Abstract
Liver resection may represent the only hope of cure for patients with intrahepatic cholangiocarcinoma (IHC) but long-term results are still far from satisfactory and the impact of prognostic factors is still controversial. Fifty-five patients underwent hepatectomy for IHC between 1997 and 2008 in our unit. Features of the patients and the tumors, operations, postoperative and long-term results were retrospectively assessed. Twenty-one patients had HBV/HCV infection, four had congenital biliary dilatation. Thirty-two patients had increased CA 19-9; 12 had multiple (≥4) tumors. Operations included 43 major resections, with 9 resections of biliary confluence, 40 regional lymphadenectomies. Operative mortality and morbidity were 0 and 27.3%, respectively. There were 44 R0-resections (80.0%). Lymphadenectomy yielded lymph node metastases in 14 cases (14/40; 35.0%). Five-year overall and disease-free survival rates were 30.2 and 27.5%, respectively. At multivariate analysis the strongest poor prognostic factor for overall survival was tumor stage. This factor, with multiplicity of lesions (≥4) and tumor grading > 2, was significant predictor of recurrence. CA19-9 > 100 IU/mL and tumor grading > 2 were found to be significantly related with early multinodular hepatic recurrence. Patients with lymph node metastases had significantly lower overall and disease-free survival but patients who underwent lymph node dissection with negative lymph nodes at final pathology showed significantly higher 5-year disease-free survival than patients who did not underwent lymphadenectomy. In conclusion, these results support the role of hepatectomy with regional lymphadenectomy as the best available treatment for IHC. Prognosis after liver resection correlates with clinical stage and multiplicity of lesions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Intrahepatic cholangiocarcinoma (IHC) is the second most common primary liver neoplasm after hepatocellular carcinoma. It arises from the intrahepatic bile ducts, and should be clearly distinguished from other types of cholangiocarcinoma arising from other sites within the biliary system, such as the main biliary confluence [1]. Risk factors such as congenital intrahepatic bile duct dilatation, intrahepatic lithiasis and fluke infection, which are widespread conditions in Southeast Asia, render these tumors much more common in Eastern countries than in the West [2, 3]. IHC accounts for only 10–20% of primary hepatic malignancies and for 6% of all cholangiocarcinomas in Western countries, although an increasing incidence during the recent years has been observed [4, 5].
The tumor tends to remain asymptomatic for long periods of time, therefore the diagnosis is often late and the treatment accordingly ineffective. Although the loco-regional extension of IHC is usually advanced at the time of diagnosis, aggressive surgical resection, when feasible, offers the best chance of prolonged survival [6–10].
The aim of this study was to review a series of 55 consecutive patients who underwent liver resection for IHC in a tertiary care center, over a period of 12 years, to assess operative results, patterns of recurrence and prognostic factors.
Methods
Between 1997 and 2008, 55 patients underwent liver resection for IHC (male/female 30/25, median age 64 years, range 43–80). All patients underwent complete physical and clinical work-up, with assessment of operability. Laboratory data included serum levels of albumin, total bilirubin, alanine and aspartate aminotransferase, alkaline phosphatase (AP), gamma glutamyl transpeptidase (gamma-GT), prothrombin activity, CA 19-9, HBV and HCV markers. Abdominal ultrasound, abdominal and chest CT scan, upper gastrointestinal endoscopy and pancolonoscopy were almost routinely performed, to exclude primary extrahepatic tumors or metastases. The assessment included magnetic resonance cholangiography in cases with the suspicion of infiltrated right or left hepatic duct or main hepatic confluence. All the exams were evaluated in a weekly multidisciplinary meeting with radiologists and oncologists. The assessment never included preoperative biopsy, unless performed before referral. The tumors were morphologically classified according to the criteria of the Liver Cancer Study Group of Japan (LCSGJ) [11], and staged according to the TNM classification [12].
Liver resections were defined according to the International Hepato-Pancreato-Biliary Association terminology derived from Couinaud’s classification [13]. Resection of three or more liver segments was classified as major hepatectomy. The surgical technique for liver resection used in our unit was previously described [14, 15]. Intraoperative ultrasound was routinely used. Parenchymal dissection was performed by Kelly-clamp, CUSA (Cavitron Ultrasonic Surgical Aspirator System 200; Valleylab Inc, Boulder, CO) and wet bipolar forceps. During resection the central venous pressure was maintained between 1 and 3 mmHg. Dissection and looping of the hepatic veins were routinely performed in major resections.
Liver resection was associated with resection of the main biliary confluence in order to obtain R0 resection in patients with periductal infiltrating tumor of the right or left hepatic duct and in case of perihilar mass forming tumor.
Lymphadenectomy around the hepatoduodenal ligament was routinely performed from January 2003; previous indication to lymphadenectomy was related to preoperative radiologic evaluation or intraoperative finding.
Patients were divided into three groups: no lymphadenectomy (L−); patients who underwent lymphadenectomy with lymph node metastases at final pathology (L+ N+), and patients who underwent lymphadenectomy without lymph node metastases (L+ N−).
The criteria for blood transfusion during surgery were hemoglobin level < 9.0 g/dL and hematocrit < 28%, or in patients over 70 or with cardiac disease, hemoglobin < 10 g/dL and hematocrit < 30%. Postoperative liver insufficiency was defined as prothrombin activity < 50% and total bilirubin > 3.0 mg/dL at postoperative day 5 [16]. Prothrombin activity < 50% was a criterion for the infusion of fresh frozen plasma.
At final pathology, diagnosis of cholangiocarcinoma in the resected specimens was based on the positive expression of low molecular weight cytokeratin CK 7 and negative expression of CK 20 [17].
The follow-up was mostly based on the serum level of CA 19-9 and appropriate blood laboratory values, hepatic ultrasound, abdominal CT scan, telephone interview and evaluation in outpatient clinics. None of the patients was lost to follow-up.
The assessed risk factors for overall and disease-free survival included demographics, associated chronic hepatitis, blood chemistries, tumor morphology and size (or size of main lesion), number of lesions, execution of major resection, associated regional lymphadenectomy, associated resection of main biliary confluence, radicality of resection, lymph node involvement, tumor grading based on degree of differentiation and UICC tumor stage.
Statistical analysis
The Chi-square test was used when appropriate. Long-term results were assessed using the Kaplan–Meier method, the log-rank test and multivariate analysis with the calculation of hazard ratios (HR, Cox proportional hazards) and 95% confidence intervals (95% CI). A P value of <0.05 was considered significant.
The statistical analysis was done using SPSS® version 13.0 for Windows® (SPSS, Chicago, IL, USA).
Results
Patient characteristics
Twenty-eight patients (50.9%) were symptomatic and abdominal pain was the most frequent symptom (16 patients; 57.1%) (Table 1). In 14 patients (25.5%) tumor was diagnosed by ultrasound during follow-up for chronic hepatitis (10 patients), for previous rectal cancer (2 patients), for chronic pancreatitis (1 patient) and for primary intrahepatic lithiasis (1 patient submitted 71 months earlier to left-hepatectomy + caudate excision + excision of biliary confluence and right‐hepaticojejunostomy). In the remaining cases (13 patients; 23.6%) tumor diagnosis was completely casual (Table 1).
IHC was associated with chronic hepatitis in 22 patients (40.0%) and with congenital bile duct dilatation in 4 patients (7.3%) (Table 1).
Preoperative biopsy showing adenocarcinoma was performed before referral in 18 patients (32.7%).
AP was elevated in 23 cases (42.6%) and gamma-GT in 27 cases (50.0%).
CA 19-9 was elevated in 32 patients (59.3%), but greater than 100 IU/mL in 20 patients (37.0%). Increases in CA 19-9 did not correlate with size, number of tumors or tumor stages, although some of the highest CA19-9 values were observed in patients with advanced tumor stage.
Increasing incidence of IHC
There was a recent increase in liver resections for IHC in our unit. Until the year 2005, we only performed for IHC ≤ 4 liver resections per year, which corresponded to 3.5–4.0% of all liver resections. Thereafter the rate rapidly increased, exceeding 10 resections per year, reaching a percentage of 12.0% of all liver resections. The trend is presently reconfirmed. This did not seem to be simply coincidental, and was not associated with a similar increase in referrals for other malignancies, or with other obvious explanations.
Surgical procedures
Major liver resections were performed in 43 patients (78.2%) (Table 2).
Resection of the main biliary confluence was associated with liver resection in seven patients with periductal infiltrating tumor of the right or left hepatic duct and in two patients with perihilar mass forming tumor (Table 2).
The dissection of regional lymph nodes in the hepato-duodenal ligament was performed in 40 cases (72.7%).
Liver resections were performed under intermittent portal pedicle clamping in 44 cases (80.0%).
Operative results
Blood transfusions were administered in 25.5% of the patients (14 patients). The mean number of units per transfused case was 2 (range 1–7).
Operative mortality was nil. Postoperative complications occurred in 15 patients (27.3%), in 13 after major resection and in 2 after minor resection. Two patients required reoperation (3.6%): both for abdominal abscess after failure of percutaneous drainage.
Pathology
The tumors were classified as “mass forming” type in 47 patients, and as “periductal infiltrating” type in 8 cases (Table 3). Mean tumor size was 6.0 ± 3.0 cm (range 1–12). A single tumor was present in 35 patients (63.6%); two lesions were present in 8 patients, and four or greater than four lesions in 12 patients (no patient had three lesions). There were 44 R0-resections (80.0%) and 11 R1-resections. R1-resections occurred in four cases with a periductal infiltrating type tumor, and in seven cases with a mass forming type tumor. There were no R2 resections.
Lymphadenectomy yielded lymph node metastases in 14 cases (14/40; 35.0%) (Table 3).
Recurrence and reresection
Recurrence occurred in 34 patients (61.8%), with a median time to recurrence of 9 months (range 3–40). Thirteen patients (38.2%) showed an early recurrence (≤6 months), of whom six patients developed multinodular recurrence on the remnant liver.
Liver-only recurrence occurred in 29.4% of the cases (10/34 patients), six of whom underwent liver reresection; both hepatic and extrahepatic recurrence in 38.2% of the cases (13/34 patients) and only extrahepatic recurrence in 32.4% (11/34 patients).
Hepatic recurrence with or without extrahepatic recurrence occurred in 23 patients (67.6%).
Most common site of extrahepatic recurrence was locoregional lymph nodes (58.3%; 14/24 cases of extrahepatic recurrence).
Univariate and multivariate analyses of prognostic factors
After a mean follow-up of 28 months, 29 patients were alive, 21 of whom without recurrence.
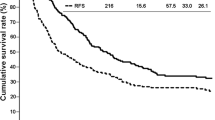
By the Kaplan–Meier method, overall survival after 12, 36 and 60 months was 83.6, 48.4 and 30.2%, with a median survival of 30 months; disease-free survival after 12, 36 and 60 months was 60.0, 31.4 and 27.5% with a median disease-free survival of 15 months (Fig. 1).
At univariate analysis, worse overall survival was significantly associated with tumor stage ≥ IIIC (P = 0.004), increased alkaline phosphatase (P = 0.005), tumor grading > 2 (P = 0.006), CA19-9 > 100 IU/mL (P = 0.012), lymph node metastases (P = 0.016), R1-resections and presence of jaundice (P = 0.020) and increased gamma glutamyl transpeptidase (P = 0.027) (Table 4).
Better overall survival, at univariate analysis, was observed in patients with chronic hepatitis, which was associated with earlier stages of tumor diagnosis (Table 4). Indeed, incidence of poor prognostic factors (multiplicity of tumors and tumor stage ≥ IIIC) was less frequent in patients with chronic hepatitis than in patients with normal liver (13.6 and 28.6% vs. 27.3 and 46.2%, respectively).
At univariate analysis, worse disease-free survival was significantly associated with multiplicity of tumors (>1 tumor, P = 0.033; ≥4 tumors, P = 0.001), tumor stage ≥ IIIC (P = 0.002), lymph node metastases (P = 0.012) and tumor grading > 2 (P = 0.036) (Table 4). The patients having two lesions showed no disease-free survival difference compared to patient with single tumor (P = 0.909); patients with ≥4 nodules showed significantly lower disease-free survival compared to patients with one and two nodules (P = 0.001) (Fig. 2).
Patients with early recurrence (≤6 months) were evaluated according to the most important potential risk factors for survival and recurrence (CA19-9 > 100; tumor grading > 2; multiplicity of tumors; lymph node metastases; tumor stage ≥ IIIC; R1 resections). None of these factors was significantly related with early recurrence. Conversely, early hepatic multinodular recurrence was significantly related with CA19‐9 > 100 and tumor grading > 2 (P = 0.025 and 0.015, respectively).
Role of lymphadenectomy was evaluated. Five-year disease-free survival was significantly different between the three groups of analyzed patients (38.6% in L+ N− group; 25.9% in L− group and 11.4% in L+ N+ group; P = 0.039) (Fig. 3).
At multivariate analysis the only strong poor prognostic factor for overall survival was tumor stage. This factor, with multiplicity of lesions (≥4) and tumor grading > 2 maintained significance at multivariate analysis for disease-free survival (Table 5).
Discussion
This article reports data on 55 consecutive patients submitted to liver resection for IHC in a tertiary care center.
Because of the advanced stage at which most patients with IHC usually present, a major hepatic resection is frequently required, often associated with additional procedures. In our patients major resection accounted for 78.2% of the operations. No mortality, low morbidity (27.3%) and a contained rate of R1 resection (20.0%) likely reflected the suitable preoperative selection of patients.
Our observation of steadily increasing referrals for IHC in recent years remains unexplained. This did not seem to be simply coincidental, and was not associated with a similar increase in referrals for other malignancies. This is very remarkable at the light of other reports of a global increase in incidence of IHC, related to the association with chronic liver disease, to the effect of industrial chemicals and pollutants or other factors, or to a reduced rate of misclassifications of IHC as metastatic adenocarcinoma or hilar cholangiocarcinoma [5, 18]. However, the large increase in incidence observed by us overrides these explanations, might be linked to specific environmental factors, and deserves very careful further assessment.
In this series the impact of prognostic factors after liver resection for IHC was evaluated.
The long-term results in our study showed that overall survival after 12, 36 and 60 months was 83.6, 48.4 and 30.2% with disease-free survival of 60.0, 31.4 and 27.5% (Fig. 1).
In our series median overall and disease-free survival were 30 and 15 months, respectively, which were higher than the 6–9 month survival reported for unresected patients [5, 19].
These outcomes allow the underscoring of an important issue in the treatment of IHC: the fact that surgery still represents the only hope of cure, while the results of surgery are far from satisfactory, as it also emerges from many published studies (Table 6) [5, 7, 9, 10, 20–37].
Some studies have recently identified HBV and HCV infections as risk factors for the development of IHC, likely related to chronic hepatic inflammation, but the evidence is not yet clear [38]. In our series IHC was associated with chronic HBV or HCV infection in 38.2% of patients. Interestingly, such patients had a better outcome. Indeed chronic hepatitis in these patients was more frequently associated with earlier tumor stage and single tumor. This was mostly related to the earlier tumor diagnosis allowed by the strict follow-up of patients with chronic hepatitis.
Differently from extrahepatic cholangiocarcinoma, jaundice is a very rare symptom upon admission. This occurs more likely with periductal infiltrating type IHC [1] and, in fact, this was the case in five of our patients who had involvement of the main biliary confluence. Moreover, jaundice is considered as a negative predictive factor, usually related to an advanced stage of disease, with bile duct hilar stricture [19]. In our series jaundice was a significant poor prognostic factor for overall survival. However, it should be emphasized that median overall survival after surgery in such patients was 14 months and that jaundice should not be considered as a contraindication for surgery.
Abdominal pain or discomfort, and anicteric cholestasis were the most common presenting features.
An increase in the enzymes of cholestasis, i.e. alkaline phosphatase and gamma-GT, was associated with poorer outcome, and this was not explained in all the cases by hilar extension of tumors or compression by large tumors. Increased CA 19-9 was observed in only 59.3% of the patients, without correlation with tumor size or number.
At multivariate analysis there was a prominent role of multiplicity of lesions, tumor grading and tumor stage in negatively affecting disease-free and overall survival. The impact of number of tumors on long-term survival after liver resection was analyzed in detail. Interestingly, 5-year disease-free survival rate of patients having two lesions was not significantly different from that of patients with a single tumor. In contrast, the UICC/AJCC classification divides these two groups of patients and discriminates only between single and multiple lesions. In our series the number of tumors ≥ 4 lesions was found to be an important prognostic factor for recurrence. On the contrary, tumor size was decidedly not a predictor for recurrence unless associated with multiple tumors.
This study demonstrates that histologic differentiation was a significant independent predictor of early recurrence. Thirteen patients (38.2%; 13/34 patients with recurrence) showed an early recurrence (≤6 months), and increased levels of CA19-9 (>100 IU/mL) and tumor grading > 2 were found to be significantly related with early hepatic multinodular recurrence.
An unresolved issue is whether hilar dissection mostly estimates tumor stage and prognosis, or there is an impact of lymphadenectomy on control of the disease, also because the relationship of positive lymph nodes with long-term results in previous studies has been inconsistent [28, 39–43]. The role of lymphadenectomy was evaluated in this series. Patients with lymph node metastases had significantly lower overall and disease-free survival. However, patients who underwent lymph node dissection with negative lymph nodes at final pathology showed significantly higher 5-year disease-free survival than patients who did not underwent lymphadenectomy. This may suggest a potential role of lymphadenectomy in enhancing disease-free survival.
There are no well-conducted studies regarding the role of adjuvant chemotherapy following resection of IHC but a multimodal approach may become mandatory, including systemic, loco-regional, targeted molecular therapies.
In conclusion, in our series worse prognosis after liver resection for IHC was significantly related with advanced clinical stage and multiplicity of lesions. However, it should be emphasized that these poor prognostic factors were associated with better long-term results than those of palliatively managed patients, and should not be considered as a contraindication to resection. Taken together, the low operative risk, the absence of impact of tumor size and the poorly defined impact of R1 resection support the role of hepatectomy with regional lymphadenectomy as the best available treatment for IHC. Finally, liver resection still represents the only hope of cure for patients with IHC but the results of surgery are far from satisfactory and there is a strong need to implement surgery with other therapeutic strategies.
References
Sano T, Shimada K, Sakamoto Y et al (2008) Prognosis of perihilar cholangiocarcinoma: hilar bile duct cancer versus intrahepatic cholangiocarcinoma involving the hepatic hilus. Ann Surg Oncol 15:590–599
Lesurtel M, Regimbeau JM, Farges O et al (2002) Intrahepatic cholangiocarcinoma and hepatolithiasis: an unusual association in Western Countries. Eur J Gastroenterol Hepatol 14:1025–1027
Lee CC, Wu CY, Chen GH (2002) What is the impact of coexistence of hepatolithiasis on cholangiocarcinoma. J Gastroenterol Hepatol 17:1015–1020
Jarnagin WR, Weber S, Tickoo SK et al (2002) Combined hepatocellular and cholangiocarcinoma: demographic, clinical, and prognostic factors. Cancer 94:2040–2046
Endo I, Gonen M, Yopp AC et al (2008) Intrahepatic cholangiocarcinoma: rising frequency, improved survival, and determinants of outcome after resection. Ann Surg 248:84–96
Cherqui D, Tantawi B, Alon R et al (1995) Intrahepatic cholangiocarcinoma. Results of aggressive surgical management. Arch Surg 130:1073–1078
Inoue K, Makuuchi M, Takayama T et al (2000) Long-term survival and prognostic factors in the surgical management of mass-forming type cholangiocarcinoma. Surgery 127:498–505
Weber SM, Jarnagin WR, Klimstra D et al (2001) Intrahepatic cholangiocarcinoma: resectability, recurrence pattern and outcomes. J Am Coll Surg 193:384–391
Lang H, Sotiropoulos GC, Sgourakis G et al (2009) Operations for intrahepatic cholangiocarcinoma: single-institution experience of 158 patients. J Am Coll Surg 208:218–228
Jonas S, Thelen A, Benckert C et al (2009) Extended liver resection for intrahepatic cholangiocarcinoma: a comparison of the prognostic accuracy of the fifth and sixth editions of the TNM classification. Ann Surg 249:303–309
The Liver Cancer Study Group of Japan (1997) Intrahepatic cholangiocarcinoma. In: Classification of primary liver cancer, 1st edn. Kanehara, Tokyo, pp 6–7
Sobin LH, Wittekind CH (2002) TNM classification of malignant tumours, 6th edn. Wiley, Hoboken
Strasberg SM, Belghiti J, Clavien PA et al (2000) Terminology committee of the IHPBA. Terminology of liver anatomy and resections. HPB Surg 2:333–339
Nuzzo G, Giuliante F, Vellone M et al (2004) Pedicle clamping with ischemic preconditioning in liver resection. Liver Transpl 10(2 Suppl 1):S53–S57
Giuliante F, Nuzzo G, Ardito F et al (2008) Extraparenchymal control of hepatic veins during mesohepatectomy. J Am Coll Surg 206:496–502
Balzan S, Belghiti J, Farges O et al (2005) The “50–50 criteria” on postoperative day 5: an accurate predictor of liver failure and death after hepatectomy. Ann Surg 242:824–828
Maeda T, Kajiyama K, Adachi E et al (1996) The expression of cytokeratins 7, 19, and 20 in primary and metastatic carcinomas of the liver. Mod Pathol 9:901–909
Shaib YH, Davila JA, McGlynn K et al (2004) Rising incidence of intrahepatic cholangiocarcinoma in the United States: a true increase? J Hepatol 40:472–477
Roayaie S, Guarrera JV, Ye MQ et al (1998) Aggressive surgical treatment of intrahepatic cholangiocarcinoma: predictors of outcomes. J Am Coll Surg 187:365–372
Kawarada Y, Yamagiwa K, Das BC (2002) Analysis of the relationships between clinicopathological factors and survival time in intrahepatic cholangiocarcinoma. Am J Surg 183:679–685
Ohtsuka M, Ito H, Kimura F et al (2002) Results of surgical treatment for intrahepatic cholangiocarcinoma and clinicopathological factors influencing survival. Br J Surg 89:1525–1531
Morimoto Y, Tanaka Y, Ito T et al (2003) Long-term survival and prognostic factors in the surgical treatment for intrahepatic cholangiocarcinoma. J Hepatobiliary Pancreat Surg 10:432–440
Terashi T, Aishima S, Taguchi K et al (2004) Decreased expression of osteopontin is related to tumor aggressiveness and clinical outcome of intrahepatic cholangiocarcinoma. Liver Int 24:38–45
Nakagawa T, Kamiyama T, Kurauchi N et al (2005) Number of lymph node metastases is a significant prognostic factor in intrahepatic cholangiocarcinoma. World J Surg 29:728–733
Miwa S, Miyagawa S, Kobayashi A et al (2006) Predictive factors for intrahepatic cholangiocarcinoma recurrence in the liver following surgery. J Gastroenterol 41:893–900
Shimada K, Sano T, Sakamoto Y et al (2007) Surgical outcomes of the mass-forming plus periductal infiltrating types of intrahepatic cholangiocarcinoma: a comparative study with the typical mass-forming type of intrahepatic cholangiocarcinoma. World J Surg 31:2016–2022
Paik KY, Jung JC, Heo JS et al (2008) What prognostic factors are important for resected intrahepatic cholangiocarcinoma? J Gastroenterol Hepatol 23:766–770
Uenishi T, Kubo S, Yamazaki O et al (2008) Indications for surgical treatment of intrahepatic cholangiocarcinoma with lymph node metastases. J Hepatobiliary Pancreat Surg 15:417–422
Yamashita Y, Taketomi A, Morita K et al (2008) The impact of surgical treatment and poor prognostic factors for patients with intrahepatic cholangiocarcinoma: retrospective analysis of 60 patients. Anticancer Res 28:2353–2359
Nakagohri T, Kinoshita T, Konishi M et al (2008) Surgical outcome and prognostic factors in intrahepatic cholangiocarcinoma. World J Surg 32:2675–2680
Weimann A, Varnholt H, Schlitt HJ et al (2000) Retrospective analysis of prognostic factors after liver resection and transplantation for cholangiocellular carcinoma. Br J Surg 87:1182–1187
DeOliveira ML, Cunningham SC, Cameron JL et al (2007) Cholangiocarcinoma: thirty-one-year experience with 564 patients at a single institution. Ann Surg 245:755–762
Konstadoulakis MM, Roayaie S, Gomatos IP et al (2008) Fifteen-year, single-center experience with the surgical management of intrahepatic cholangiocarcinoma: operative results and long-term outcome. Surgery 143:366–374
Tamandl D, Herberger B, Gruenberger B et al (2008) Influence of hepatic resection margin on recurrence and survival in intrahepatic cholangiocarcinoma. Ann Surg Oncol 15:2787–2794
Portolani N, Baiocchi GL, Coniglio A et al (2008) Intrahepatic cholangiocarcinoma and combined hepatocellular-cholangiocarcinoma: a Western experience. Ann Surg Oncol 15:1880–1890
Yedibela S, Demir R, Zhang W et al (2009) Surgical treatment of mass-forming intrahepatic cholangiocarcinoma: an 11-year Western single-center experience in 107 patients. Ann Surg Oncol 16:404–412
Guglielmi A, Ruzzenente A, Campagnaro T et al (2009) Intrahepatic cholangiocarcinoma: prognostic factors after surgical resection. World J Surg 33:1247–1254
Lee CH, Chang CJ, Lin YJ et al (2009) Viral hepatitis-associated intrahepatic cholangiocarcinoma shares common disease processes with hepatocellular carcinoma. Br J Cancer 100:1765–1770
Kokudo N, Makuuchi M (2002) Extent of resection and outcome after curative resection for intrahepatic cholangiocarcinoma. Surg Oncol Clin N Am 11:969–983
Shirai K, Ebata T, Oda K et al (2008) Perineural invasion is a prognostic factor in intrahepatic cholangiocarcinoma. World J Surg 32:2395–2402
Nathan H, Aloia TA, Vauthey JN et al (2009) A Proposed staging system for intrahepatic cholangiocarcinoma. Ann Surg Oncol 16:14–22
Tamandl D, Kaczirek K, Gruenberger B et al (2009) Lymph node ratio after curative surgery for intrahepatic cholangiocarcinoma. Br J Surg 96:919–925
Choi SB, Kim KS, Choi JY et al (2009) The prognosis and survival outcome of intrahepatic cholangiocarcinoma following surgical resections: association of lymph node metastasis and lymph node dissection with survival. Ann Surg Oncol 16:3048–3056
Acknowledgments
We are sincerely grateful to Professor Masatoshi Makuuchi for his very helpful and constructive advice in the preparation of this manuscript. This study was supported by a contribution from the Catholic University and the Italian Ministry for University and Scientific Research (D.1 Funds).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nuzzo, G., Giuliante, F., Ardito, F. et al. Intrahepatic cholangiocarcinoma: prognostic factors after liver resection. Updates Surg 62, 11–19 (2010). https://doi.org/10.1007/s13304-010-0007-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13304-010-0007-x