Abstract
The present study evaluated the effect of different pretreatments on unlocking the lignocellulosic composition of pine needle litter biomass followed by performance under aerobic/anaerobic digestion. The pretreatment strategies employed were milling, steam explosion, and chemical treatment with acid–base–acid. The results revealed that the most effective method of pretreatment of pine needle litter was the physico-chemical treatment (milling + steam treatment + acid–base–acid hydrolysis), which resulted in 65.92% increase in holocellulose content with a corresponding 21.34% decrease in total lignin content as compared to untreated control. The effect of pretreatments on anaerobic digestion using digested sludge as inoculums was evaluated through biochemical methane generation. During the liquid-phase anaerobic digestion process of different pretreated pine needle litter, the mechanically milled pine litter yielded a methane level of 5.53 ± 0.11 mmol CH4/g VSin in 40 days, which was 40.7% higher than during anaerobic digestion of untreated pine needle litter. The physico-chemical pretreatment resulted in 21.4% higher methane levels as compared to untreated pine litter. Additionally, the aerobic digestion of pretreated pine needle litter with white rot fungal strain, Cotylidia pannosa for 60 days at 30 °C, resulted in 95.5% decrease in holocellulose content and 52.3% decrease in total lignin content. Findings in the present study suggest that in spite of significant reduction in lignin content during the pretreatment, the addition of pine litter as a substrate inhibits anaerobic digestion process.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Lignocellulosic complexities and autochthonous microflora associated with pine needle litter account for extremely slow rates of natural degradation of biomass on the forest floors (Mahajan et al. 2016). As a result of its accumulation, it has long been considered as an unutilized biomass (Tsavkelova et al. 2012; Azman et al. 2015). However, the recent scientific breakthroughs, especially in the last decade, have resulted in paradigm shift in thinking and action, especially in forest residue management policies world over, whereby the forest biomass is now considered as potential feedstock for meeting global renewable bioenergy needs (Paredes-Sánchez et al. 2016).
The Himalayan subtropical Pine forests are the largest in the Indo-Pacific region and are widely spread in different regions of India with Chir pine (Pinus roxburghii), as the dominant species. In India, the total area under Chir forests is estimated to be 8,90,000 hectares and occurs between 450 and 2300 m altitude. Considering the Indian scenario, estimates suggest that biomass production from agricultural and forest residues accounts for about 565 million tonnes per year. An estimated 189 million tones of this biomass remains unutilized, which, if used judiciously, can result in renewable power that in turn can sustain up to 25 GW of installed capacity of power generation (Joshi et al. 2016).
The present study focuses on the effective utilization of pine forest needle residue, which can significantly contribute to sustainable forest litter management. The pine needle residue used in the present study was selected because of the environmental concerns related to its excessive accumulation on forest floors, which often becomes a reason of extensive ground fires, especially near settlements as reported in a published report from the Ministry of Renewable Energy, Government of India (Jain and Srinivas 2012). Under natural conditions, pine litter decomposes very slowly because of the release of allelochemicals; there is a significant hindrance in the establishment of other plant species in forest ecosystem. The chemical composition of pine litter of different coniferous species varies, but its main components are lignin, cellulose, and hemicellulose. As a substrate for the microbiological processes, pine litter is characterized by the presence of a large number of essential oils, resin acids, tannins, phenolic and terpene compounds, and a high acidity (pH 4–5) (Isidorov et al. 2010; Lacourt 2011). The tannins, terpenes, resin acids, and products of their hydrolysis have a significant toxic effect on the aerobic and anaerobic microorganisms (Kayembe et al. 2013). The high amount of aromatic compounds complicates the treatment of pine litter and limits the applicability of the standard pretreatment and further use of litter biomass as a substrate for microbial processes (Isidorov et al. 2010; Lacourt 2011).
The anaerobic digestion of pine needles without any pretreatment resulted in lower biogas yield (Brown et al. 2012); therefore, this study primarily evaluated the effect of different pretreatments on substrate availability followed by its performance in anaerobic digestion. The findings in the present manuscript, under the joint research initiative from Government of India and RFBR (Russian Foundation for of Basic Research), are primarily aimed at developing microbial strategies for unlocking the hidden potential of lignocellulosic-rich forest residue for conversion of biomass to renewable bioenergy (biogas). The present study was conducted in two phases: (a) evaluation of different pretreatment strategies to unlock the lignocellulosic complexities in the pine needle and (b) evaluation of pretreated pine needle as substrate under aerobic solid-state fermentation and anaerobic digestion.
The study was carried out jointly at Department of Biotechnology and Bioinformatics, Jaypee University of Information Technology, Waknaghat, Solan, India (June 2014–December 2015) and Winogradsky Institute of Microbiology, Research Center of Biotechnology of the Russian Academy of Sciences, Moscow, Russia (November 2014–January 2016).
Materials and methods
Lignocellulosic substrate
The study was conducted in subtropical Kandaghat pine forest range, one of the five forest ranges of Solan forest Division of Himachal Pradesh, known for frequent forest fires, where pine needles being the primary cause, especially during summers. Naturally dried pine needle litter of Pinus roxburghii was used as a substrate for microbial degradation. The pine litter was collected during the summer in the area of Kandaghat (India, 31°00′57″N; 77°04′75 17 ″E).
Pretreatment of pine needles
The overall pretreatment procedure was divided into three stages (“Stage I”: milling pretreatment; “Stage II”: stream explosion pretreatment; and “Stage III”: acid–base–acid pretreatment process), and changes (percent increase/decrease) in lignin and holocellulose content of pine needle litter were monitored.
The collected pine needle litter was milled using a blender and then sieved through a sieve No 20 (skip particle size ≤ 0.85 mm). The mechanically milled litter was treated by steam explosion involving the treatment of pine needle aliquots of 250 g at 121 °C for 30 min. The physically pretreated substrate was treated with 1N HCl for 30 min at 70 °C, followed by treatment with 1N NaOH, for 30 min at 70 °C. The step with NaOH treatment was repeated until a clear solution was obtained. The substrate was once again treated with 1N HCl for 30 min at 70 °C followed by repeated washings with water (10 min at 70 °C) until pH reached neutrality (Demirbas 2007) and as reviewed by Jonsson and Martín (2016).
Holocellulose and lignin estimations
In order to evaluate the effectiveness of pretreatment methodologies, pine needles were evaluated for holocellulose content as described in TNI-A-9 test method; laboratory manual of CPPRI (TAPPI Standard Methods 2009) and lignin content: acid-soluble lignin (ASL) and acid-insoluble lignin (AIL) as described in method T222 of the Technical Association of the Pulp and Paper Industry (TAPPI, Atlanta, GA, USA). The total lignin content in a sample was expressed as the sum of ASL and AIL and calculated in mg/g of dry weight or in %. The holocellulose content was expressed as a percentage per gram of dry weight (DW) (ASTM D1104-56 1978; Johnson et al. 1961; Tappi 2002).
Anaerobic liquid-phase digestion
To evaluate the impact of pine needle litter on anaerobic digestion, the untreated pine needle litter (control) along with the pretreated pine needles was used as substrate. Pretreated substrate samples were mixed with distilled water in the ratio 1:5–1:10 (on the basis of weight) to achieve moisture up to 93–94%. Food waste (FW) was thoroughly blended and used as a co-substrate for anaerobic digestion. A thermophilically digested sewage sludge (DSS) previously used for the digestion of the organic fraction of municipal solid waste (OFMSW) and FW (Nikitina et al. 2015) was used as inoculum.
The experiments were carried out in glass bottles (500 mL) containing 100 mL volume of digesting mixture, which were tightly closed with rubber stoppers and aluminum caps. The inoculum to substrate ratio (ISR) of 1:1 was selected based on the total organic content of the mixture. On addition of FW as co-substrate, the ratio of inoculum:pine needle litter:food waste was 2:1:1. The bottles were flushed with argon gas to create anaerobic conditions, and incubation was carried out at 50 °C for 60 days (Nikitina et al. 2015). The composition of gas phase (CH4, H2, CO2), the amount of volatile fatty acids (VFA) in the liquid phase, pH, dry weight (DW), and volatile solids (VS) content, and humidity of mixtures were measured regularly in accordance with the procedures described earlier (Nikitina et al. 2015). The author’s modification of the method of preparation of samples for measurement of VFA by gas chromatography was created in order to prevent contamination of sites of chromatograph with products of the pine litter degradation, which adversely affect the reproducibility of the analysis results. Samples for VFA analysis were centrifuged at 10,000 rpm for 5–10 min, and then, the supernatant was transferred to a clean test tube and acidified with 10% solution of H3PO4 up to pH of 1.0–2.0. The absorbents added were: 0.01 g of aerosil (SiO2, amorphous silica with a specific surface area of 300 m2/g) and 0.01 g of silica gel (high-dispersive powder, mSiO2*nH2O) in 0.2 mL of sample and vortexed. The obtained suspension was centrifuged at 10,000 rpm for 5 min, after which the supernatant was transferred to a clean tube. It was shown that aerosil and silica gel do not absorb VFA and alcohols (C1–C5), and therefore, there was no adverse effect on the accuracy of the analysis.
Aerobic solid-state co-digestion of pine needle litter and digested sludge
To increase the efficiency of degradation, aerobic solid-state co-digestion was set up using pretreated pine needle litter and digested sludge (DS) from thermophilic bioreactor, digesting the mixture of organic fraction of municipal solid waste (OFMSW) and sewage sludge. For inoculum in solid substrate cultivation (SSC), a mixed suspension (1:1) of soil (from a composting pile in a farm yard) and dry mature, industrial compost [produced from a mixture of pine sawdust and solid substrate (SS) (Lipetsk, Russia)] was used. Inoculum was preactivated with addition of cardboard (1.5%) and NH4Cl (0.25%) and incubated at 30 ± 2 °C for a duration of 10 days (Nikitina et al. 2015). The pine needle litter was mixed with digested sludge in the ratio of 2:3 by weight (6:1 depending upon VS), and then, 20–25 mL of inoculum was added. The nitrogen was added as a solution of ammonium chloride, in the quantity required for optimum ratio of C/N = 25–30/1, based on the theoretical results that C/N ratio of pine litter is 80/1, and C/N ratio of digested sludge is 10/1 (Hubbe et al. 2010; Kuhad et al. 2011). The initial moisture of the mixtures was kept at 67–70%. The experiment was carried out at a temperature of 30 ± 2 °C for a period of 60 days. During the experiment, the distilled water was added regularly to maintain a humidity of mixtures up to 65–75%, and the mixtures were stirred to improve mass transfer and aeration.
Aerobic solid-state fermentation of pine needle litter by Cotylidia pannosa
The culture of white rot fungus Cotylidia pannosa F6 isolated from soil samples of the Himalayas, having lignolytic and cellulolytic activity (Sharma et al. 2015), was used as an inoculum. The culture of C. pannosa was grown on potato dextrose agar at 30 °C until full sporulation and then washed with distilled water. The resulting suspension was used as inoculum at a spore concentration of about 3 × 108 spores/mL. Experiments were performed in flat-bottomed 250-mL flasks with cotton plugs according to the procedure described Sharma et al. (2015). The pretreated pine needle litter was moistened with buffer solution containing (g/L): (NH4)2SO4, 10.0; KH2PO4, 3.0; MgSO4*7H2O, 0.5; CaCl2*H2O, 0.5 and then mixed with the digested sludge (2:3 by weight), and inoculum was applied (ISR 1:1 by weight). The initial moisture content of the experimental mixtures was 60%. The experiment was carried out for 60 days at a temperature of 30 ± 2 °C.
Results and discussion
Pretreatment of pine needle litter and evaluation of the methodology for improved substrate availability
The lignocellulosic complexities associated with pine needle litter have been previously discussed and reported from our laboratory (Mahajan et al. 2016). The extraordinarily resistant nature of lignocellulose to biodegradation is often attributed to the lignin, which exists in a highly polymerized state; therefore, extensive pretreatment strategies, viz. mechanical grinding, chemical pulping, and physico-chemical processes like steam treatment in the presence of alkali or acid, are required in order for the cellulose and hemicelluloses to be broken down. In the present study, therefore, a comparative evaluation of the pretreatment strategies for pine needle litter was carried out. All of the pretreatment methods were tested under mild conditions to avoid the formation of the toxic intermediates due to the presence of aromatic compounds in pine needles. The application of higher temperature and harsh chemical treatments may result in the production of other metabolites having inhibitory effect on microorganisms (Cheng et al. 2011; Lacourt 2011).
Obtained results demonstrate “Stage I” of pretreatment, i.e., milling itself is a rather effective and simple way of pretreatment and promotes remarkable release of cellulose from the lignocellulosic fibers; a 84.82% increase in holocellulose content was observed with a corresponding 0.32% decrease in total lignin content over untreated pine needle litter. “Stage II” of the pretreatment, which involved additional stream explosion to the milled pine needle litter, resulted in 5.09% increase in holocellulose content and a 4.46% decrease in total lignin content over the “Stage I” pretreated substrate. Implementation of “Stage III” pretreatment process, i.e., the use of acid–base–acid treatment on “Stage II” pretreated substrate, resulted in 65.92% increase in holocellulose content with a corresponding 21.34% decrease in total lignin content over the holocellulose and lignin content of “Stage II” pretreated substrate.
According to previously documented reports, milling allows significant increase in surface area of lignocellulosic biomass, thereby making it more accessible to enzymes and microorganisms. The milling-based pretreatment also decreases the crystallinity of the cellulose without loss of low molecular organic substrates and use of toxic and expensive reagents (Lin et al. 2010). Milling process promotes facilitation of mass transfer and increase in the effectiveness of enzyme adsorption (Azman et al. 2015). Mechanical milling of lignocellulosic wastes promotes increase in hydrolysis rate to 5–25% and biogas production to 10–60%; therefore, many researches consider that this method of pretreatment is sufficient for the subsequent aerobic treatment of pine needle litter (Hubbe et al. 2010; Kuhad et al. 2011).
Pretreatment of lignocellulosic biomass with hot steam promotes the release of hemicellulose and their partial hydrolysis, making cellulose more accessible to subsequent utilization. It also improves adsorption of water by the substrate, thereby increasing its accessibility to microorganisms (Elliston et al. 2015). Previously documented reports by Silva et al. (2012), in their study, suggested that pretreatment of milled wheat straw with hot steam leads to 40% increase in hydrolysis rate. According to Liu et al. (2002), hot steam treatment of lignocellulosic fraction of OFMSW under pressure helps to increase methane formation to 40%. In the present study, the results were not much encouraging when compared to the results obtained using only mechanical milling. The findings are in corroboration with earlier reports of Lawther et al. (1996), who demonstrated that prolonged (6 h) hot steam treatment of wheat straw at low pressure did not cause significant increase in anaerobic digestion process of the feedstock. There are also reports on 3–12% loss in VS when this method of pretreatment was used (Elliston et al. 2015).
In the present study, the most effective method of pretreatment of pine needle litter was the physico-chemical treatment (milling + steam explosion + acid–base–acid hydrolysis). Obtained data correlate with the results of Lin et al. (2010), who demonstrated that up to 38% of total lignin was removed due to chemical pretreatment of rice straw. However, application of this method has a number of limitations: loss of organics, necessity of subsequent washing of the treated feedstock, and implementation of corrosion-resistant equipment. Also, the reagents that are used in this method are expensive and require further utilization (Lin et al. 2010). Kim et al. (2013) showed that 14–34% of the biomass is lost due to washing after chemical pretreatment.
Aromatic substances and lignin derivatives, which have inhibitory effect on the microorganisms, may be removed using various methods of detoxification (Jonsson and Martín 2016). However, carrying out the procedure of detoxification requires implementation of additional steps in the technical plan, and often, it is an expensive and energy-intensive process. In addition, the effect of detoxification on the anaerobic digestion is poorly understood (Matsakas et al. 2015). Therefore, the search of active microbial agents that can increase the bioavailability of cellulose in lignin-containing waste without the formation of inhibitory substances or degrade produced toxicants remains an urgent task.
Liquid-phase anaerobic digestion of the pine needle litter
Organic fraction of municipal solid waste is known to contain up to 40–53% of cellulose, 9–12% of hemicelluloses, and 10–17% of lignin per DW (Staley et al. 2012). Therefore, using digested sludge (DS) as inoculum for anaerobic digestion was hypothesized to be a suitable strategy. It has been shown that DS contains consortium of anaerobic microorganisms, necessary for the decomposition of lignocellulose-containing substrates (Tsavkelova et al. 2012; Nikitina et al. 2015). Therefore, in the present study, DS was used as inoculum for initiation of thermophilic anaerobic digestion of untreated and pretreated pine needle litter. Findings presented in Table 1 reveal that 60 days after the experimental setup, significantly lower amount of lignin was biologically degraded irrespective of the nature of the pretreated substrate. Digestion mixtures of DS and milled pine needles after digestion process resulted in 5.9% lignin reduction. It is important to note that degradation of acid-soluble fraction of lignin was observed, while acid-insoluble lignin was not degraded. These values for reduction in lignin content were further reduced to 2.9% when food waste was added into the digestion mixture. Similar observations were recorded when physicochemically pretreated substrate was used (Table 1). A lower decrease in lignin content when food wastes were added was probably due to the presence of a high number of more preferred organic substrates for microorganisms.
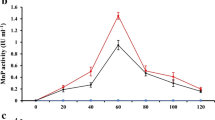
The overall anaerobic digestion process (in terms of methane yields) also seemed to have not worked effectively when pine needles were added to the digestion mixture. Anaerobic digestion of untreated pine needle litter (control) during 40 days resulted in the formation of 3.93 ± 0.08 mmol CH4/g VSin. Only 0.99 ± 0.02 mmol CH4/g VSin or 22.2 L CH4/kg VSin was formed from VS of the litter. These results demonstrate that untreated pine litter has a rather low methane potential (22.2 L CH4/kg VS), as compared to mechanically milled pine pulp (21–28 L CH4/kg VS) (Steffen et al. 2016) and birch sawdust (17.5 L CH4/kg VS) (Matsakas et al. 2015). Previously documented reports reveal that methane yields in other lignin-containing wastes are remarkably higher and reach 217–360 L CH4/kg VS for rice straw, 290–336 L CH4/kg VS for wheat straw, 375 L CH4/kg VS for pine pulp after chemical hydrolysis, and 443 L CH4/kg VS for pig manure (Cu et al. 2015). Mechanical milling was more effective from the point of CH4 yield and methanogenesis rate than complex physico-chemical pretreatment (Fig. 1, Table 1).
Accumulation of methane during anaerobic digestion of pine litter per gram of initial volatile solids (mmol CH4/g VSin): 1—control-digested sludge, 2—control-untreated pine litter + digested sludge (1:1 on VS), 3—milled pine litter + digested sludge (1:1 on VS), 4—physicochemically treated pine litter + digested sludge (1:1 on VS). The error of the calculations was 0.01–0.25 mmol/g VS
During the anaerobic digestion process of different pretreated pine needle litter, the mechanically milled pine litter along with DS yielded a methane level of 5.53 ± 0.11 mmol CH4/g VSin in 40 days, which was 40.7% higher than during the digestion of untreated pine needle litter. The physico-chemical pretreatment resulted in 21.4% higher methane levels as compared to untreated pine litter. The results indicate that the major contribution toward methane generation in both cases is from the organic fraction of pretreated pine litter contributing about 4.19 ± 0.08 mmol CH4/g VSin and 2.67 ± 0.05 mmol CH4/g VSin, respectively. In both cases, the maximum accumulation of VFA (acetate, propionate, and butyrate) was observed on day 6 of incubation; the acetate and butyrate were actively consumed by anaerobic microorganisms; and propionate was accumulating. There was a slight accumulation of hydrogen (0.1–0.2 vol%). Acidification of digesting mixtures was not observed; however, the pH level increased during the experiment from 6.0–6.5 to 7.5 units.
In the present study, addition of the pine needle litter in ratio 1:1 inoculum adversely affected methanogenesis. Methane yield and the percent of utilized organic matter were higher in case of AD of milled pine litter than in case of digestion of physicochemically treated litter, indicating the absence of the positive effect of the complex pretreatment on the anaerobic digestion of the pine litter. The possible reasons could be the loss of available organics (Kim et al. 2013; Elliston et al. 2015) and formation intermediates, toxic for anaerobic microorganisms during physico-chemical pretreatment (Cheng et al. 2011). This is further confirmed by the comparison of dynamics and rates of methane formation in experimental variants and control (DS without litter). It can be seen from the Fig. 1 and Table 1 that yield and formation rate of methane are lower in experimental variants indicating the negative effect of the litter content on methanogenic community of DS. Obtained results correlate with the results of Lacourt (2011), who reported the negative effect of pine litter on anaerobic process. Brown et al. (2012) reported lower methane yields of 2.5 L/kg VS in anaerobic digestion experimentation with pine for 30 days. Previously documented literature shows that the toxic effect of aromatic compounds and products of the partial hydrolysis of lignin is the main reason of inhibition of anaerobic digestion (Cheng et al. 2009).
During the anaerobic co-digestion of pine litter with food waste, the total yield of methane increased only due to the digestion of food waste as the more accessible substrate for microorganisms of inoculum (Fig. 2, Table 1). Unlike digestion of pine needle litter, co-fermentation with food waste smoothened the difference between the applied pretreatment methods. Additionally, the food waste also possesses high organic matter, appropriate moisture content, and volatile solids (> 95%) rendering it easily biodegradable (An et al. 2012). Dynamics and rates of methane formation at co-digestion of both milled litter and litter after complex pretreatment did not vary significantly. Methane yield on the fortieth day reached 7.88 ± 0.25 mmol/g VSin, regardless of the method of pretreatment. Some authors have suggested that the low efficiency of the anaerobic degradation of lignocellulosic feedstock may be associated with unsuitable inoculum. When comparing the yield of methane in the thermophilic AD of cellulosic feedstock, depending on the inoculum used, it was shown that the microbial community taken from cattle manure is the most effective inoculum (Tsavkelova et al. 2012). Of great interest from the point of view of future research initiatives is the search of active lignolytic and cellulolytic anaerobic microorganisms, especially from the gastrointestinal tract of wild ruminants, such as moose and beavers (Lacourt 2011). Using anaerobic fungi of genera Neocallimastix, Piromyces, Ontomyces, and other representatives of the family Neocallimastigaceae, together with the bacteria, could also increase the production of biogas, since micromycetes due to their mycelial growth and large enzymatic complexes can efficiently break down lignocellulose structure, thus making it more accessible to bacteria (Cheng et al. 2009; Dollhofer et al. 2015). To test this hypothesis, we further carried out experiments using cow dung and digested sludge of bioreactor digesting manure, cattle litter, and food waste as inoculum. However, the results obtained (data not shown) indicated the inhibition of anaerobic digestion of the pine litter regardless of the inoculum used.
Accumulation of methane during anaerobic co-digestion of pine litter and food waste per gram of initial volatile solids (mmol CH4/g VSin): 1—control-digested sludge, 2—food waste + digested sludge (1:1 on VS), 3—milled pine litter + food waste + digested sludge (1:1:2 on VS), 4—physicochemically treated pine litter + food waste + digested sludge (1:1:2 on VS). The error of the calculations was 0.01–0.48 mmol/g VS
Aerobic co-fermentation of the pine litter and digested sludge
Aerobic fermentation is a reliable way to manage lignocellulosic waste and provides a bio-humus as a useful product. To optimize and intensify the process of aerobic treatment of organic wastes is necessary to maintain a favorable ratio of C/N in the substrate mixture (25–30/1) and the humidity not lower than 40%. The lignocellulosic wastes are characterized by a high C/N ratio that makes them difficult to recycle (Hubbe et al. 2010; Kuhad et al. 2011). Addition of other organic wastes with low C/N ratio, for example, DS, waste of meat, and dairy industry, is reported to improve the rate and efficiency of degradation of lignocellulosic waste (Kuhad et al. 2011; Nikitina et al. 2015) and increase humidity of substrate mixture.
In the present study, experiments on aerobic fermentation revealed that in samples containing a mixture of mechanically milled pine litter or the litter after complex pretreatment and DS, the removal of VS in 30 days reached 14.5 and 13.3%, respectively. This was 2–3 times lower than in the mixtures without addition of sludge. Thus, the addition of sludge stimulated the removal of VS, but despite this, the overall process of degradation of the pine needle litter was extremely slow. The findings, therefore, indicated the use of a specialized active inoculum that could possibly speed up the process. Based on the leads from this experiment, we explored the potential of fungi C. pannosa strain F6 in degrading pine needle litter. The results are discussed in the following section.
Aerobic solid-state fermentation of the pine needle litter using culture of white rot fungi Cotylidia pannosa
The efficiency of decomposition of recalcitrant lignocellulosic waste under aerobic conditions could be enhanced by the application of suitable culture (or consortia) of aerobic microorganisms with high lignolytic and cellulolytic potential. Recently, a strain of basidial micromycetes, white rot fungi C. pannosa F6, has been reported from our group as a potential producer of lignocellulolytic enzymes involved in the decomposition of lignin. This laccase, producing strain, does not lose its activity at low pH (Sharma et al. 2015), which is extremely important in the processing of substrates such as pine litter, because they have a high acidity. In addition, the strain C. pannosa F6 is indigenous, as it was isolated from forest soil of the Himalayas. The efficiency of using of culture C. pannosa F6 as inoculum was evaluated in experiments on aerobic solid-state fermentation of the pretreated pine needle litter and has been documented in Table 2.
Perusal of data appended in Table 2 indicates ability of the C. pannosa strain F6 to effectively degrade pine needle litter because of its high lignolytic and cellulolytic activity (Sharma et al. 2015). Pretreatment of the pine needle litter resulted in better availability of cellulosic and lignocellulosic components of the pine needle for degradation. The physicochemically treated pine needle litter was the most biodegradable substrate. Out of the 58.17% available holocellulose after treatment with C. pannosa strain F6, only 2.57% remained unutilized after 60 days, thus resulting in 95.58% decrease in holocellulose content. The physico-chemical pretreatment resulted in the separation of 37.33% lignin from the pine needle. At end of 60 days of experimentation, C. pannosa strain F6 was able to reduce this available lignin to 17.80%, thus accounting for 52.32% decrease in total lignin content. As representatives of other species of micromycetes, agents of white rot are also capable for efficient decomposition of different plant waste. Zhi and Wang (2014) showed that 28.5% of lignin was removed after inoculation of wheat straw with culture of Phanerochaete chrysosporium in 12 days. Gupta et al. (2011) observed the removal of 7.7–11.9% lignin in aerobic solid-state fermentation of the crushed residues of woody plants Prosopis juliflora with the culture of Pycnoporus cinnabarinus in 15 days. Thus, the use of C. pannosa strain F6 in the present study is highly effective when compared to other fungal cultures as reported earlier. The effectiveness of the decomposition of lignin by white rot fungi is directly related to the activity of laccase, and a maximum enzymatic activity is achieved at 10–30 days of cultivation. Keeping this in mind, the experiment in the present study was carried out for 60 days. It should be emphasized that aerobic fungi promote the release (emission) of terpenes, essential oils, and resins in the form of volatile compounds during the decomposition of coniferous litter, and some species are able to degrade aromatic compounds (Isidorov et al. 2010), thereby reducing the toxicity of the litter to aerobic and anaerobic microorganisms.
Conclusion
Pine needle litter is a complex substrate for microbial degradation due to the high content of lignin and aromatics and therefore requires mandatory pretreatment. The most efficient method of pretreatment in terms of the removal of lignin and release of free cellulose from pine needle litter is physico-chemical treatment. However, at the same time, in terms of simplicity, cost, and comparable efficacy, the best way to pretreat the pine litter is a mechanical milling. In spite of significant reduction in lignin content during the pretreatment, the addition of pine litter as a substrate inhibited the overall anaerobic digestion process, regardless of the inoculum used. This warrants for future endeavors, especially in the area of development of specific microbial consortia that could be used as inoculum in anaerobic digestion. During the digestion process of different pretreated pine needle litter, the mechanically milled pine litter along with DS yielded higher methane levels than digestion of untreated pine needle litter. The results suggested that the co-digestion of pine needles with other organic waste (FW, OFMSW, and others) is an effective strategy to degrade these recalcitrant wastes with an active inoculum.
References
An CH, Huang GH, Yao Y, Sun W, An K (2012) Performance of in-vessel composting of food waste in the presence of coal ash and uric acid. J Hazard Mater 203–204:38–45
ASTM D1104-56 (1978) Method of test for holocellulose in wood. http://www.astm.org/Standards/D1104.htm. Accessed 28 June 2014
Azman S, Khadem AF, Van Lier JB, Zeeman G, Plugge CM (2015) Presence and role of anaerobic hydrolytic microbes in conversion of lignocellulosic biomass for biogas production. Crit Rev Environ Sci Technol 45:2523–2564
Brown D, Shi J, Li Y (2012) Comparison of solid-state to liquid anaerobic digestion of lignocellulosic feedstocks for biogas production. Bioresour Technol 124:379–386
Cheng YF, Edwards JE, Allison GG, Zhu WY, Theodorou MK (2009) Diversity and activity of enriched ruminal cultures of anaerobic fungi and methanogens grown together on lignocellulose in consecutive batch culture. Bioresour Technol 100:4821–4828
Cheng CL, Lo YC, Lee KS, Lee DJ, Lin CY, Chang JS (2011) Biohydrogen production from lignocellulosic feedstock. Bioresour Technol 102:8514–8523
Cu TT, Nguyen TX, Triolo JM, Pedersen L, Le VD, Le PD, Sommer SG (2015) Biogas production from Vietnamese animal manure, plant residues and organic waste: influence of biomass composition on methane yield. Asian-Australas J Anim Sci 28:280
Demirbas A (2007) Products from lignocellulosic materials via degradation processes. Energy Sources Part A Recovery Util Environ Eff 30(1):27–37
Dollhofer V, Podmirseg SM, Callaghan TM, Griffith GW, Fliegerová K (2015) Anaerobic fungi and their potential for biogas production. In: Guebitz GM, Bauer A, Bochmann G, Gronauer A, Weiss S (eds) Biogas science and technology. Springer International Publishing, Heidelberg, pp 41–61
Elliston A, Wilson DR, Wellner N, Collins SR, Roberts IN, Waldron KW (2015) Effect of steam explosion on waste copier paper alone and in a mixed lignocellulosic substrate on saccharification and fermentation. Bioresour Technol 187:136–143
Gupta R, Mehta G, Khasa YP, Kuhad RC (2011) Fungal delignification of lignocellulosic biomass improves the saccharification of cellulosics. Biodegradation 22:797–804
Hubbe MA, Nazhad M, Sánchez C (2010) Composting as a way to convert cellulosic biomass and organic waste into high-value soil amendments: a review. BioResources 5:2808–2854
Isidorov VA, Smolewska M, Purzyńska-Pugacewicz A, Tyszkiewicz Z (2010) Chemical composition of volatile and extractive compounds of pine and spruce leaf litter in the initial stages of decomposition. Biogeosciences 7:2785–2794
Jain VK, Srinivas SN (2012) Empowering rural India the RE way: inspiring success stories. Ministry of New and Renewable Energy, Government of India, Navi Mumbai. ISBN 978-81-920040-0-6
Johnson DB, Moore WE, Zank LC (1961) The spectrophotometric determination of lignin in small wood samples. Tappi 44(11):793–798
Jonsson LJ, Martín C (2016) Pretreatment of lignocellulose: formation of inhibitory by-products and strategies for minimizing their effects. Bioresour Technol 199:103–112
Joshi P, Sharma N, Manab Sarma P (2016) Assessment of biomass potential and current status of bio-fuels and bioenergy production in India. Curr Biochem Eng 3:4–15
Kayembe K, Basosila L, Mpiana PT, Sikulisimwa PC, Mbuyu K (2013) Inhibitory effects of phenolic monomers on methanogenesis in anaerobic digestion. Br Microbiol Res J 3:32
Kim HJ, Lee S, Kim J, Mitchell RJ, Lee JH (2013) Environmentally friendly pretreatment of plant biomass by planetary and attrition milling. Bioresour Technol 144:50–56
Kuhad RC, Chandna P, Singh A (2011) Composting of lignocellulosic waste material for soil amendment. In: Singh A, Parmar N, Kuhad RC (eds) Bioaugmentation, biostimulation and biocontrol. Springer, Berlin, pp 107–128
Lacourt W (2011) Enrichment of methanogenic microcosms on recalcitrant lignocellulosic biomass (Doctoral dissertation)
Lawther JM, Sun R, Banks WB (1996) Effect of steam treatment on the chemical composition of wheat straw. Holzforsch-Int J Biol Chem Phys Technol Wood 50:365–371
Lin Z, Huang H, Zhang H, Zhang L, Yan L, Chen J (2010) Ball milling pretreatment of corn stover for enhancing the efficiency of enzymatic hydrolysis. Appl Biochem Biotechnol 162:1872–1880
Liu HW, Walter HK, Vogt GM, Vogt HS, Holbein BE (2002) Steam pressure disruption of municipal solid waste enhances anaerobic digestion kinetics and biogas yield. Biotechnol Bioeng 77:121–130
Mahajan R, Nikitina A, Litti Y, Nozhevnikova A, Goel G (2016) Autochthonous microbial community associated with pine needle forest litterfall influences its degradation under natural environmental conditions. Environ Monit Assess 188:417
Matsakas L, Rova U, Christakopoulos P (2015) Sequential parametric optimization of methane production from different sources of forest raw material. Front Microbiol 6:1163
Nikitina AA, Kevbrina MV, Kallistova AY, Nekrasova VK, Litti YV, Nozhevnikova AN (2015) Intensification of microbial decomposition of organic fraction of municipal waste: laboratory and field experiments. Appl Microbiol Biotechnol 51:393–401
Paredes-Sánchez JP, García-Elcoro VE, Rosillo-Calle F, Xiberta-Bernat J (2016) Assessment of forest bioenergy potential in a coal-producing area in Asturias (Spain) and recommendations for setting up a Biomass Logistic Centre (BLC). Appl Energy 171:133–141
Sharma D, Goel G, Sud A, Chauhan RS (2015) A novel laccase from newly isolated Cotylidia pannosa and its application in decolorization of synthetic dyes. Biocat Agric Biotechnol 4:661–666
Silva GG, Couturier M, Berrin JG, Buléon A, Rouau X (2012) Effects of grinding processes on enzymatic degradation of wheat straw. Bioresour Technol 103:192–200
Staley BF, Francis L, Barlaz MA (2012) Comparison of bacteria and Archaea communities in municipal solid waste, individual refuse components, and leachate. FEMS Microbiol Ecol 79:465–473
Steffen F, Requejo A, Ewald C, Janzon R, Saake B (2016) Anaerobic digestion of fines from recovered paper processing—influence of fiber source, lignin sand ash content on biogas potential. Bioresour Technol 200:506–513
Tappi T (2002) 222 om-02: acid-insoluble lignin in wood and pulp 2002–2003 TAPPI test methods
TAPPI Standard Methods (2009) Fibrous materials and pulp testing. Technical Association of Pulp and Paper Industry, Atlanta
Tsavkelova EA, Egorova MA, Petrova EV, Netrusov AI (2012) Thermophilic anaerobic microbial communities that transform cellulose into methane (biogas). Mosc Univ Biol Sci Bull 67:75–81
Zhi Z, Wang H (2014) White-rot fungal pretreatment of wheat straw with Phanerochaete chrysosporium for biohydrogen production: simultaneous saccharification and fermentation. Bioprocess Biosyst Eng 37:1447–1458
Acknowledgments
The research was funded by the Department of Science and Technology, Government of India, for Indo-Russian collaborative project “Elucidating the linkage between key limiting processes and microorganisms during anaerobic degradation of lignocellulosic waste” INT/RUS/RFBR/P-175. The authors wish to thank the collaborating institutes for providing research facilities to execute this research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editorial responsibility: M. Abbaspour.
Rights and permissions
About this article
Cite this article
Mahajan, R., Nikitina, A., Litti, Y. et al. Evaluating anaerobic and aerobic digestion strategies for degradation of pretreated pine needle litter. Int. J. Environ. Sci. Technol. 16, 191–200 (2019). https://doi.org/10.1007/s13762-017-1601-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-017-1601-y