Abstract
The wild boar is a native animal species of the Czech Republic that has significantly increased its population density in recent years. Objective methods of population density estimation are required to inform the intensity of regulation needed for the sustainable management of the wild boar. Population estimation and regulation of the abundance of the wild boar are problematic in comparison to those of other ungulates, and the only widely used method of control is intensive hunting. The purpose of this study was to test if faecal pellet group (FPG) counting was a reliable method of wild boar population censusing in a forest environment. The density of wild boars was evaluated in an area of 22.56 km2, circumscribed by both natural and man-made barriers that restrict wild boar migration. As it is essential for proper application of FPG count methods, a separate trial was carried out to determine the daily defecation rate of wild boars. The distribution of faeces in different types of habitat in a forest environment was tested. As the period of faeces accumulation fell within the intensive hunting season, the result is biased by the fact that the calculation includes FPGs produced by animals removed from the population through hunting during winter. The average daily defecation rate of adults and piglets was 4.29 and 5.82, respectively. The corrected (without shot animals) average density of wild boars in the study area estimated by the FPG count at the time of the census was 6.08 individuals (ind.)/km2. We have confirmed that an FPG count conducted in winter is applicable for estimation of wild boar abundance on a forested area.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The wild boar is a native and important species of the Czech Republic fauna. The abundance of the wild boar has varied significantly throughout the last centuries, and early in the nineteenth century, the wild boar was eradicated in order to prevent extensive damage to agricultural crops (Hladíková et al. 2008, Apollonio et al. 2010). After the Second World War, the wild boar began recolonising the Czech Republic, and today, its growing population presents an economic, ecological and social threat not only in the Czech Republic but also across Central Europe (Keuling et al. 2013, Acevedo et al. 2014). The most serious consequences of the increase in wild boar density are the damage to agricultural crops, road accidents, transmission of infectious diseases and the destruction of managed green spaces in populated areas (Geisser 1998, Brauer et al. 2006, Herrero et al. 2006, Acevedo et al. 2007).
The main goal for wildlife managers is to halt the continual increase of the wild boar population and manage it sustainably. Although there are several possibilities for wild boar population control including poisoning, sterilization of females and trapping (Cruz et al. 2005, Killian et al. 2006, West et al. 2009, Braga et al. 2010), in Central Europe, practically the only widely used method is intensive hunting (Fruzinski and Labudzki 2002, Keuling et al. 2013). The success of wild boar population control by hunting depends on a number of variables including the annual reproduction rate, the distribution of attractive food sources and cover and the skills of hunters and hunting pressure (using efficient hunting methods, precise estimation of wild boar abundance and an annual harvest plan).
Wild boars generally start to conceive at 8–12 months and can continue to produce approximately four to seven young throughout their adult lives (Gethöffer et al. 2007, Fonseca et al. 2011). If the population is to be successfully reduced, hunting pressure has to eliminate the annual increment and a percentage of the adult females from the breeding population. The intensity of hunting pressure in recent years has apparently allowed the wild boar population to expand, whilst efforts to stop or slow population growth have been limited by the low accuracy of current abundance estimations (Merli and Meriggi 2006). Wildlife managers therefore need accurate estimations of wild boar density to inform decisions on the numbers of individuals that need to be removed. For this reason, improved methods to determine wild boar abundance are a key pillar in the management of this species (Engeman et al. 2013).
The accuracy of population estimation depends on a number of factors such as species, method used, experience of researchers, season of the year and many others (Mayle et al. 1999). Determination of wild boar abundance is, compared to other free-living ungulates, very problematic due to their secretive habits, migrations between fields and forests and highly variable reproductive rate (Engeman et al. 2013). Therefore, the only reliable source of data for wild boar populations comes from steadily increasing hunting records (Hladíková et al. 2008). Hunting records show population trends in large areas and over long periods, but they are not suitable for adaptative management in small areas, where the density of wild boars can significantly fluctuate during the year. More reliable methods to estimate wild boar density have therefore been developed. Estimating abundance and regulating a wild boar population, in comparison to other wild living ungulates, are problematic. A variety of observation techniques have been used in studies to determine various wild boar populations in Europe (Focardi et al. 2002); however, they are simple but often inaccurate (Mayle et al. 1999). In other instances, wild boar numbers have been assessed by the repeat counting of marked individuals (Waithman et al. 1999), counting of tracks on snow cover (Fonseca et al. 2007) and phototrapping combined with marked individuals (Sweitzer et al. 2000, Hebeisen et al. 2008, Franzetti et al. 2012) or without the need for individual recognition (Rowcliffe et al. 2008) used on wild boars by Passon et al. (2012). Some of these methods have low accuracy whilst others require significant investment in time and money.
Faecal pellet group counting is one of the most common (Neff 1968, Hemami and Dolman 2005) and the most accurate (Barnes 2002, Campbell et al. 2004) methods of abundance estimation in free-living animals. The method originated in the late 1930s (Neff 1968), and there are several modifications now widely used and considered as both reliable and economic. Furthermore, the technique has also been successfully applied to non-ruminant species including the snowshoe hare (Prugh and Krebs 2004) and red fox (Webbon et al. 2004).
Faecal pellet group counting has been neglected so far in wild boar population density estimation principally for three reasons: (1) uneven distribution of wild boar faeces in the environment, (2) the lack of data on its defecation rate and (3) low defecation rate in comparison to that of ruminants.
The distribution of wild boar faeces is influenced by the tendency of wild boars to predominantly defecate in places where they feel safe and during eating (Musial et al. 1999). Therefore, it is not possible to use FPG count to determine the distribution of wild boars across the habitats that they utilize, including their preferential biotopes, or to accurately estimate their abundance in small areas. Nevertheless, FPG counts could be employed to census wild boar populations in larger areas under certain conditions. Generally, it is not possible to determine the daily defecation rate in free-living animals (Košnář and Rajnyšová 2012); therefore, defecation rate is mostly determined experimentally using animals held in captivity. These experiments simulate natural feeding conditions using food sources that would be available to free-living animals. However, both the quality and quantity of food accessible to free-living wild boars vary markedly throughout the year (Massei et al. 1996, Schley and Roper 2003); hence, the defecation rate can also fluctuate. Food supply can be markedly affected by supplementary feeding and baiting (Geisser and Reyer 2005) whilst seasonality can affect the results of defecation decomposition rate experiments. Reduced levels of vegetative cover associated with the winter period simplify FPG observations (Campbell et al. 2004) whilst an absence of dung beetle activity (Massei et al. 1998) prolongs the availability of FPGs for counting, making it simpler to undertake this type of work than would be the case at other times of the year.
The main aim of this study was to assess the applicability of FPG counting to estimate a wild boar population in winter. To achieve this, we undertook, firstly, an analysis of wild boar FPG distribution in the forest environment and, secondly, an assessment of wild boar defecation rate.
Study area
The study area of 22.56 km2 is situated in the Drahanská vrchovina highlands in the south-eastern part of the Czech Republic, at an average altitude of 450 m a.s.l. (Fig. 1). The selected area is completely afforested and is enclosed within natural borders and man-made barriers that limit the migration of wild boars. From the south and the west, it is surrounded by extensive areas of agricultural land and a water basin and from the north and the east by a busy public road. Although the fields and the road do not prevent migration of wild boars, the movement of wild boars from the study area during winter is believed to be extremely limited. During winter, the wild boars were induced to remain in the study area by placing supplementary feed on permanent sites whilst the surrounding areas remained covered with deep snow without any food and cover. For a detailed description of the study area, see Plhal et al. (2011).
Material and methods
Defecation rate
A separate trial aimed to determine the daily defecation rate of wild boars was conducted in two enclosures of approx. 800 m2 near the study area, with very similar natural and climatic conditions, in the period from December 2010 to February 2011. The first enclosure (enclosure 1) contained two tame adult 3-year-old boars (one boar, one sow). Approximately half of the area of the enclosure was covered with a 30-year-old pine stand, the other half with a thin stand of mature broadleaves with an associated understorey of thin undergrowth. In the second enclosure (enclosure 2), 6-month-old piglets were kept (Table 1). The entire area of enclosure 2 was situated in a mature broadleaved stand without undergrowth. Throughout the experiment, the animals in both enclosures were offered a limited amount of a mix of cereal grains in order to simulate the feeding conditions of the free-living wild boars in the Czech Republic that feed mainly on cereals at feeding stations (Plhal et al. 2011). Natural sources of food were not available in either of the enclosures, and all the animals had free access to water. The experiment was carried out during optimal climatic conditions when dry frosty weather prevented destruction of the faeces by decomposition, dilution, burying or trampling.
To estimate the defecation rate, we firstly removed all the faeces from the enclosures; after a set period, we counted all the faecal pellet groups found and then removed all the faeces again to allow a repeat of the process. The period of exposition had to take account of the number of animals present in order to prevent destruction of FPGs by trampling or their overlaying in places with faeces accumulation. In the enclosure with adult boars, the period of exposition was 3–6 days; in the enclosure with piglets, it was 1–2 days (Table 1).
The daily defecation rate of one animal was determined by the equation:
- DR:
-
Daily defecation rate (number of FPGs per animal per day)
- N :
-
Number of FPGs in the enclosure
- a :
-
Accumulation period (days)
- x :
-
Number of individuals in the enclosure
Subsequently, we calculated the mean defecation rate from all the counts conducted, separately for piglets and for adult boars. To estimate the wild boar population density in the study area, we used the mean defecation rate of wild boars, as the weighted average in relation to the share of piglets and adults in our population, which was 1.0:1.3 (Plhal et al. 2011). Experiments were carried out in accordance with applicable legislation in the Czech Republic.
Population density estimation by FPG count
The study area was divided into seven basic types of habitat (clearing, young broadleaved growth, young coniferous growth, broadleaved pole stand, coniferous pole stand, mature broadleaved stand, mature coniferous stand). The percentage of each habitat in the total area was estimated from forestry records and a field inspection (Table 2). Ten localities, evenly distributed over the study area, were chosen for FPG counting. Data were collected in all seven types of habitat at each of the ten localities (Fig. 1), for 6 days immediately after snowmelt in spring (18–23 March 2010). In each habitat type, we counted FPGs on eight single-sampling uncleared strip transects of 100 × 2 m (200 m2). Hence, we sampled 56 transects at each of the ten localities (560 transects in total) which equate to 0.5 % of the whole study area. Eighty transects were situated in each of the seven types of habitat. The period of faeces exposition, chosen with respect to the local climatic and natural conditions, lasted from 1 November 2009 until the day of counting, i.e. 137–143 days (140 days on average).
For each type of habitat, we calculated the mean number of pellet groups for the whole area it represented. Consequently, the absolute number of wild boars was estimated separately for each habitat according to the equation:
- PD i :
-
Population density for habitat i (individuals (ind.)/km2)
- x i :
-
Number of FPGs on transects in habitat i
- AP:
-
Accumulation period
- DR:
-
Daily defecation rate
- P i :
-
Total area of transects in habitat i (km2)
As the period of faeces accumulation fell within the intensive hunting season, the result is biased by the fact that the calculation includes FPGs produced by the animals subsequently removed during the winter. To achieve a more accurate estimation of wild boar abundance at the time of the census, we subtracted the number of FPGs equivalent to the number of the shot individuals from the total number of the recorded FPGs. The number of pellet groups produced by the shot individuals was determined from the defecation rate and the date of shooting of the individual animals according to the equation:
- x :
-
Number of FPGs produced by the shot animals within the study area
- n :
-
Number of events (140 days)
- DR1 :
-
Defecation rate of piglets
- DR2 :
-
Defecation rate of adults
- a k :
-
Number of shot piglets on a specific day
- b k :
-
Number of shot adults on a specific day
Subsequently, the estimated total number of pellet groups produced by the shot individuals was divided proportionally between the individual habitats and subtracted from the number of FPGs found in each habitat. This corrected number of FPGs was used in the calculation of the final population density of wild boars in the study area at the time of the census.
Preference of habitats (P x ) was expressed with the Jacobs index (Hansen 1996):
where x1 is the number of faeces counted in habitat x, x2 is the size of habitat x, y1 is the total number of counted faeces and y2 is the size of the total area.
The index can take on values from −1 to +1, the positive values indicating positive selection and the negative values negative selection of the respective habitat. Statistical analysis was performed using SPSS statistical software.
Results
Defecation rate
Throughout the 32 days of the experiment, the wild boars produced 549 FPGs in total (adults, 180 groups; piglets, 369 groups; see Table 1). The average daily defecation rate of adults was 4.29 FPGs (3.00–5.75; n = 5); for piglets, it was 5.82 FPGs (5.08–6.37; n = 6). The difference between the defecation rate of adults and that of piglets was statistically significant (Mann-Whitney U test; p = 0.018). The modified average winter defecation rate of wild boars in the study area (weighted average reflecting the share of piglets and adult boars in the local population) was 4.95 FPGs/ind./day.
Population density estimation by FPG count
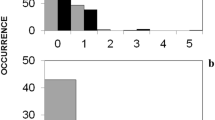
The distribution of FPGs on the transects of 200 m2 was uneven, and the percentage of sites with a zero value was high. Therefore, we pooled the data from every four adjacent transects (in each habitat at each locality, there were two transects of 800 m2). The resulting set of data showed normal distribution within the individual types of habitat (one-sample Kolmogorov-Smirnov test; p > 0.05; N = 20; for all cases), except for habitat 1 (p = 0.042). The density of faeces was uneven in the individual types of habitat (ANOVA; F 6–133 = 16.07; p < 0.001). The highest density of faeces was in young coniferous and broadleaved growths (habitats 2 and 3), as there were more FPGs than in other types of habitat (Games-Howell post hoc test; p < 0.05; for all cases). The lowest density of faeces was found in older stands, both broadleaved and coniferous (p < 0.05; Fig. 2). According to the index of preference based on defecation rate, wild boars preferentially selected young growth during winter (P x > +0.5) and, by contrast, avoided the mature stands (P x < −0.6); as for the other types of habitat, the animals did not show a preference for any of them (+0.05 > P x > −0.12).
Density and distribution of FPGs within seven types of forest habitat (for a description of the habitats, see Table 2). In the box are shown the median, quartiles, outlier (circle) and extreme (asterisk) values
Based on all the FPGs collected, the number of wild boars in the study area was estimated at 171 individuals (±59; 95 % confidence interval (CI); Table 2). During the period of faeces accumulation, 71 wild boars were shot in the study area in total, of which 62 were piglets and 9 were adults. From 1 November 2009 until being shot, these animals produced an estimated 23,640 FPGs, which make approx. 20 % of the total number of wild boar pellet groups found in the study area. After subtraction of the pellet groups left in the area by the shot boars, the number of individuals present at the time of the FPG count decreased to 137 (±36; 95 % CI). The corrected average density of wild boars in the whole area at the time of the census was 6.08 ind./km2 (±1.59; 95 % CI).
Discussion
The wild boar defecation rate determined in this study (4.95 FPGs) was very similar to the value 4.5 (Tottewitz et al. 1996), mentioned in the works of Cristescu and Ion (2007) and Heinze et al. (2011). The same value, 4.5, in average (varying from 3 to 6.25) is published by Briedermann (1986). Rather higher values are reported in the work of Musial et al. (1999) who had determined the number of defecations in experimental miniature pigs between 4.8 and 9.4 per individual and day. Similar results (ten defecations per individual and day) were found in Iberian pigs during the autumn mast period (Rodríguez-Estévez et al. 2005). Such high values, however, may have been caused by laboratory environment and feeding ad libitum.
A correct determination of the decomposition rate is very important when applying FPG counting to estimate wild boar population density. The values of decomposition rate can vary greatly depending on vegetation, air temperature, humidity, precipitation and dung beetle activity (Massei et al. 1998, Laing et al. 2003, Hemami and Dolman 2005). The most intensive decomposition occurs in the vegetative growing period and during full dung beetle activity in habitats with dense ground vegetation; in contrast, the longest decomposition has been observed in coniferous stands without ground vegetation (Tsaparis et al. 2009). When determining the faeces exposition period in broadleaved forest stands, attention must be paid also to the fact that older pellet groups get covered with autumnal leaf fall (Tsaparis et al. 2009). For this reason, we scheduled the beginning of the faeces exposition period in the study area for the time after leaf fall. Winter is an ideal period for faeces counting (Massei et al. 1998). During this season, faeces are subject to minimum decomposing factors and so they remain in the environment in an almost unchanged state and are suitable for counting before the start of the growing season. Massei et al. (1998) determined the period of wild boar faeces disappearance in winter in an area of Mediterranean climate to be approx. 120–240 days depending on the type of habitat.
The density of the wild boar population that we have estimated by FPG count in the study area was 6.08 ind./km2. This value corresponds to the values obtained from the same area and the same period determined by phototrapping (6.8 ind./km2) and by snow track count (6.3 ind./km2, Plhal et al. 2011). Additionally, the derived figures are very similar to the abundances of wild boars noted in other studies, i.e. in the Bialowieza National Park (3.5–5.9 ind./km2; Jedrzejewska et al. 1994) or in a similar forest environment, southwestern Germany (3.5 and 5.0 ind./km2; Ebert et al. 2012). Some other studies report a large variation range of population densities, i.e. in the forests of other European countries (1.5–12 ind./km2; Hebeisen et al. 2008) or in western Eurasia (0.01–10 ind./km2; Melis et al. 2006). The fact that FPG count produced results comparable to the other methods indicates that its application in optimum conditions (stable non-migrating population, sufficiently high population density of wild boars, sufficiently large area and persistence of faeces in the environment) is possible. An undeniable advantage of FPG count is that it does not require expensive equipment such as phototraps and does not rely on optimum snow conditions (in contrast to snow track counting) (Table 3).
The accuracy of wild boar abundance estimation by FPG count is significantly affected by an uneven distribution of faeces in different types of habitat; therefore, the deviation from the mean value is markedly higher than in the other methods (Table 3). This variation is caused by the fact that (1) wild boars defecate less often than ruminants, (2) they prefer places where they feel safe and (3) they often defecate together. Putten (2000) reports that wild boars never defecate directly in their resting area but only at a certain distance from it. The theory of uneven faeces distribution corresponds with the observation of Musial et al. (1999) that defecation of wild boars usually happens during food intake. This is the reason why, in wild boars, the distribution of FPGs is markedly more patchy than that in ruminant ungulate species (Abaigar et al. 1994), invoking the need to monitor the population on a sufficiently large area, to define the habitat categories precisely and to determine the percentage of each in the total area of the studied region. In our study area, the highest concentration of faeces was found in young stands that provided good shelter for wild boars (11.7–12.3 FPGs per 200 m2) and about ten times lower in places without undergrowth (1.15–1.95 FPGs per 200 m2). Our results correspond with previously published data. For example, Gerard et al. (1991), Lemel et al. (2003) and Keuling et al. (2008) report that during daylight, wild boars usually seek shelter and rest in young dense tree stands. Wild boars prefer mainly the beech-hornbeam and oak stands where they have the opportunity to feed on the mast crop (Abaigar et al. 1994, Fonseca 2008). On the other hand, Thurfjell et al. (2009) confirm that young boars favour young coniferous stands to broadleaved stands in winter in conditions very similar to those of our study area—including intensive additional feeding—except in a mast year. In our study area, there were very few natural sources of food for wild boars available at the time of data collection and so the animals depended upon the food from feeding stations (Plhal et al. 2011).
The significant variation in the spatial distribution of faeces could be turned into an advantage through considerable simplification of data collection. In our study, 82 % of wild boar defecations were localized on 50 % of the study area in habitats 1–5. Stands without undergrowth (habitats 6 and 7) formed one half of the area, but only 18 % of faeces were found there. Provided that the percentage of faeces found in the areas without shelter is constant, it would be possible to limit FPG counting to the localities where they are more abundant and to reduce the labour input associated with material collection markedly without loss of accuracy. However, the distribution of faeces will always depend on the specific environment, distribution of food sources and percentage and proximity of the areas providing shelter, all of which we would recommend considering when planning this method of population estimation.
Management implications
We have confirmed that FPG counting conducted in winter is an appropriate and applicable method to estimate wild boar abundance over a moderately large forested area where the animals are actively hunted. Moreover, FPG counting involves no expensive equipment and the level of training required to achieve competency compares favourably with that of other methods. To improve the accuracy of the population estimation, it is our opinion that the initial FPG counts require sample sizes obtained from as large an area as possible across a range of habitats to minimize the errors due to the uneven distribution of faeces. However, having verified the percentage of faeces found in areas devoid of undergrowth, it should be possible to count FPGs on significantly reduced areas (approximately half in our study area) without compromising population estimation accuracy. Further research is required to determine the distribution of faeces in different environments, to verify the defecation rate of wild boars in relation to the type of diet and to further test the accuracy of the wild boar abundance estimation by FPG counts compared to that of other methods.
References
Abaigar T, Delbarrio G, Vericad JR (1994) Habitat preference of wild boar (Sus scrofa L, 1758) in a Mediterranean environment—indirect evaluation by signs. Mammalia 58:201–210
Acevedo P, Quirós-Fernández F, Casal J, Vicente J (2014) Spatial distribution of wild boar population abundance: basic information for spatial epidemiology and wildlife management. Ecol Indic 36:594–600
Acevedo P, Vicente J, Höfle U, Cassinello J, Ruiz-Fons F, Gortazar C (2007) Estimation of European wild boar relative abundance and aggregation: a novel method in epidemiological risk assessment. Epidemiol Infect 135:519–527
Apollonio M, Andersen R, Putman R (2010) European ungulates and their management in the 21st century. Cambridge University Press, Cambridge
Barnes RFW (2002) The problem of precision and trend detection posed by small elephant populations in West Africa. Afr J Ecol 40:179–185
Braga C, Alexandre N, Fernandez-Llario P, Santos P (2010) Wild boar (Sus scrofa) harvesting using the espera hunting method: side effects and management implications. Eur J Wildl Res 56:465–469
Brauer A, Lange E, Kaden V (2006) Oral immunisation of wild boar against classical swine fever: uptake studies of new baits and investigations on the stability of lyophilised C-strain vaccine. Eur J Wildl Res 52:271–276
Briedermann L (1986) Schwarzwild. VEB Deutscher Landwirtschaftsverlag, Berlin
Campbell D, Swanson GM, Sales J (2004) Comparing the precision and cost-effectiveness of faecal pellet group count methods. J Appl Ecol 41:1185–1196
Cristescu B, Ion I (2007) Density of ungulates at preferential feeding in two hunting grounds of Vrancea County (Romania), with comparison between two study methods. Analele Stiintifice ale Universitatii “Al. I. Cuza” din Iasi Sectiunea Biologie Animala, Tom LIII:245–250.
Cruz F, Donlan CJ, Campbell K, Carrion V (2005) Conservation action in the Galapagos: feral pig (Sus scrofa) eradication from Santiago Island. Biol Conserv 121:473–478
Ebert C, Knauer F, Spielberger B, Thiele B, Hohmann U (2012) Estimating wild boar Sus scrofa population size using faecal DNA and capture-recapture modelling. Wildl Biol 18:142–152
Engeman RM, Massei G, Sage M, Gentle MN (2013) Monitoring wild pig populations: a review of methods. Environ Sci Pollut Res 20:8077–8091
Focardi S, Isotti R, Pelliccioni ER, Iannuzzo D (2002) The use of distance sampling and mark-resight to estimate the local density of wildlife populations. Environmetrics 13:177–186
Fonseca C (2008) Winter habitat selection by wild boar Sus scrofa in southeastern Poland. Eur J Wildl Res 54:361–366
Fonseca C, Alves J, Vingada J, Soares AMVM (2011) Reproductive performance of wild boar females in Portugal. Eur J Wildl Res 57:363–371
Fonseca C, Kolecki M, Merta D, Bobek B (2007) Use of line intercept track index and plot sampling for estimating wild boar, Sus scrofa (Suidae), densities in Poland. Folia Zool 56:389–398
Franzetti B, Ronchi F, Marini F, Scacco M, Calmanti R, Calabrese A, Paola A, Paolo M, Focardi S (2012) Nocturnal line transect sampling of wild boar (Sus scrofa) in a Mediterranean forest: long-term comparison with capture-mark-resight population estimates. Eur J Wildl Res 58:385–402
Fruzinski B, Labudzki L (2002) Management of wild boar in Poland. Z Jagdwiss 48:201–207
Geisser H, Reyer HU (2005) The influence of food and temperature on population density of wild boar Sus scrofa in the Thurgau (Switzerland). J Zool 267:89–96
Geisser H (1998) The wild boar (Sus scrofa) in the Thurgau (Northeastern Switzerland): population status, damages and the influence of supplementary feeding on damage frequency. Gibier Faune Sauvage 15:547–554
Gerard JF, Cargnelutti B, Spitz F, Valet G, Sardin T (1991) Habitat use of wild boar in a French agroecosystem from late winter to early summer. Acta Theriol 36:119–129
Gethöffer F, Sodeikat G, Pohlmeyer K (2007) Reproductive parameters of wild boar (Sus scrofa) in three different parts of Germany. Eur J Wildl Res 53:287–297
Hansen K (1996) Impact of modern farming on food supply of hares (Lepus europaeus, Pallas) during the summer period. Proceedings of the Ist European Congress of Mammalogy. Museu Bocage, Lisboa:115–124.
Hebeisen C, Fattebert J, Baubet E, Fischer C (2008) Estimating wild boar (Sus scrofa) abundance and density using capture-resights in Canton of Geneva, Switzerland. Eur J Wildl Res 54:391–401
Heinze E, Boch S, Fischer M, Hessenmoller D, Klenk B, Muller J, Prati D, Schulze ED, Seele C, Socher S, Halle S (2011) Habitat use of large ungulates in northeastern Germany in relation to forest management. For Ecol Manag 261:288–296
Hemami MR, Dolman PM (2005) The disappearance of muntjac (Muntiacus reevesi) and roe deer (Capreolus capreolus) pellet groups in a pine forest of lowland England. Eur J Wildl Res 51:19–24
Herrero J, García-Serrano A, Couto S, Ortuno VM, García-González R (2006) Diet of wild boar Sus scrofa L. and crop damage in an intensive agroecosystem. Eur J Wildl Res 52:245–250
Hladíková B, Zbořil J, Tkadlec E (2008) Population dynamics of the Wild Boar (Sus scrofa) in central Moravia, Czech Republic (Artiodactyla: Suidae). Lynx 39:55–62
Jedrzejewska B, Okarma H, Jedrzejewski W, Milkowski L (1994) Effects of exploitation and protection on forest structure, ungulate density and wolf predation in Bialowieza Primeval Forest, Poland. J Appl Ecol 31:664–676
Keuling O, Baubet E, Duscher A, Ebert C, Fischer C, Monaco A, Podgórski T, Prevot C, Ronnenberg K, Sodeikat G, Stier N, Thurfjell H (2013) Mortality rates of wild boar Sus scrofa L. in central Europe. Eur J Wildl Res 59:805–814
Keuling O, Stier N, Roth M (2008) Annual and seasonal space use of different age classes of female wild boar Sus scrofa L. Eur J Wildl Res 54:403–412
Killian G, Miller L, Rhyan J, Doten H (2006) Immunocontraception of Florida feral swine with a single-dose GnRH vaccine. Am J Reprod Immunol 55:378–384
Košnář A, Rajnyšová R (2012) Assessment of a daily defecation rate of fallow deer upon a closed population study. Acta Univ Agric Et Silvic Mendel Brun 60:173–180
Laing SE, Buckland ST, Burn RW, Lambie D, Amphlett A (2003) Dung and nest surveys: estimating decay rates. J Appl Ecol 40:1102–1111
Lemel J, Truve J, Soderberg B (2003) Variation in ranging and activity behaviour of European wild boar Sus scrofa in Sweden. Wildl Biol 9:29–36
Massei G, Bacon P, Genov PV (1998) Fallow deer and wild boar pellet group disappearance in a Mediterranean area. J Wildl Manag 62:1086–1094
Massei G, Genov PV, Staines BW (1996) Diet, food availability and reproduction of wild boar in a Mediterranean coastal area. Acta Theriol 41:307–320
Mayle BA, Peace AJ, Gill RMA (1999) How many deer? Forestry Commission, Edinburgh
Melis C, Szafrańska PA, Jedrzejewska B, Bartoń K (2006) Biogeographical variation in the population density of wild boar (Sus scrofa) in western Eurasia. J Biogeogr 33:803–811
Merli E, Meriggi A (2006) Using harvest data to predict habitat-population relationship of the wild boar Sus scrofa in Northern Italy. Acta Theriol 5:383–394
Musial F, Kowalski A, Enck P, Kalveram KT (1999) A computer-controlled, long-term recording system for studying eating, drinking, and defecation behavior in miniature pigs. Physiol Behav 68:73–80
Neff DJ (1968) Pellet-group count technique for big game trend census and distribution—review. J Wildl Manag 32:597–614
Passon Ch, Keuling O, Neubauer D, Gräber R, Brün J (2012) Estimating wild boar (Sus scrofa) density using camera traps and distance sampling. In: Book of abstracts—9th International Symposium on Wild Boar and Other Suids, 2–6 September 2012 Hannover, Germany, pp 9
Plhal R, Kamler J, Homolka M, Adamec Z (2011) An assessment of the applicability of photo trapping to estimate wild boar population density in a forest environment. Folia Zool 60:237–246
Prugh LR, Krebs CJ (2004) Snowshoe hare pellet-decay rates and aging in different habitats. Wildl Soc B 32:386–393
Putten G (2000) An ethological definition of animal welfare with special emphasis on pig behaviour. In: Hovi M, Garcia TR (eds) Diversity of livestock systems and definition of animal welfare. Proceedings of the Second NAHWOA Workshop. Palacio de la Merced in Córdoba, 8–11 January 2000, Córdoba, Spain, pp 120–134
Rodríguez-Estévez V, Martínez A, Gomez AG, Mata C (2005) Feeding activity and other activities during the grazing journey of the Iberian pig in the Montanera fattening period. In: Proceedings of EAAP—56th Annual Meeting. European Association for Animal Production, 5–8 June 2005. Uppsala, Sweden, p 239
Rowcliffe JM, Field J, Turvey ST, Carbone C (2008) Estimating animal density using camera traps without the need for individual recognition. J Appl Ecol 45:1228–1236
Schley L, Roper TJ (2003) Diet of wild boar Sus scrofa in Western Europe, with particular reference to consumption of agricultural crops. Mammal Rev 33:43–56
Sweitzer RA, Van Vuren D, Gardner IA, Boyce WM, Waithman JD (2000) Estimating sizes of wild pig populations in the north and central coast regions of California. J Wildl Manag 64:531–543
Thurfjell H, Ball JP, Ahlen PA, Kornacher P, Dettki H, Sjoberg K (2009) Habitat use and spatial patterns of wild boar Sus scrofa (L.): agricultural fields and edges. Eur J Wildl Res 55:517–523
Tottewitz F, Stubbe C, Ahrens M, Dobias K, Goretzki J, Paustian KH (1996) Counting droppings as a method of estimating the population of ruminant game. Z Jagdwiss 42:111–122
Tsaparis D, Katsanevakis S, Ntolka E, Legakis A (2009) Estimating dung decay rates of roe deer (Capreolus capreolus) in different habitat types of a Mediterranean ecosystem: an information theory approach. Eur J Wildl Res 55:167–172
Waithman JD, Sweitzer RA, Van Vuren D, Drew JD, Brinkhaus AJ, Gardner IA (1999) Range expansion, population sizes, and management of wild pigs in California. J Wildl Manag 63:298–308
Webbon CC, Baker PJ, Harris S (2004) Faecal density counts for monitoring changes in red fox numbers in rural Britain. J Appl Ecol 41:768–779
West BC, Cooper AL, Armstrong JB (2009) Managing wild pigs: a technical guide. Hum Wildl Interact Monogr 1:1–55
Acknowledgments
This study was supported by the Ministry of Education, Youth and Sports of the Czech Republic—project number: MSM 6215648902. We would like to thank the members of the Hunting Club in Prace and staff of the Game Preserve Soutok for their support during the field work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Dries Kuijper
Rights and permissions
About this article
Cite this article
Plhal, R., Kamler, J. & Homolka, M. Faecal pellet group counting as a promising method of wild boar population density estimation. Acta Theriol 59, 561–569 (2014). https://doi.org/10.1007/s13364-014-0194-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13364-014-0194-9