Abstract
The intrahepatic biliary cystadenoma is a rare benign tumor of the liver, originating from an intrahepatic bile duct: it becomes symptomatic only when it causes obstruction of the bile duct itself. Regardless of the various diagnostic modalities available, it is difficult to distinguish preoperatively the cystadenoma both from a simple liver cyst, and from a cystic carcinoma of the bile duct. An incomplete surgical removal of the cyst often results in a higher risk of size increase and recurrence, even considering that the lesion may degenerate into a cystadenocarcinoma. Between January 2004 and May 2011, 1,173 liver resections were carried out at the Hepatobiliary Surgery Unit of San Raffaele Hospital: 12 of these were performed for cystadenoma. Forty-six patients underwent laparoscopic liver cysts deroofing: definitive histological examination in six of these patients revealed instead the diagnosis of cystadenoma. In 50% of cases, the diagnosis of cystadenoma was therefore acquired as a result of an incidental finding. The patients were all female, median age 45 years. The liver resection included six cases of left hepatectomy, three left lobectomies, and three of the right hepatectomy. The operations were performed by laparotomy, with the exception of two left lobectomies completed laparoscopically. In all cases, the postoperative course was without major complications. The resection was radical in all cases and the median hospital stay was 5 days. At a median follow-up of 16 months (range 7–30), all patients are alive and disease free. Biliary cystadenomas can easily be misunderstood and interpreted as simple hepatic cysts. Radical surgical resection is necessary and provides good short- and long-term outcomes.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Biliary cystadenoma (CAB) is a rare cystic tumor arising within the liver parenchyma or, more rarely, along the extrahepatic bile ducts making up about 5% of cystic liver disease [1]. It occurs more frequently in adult females, in the form of an unique and large cyst, with a multi-lobulated surface outside and internal septa [2, 3]. The presence of mesenchymal stromal tissue within the cyst is a risk factor for the degeneration of the CAB into cystadenocarcinoma (BCAC) [4, 5].
The differential diagnosis may be often difficult, especially with other cystic liver diseases, such as simple cysts and hydatid cysts, but it is essential since the treatment of CAB requires a radical liver resection to prevent the risk of size increasing (associated with complications such as rupture and infection), recurrence and malignant degeneration [6]. The diagnosis of CAB and BCAC, however, is often incidental, being acquired during histological examination performed after laparoscopic or laparotomic fenestration of liver cysts [3].
This study reports the series of CAB in a single center, analyzing the short- and long-term outcomes of liver resection in patients with this pathology, and in particular in patients with an incidental diagnosis of CAB.
Materials and methods
Patients
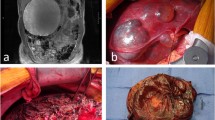
From January 2004 to May 2011, 1,173 liver resections were performed at the Hepatobiliary Surgery Unit of San Raffaele Hospital in Milan. Data about these patients were prospectively collected in a database and are now retrospectively reviewed. Eighty-eight patients underwent surgery for cystic liver disease: final histological examination revealed simple cysts in 49 cases (46 patients operated by laparoscopy and 3 by laparotomy), hydatid cyst in 27 cases (all by laparotomy) and CAB in 12 cases (Fig. 1). This latter group of patients is the subject of this study.
In 6 of the 12 patients with CAB, detection of the disease was an incidental finding at the histological examination performed after laparoscopic deroofing of cysts that were considered simple cysts at the preoperative imaging.
Patients were referred to our attention for non-specific abdominal pain, epigastric heaviness and in two cases for palpation of an epigastric mass during self-examination. No patient presented jaundice or other complications at diagnosis.
Preoperative investigations
All patients were assessed by ultrasound of the abdomen, contrast-enhanced abdominal CT scan and in four cases MRI of the upper abdomen. In the six patients with incidental CAB (none of whom had undergone MR), imaging studies were in agreement in the preoperative diagnosis of simple hepatic cysts: all of these patients were then re-evaluated with additional CT study after histological founding.
Radiological CT findings in each patient are summarized in Table 1.
In all patients, routine blood tests were also performed, including the dosage of anti-echinococcus antibody titers and tumor markers (CEA and GICA). Chemical and physical evaluation or markers dosage on the cystic fluid were not performed.
Outcome
The short-term outcome was evaluated in terms of postoperative morbidity and mortality, operative time, blood loss, intraoperative and postoperative transfusions, use of portal clamping, duration of hospital stay, surgical margins, postoperative functional recovery (resumption of oral nutrition and ductwork, suspension of infusion therapy and pain relief, walking independently). The follow-up was performed 1 month after surgery with outpatient visits and through CT re-evaluation CT after 3–6 months. The long-term outcome was evaluated in terms of disease recurrence.
Statistical analysis
Retrospective analysis was performed from a prospectively collected database of patients. The average values are expressed as mean ± standard deviation from the mean or as median and range when appropriate. Statistical analysis was performed using SPSS software version 18.0 (SPSS Inc., Chicago, IL). The difference was considered significant when the P value was <0.05.
Results
The patients were all female, with a mean age of 45 years (±9.4) and, in most cases had an ASA score of 2. Blood tests showed no significant alterations: in particular, in all patients the dosage of anti-echinococcus antibody titer was negative and tumor markers were normal.
In all six cases of incidental diagnosis of CAB, the patients had undergone laparoscopic deroofing of cysts with no postoperative complications and a median hospital stay of 2 days (range 1–3). In two cases, extemporaneous histological examination that had been performed intraoperatively resulted suggestive for simple liver cysts. The median interval between the first and second surgery was 31 days (range 20–45).
Six patients underwent left hepatectomy, three left lateral sectionectomy, and three right hepatectomy. The operations were performed by laparotomy, except for two left lateral sectionectomies carried out laparoscopically in patients with incidental cystadenoma and who had therefore already undergone laparoscopic cyst deroofing.
The mean length of surgery was 209 ± 78 min. In particular, there were no significant differences in terms of duration of surgery among patients with incidental cystadenoma and the others. Mean intraoperative blood loss was 260 ± 130 mL. No patient required intra-or postoperative blood transfusion. Portal clamping (Pringle maneuver intermittently) was performed in 9 of 12 patients for a mean duration of 30 ± 10 min.
Postoperative mortality was nil. Two patients developed postoperative minor complications: one developed a surgical wound infection and the second a wound infection and a urinary tract infection, which resolved after antibiotic therapy. There were no major complications.
The median hospital stay was 5 days (range 4–8). The functional recovery occurred at a median of 4 days after surgery, except for patients operated by laparoscopy who had an earlier functional recovery, in the third postoperative day.
Data regarding patients and surgery characteristics are summarized in Table 2.
Histological examination confirmed the diagnosis of cystadenoma in 12 cases. The surgical resection margins were negative in all cases.
At a median follow-up of 16 months (range 7–30), no patient showed disease recurrence or presence of symptoms.
Discussion
The number of CAB and BCAC reported over a long period is small, so that identification of pathognomonic clinical, biochemical and radiologica characteristics is difficult. A literature review of last published series is reported in Table 3.
Biliary cystadenoma occurs more frequently in the form of an intrahepatic lesion. Instead it originates from the biliary tree in <10% of cases. The origin of these lesions is probably due to proliferation of ectopic embryonic tissue, with histological features of ovarian stroma [4, 5]. The CAB consists of multiloculated cystic lesions with an epithelial lining composed of biliary-type columnar or cuboidal non-ciliated cells, surrounded by stromal tissue that often (85–90%) mimics the ovarian tissue. The presence of baseline membrane invasion allows the diagnosis of BCAC [1–3].
Presenting symptoms are often abdominal pain and the sense of occupation, caused by the mass forming effect of the disease. Clinical and biochemical signs of jaundice and cholangitis suggest instead the presence of biliary obstruction, secondary to the lesion itself and to mucin production. Most lesions occur in adult women, in the absence of identified risk factors, although a hypothetical hormonal factor may be implied in the pathogenesis [7].
Lesions tend to have a slow growth pattern, but are considered precancerous lesions with a malignant transformation rate of about 30% [8].
The diagnosis is based primarily on radiological features, but it is often difficult because other nosological entities enter the differential diagnosis with the CAB as they have similar characteristics. The differential diagnosis includes simple hepatic cysts, the cystic hydatid lesions, liver abscesses, malignant cystic lesions (BCAC above all), Caroli’s disease, post-traumatic cysts, and polycystic disease. Obtaining a conclusive diagnosis is critical because treatment modalities are very different depending on the histology of the cystic lesion, even considering that any approach that does not provide a complete removal of the CAB exposes the patient to a high risk of malignant transformation (30%) and disease recurrence (90%) [9].
The laboratory findings may help in the differential diagnosis of infectious cystic disease (serum level of anti-Echinococcus and anti-Amoeba antibodies); on the contrary, tumor markers are usually normal in cases of CAB [10] and cannot be used to distinguish CAB from simple cysts, indeed none of the patients in our series showed high levels of CEA, CA 19.9 or α-fetoprotein. Serum levels of CEA and Ca 19.9 may be increased in patients with BCAC, but an accurate differential diagnosis between CAB and BCAC can be done only after histological examination since reliable pathognomonic signs are lacking.
Some authors report an analysis of the role of Ca 19.9 and CEA levels within the cyst fluid, in particular Koffron et al. [11] reported high levels of Ca 19.9 and CEA in cystic fluid obtained with a laparoscopic approach in all 22 patients with cystadenoma of his series11.
Radiological images are an important diagnostic tool, both the abdomen ultrasound and the CT scan, and possibly MRI. Typical characteristics include the presence of multiloculated cystic lesions with internal septa, thickened and irregular surface and wall nodules of papillary protrusions, calcification and enhancement of the wall. Ultrasound is usually more sensitive to the presence of septa, while the CT is necessary in order to delineate the anatomic relationships of the lesion within the liver parenchyma [12, 13].
The above criteria are not exclusive for the CAB, however, and this accounts for the difficulty in differential diagnosis. In 50% of cases of this series, combining different imaging studies allowed to suspect the diagnosis of cystadenoma preoperatively, while in the remaining 50% CAB diagnosis was incidental in patients with a false diagnosis of simple cyst (false-negatives for CAB): indeed, this latter group of patients had no one of the above cited CT characteristics. In contrast, Thomas et al. [14] reported a 95% accuracy in diagnosis of CAB using only CT scan: in the same study, however, cases of false-positive are not reported and the diagnostic criteria of CAB were not indicated14.
Percutaneous biopsy for the purpose of histological characterization of liver cystic lesions is not considered among standard diagnostic procedures in our Center because of limited diagnostic value of a sample for cytology or micro histology in the diagnosis of CAB.
Two authors reported the role of extemporaneous histological examination in the intraoperative diagnosis of CAB [15, 16]. Vogt et al. [15], in particular, report the case of a patient with a histological diagnosis of simple cyst performed on six samples taken during laparoscopic cyst deroofing: the definitive histological diagnosis was instead reported to be of CAB15.
In the present series, two patients underwent laparoscopic deroofing of liver cyst with a frozen section histological examination, which resulted suggestive for simple cyst: on the contrary, the final examination revealed the diagnosis of CAB. There is no data in the literature regarding the actual reliability of the frozen section examination and how this tool can help when the diagnosis is uncertain, but the impression is that the examination cannot be taken into account as diriment for diagnosis. This fact explains the high incidence of incidental CAB and certainly more efforts should be made to develop an instrument or diagnostic criteria to make this diagnosis more reliable. The radical excision of the tumor is the treatment of choice for both the CAB and the BCAC. Any more conservative treatment is burdened by the risk of local recurrence, malignant degeneration and complications, as demonstrated in all studies of literature. The removal of the disease makes it necessary, in most cases, a formal liver resection [7, 15], even if a more conservative approach by lesion enucleation may be taken into account if negative margins can be achieved.
In patients with diagnosis of CAB, the prognosis is very good after complete removal, as demonstrated in this series, with excellent results in the long-term. Long-term outcome in patients affected by BCAC is worse and is comparable to that of intrahepatic cholangiocarcinoma with a poor long-term survival, mainly affected by neoplasm grading, lymph node status and resection margin [7, 16].
In conclusion, CAB is frequently misdiagnosed at primary observation and treated inadequately by deroofing as simple cysts of the liver. Even intraoperative diagnosis on frozen section is difficult. On the other hand, incomplete resection of CAB carries the risk of recurrence or development of BCAC. Therefore, whenever the diagnosis of CAB is suggested by preoperative studies, radical resection instead of deroofing is strongly suggested; in all other cases, a postoperative diagnosis of CAB after partial resection (deroofing) has to be considered, thus requiring a second surgery for radical resection. All patients undergoing surgery for simple cysts of the liver have to be informed about the potential final diagnosis of CAB with the resulting need of further surgery.
References
Davies W, Chow M, Nagorney D (1995) Extrahepatic biliary cystadenomas and cystadenocarcinomas: report of seven cases and review of the literature. Ann Surg 222:619–625
Dixon E, Sutherland FR, Mitchell P, McKinnon G, Nayak V (2001) Cystadenomas of the liver: a spectrum of disease. Can J Surg 44:371–376
Fiamingo P, Veroux M, Cillo U, Basso S, Buffone A, D’Amico DF (2004) Incidental cystadenoma after laparoscopic treatment of hepatic cysts: which strategy? Surg Laparosc Endosc Percutan Tech 14:282–284
Ishak KG, Willis GW, Cummins SD, Bullock AA (1977) Biliary cystadenoma and cystadenocarcinoma. Report of 14 cases and review of the literature. Cancer 39:322–328
Subramony C, Herrera GA, Turbat-Herrera EA (1993) Hepatobiliary cystadenoma: a study of five cases with reference to histogenesis. Arch Pathol Lab Med 117:1036–1042
Delis SG, Touloumis Z, Bakoyiannis A, Tassopoulos N, Paraskeva K, Athanassiou K, Safioleas M, Dervenis C (2008) Intrahepatic biliary cistoadenoma: a need for radical resection. Eur J Gastroenterol Hepatol 20:10–14
Thomas KT, Welch D, Trueblood A, Sulur P, Wise P, Gorden L, Chari RS, Wright JK, Washington K, Pinson W (2005) Effective treatment of biliary cistadenoma. Ann Surg 241:769–775
Devaney K, Goodman ZD, Ishak KG (1994) Hepatobiliary cystadenoma and cystadenocarcinoma: a light microscopic and immunohistochemical study of 70 patients. Am J Surg Pathol 18:1078–1091
Teoh AYB, Ng SSM, Lee KF, Lai PBS (2006) Biliary cistadenoma and other complicated cystic lesions of the liver: diagnostic and therapeutic challenges. World J Surg 30:1560–1566
Lee JH, Chen DR, Pang SC, Lai YS (1996) Mucinous biliary cystadenoma with mesenchymal stroma: expressions of CA 19–9 and carcinoembryonic antigen in serum and cystic fluid. J Gastroenterol 31:732–736
Koffron A, Rao S, Ferrario M, Abecassis M (2004) Intrahepatic biliary cystadenoma: role of cyst fluid analysis and surgical management in the laparoscopic era. Surgery 136:926–936
Mortele KJ, Ros PR (2001) Cystic focal liver lesions in the adult: differential CT and MR imaging features. Radiographics 21:895–910
Palacios E, Shannon M, Solomon C, Guzman M (1990) Biliary cystadenoma: ultrasound, CT, and MRI. Gastrointest Radiol 15:313–316
Thomas KT, Welch D, Trueblood A, Sulur P, Wise P, Gorden DL, Chari RS, Wright JK Jr, Washington K, Pinson CW (2005) Effective treatment of biliary cystadenoma. Ann Surg 241:769–775
Vogt DP, Henderson JM, Chmielewski E (2005) Cystadenoma and cystadenocarcinoma of the liver: a single centre experience. J Am Coll Surg 200:727–733
Hansman MF, Ryan JA, Holmes JH, Hogan S, Lee FT, Kramer D, Biehl T (2001) Management and long-term follow-up of hepatic cysts. Am J Surg 181:404–410
Gadzijev E, Ferlan-Marolt V, Grkman J (1996) Hepatobiliary cystoadenomas and cystadenocarcinoma. Report of five cases. HPB Surg 9:83–92
Choi HK, Lee JK, Lee KH, Lee KT, Rhee JC, Kim KH, Jang KT, Kim SH, Park YP (2010) Differential diagnosis for intrahepatic biliary cystadenoma and hepatic simple cyst. Significance of cystic fluid analysis and radiologic findings. J Clin Gastroenterol 44:289–293
Erdogan D, Kloek J, Lamers WH, Offerhaus GJA, Busch ORC, Gouma DJ, van Gulik TM (2010) Mucinous cystadenomas in liver: management and origin. Dig Surg 27:19–23
Seo JK, Kim SH, Lee SH, Park JK, Woo SM, Jeong JB, Hwang JH, Ryu JK, Kim JW, Jeong SH, Kim YT, Yoon YB, Lee KU, Kim SH, Kim MA (2010) Appropriate diagnosis of biliary cystic tumors: comparison with atypical hepatic simple cysts. Eur J Gastroenterol Hepatol 22:989–996
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ratti, F., Ferla, F., Paganelli, M. et al. Biliary cystadenoma: short- and long-term outcome after radical hepatic resection. Updates Surg 64, 13–18 (2012). https://doi.org/10.1007/s13304-011-0117-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13304-011-0117-0