oleic (C18:1), and linolenic acid (C18:2) which have been reported to be the most common FA in biodiesel should have the highest content in the selected microalgal strain. Nannochloropsis sp. PTCC 6016 has been reported to be desirable for biodiesel production (David et al. 2005), and as Table 4 shows, the higher amounts of fatty acids essential for biodiesel production are observable for C16:0, C18:0, C18:1, and C18:2. Comparison of oleic acid as the most element for biodiesel (Furuki et al. 2003; Mofijur et al. 2017) for the cells at stationary phase and cells after 90 days of cultivation shows that cell age improved the C18:1 by 82% in ultrasonication method, while this content was decreased for the Soxhlet method.
Modeling and prediction
To determine the most appropriate model, 28 datasets of lipid extraction of Bligh and Dyer method with various learning algorithms such as combined conjugate gradient algorithm (Traincgb), Bayesian algorithm (Trainrp), and Levenberg–Marquardt algorithm (Trainlm) were used for training MLP models (Hoskins and Himmelblau 1988; Rabbani et al. 2017; Maroofi et al. 2020). The results show that Levenberg–Marquardt with various structures and different numbers of layers and neurons is the best algorithm for simulations. According to the evaluation criteria, the best structure for the lipid extraction system includes a network with two hidden layers and 4, 10 neurons in the hidden layers. The experimental lipid extraction system was divided into three distinct sets including training (70%), validation (15%), and test data (15%) (Hoskins and Himmelblau 1988; Campbell and Ying 2011). The values of %AARE, RMSE, and R2 for the network of the lipid extraction system were 2.3042, 0.0013, and 0.98107. Adaptation between the experimental data and the MLP model predictions for the lipid extraction system for train data is given in Fig. 17
WEKA 3.7 software was used to create the SVR model. In this method, similar to the MLP method, 28 existing laboratory datasets have been used for modeling where 70% was used to teach the model and the rest 30% to test the model (Hoskins and Himmelblau 1988; Campbell and Ying 2011). The parameters γ, C, and ε were obtained by trial and error for different kernels (linear, quadratic, Gaussian, logistic). The parameters of this kernel must be obtained so that the SVR model has the most desired values for the evaluation parameters. By comparing the evaluation criteria, the best values for the parameters of the SVR model are obtained in Table 5.
The results of SVR and MLP modeling showed that the use of two models has accurate estimates of experimental results. The values of RMSE, and R2 for the MLP were, 0.035, 0.9991 and for the SVR were 0.0499 and 0.993, respectively. However, the results related to MLP are more accurate. The following table compares the modeling results with the experimental results, and the absolute error is also reported (Table 6).
As can be seen in Fig. 18, the effect of different parameters on the percentage of lipid extraction was investigated according to the modeling results. Lipid extraction increases with time reduction and increases in mixing speed. The increase in time has been somewhat effective, and after 3 s, this increase has a negative effect on lipid extraction.
Conclusion
Lipid extraction from microalgal cells is cumbersome due to its thick cell wall, and therefore, one of the well-known challenges of biodiesel production from microalgae is the development of an economical technique to obtain a high lipid extraction efficiency. To improve the lipid extraction efficiency of microalgae Nannochloropsis sp., optimization of the operational parameters for Soxhlet, Bligh and Dyer, and ultrasonication methods was accomplished in this work. In the Soxhlet method, chloroform–methanol (1:2) yielded the highest extraction of 21%. In Bligh and Dyer method, effects of biomass/solvent and solvent/solvent ratios, mixing time, mixing frequency, and pre-treatment method were investigated and optimized. In the ultrasonication method, effects of biomass to solvent ratios, solvent ratios and cavitation time, power and cycle frequency were accomplished. Finally, the effect of cell age on lipid extraction demonstrated the highest yield for all methods after 90 days with 56%, 24%, and 45% for Soxhlet, Bligh and Dyer, and ultrasonication methods, respectively. MLP neural network modeling was used by dividing the data into 70, 15, and 15 for training, validation and testing data, and the network was trained for different algorithms, the number of layers, and neurons. Using the Levenberg–Marquardt algorithm, one layer and 7 neurons with correlation coefficient value of 0.9991 and square root mean error of 0.035 were selected as the optimal network. In the SVR model, 70% of the data was used for training and 30% for testing, and various kernels and parameters such as γ, C and ε were used by trial and error. The results showed that the Gaussian kernel had the best result and the values of γ, C and ε parameters were determined with RMSE and R2 evaluation criteria of 0.0499 and 0.993, respectively. Although the highest extraction was obtained for the Soxhlet method, considering the 10 times higher solvent used in this method compared to other methods, the ultrasonication method was the most desired option for lipid extraction efficiency of Nannochloropsis sp. in large-scale operations for the purpose of biodiesel production. In this regard, the focus of the future studies should be the cost-effective biofuel production from microalgae and cyanobacteria to achieve sustainability, energy security and addressing the climate change due to high greenhouse gas emissions.
References
Abou-Shanab RAI et al (2014) Cultivation of a new microalga, Micractinium reisseri, in municipal wastewater for nutrient removal, biomass, lipid, and fatty acid production. Biotechnol Bioprocess Eng 19(3):510–518
Adholeya A, Dadhich PK (2008) Production and technology of bio-diesel: seeding a change. Prod Technol Bio-Diesel Seeding a Chang
Ahmad AL, Yasin NHM, Derek CJC, Lim JK (2011) Microalgae as a sustainable energy source for biodiesel production: a review. Renew Sustain Energy Rev 15(1). Pergamon, pp. 584–593, https://doi.org/10.1016/j.rser.2010.09.018
Anto S et al (2020) Algae as green energy reserve: Technological outlook on biofuel production. Chemosphere 242:125079
Asgharnejad H, Khorshidi Nazloo E, Madani Larijani M, Hajinajaf N, Rashidi H (2021) Comprehensive review of water management and wastewater treatment in food processing industries in the framework of water-food-environment nexus. Compr Rev Food Sci Food Saf 20(5):4779–4815
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917
Boni J, Aida S, Leila K (2018) Lipid extraction method from microalgae botryococcus braunii as raw material to make biodiesel with soxhlet extraction. J Phys Conf Ser 1095(1):12004
Breil C, Abert Vian M, Zemb T, Kunz W, Chemat F (2017) ‘Bligh and Dyer’ and Folch methods for solid–liquid–liquid extraction of lipids from microorganisms. Comprehension of solvatation mechanisms and towards substitution with alternative solvents. Int J Mol Sci 18(4):708
Campbell C, Ying Y (2011) Learning with support vector machines. Synth Lect Artif Intell Mach Learn 5(1):1–95
Cancela A, Maceiras R, Alfonsin V, Sanchez A (2017) A study on techniques for microalgae separation and lipid extraction for Desmodesmus subspicatus. Int J Environ Res 11(3):387–394
Chatsungnoen T, Chisti Y (2016) Optimization of oil extraction from Nannochloropsis salina biomass paste. Algal Res 15:100–109. https://doi.org/10.1016/j.algal.2016.02.015
Chisti Y (2007) Biodiesel from microalgae. Biotechnol Adv 25(3):294–306. https://doi.org/10.1016/j.biotechadv.2007.02.001
Chowdhury H, Loganathan B (2019) Third-generation biofuels from microalgae: a review. Curr Opin Green Sustain Chem 20:39–44
Corrêa PS, Morais Júnior WG, Martins AA, Caetano NS, Mata TM (2021) Microalgae biomolecules: Extraction, separation and purification methods. Processes 9(1):10
David F, Sandra P, Vickers AK (2005) “Column selection for the analysis of fatty acid methyl esters. Food Anal Appl Palo Alto CA Agil Technol 19:19
Demirbas A (2009) Biofuels securing the planet’s future energy needs. Energy Convers Manag 50(9):2239–2249. https://doi.org/10.1016/j.enconman.2009.05.010
Ellison CR, Overa S, Boldor D (2019) Central composite design parameterization of microalgae/cyanobacteria co-culture pretreatment for enhanced lipid extraction using an external clamp-on ultrasonic transducer. Ultrason Sonochem 51:496–503
Engelbrecht AP (2007) Computational intelligence: an introduction. Wiley, New York
Fallahi A, Hajinajaf N, Tavakoli O, Sarrafzadeh MH (2020) Cultivation of mixed microalgae using municipal wastewater: biomass productivity, nutrient removal, and biochemical content. Iranian J Biotech 18(4):88–97
Fallahi A, Hajinajaf N, Tavakoli O, Mehrabadi A (2021) Effects of Simultaneous CO2 addition and Biomass Recycling on Growth Characteristics of Microalgal Mixed Culture. J Chem Technol Biotechnol
Fallahi A, Rezvani F, Asgharnejad H, Khorshidi E, Hajinajaf N, Higgins B (2021) Interactions of microalgae-bacteria consortia for nutrient removal from wastewater: a review. Chemosphere 129878
Figueiredo ARP, da Costa E, Silva J, Domingues MR, Domingues P (2019) The effects of different extraction methods of lipids from Nannochloropsis oceanica on the contents of omega-3 fatty acids. Algal Res 41:101556. https://doi.org/10.1016/j.algal.2019.101556
Furuki T et al (2003) Rapid and selective extraction of phycocyanin from Spirulina platensis with ultrasonic cell disruption. J Appl Phycol 15(4):319–324. https://doi.org/10.1023/A:1025118516888
Greenspan P, Mayer EP, Fowler SD (1985) Nile red: a selective fluorescent stain for intracellular lipid droplets. J Cell Biol 100(3):965–973
Hagan MT, Menhaj MB (1994) Training feedforward networks with the Marquardt algorithm. IEEE Trans Neural Networks 5(6):989–993
Hajinajaf N, Mehrabadi A, Tavakoli O (2021) Practical strategies to improve harvestable biomass energy yield in microalgal culture: A review. Biomass Bioenerg 145:105941. https://doi.org/10.1016/j.biombioe.2020.105941
Heinrich M, Barnes J, Gibbons S, Williamson E (2012) Production, standardization and quality control. Fundam. Pharmacogn. Phyther. (2nd Ed. Churchill Livingstone Elsevier
Hill J, Nelson E, Tilman D, Polasky S, Tiffany D (2006) Environmental, economic, and energetic costs and benefits of biodiesel and ethanol biofuels. Proc Natl Acad Sci U S A 103(30):11206–11210. https://doi.org/10.1073/pnas.0604600103
Hoskins JC, Himmelblau DM (1988) Artificial neural network models of knowledge representation in chemical engineering. Comput Chem Eng 12(9–10):881–890
Jensen SK (2008) Improved Bligh and Dyer extraction procedure. Lipid Technol 20(12):280–281
Khoo KS et al (2020) Recent advances in downstream processing of microalgae lipid recovery for biofuel production. Bioresour Technol 304:122996
Kumar A, Ergas S, Yuan X, Sahu A, … Q. Z.-T. in, and undefined 2010, “Enhanced CO2 fixation and biofuel production via microalgae: recent developments and future directions. Elsevier, Accessed: Dec. 09, 2018. [Online]. Available: https://www.sciencedirect.com/science/article/pii/S0167779910000715
Kumar V, Arora N, Nanda M, Pruthi V (2019) Different cell disruption and lipid extraction methods from microalgae for biodiesel production. In: Microalgae biotechnology for development of biofuel and wastewater treatment, Springer, pp 265–292
Kwon MH, Yeom SH (2015) Optimization of one-step extraction and transesterification process for biodiesel production from the marine microalga Nannochloropsis sp. KMMCC 290 cultivated in a raceway pond. Biotechnol Bioprocess Eng 20(2):276–283
Lee MJ, Chen JT (1993) Fluid property predictions with the aid of neural networks. Ind Eng Chem Res 32(5):995–997
Lee JY, Yoo C, Jun SY, Ahn CY, Oh HM (2010) Comparison of several methods for effective lipid extraction from microalgae. Biores Technol 101(1 SUPPL.):S75–S77. https://doi.org/10.1016/j.biortech.2009.03.058
Lee SJ, Yoon BD, Oh HM (1998) Rapid method for the determination of lipid from the green alga Botryococcus braunii. Biotechnol Tech 12(7):553–556. https://doi.org/10.1023/A:1008811716448
Li Y et al (2014) A comparative study: the impact of different lipid extraction methods on current microalgal lipid research. Microb Cell Fact 13(1):1–9
Maggio G, Cacciola G (2012) When will oil, natural gas, and coal peak? Fuel 98:111–123. https://doi.org/10.1016/j.fuel.2012.03.021
Maroofi J, Hashemabadi SH, Rabbani Y (2020) Investigation of the chain formation effect on thermal conductivity of magnetorheological fluids. J Thermophys Heat Transf 34(1):3–12
Mason TJ, Lorimer JP (1989) An introduction to sonochemistry. Endeavour 13(3):123–128. https://doi.org/10.1016/0160-9327(89)90086-0
Mercer P, Armenta RE (2011) Developments in oil extraction from microalgae. Eur J Lipid Sci Technol 113(5):539–547. https://doi.org/10.1002/ejlt.201000455
Moazami N, Ranjbar R, Ashori A, Tangestani M, Nejad AS (2011) Biomass and lipid productivities of marine microalgae isolated from the Persian Gulf and the Qeshm Island. Biomass Bioenergy 35(5):1935–1939
Mofijur M, Rasul MG, Hassan NMS, Masjuki HH, Kalam MA, Mahmudul HM (2017) Assessment of physical, chemical, and tribological properties of different biodiesel fuels. Clean Energy Sustain Develop, 441–463
Nagappan S, Devendran S, Tsai P-C, Dinakaran S, Dahms H-U, Ponnusamy VK (2019) Passive cell disruption lipid extraction methods of microalgae for biofuel production–a review. Fuel 252:699–709
Patil V, Källqvist T, Olsen E, Vogt G, Gislerød HR (2007) Fatty acid composition of 12 microalgae for possible use in aquaculture feed. Aquac Int 15(1):1–9. https://doi.org/10.1007/s10499-006-9060-3
Pernet F, Tremblay R (2003) Effect of ultrasonication and grinding on the determination of lipid class content of microalgae harvested on filters. Lipids 38(11):1191–1195. https://doi.org/10.1007/s11745-003-1178-6
Rabbani Y, Shariaty-Niassar M, Ebrahimi SAS (2021) The effect of superhydrophobicity of prickly shape carbonyl iron particles on the oil-water adsorption. Ceram Int 47(20):28400–28410
Rabbani Y, Shirvani M, Hashemabadi SH, Keshavarz M (2017) Application of artificial neural networks and support vector regression modeling in prediction of magnetorheological fluid rheometery. Colloids Surfaces A Physicochem Eng Asp 520:268–278. https://doi.org/10.1016/J.COLSURFA.2017.01.081
Rabbani Y, Shariaty-Niassar M, Seyyedebrahimi SA (2020) An investigation of the effects of dopamine on hydrophobicity of carbonyl iron particles with stearic acid. Iran J Chem Eng 17(4)
Ramluckan K, Moodley KG, Bux F (2014) An evaluation of the efficacy of using selected solvents for the extraction of lipids from algal biomass by the soxhlet extraction method. Fuel 116:103–108. https://doi.org/10.1016/j.fuel.2013.07.118
Ranjan A, Patil C, Moholkar VS (2010) Mechanistic assessment of microalgal lipid extraction. Ind Eng Chem Res 49(6):2979–2985. https://doi.org/10.1021/ie9016557
Ranjith Kumar R, Hanumantha Rao P, Arumugam M (2015) Lipid extraction methods from microalgae: a comprehensive review. Front Energy Res 2:61
Rodolfi L et al (2009) Microalgae for oil: Strain selection, induction of lipid synthesis and outdoor mass cultivation in a low-cost photobioreactor. Biotechnol Bioeng 102(1):100–112. https://doi.org/10.1002/bit.22033
Rumin J et al (2015) The use of fluorescent Nile red and BODIPY for lipid measurement in microalgae. Biotechnol Biofuels 8(1):1–16
Ryckebosch E, Muylaert K, Foubert I (2012) “Optimization of an analytical procedure for extraction of lipids from microalgae. JAOCS J Am Oil Chem Soc 89(2):189–198. https://doi.org/10.1007/s11746-011-1903-z
Scott SA et al (2010) Biodiesel from algae: Challenges and prospects. Current Opin Biotechnol 21(3):277–286. https://doi.org/10.1016/j.copbio.2010.03.005
Shanmugam S, Mathimani T, Anto S, Sudhakar MP, Kumar SS, Pugazhendhi A (2020) Cell density, Lipidomic profile, and fatty acid characterization as selection criteria in bioprospecting of microalgae and cyanobacterium for biodiesel production. Bioresour Technol 304:123061
Sharma YC, Singh B, Upadhyay SN (2008) Advancements in development and characterization of biodiesel: a review. Fuel 87(12):2355–2373. https://doi.org/10.1016/j.fuel.2008.01.014
Sivaramakrishnan R, Incharoensakdi A (2018) Microalgae as feedstock for biodiesel production under ultrasound treatment–A review. Bioresour Technol 250:877–887
Stanley GHS, Folch J, Lees M (1957) A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem 226(1):497–509
Thawechai T, Cheirsilp B, Louhasakul Y, Boonsawang P, Prasertsan P (2016) Mitigation of carbon dioxide by oleaginous microalgae for lipids and pigments production: effect of light illumination and carbon dioxide feeding strategies. Bioresour Technol 219:139–149
Wahidin S, Idris A, Shaleh SRM (2013) The influence of light intensity and photoperiod on the growth and lipid content of microalgae Nannochloropsis sp. Bioresour Technol 129:7–11. https://doi.org/10.1016/j.biortech.2012.11.032
Wang L, Weller CL (2006) Recent advances in extraction of nutraceuticals from plants. Trends Food Sci Technol 17(6):300–312. https://doi.org/10.1016/j.tifs.2005.12.004
Xiao Y, Zhang J, Cui J, Feng Y, Cui Q (2013) Metabolic profiles of Nannochloropsis oceanica IMET1 under nitrogen-deficiency stress. Bioresour Technol 130:731–738. https://doi.org/10.1016/j.biortech.2012.11.116
Yadav G, Dash SK, Sen R (2019) A biorefinery for valorization of industrial waste-water and flue gas by microalgae for waste mitigation, carbon-dioxide sequestration and algal biomass production. Sci Total Environ 688:129–135
Acknowledgements
The authors would like to thank the technical and analytical support of both Green Technology Laboratory (School of Chemical Engineering, University of Tehran) and Department of Biotechnology of IROST (Iranian Research Organization for Science and Technology).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no financial or commercial conflict of interest.
Additional information
Editorial responsibility: Senthil Kumar Ponnusamy.
Rights and permissions
About this article
Cite this article
Hajinajaf, N., Rabbani, Y., Mehrabadi, A. et al. Experimental and modeling assessment of large-scale cultivation of microalgae Nannochloropsis sp. PTCC 6016 to reach high efficiency lipid extraction. Int. J. Environ. Sci. Technol. 19, 5511–5528 (2022). https://doi.org/10.1007/s13762-021-03760-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-021-03760-x