Abstract
In this work, the anaerobic digestion of three microalgae (Chlorella sp., Nannochloropsis sp., and Scenedesmus sp.) and their residues, resulting from the oil extraction process and the in situ transesterification reaction for biodiesel production, using two inoculums (sewage sludge and poultry manure) for biogas production was investigated. It was found that the biogas production from digestion of oil-extracted microalgae residue with sewage sludge reached values similar to those obtained with raw microalgae (around 500 NL kg−1 VS). Both the volume of biogas generated from the microalgae residue from the extraction process of its oil and the quality of the biogas produced reflect the value of this residue to be valorized by anaerobic digestion. This approach based on a biorefinery concept and focusing on the anaerobic digestion process could be a key technology for energy production from biomass.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
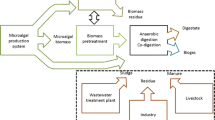
In the production of biodiesel from microalgae, both the process of obtaining biodiesel in two stages (extraction reaction) and the one-stage process (direct or in situ) generate solid residues of microalgae that are susceptible of being valued by other conversion processes for energy recovery or for the generation of raw materials for other processes. Specifically, biodiesel production from the lipid fraction of high lipid containing microalgae species results roughly 65% residues of total biomass that are rich in proteins and carbohydrates [1]. The residues produced after biofuel production from microalgae could be used for biogas production. This integrated approach minimizes costs and energy required for cultivation and harvesting of microalgae.
Biogas production through anaerobic digestion is a simple and low-cost method to convert biomass feedstocks into a renewable source of energy. However, anaerobic digestion of microalgal biomass often suffers from low methane yields due to their rigid cell walls, but also to their composition (low C/N ratio) and to their high protein content that causes a release of ammonia that inhibits the digestion process [2,3,4,5,6]. Specifically, the high protein content of the algal biomass usually leads to a low C/N ratio, which is imbalanced for anaerobic digestion. A C/N ratio of 25 to 32 was reported to have a positive effect on the methane yield [7]. At lower C/N ratios, the risk of excess in nitrogen becomes inhibitory. Therefore, digestion of microalgal biomass can be overcome through digestion with a carbon-rich waste as co-substrate, increasing their methane yield [8,9,10]. The favorable effect of anaerobic digestion of microalgae was recently reported by Solé-Bundó et al. [11].
Besides, several authors have studied sewage sludge as inoculum in anaerobic digestion processes with different types of substrates, including microalgae. Sewage sludge is the most researched inoculum for microalgae [12,13,14,15,16,17]. Animal manure and microalgae anaerobic digestion have received less attention than microalgae digestion with sewage sludge [16]; however, in the large volume of manure produced in poultry farms, its high polluting power and its high degree of biodegradability make the digestion of microalgae with poultry manure worth investigating.
Although numerous studies have paid attention to the anaerobic digestion of raw microalgae for biogas production, limited amount of study investigated the utilization of microalgae residue resulted from oil extraction process or in situ transesterification reaction for biodiesel production [8, 9, 18,19,20,21,22,23,24,25,26]. This work is focused on the study of anaerobic digestion of three microalgae and their residues resulting from microalgae utilization processes (oil extraction process and in situ transesterification reaction for biodiesel production) using two inoculums, sewage sludge (SS) and poultry manure (PM), for biogas production. Firstly, the production and composition of biogas obtained in the digestion of the two types of inoculums with three different raw microalgae species (RM) were studied. Subsequently, the digestion of different microalgae solid residues obtained from the oil extraction processes (ER) and from the in situ transesterification reactions (TR) was carried out with the inoculum that generated the highest methane production.
2 Materials and methods
2.1 Substrates for anaerobic digestion
Three raw microalgae, Chlorella sp. (C), Nannochloropsis sp. (N), and Scenedesmus sp. (S), their residues from the oil extraction processes (ER-C, ER-N, and ER-S, respectively), and the in situ transesterification reactions for biodiesel production (TR-C, TR-N, and TR-S, respectively) were studied as substrates in anaerobic digestion for biogas production.
The species Chlorella sp., Nannochloropsis sp., and Scenedesmus sp. were grown during summertime in a raceway pond, pilot-scale plant located in the facilities of the Department of Biotechnology of the Technological Institute of the Canary Islands (ITC) in the southeast of Gran Canaria. These strains belong to the ITC microalgae culture collection. The species were cultured in f/2 medium [27], Chlorella sp. and Scenedesmus sp. in fresh water and Nannochloropsis sp. in saline water.
Cultures reaching the stationary growth phase, characterized by insignificant variations in cell density, were harvested and centrifuged to eliminate the culture medium. Then, they were dried, by means of a spray drier device, to become a microalgal powder with no more than 5% of humidity. Finally, they were vacuum-packed and stored at 6 °C, protected from light and humidity. Once the containers were opened, it was necessary to keep them cold (4 °C) and protect from light to avoid oxidation.
Microalgae residues were obtained from previous research carried out by the authors, corresponding to two processes: extraction of microalgae oil via solvent extraction, which is part of the first stage of the biodiesel production process when it is carried out in two stages (extraction-reaction), and in situ transesterification reaction for biodiesel production in one stage. Oil extraction process was carried out using a Soxhlet extractor and methanol as organic solvent. From this process, a liquid phase is obtained (oil) for the second stage of the biodiesel production process and a solid residue (ER). In situ transesterification reactions were carried directly on the biomass using methanol as reagent and sulfuric acid as acid catalyst. From this process, a liquid phase that contains the products, reaction by-products, excess reagents, and the catalyst used is obtained. A solid phase is also obtained (TR), which constitutes the main residue of the process. Both residues, ER and TR, were dried at 40 °C for 24 h to evaporate the residual solvent impregnated in them and kept at 4 °C in sterilized containers, protected from light.
2.2 Inoculums for anaerobic digestion
Two inoculums were employed for anaerobic digestion: sewage sludge and poultry manure. Sewage sludge was obtained from a pilot-scale anaerobic membrane biorreactor (AnMBR) located in the wastewater treatment plant (WWTP) of Valle Guerra (Tenerife, Canary Islands, Spain). Poultry manure was hand-collected from a local poultry farm located in La Esperanza (Tenerife, Canary Islands, Spain) and was stored in sterilized containers. The manure presented remnants of shavings, dry grass, husks, food residues, and feathers, among others. After collections, the inoculums were then kept in closed containers, under shade at room temperature for subsequent use.
2.3 Anaerobic digestion tests
Batch anaerobic digestion tests for biogas production were carried out using the procedure described by García et al. [28]. A 500-mL volume amber glass flask was used as digester. In each digester, 6 ± 0.05 g of substrate was introduced to which 6 mL of 1.5 M NH4Cl solution (purity 99.5%, Panreac) was subsequently added to provide the medium with a nitrogen source, 1.5 mL of buffer solution (pH = 7.00 (20 °C), Scharlau) and 50 mg of CaCO3 (purity 99.0%, Panreac) that acted as a buffer. Anaerobic digestion tests were carried out with inoculum and in the absence of this. When the inoculum (SS or PM) was introduced, a 50:50 inoculum:substrate ratio (based on the volatile solid (VS) content) was used. When the poultry manure was used as inoculum, 250 mL of distilled water was added to the digester due to its solid nature (not for the sewage sludge due to its semisolid nature) to promote the homogenization of the mixture and to have the necessary aqueous medium for the anaerobic digestion process. Table 1 shows the list of experiments carried out. The substrate and inoculum used are indicated for each experiment.
The content of the digester was mixed, and, before starting the digestion process, pH was measured and adjusted to values around 7.0–8.0 with a NaOH solution (purity 99.0%, Scharlau). Conductivity was also measured. Then, the biodigesters were hermetically sealed with a silicone septum equipped with a gas sampler system, for the control of the gas quality, and an outlet tubing with an on/off valve connected to a volumetric flask. The volumetric flasks allow to monitor the volume of gas produced by liquid displacement. Once each digester was sealed, nitrogen was bubbled to displace the oxygen contained in the digester. The volumetric flasks were filled with distilled water acidified (HCl, purity 37%, Merck) with pH = 2 to shift the CO2 ↔ HCO3− equilibrium to the left. Each volumetric flask was also previously bubbled with nitrogen to displace the oxygen contained in the gas chamber. Additionally, reactors with only inoculum (and distilled water added for PM) were used as control system (SS-blank and PM-blank).
The systems were incubated in mesophilic conditions (31.5 °C) until biogas production ceased. Each system was manually mixed once per day, just before the volume readings of displaced liquid to determine gas production and the gas sampling to determine its composition. In addition, ambient pressure and temperature measurements were taken to normalize the gas volume data.
The effect of the type of inoculum was studied using the raw microalgae as a substrate. Once the best inoculum was selected, digestions were carried out with the raw microalgae and its residues. These tests were performed in duplicate.
Biogas production (NL kg−1 VS) was calculated by normalizing the produced biogas volume (0 °C and 1 atm) per kg of volatile solids of the substrate introduced in the digester. Moreover, the biogas composition was determined by a gas chromatography. At the end of the digestion tests, the conductivity and pH values were measured.
2.4 Analytical methods
Total solid (TS) and volatile solid content were determined according to the standard methods 2540 G (for solid and semisolid samples) and 2540 B (for the determination of total solids in sewage sludge) [29]. TS and VS content of the nine substrates and the two inoculums are depicted in Table 2. Besides, the elemental composition of each substrate (CHNS) was analyzed by a Thermo Scientific™ Elemental Analyzers (Flash EA 1112), and the C/N ratios were determined (Table 3).
Conductivity and pH measurements were performed with a Crison GLP31 conductimeter (Crison Instrument, Barcelona, Spain) with automatic compensation of the readings to 25 °C standard temperature and Crison pH meter basic 20 (Crison Instrument, Barcelona, Spain), respectively.
Biogas composition was analyzed by gas chromatography (GC) (Agilent 7820A GC System) with a thermal conductivity detector (TCD), a split/splitless injector and two capillary columns working in parallel, CP-molsieve 5 Å (30 m, 0,53 mm DI, 0,53 μm) and CP-PoraBond Q (packed column), to enable the measurement to CH4, CO2, CO, O2, and N2. The gas chromatography operating conditions were as follows: (a) oven temperature isothermal at 40 °C; (b) injector temperature 175 °C; (c) TCD temperature 180 °C; and (d) helium was used as carrier gas at 9 psi pressure, a flow rate of 36 mL min−1. The sample injection was performed in split mode (5:1 split ratio) and an injected sample volume of 0.5 mL.
3 Results and discussion
3.1 Effect of inoculum type on biogas production
Prior to the anaerobic digestion tests with inoculum, digestions were carried out with each of the nine substrates already described and in the absence of inoculum. The results obtained are shown in the Appendix Figs. 5, 6, and 7. In all cases, absence of methane was observed. The gas generated was composed of CO2. Although the main goal of this work is the production of biogas, this CO2 could be used as a raw material for the production of other biofuels such as bio-dimethyl ether (DME).
The effect of the inoculum type on biogas production was studied using the raw microalgae as substrate due to their greater availability. The solid content of the substrates and inoculums correspond to those shown in Table 2.
Figure 1 shows accumulated production of CH4 and CO2, for 40 days, for the three species of microalgae using sewage sludge and poultry manure as inoculums. In addition, Table 4 shows the total biogas production generated in each biodigester (both, including and not including the blank production), as well as the corresponding percentage of CH4 and CO2.
Digestion with sewage sludge showed a higher biogas production (both CH4 and CO2) than when poultry manure was used; this behavior is independent of the microalgae species used as substrate. Total biogas productions around 360–480 NL kg−1 VS were reached with SS compared to the 24–40 NL kg−1 VS reached with PM.
The low gas production in the digestion of microalgae with poultry manure may be due to lack of water. The negative values obtained in the production of biogas by subtracting the production of the blank indicate the antagonistic effect generated by the substrate-PM mixture in anaerobic digestions. Sadaka and Engler [30] reported that solid and water content is one of the important parameters in the biogas production which directly affects the anaerobic digestion. Water makes possible route for the movement and growth of bacteria by facilitating the transport of nutrient and reduces the limitation of mass transfer. Deepanraj el al. [31] reported that the optimum TS concentration for biogas production using food waste as feed and cow dung as inoculum was 7.5%. In the present study, the average TS concentration of the biodigesters in which the digestion of microalgae with PM was carried out was 10.3%. High TS concentration could be negatively affecting biogas production. However, the TS concentration of the PM-blank is lower (8.49%), and a higher biogas production is achieved (Table 4).
In Table 4, the biogas obtained from the digestions carried out with SS is rich in CH4 (with an average value of 65%), indicating that it is a quality biogas. However, biogas obtained from the digestions carried out with PM as inoculum is rich in CO2 (with an average value of 91%).
In brief, anaerobic sewage sludge proved to be the most appropriate inoculum for the digestions of microalgae, due to both the production levels reached and the quality of the gas produced. Therefore, SS has been selected as inoculum in the following digestions. The highest total biogas production was achieved with Nannochloropsis sp., with a production of 483.56 NL kg−1 VS (407.94 NL kg−1 VS not including the blank production) for 40 days (Table 4).
3.2 Biogas production from anaerobic digestion of raw microalgae and microalgae residues with sewage sludge
Before proceeding to the study of the digestion of the microalgae and their residues from the oil extraction and transesterification process, with sewage sludge as inoculum, the pH and conductivity values of the biodigesters were determined. The corresponding values for each biodigester before digestion process are shown in the Table 5.
The pH initial values in all reactors ranged from 7.76 to 8.35, which could be suitable for the anaerobic digestion process. The main drawback of the anaerobic digestion process could be the low pH, which negatively affects the methanogenesis phase, since the microorganisms that carry out the digestive process are highly sensitive to it [32]. In the literature, contradictory results have been found at alkaline pH. Chandra et al. [33] reported that if the C/N ratio is very low, nitrogen will be liberated and accumulated in the form of ammonium ion, then the presence of excess NH4 will increase the pH of the biodigestate in the digester, and thus a pH higher than 8.5 will start showing a toxic effect on methanogens population. However, Rincón-Pérez et al. [34] demonstrated that anaerobic digestion at alkaline condition (pH = 9) produced biogas with higher methane content (83%) that at neutral pH (61%).
Conductivity is other parameter that should be controlled during the anaerobic digestion process because ammonium (NH4+) can act as an inhibitor if it is found at high concentrations, reducing methanogenic productivity and the possible cessation of production methane [35, 36]. The biodigesters that contain microalgae residues from the transesterification reaction to produce biodiesel are those with the highest conductivity, which could negatively affect biogas production. The highest conductivity associated with this residue is independent of the microalgae from which it comes from.
The biogas produced for each of the microalgae species and their residues was studied, and Fig. 2, Fig. 3, and Fig. 4 show the accumulated gas productions for the RM, ER, and TR digestion tests, respectively, using sewage sludge as inoculum. All digestions were performed in duplicate. Moreover, Table 6 shows the average total biogas production (both, including and not including the blank production) from each biodigester, as well as the corresponding percentage of CH4 and CO2. In general, the reproducibility of the experiments was high despite the heterogeneous nature of the samples.
Figure 2 shows accumulated production of CH4 and CO2 from RM anaerobic digestion for the three species of microalgae using SS as inoculum. All digestion process presented the same evolution in the gas accumulated production: a stage of initiation, characterized by low gas production speed; a stage of maximum gas production speed; and a stabilization stage where the gas production speed decreased until it became null. In biodigesters containing Chlorella sp., this trend was observed more smoothly (Fig. 2a). The average total biogas production was 473.87, 518.42, and 530.07 NL kg−1 VS for RM-C, RM-N, and RM-S, respectively (Table 6). Therefore, the maximum biogas production was reached in the biodigester that contained Scenedesmus sp. (RM-S). The biogas obtained showed an average CH4 composition of 68.16%, 68.59%, and 63.40% for RM-C, RM-N, and RM-S, respectively.
Figure 3 shows accumulated production of CH4 and CO2 from ER anaerobic digestion for the three species of microalgae using SS as inoculum. All digestion process presented the same evolution in the accumulated gas production than that observed for the RM digestions (a stage of initiation, a stage of maximum gas production speed, and a stabilization stage), although presenting a smoother evolution. The average total biogas productions achieved were similar to those obtained in the digestions of RM, 484.30, 510.69, and 455.30 NL kg−1 VS for ER-C, ER-N, and ER-S, respectively (Table 6). However, the maximum biogas production was reached in the biodigester that contained Nannochloropsis sp. residues from oil extraction process (ER-N). Further, the biogas obtained showed an average CH4 composition of 71.07%, 67.38%, and 65.56% for ER-C, ER-N, and ER-S, respectively.
Both the volume of biogas generated from the microalgae residue from the extraction process of its oil and the quality of the biogas produced reflect the potential of this residue to be valorized by anaerobic digestion. Carrying out an oil extraction process from microalgae, obtaining a liquid of great added value for other applications (including the biodiesel production) and a solid with the ability to produce biogas through anaerobic digestion, it is very advantageous and more profitable, than only obtaining biogas from raw microalgae, because the liquid product, that has a great added value for multiple applications, could be lost.
Figure 4 shows accumulated production of CH4 and CO2 from TR anaerobic digestion for the three species of microalgae using SS as inoculum. All digestion process presented the same evolution in the gas accumulated production than that observed for the RM and ER digestion; however, the maximum production stage was significantly lower than those obtained from the RM and ER digestions. Moreover, the total biogas productions were much lower than those achieved in the RM and ER digestions: 134.25, 253.93, and 172.60 NL kg−1 VS for TR-C, TR-N, and TR-S, respectively (Table 6). The low biogas productions obtained may be due to the process where the residue (TR) comes from, which is carried out under highly aggressive conditions; a strong acid (H2SO4) is used as catalyst. The sulfate reduction to sulfide may be the cause of the low biogas production. The presence of sulfur in this substrate is shown in Table 3. The properties of this substrate do not seem to be the most suitable for the anaerobic digestion process. In Table 5, the high initial conductivities of the biodigesters that contained this type of substrate were observed and may be the cause of the inhibition in the biogas production.
In the TR digestion tests, the maximum biogas production was reached in the biodigester that contained Nannochloropsis sp. residues from transesterification reaction for biodiesel production (TR-N). Despite the low biogas production, the average composition of methane was over 60% (Table 6), except for the Scenedesmus sp. residue from transesterification reaction (TR-S). The average CH4 composition was 70.55%, 68.36%, and 59.31% for TR-C, TR-N, and TR-S, respectively.
According to Table 6, the digestions of the RM and ER substrates with SS presented total biogas productions much higher than those reached with SS-blanks; however, the biogas production values from the digestions of TR with SS were equal to or lower than those reached with SS-blanks (134.25, 253.93, and 172.60 NL kg−1 VS for TR-C, TR-N, and TR-S, respectively), indicating that TR is not a suitable substrate for anaerobic digestion.
Overall, it can be concluded that the anaerobic digestion is a process suitable for the valorization of microalgae residues from oil extraction process, since biogas productions similar to those achieved with raw microalgae are reached. In addition, the anaerobic digestion of residues generated in the biodiesel production process from microalgae in two stages (extraction reaction) would allow to benefit not only from the lipid extract of microalgae for obtaining biodiesel but also from solid residues generated throughout the process. Table 7 shows a mass balance of each of the processes from 1 kg of dry microalgae. This approach based on a biorefinery concept and focusing on the anaerobic digestion process could be a key technology for energy production from biomass.
The pH and conductivity values of the biodigesters measured after the anaerobic digestion tests are shown in Table 5. The final pH values (7.06–7.90) showed a slight reduction after the anaerobic digestion process, although they remained around the initial values; therefore, the microbial activity was not affected in any case by the pH of the medium, and as a result the biogas production rate was not inhibited by this parameter.
For all the digestions tests carried out, the final conductivity values were higher than the initial (Table 5). Researchers have already demonstrated that the increase in conductivity can be directly correlated to ammonium nitrogen released during digestion of protein rich biomass [35, 36]. This in turn enables it to be used as an indicator of biomass hydrolysis [37]. The highest conductivity increase was 13.3 mS cm−1 for ER-C2 biodigester, which generated a biogas production of 494.18 NL kg−1 VS. Even though an increase in conductivity could indicate an increase in ammonium nitrogen concentration, which can act as an inhibitor of the methanogenic process if it is found at high concentrations, it is possible that the ammonium concentrations reached were not very high since relatively high biogas productions were obtained.
The increase in conductivity seems to be independent of the type of microalgae used (Chlorella sp., Nannochloropsis sp., or Scenedesmus sp.); however, it seems to be influenced by the type of substrate used (RM, ER, or TR). The average increase in conductivity in the biodigesters that used RM, ER, and TR as substrate was 7.5, 9.9, and 3.2 mS cm−1, respectively.
The smallest increases in conductivity were observed for the biodigesters that contained post transesterified microalgae residues, which were those that presented a lower average biogas production (Table 6). Therefore, the observed increase in conductivity seems not to inhibit the methanogenic process; in this case, that the low increase in conductivity does not favor the biogas production may be due to the severe treatment to which the microalgae are subjected during transesterification; the reaction was carried out with a strong acid, sulfuric acid, as catalyst. Another potential reason could be the sulfate reduction as it was mentioned previously or the presence of non-hydrolysable particulate matter. In the latter case, the residues could be pretreated to increase digestibility.
Table 8 shows comparative data on methane production from the digestion in batch of raw microalgae, microalgae residues from the oil extraction process, and post transesterified microalgae residues from studies found in the literature in recent years, and they are compared with the methane productions obtained in the present study.
The methane productions, found in the literature, from the digestion of microalgae not subjected to any type of prior treatment, are lower than those found in this study. For RM subjected to a previous treatment, in general, it is observed that methane productions are equal to or lower than those found in this study, except that achieved by Park et al. [18], 432.17 NmLCH4 g−1VS, from the digestion of pretreated Chlorella sp. with sludge.
Little current literature about digestion of microalgae residues was found. Meneses-Reyes et al. [25] carried out the co-digestion of Chlorella vulgaris with chicken litter and glycerol, achieving 131.1 NmLCH4 g−1VS; in this study, methane production exceeded twice the value that was achieved: 344.19, 344.10, and 298.49 NmLCH4 g−1VS for ER-C, ER-N, and ER-S, respectively.
On the other hand, Ehimen et al. [26] studied the TR co-digestion and achieved higher methane productions (308 NmLCH4 g−1VS) than those obtained in this study. However, glycerol from biodiesel production for the anaerobic digestion of TR was added. Increasing the substrate C/N ratio to 12.44 by co-digesting the microalgae residues with glycerol was observed to increase CH4 production by > 50%, compared with the CH4 production when the residues were digested alone. Therefore, the integration of glycerol in the digestion process of the studied TR could increase the biogas production considerably.
4 Conclusions
The sewage sludge proved to be better inoculum for the anaerobic digestion process than poultry manure. For using poultry manure as inoculum, it should be previously acclimatized. In addition, the concentration of solids in the biodigester should be taken into account, since high concentrations can inhibit the biogas production.
For the three types of substrates studied (RM, ER, and TR), the highest biogas productions were reached with raw microalgae and with oil-extracted microalgae residues, with similar productions; this indicates that ERs are highly recoverable residues. Both the volume of biogas generated and the quality of the biogas produced reflect the potential of this residue to be valorized by anaerobic digestion.
Post transesterified microalgae residues are not a suitable substrate for anaerobic digestion since they generated low biogas productions, probably due to the severe conditions they are subjected to during the transesterification reaction. The sulfate reduction to sulfide may be the cause of the low biogas production. Another possible reason could be the presence of non-hydrolysable particulate matter.
The results highlight the promising potential of oil-extracted microalgae residues through anaerobic digestion process. In addition, the anaerobic digestion of residues generated in the process of obtaining biodiesel from microalgae in two stages (extraction reaction) would allow to benefit not only from the lipid extract of microalgae for obtaining biodiesel but also from solid residues generated throughout the process. With this residue solid, highly reusable via anaerobic digestion inoculating with sewage sludge, a total biogas production of 510.69 NL kg−1 VS was achieved. This approach based on a biorefinery concept and focusing on the anaerobic digestion technique could be a key technology for energy production from biomass.
References
Cherubini F (2010) The biorefinery concept: using biomass instead of oil for producing energy and chemicals. Energy Convers Manag 51:1412–1421
Ward AJ, Lewis DM, Green FB (2014) Anaerobic digestion of algae biomass: a review. Algal Res 5:204–214
Zamorano-López N, Borrás L, Seco A, Aguado D (2020) Unveiling microbial structures during raw microalgae digestion and co-digestion with primary sludge to produce biogas using semi-continuous AnMBR systems. Science of the Total Environment 699:134365
Cuellar-Bermudeza SP, Magdalenab JA, Muylaerta K, Gonzalez-Fernandez C (2019) High methane yields in anaerobic digestion of the cyanobacterium Pseudanabaena sp. Algal Research 44:101689
Chen Y, Cheng JJ, Creamer KS (2008) Inhibition of anaerobic digestion process: a review. Biores Technol 99:4044–4064
Leadbeater BSC (2006) The ‘D. roop equation’-Michael Droop and the legacy of the ‘cell-quota model’ of phytoplankton growth. Protist 157(3):345–58
Kayhanian M, Tchobanoglous G (1992) Computation of C/N ratios for various organic fractions. Biocycle 33:58–60
Solé-Bundó M, Garfí M, Ferrer I (2020) Pretreatment and co-digestion of microalgae, sludge and fat oil and grease (FOG) from microalgae-based wastewater treatment plants. Bioresource Technology 298:122563
Lu D, Liu X, Apul OG, Zhang L, Ryan DK, Zhang (Jackie) X (2019) Optimization of biomethane production from anaerobic co-digestion of microalgae and septic tank sludge. Biomass and Bioenergy 127:105266
Sialve B, Bernet N, Bernard O (2009) Anaerobic digestion of microalgae as a necessary step to make microalgal biodiesel sustainable. Biotechnol Adv 27:409–416
Solé-Bundó M, Passos F, Romero-Güiza MS, Ferrer I, Astals S (2019) Co-digestion strategies to enhance microalgae anaerobic digestion: a review. Renew Sustain Energy Rev 112:471–482
Wang M, Sahu AK, Rusten B, Park C (2013) Anaerobic co-digestion of microalgae Chlorella sp. and waste activated. Bioresource Technology 142:585–590
Olsson J, Feng XM, Ascue J, Gentili FG, Shabiimam MA, Nehrenheim E et al (2014) Codigestion of cultivated microalgae and sewage sludge from municipal waste wáter treatment. Bioresour Technol 171:203–210
Caporgno MP, Trobajo R, Caiola N, Ibáñez C, Fabregat A, Bengoa C (2015) Biogas production from sewage sludge and microalgae co-digestion under mesophilic and thermophilic conditions. Renew Energy 75:374–380
Hidaka T, Takabe Y, Tsumori J, Minamiyama M (2017) Characterization of microalgae cultivated in continuous operation combined with anaerobic co-digestion of sewage sludge and microalgae. Biomass Bioenergy 99:139–146
Solé-Bundó M, Cucina M, Folch M, Tàpias J, Gigliotti G, Garfí M et al (2017) Assessing the agricultural reuse of the digestate from microalgae anaerobic digestion and codigestion with sewage sludge. Sci Total Environ 586:1–9
Thorin E, Olsson J, Schwede S, Nehrenheim E (2018) Co-digestion of sewage sludge and microalgae – biogas production investigations. Apply Energy 227:64–72
Park J, Kumar AN, Cayetano RDA, Kim SH (2020) Assessment of Chlorella sp. as a potential feedstock for biological methane production. Bioresource Technology 305:123075
Ekinci K, Erdal I, Uysal Ö, Uysal FÖ, Tunce H, Dogan A (2019) Anaerobic digestion of three microalgae biomasses and assessment of digestates as biofertilizer for plant growth. Environmental progress & sustainable energy 38(3):1–9
Arias DM, Solé-Bundó M, Garfí M, Ferrer I, García J, Uggetti E (2018) Integrating microalgae tertiary treatment into activated sludge systems for energy and nutrients recovery from wastewater. Bioresour Technol 247:513–519
Olsson J, Forkman T, Gentili FG, Zambrano J, Schwede S, Thorin E et al (2018) Anaerobic co-digestion of sludge and microalgae grown in municipal wastewater - a feasibility study. Water Sci Technol 77:682–694
Solé-Bundó M, Salvadó H, Passos F, Garfí M, Ferrer I (2018) Strategies to optimize microalgae conversion to biogas: co-digestion, pretreatment and hydraulic retention time. Molecules 23(9):2096
Passos F, Cordeiro PHM, Baeta BEL, de Aquino SF, Perez-Elvira SI (2018) Anaerobic codigestion of coffee husks and microalgal biomass after thermal hydrolysis. Bioresour Technol 253:49–54
Carminati P, Gusmini D, Pizzera A, Catenacci A, Parati K, Ficara E (2018) Biogas from mono- and co-digestion of microalgal biomass grown on piggery wastewater. Water Sci Technol 78:103–113
Meneses-Reyes JC, Hernández-Eugenio G, Huber DH, Balagurusamy N, Espinosa-Solares T (2017) Biochemical methane potential of oil-extracted microalgae and glycerol in co-digestion with chicken litter. Biores Technol 224:373–379
Ehimen EA, Sun ZF, Carrington CG, Birch EJ, Eaton-Rye JJ (2011) Anaerobic digestion of microalgae residues resulting from the biodiesel production process. Appl Energy 88:3454–3463
R.R.L. Guillard (1975) Culture of phytoplankton for feeding marine invertebrates. In Smith W.L. and Chanley M.H (Eds.) Culture of marine invertebrate animals. Plenum Press, New York, USA 26–60
García Peña EI, Parameswaran P, Kang DW, Canul-Chan M, Krajmalnik-Brown R (2011) Anaerobic digestión and co-digestion processes of vegetable and fruit residues: process and microbial ecology. Bioresource Technology 102:9447–9455
APHA (American Public Health Association), AWWA (American Water Works Association), WPCF (Water Pollution Control Federation). R.B. Baird, A.D. Eaton & E.W. Rice (Eds.), Standard methods for examination of water & wastewater, 2017 (23nd Edition), Washington, DC: American Public Health Association.
Sadaka SS, Engler CR (2003) Effect of initial total solids on compositing of raw manure with biogas recovery. Compost Science & Utilization 11:361–369
B. Deepanraj, V. Sivasubramanian, S. Jayaraj. Solid concentration influence on biogas yield from food waste in an anaerobic batch digester, International Conference and Utility Exhibition 2014 on Green Energy for Sustainable Development (ICUE 2014). DOI: https://doi.org/10.13140/2.1.2309.4405
Teng Z, Changsong JH, Lu WX (2014) Design and optimization principles of biogas reactors in large scale applications. Reactor and process design in sustainable energy technology-chapter 4:99–134
Chandra R, Takeuchi H, Hasegawa T (2012) Methane production from lignocellulosic agricultural crop wastes: a review in context to second generation of biofuel production. Renew Sustain Energy Rev 16(3):1462–1476
Rincón-Pérez J, Celis LB, Morales M, Alatriste-Mondragón F, Tapia-Rodríguez A, Razo-Flores E (2021) Improvement of methane production at alkaline and neutral pH from anaerobic co-digestion of microalgal biomass and cheese whey. Biochemical Engineering Journal 169:107972
Adamietz T, Jurkowski W, Adolph J, Brück TB (2019) Biogas yields and composition from oil-extracted halophilic algae residues in conventional biogas plants operated at high salinities. Bioprocess Biosyst Eng 42:1915–1922
Micolucci F, Gottardo M, Bolzonella D et al (2014) Automatic process control for stable bio-hythane production in two-phase thermophilic anaerobic digestion of food waste. Int J Hydrogen Energy 39(31):17563–17572
Klein R, Slaný V, Krčálová E (2018) Conductivity measurement for control of a biogas plant. Acta Univ Agric Silvic Mendelianae Brun 66(5):1151–1156
Funding
The authors acknowledge to the Agencia Canaria de Investigación, Innovación y Sociedad de la Información by a research grant support by “Programa de ayuda de Formación del Personal Investigador.”
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Torres, A., Padrino, S., Brito, A. et al. Biogas production from anaerobic digestion of solid microalgae residues generated on different processes of microalgae-to-biofuel production. Biomass Conv. Bioref. 13, 4659–4672 (2023). https://doi.org/10.1007/s13399-021-01898-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-021-01898-9