Abstract
Chronic myeloid leukemia (CML) results from malignant transformation of hematopoietic stem cells induced by the BCR-ABL oncogene. Transformation from chronic to blastic phase is the lethal step in CML. Leukemic stem cells (LSCs) are the basic reason for blastic transformation. It has been shown that Wnt/β-catenin signaling contributes to the self-renewal capacity and proliferation of LSCs in CML. However, the role of Wnt/β-catenin signaling in blastic transformation of CML is still obscure. Here, we explored the relationship between BCR-ABL and β-catenin signaling in vitro and in vivo. We found that BCR-ABL stimulated β-catenin via activation of PI3K/AKT signaling in blastic phase CML cells. Inhibition of the kinase activity of BCR-ABL, PI3K, or AKT decreased the level of β-catenin in both K562 cells and a CML mouse model and suppressed the transcription of downstream target genes (c-myc and cyclin D1). In addition, inhibition of the BCR-ABL/PI3K/AKT pathway delayed the disease progression in the CML mouse model. To further explore the role of β-catenin in the self-renewal and survival of CML LSCs, we established a secondary transplantation CML mouse model. Our data revealed that inhibition of the BCR-ABL/PI3K/AKT pathway reduced the tumor-initiating ability of K562 cells, decreased leukemia cell infiltration into peripheral blood and bone marrow, and prolonged the survival of mice. In conclusion, our data indicate a close relationship between β-catenin and BCR-ABL/PI3K/AKT in blastic phase CML. β-Catenin inhibition may be of therapeutic value by targeting LSCs in combination with a tyrosine kinase inhibitor, which may delay blastic transformation of CML.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Chronic myeloid leukemia (CML) is believed to originate from leukemic stem cells (LSCs) harboring the Philadelphia chromosome formed through reciprocal translocation between chromosomes 9 and 22 [1–3]. Human CML is induced by the BCR-ABL oncogene that produces the chimeric BCR-ABL protein as a constitutively active tyrosine kinase in leukemogenesis [4, 5]. CML usually presents as the chronic phase (CP) that progresses to the accelerated phase (AP) and terminal blastic phase (BP) [6, 7]. BCR-ABL tyrosine kinase inhibitors (TKI) imatinib (IM), nilotinib, and dasatinib are effective to treat CP CML patients but are less effective against BP CML [8–11]. The basic reason is that they cannot completely eradicate LSCs [12]. It has been shown that IM is highly toxic to differentiated CML progenitors, but not to LSCs that remain viable in a quiescent state [9, 13]. Therefore, it is very important to prevent the transition from CP to AP or BP CML, which would depend on the mechanism of CML blastic transformation.
Several signaling pathways have been found to play important roles in CML development and specifically in LSC survival, including Wnt/β-catenin, Hedgehog, Alox5, and SRC family kinases [14]. The Wnt/β-catenin signaling pathway is crucial for LSC self-renewal [15]. Deregulation of the Wnt/β-catenin signaling pathway could be an underlying mechanism for the existence of LSCs, and disruption of β-catenin in mouse hematopoietic cells significantly reduces BCR-ABL-mediated leukemic transformation [16, 17]. Another study showed that activation of the Wnt/β-catenin pathway enables progenitor cells to regain the ability of self-renewal, leading to tumor occurrence, development, recurrence, and drug resistance [18–20]. Studies have shown that blastic transformation in CML is related to overexpression of BCR-ABL and activation of β-catenin in granulocyte macrophage precursors from CML patients [15, 21]. However, the underlying molecular relationship between BCR-ABL and β-catenin in blast crisis CML remains unknown.
The BCR-ABL fusion protein is crucial for LSCs to maintain their unlimited proliferative capacity via activation of multiple downstream signaling pathways [22]. PI3K/AKT is one of the most important signaling pathways. Activation of AKT inhibits glycogen synthase kinase-3β (GSK-3β) kinase activity, and inhibition of GSK-3β activates β-catenin and its downstream pathways in blast crisis CML cells [21]. Moreover, the level of GSK-β decreases, while the level of β-catenin increases in blast crisis CML patients [23]. Accumulating evidence indicates a close relationship between PI3K/AKT and β-catenin. Deletion of phosphatase with tensin homology (PTEN), a negative regulator of PI3K/AKT signaling, promotes nuclear localization of β-catenin and enhances intestinal stem cell self-renewal and proliferation and thus initiates the formation of precancerous intestinal polyps in mice [24]. In addition, activation of β-catenin signaling has been found in blast crisis CML cells of a conditional Pten knockout mouse model [25]. However, it is still unknown whether BCR-ABL activates the Wnt/β-catenin signaling pathway via activation of PI3K/AKT in BP CML. Based on the previous studies, we hypothesized that Wnt/β-catenin signaling is activated by BCR-ABL, and this activation is related to the PI3K/AKT pathway.
Here, we investigated the relationship between BCR-ABL, PI3K/AKT, GSK-3β and β-catenin in CML-BP patients and CML-BP cell lines. We also examined the role of the BCR-ABL/PI3K/AKT/β-catenin pathway in LSC self-renewal and tumorigenesis in a CML mouse model.
Materials and methods
Cell lines and cell culture
K562, KU812, BaF3, and 32D cell lines were obtained from the American Type Culture Collection (China, Beijing). BaF3-p210 (BP210) cells were established in our laboratory. The 32D-p210 (32DP) cell line was obtained from Dr. Ri Zhang (The First Affiliated Hospital of Soochow University, China). Cells were grown in RPMI 1640 medium containing 10 % fetal bovine serum (Gibco, USA). All cells were cultured and maintained in a 37 °C incubator with 5 % CO2. BP210 and 32DP cell lines were generated from BaF3 and 32D cell lines transformed by p210 BCR-ABL, respectively.
Bone marrow samples
Bone marrow samples were obtained from newly diagnosed CML patients (CP and BP) and healthy volunteers. Mononuclear cells were separated by density gradient centrifugation with Ficoll (Tianjin Haoyang Biological Manufacture Co., Ltd., China). This study was authorized by the Human Ethics Committee of Chongqing Medical University. All participants provided written informed consent.
Antibodies
Antibodies against c-Abl, β-catenin, phosphorylated (p)-c-Abl (Y177), p-β-catenin (Ser33/37/Thr41), GSK-3β (27C10), p-AKT (Thr308), p-GSK-3β (27C10), H2AX, and actin were purchased from Cell Signaling Technology. Anti-Human/Mouse beta-Catenin eFluor 660 was purchased from eBioscience. An anti-human CD45-FITC antibody was purchased from Biolegend.
Western blot analysis
Protein lysates were obtained by lysing cell pellets with RIPA lysis buffer. A total of 100 μg of each protein sample was loaded into each well, separated, and transferred onto a polyvinylidene fluoride membrane. The membrane was blocked in 5 % dry nonfat milk/TBST and then incubated with primary antibodies overnight. The membrane was washed, incubated with a horseradish peroxidase-conjugated secondary antibody, and then developed with enhanced chemiluminescence substrate (Millipore, USA). Chemiluminescent bands were visualized on a cool image workstation II (Viagene, USA).
Real-time RT-PCR
Total RNA was isolated from cells using RNAiso Plus (Takara, Japan). The total RNA (1 μg) was reverse transcribed into cDNA using a PrimeScript RT reagent Kit with gDNA Eraser (Takara). Real-time PCR was performed using a SYBR premix ExTaq™ kit (Takara) on the MiniOpticon real-time PCR detection System (Bio-Rad, USA). All experiments were performed in triplicate. β-Actin was used as the internal control. The primer sequences were as follows:
BCR-ABL sense, 5′-CCACAAGGAGTTCTATGATG-3′;
BCR-ABL antisense, 5′-CAGCACTTCTCAGCCATT-3′;
β-catenin sense, 5′-GTGTGGCGACATATGCAGCT-3′;
β-catenin antisense, 5′-CAAGATCAGCAGTCTCATTC-3′;
c-myc sense, 5′-ACACATCAGCACAACTACG-3′;
c-myc antisense, 5′-GTTCGCCTCTTGACATTCT-3′;
cyclin D1 sense, 5′-CCTCGGTGTCCTACTTCA-3′;
cyclin D1 antisense, 5′-CTCCTCGCACTTCTGTTC-3′;
β-actin sense, 5′-CTGAAGTACCCCATCGAGCACGGCA-3′;
and β-actin antisense, 5′-GGATAGCACAGCCTGGATAGCAACG-3′.
Immunofluorescence microscopy
Cells were collected, washed three times with PBS, and fixed in ice-cold acetone for 15 min. Then, the cells were permeabilized with 1 % Triton X-100/PBS at 37 °C for 10 min and preblocked in 5 % goat serum. The cells were incubated with an anti-β-catenin antibody (Bioworld, USA) diluted in 1 % bovine serum albumin (BSA)/0.05 % Triton X-100/PBS at 4 °C overnight. Next, the cells were incubated with a fluorochrome-conjugated secondary antibody (Invitrogen, USA) diluted in 1 % BSA/0.05 % Triton X-100/PBS at 37 °C for 1 h. The nucleus was stained with 1 μg/ml 4,6-diamidino-2-phenylindole (DAPI; Beyotime, China) for 10 min. Images were acquired by fluorescence microscopy (ECLIPSE 80i, Nikon, Japan).
Hematoxylin-eosin and Wright’s staining
Mouse liver, lung, and spleen were fixed in 4 % (w/v) paraformaldehyde and embedded in paraffin. Sections (4 μm thick) were prepared, dewaxed in xylene, rehydrated in a descending alcohol series, and stained using a routine hematoxylin-eosin staining technique as described previously [26]. Air-dried cell smears were stained with Wright’s staining solution A (Baso, China) for 30 s and then solution B for 45 s, followed by rinsing with tap water for observation.
Immunohistochemical staining
Paraffin-embedded sections were dewaxed in xylene, incubated with the anti-β-catenin antibody at 4 °C overnight, and then incubated with a biotin-streptavidin HRP secondary antibody using a SPlink Detection Kit (ZSGB-Bio, China). Lastly, the sections were stained with DAB chromogen (ZSGB-Bio, China).
Flow cytometric analysis
Cells were collected from peripheral blood and bone marrow of mice. Red blood cells were lysed with red blood cell lysis buffer (Bioflux, Japan). The remaining cells were washed with PBS and incubated with CD45-FITC. Then, the cells were treated with permeabilization wash buffer (Biolegend, USA) and stained with β-catenin eFluor 660 (eBioscience, USA). After washing, the cells were subjected to flow cytometric analysis.
Methylcellulose colony formation assay
Colony formation assays were performed according to Zhao et al. [7]. Briefly, K562 cells were pretreated with IM, LY294002, and AKTi IV for the indicated times, collected, and then cultured in methylcellulose (500 cells/well) at 37 °C in a humidified atmosphere for 7 days. The colonies were counted under an inverted microscope (Olympus, Japan). All analyses were performed three times.
Mice
NOD/SCID mice were obtained from Chongqing Medical University Laboratory Animal Centre. The mice were maintained in a temperature- and humidity-controlled environment and given acidified water. All animal care and experimental procedures were conducted according to protocols from the Biomedical Ethics Committee at Chongqing Medical University.
Induction of CML in mice
Six- to 8-week-old NOD/SCID mice were injected with K562 cells. To establish K562-NOD/SCID CML mice, K562 cells were treated with 5 μM IM for 24 h, 20 μM LY294002 for 24 h, 5 μM AKTi IV for 6 h, or DMSO. About 5 × 106 K562 cells from each group were intravenously injected into 300 cGy lethally irradiated recipient NOD/SCID mice. For secondary transplantation, equal numbers of CD45+ bone marrow cells from primary K562-NOD/SCID CML mice were transplanted into 300-cGy-irradiated recipient NOD/SCID mice.
In vivo drug treatment
IM (Novartis, China) dissolved in water directly at a concentration of 10 mg/mL was administered orally by gavage in a volume of less than 0.5 mL once a day at 100 mg/kg body weight. LY294002 (Cell Signaling Technology, USA) and AKTi IV (Calbiochem, Germany) were dissolved in DMSO directly at concentrations of 0.5 and 0.3 mg/mL, respectively, and administered by intraperitoneal injection in a volume of less than 0.5 mL once a day at 5 or 3 mg/kg body weight. Drug treatments began at 8 days after K562 cell injection and were administered as two doses/week for 3 weeks.
Statistical analysis
Results are presented as means ± the standard error. Statistical analysis was performed using SPSS 17.0 Statistical software. Data were analyzed by the Student’s t test or one-way analysis of variance. Statistical significance was defined as P < 0.05.
Results
BCR-ABL activates β-catenin in CML-BP cells
Bone marrow mononuclear cells were collected from CML-CP patients (n = 6), CML-BP patients (n = 6), and healthy volunteers (n = 6). BCR-ABL and β-catenin expression was measured by real-time reverse transcription-polymerase chain reaction (RT-PCR) and normalized to the housekeeping gene β-actin. As the BCR/ABL level increased in CML-BP patients compared with CML-CP patients, a high level of β-catenin was observed in CML-BP patients compared with CML-CP patients (Fig. 1a, b). β-Catenin expression in CML-BP patients was found to increase by at least twofold compared with the mean β-catenin expression in CML-CP patients and healthy volunteers (P < 0.05). Next, western blotting was performed to assay the protein levels of β-catenin in four CML-CP samples, three CML-BP samples, and three healthy samples. Consistent with the messenger RNA (mRNA) observations, the protein levels of BCR/ABL and β-catenin were also significantly increased in CML-BP patients (Fig. 1c).
BCR-ABL activates β-catenin in CML-BP cells. a Real-time RT-PCR analysis of BCR-ABL expression in CML-CP and CML-BP patients. b Real-time RT-PCR analysis of β-catenin expression in CML-CP patients, CML-BP patients, and healthy volunteers. c Western blot analysis of BCR-ABL and β-catenin expression in CML-CP patients, CML-BP patients, and healthy volunteers. d Western blot analysis of β-catenin expression in six cell lines. e Western blot analysis of BCR-ABL, p-AKT, and β-catenin expression in cell lines
To examine the relationship between BCR-ABL and β-catenin, we detected their protein levels in six cell lines including CML-BP cell lines (KU812 and K562), p210 (BCR-ABL)-transformed cell lines (32DP and BP210), and normal cell lines (32D and BaF3). As expected, the levels of β-catenin in KU812 and K562 cells were significantly higher than those in the other cell lines (Fig. 1d). Furthermore, we found that the expression of p-AKT (Thr308) and β-catenin in p210 (BCR-ABL)-transformed cell lines was higher than that in normal cell lines (Fig. 1e).
Inhibition of BCR-ABL, PI3K, and AKT decreases the expression of β-catenin in K562 cells
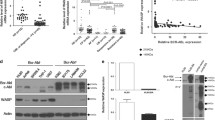
To investigate the relationship between the PI3K/AKT pathway and β-catenin in CML, we used IM, LY294002, and AKTi IV to inhibit BCR/ABL, PI3K, and AKT, respectively, in K562 cells. We found that these inhibitors all inhibited their corresponding proteins using the appropriate concentration and treatment time (5 μM IM for 24 h, 20 μM LY294002 for 24 h, and 5 μM AKTi IV for 6 h) (Fig. 2a). It has been reported that stabilized cytoplasmic β-catenin has the ability to translocate to the nucleus. Therefore, we tested whether IM, LY294002, or AKTi IV had any effect on the distribution of β-catenin. K562 cells were treated with IM, LY294002, or AKTi IV. Cytoplasmic and nuclear proteins isolated from the total lysate were analyzed by western blotting. Independent experiments were repeated three times. We observed a significant decrease of β-catenin protein levels in both the cytoplasm and nucleus compared with the control group (Fig. 2b). To confirm the changes of β-catenin protein levels in the cytoplasm and nucleus, immunofluorescence analysis was performed to determine the distribution of β-catenin (red). Nuclei were stained with DAPI (blue). β-Catenin was localized in both the cytoplasm and nucleus, especially the cytoplasm, in K562 cells. After inhibitor treatments, the expression of β-catenin in both the cytoplasm and nuclear was decreased as shown by western blot analyses (Fig. 2c).
Inhibition of BCR-ABL, PI3K, and AKT decreases the expression of β-catenin in K562 cells. a Western blot analysis of the corresponding protein levels in K562 cells treated with IM, LY294002, or AKTi IV at various concentrations and times. b Western blot analysis of β-catenin protein levels in the cytoplasm and nucleus of K562 cells treated with IM, LY294002, or AKTi IV. c Immunofluorescence analysis of the distribution of β-catenin in the cytoplasm and nucleus of K562 cells treated with IM, LY294002, or AKTi IV. d Colony numbers per 500 K562 cells treated with IM, LY294002, or AKTi IV at day 7 as determined by a methylcellulose colony formation assay (*P < 0.05)
The progression of CP CML to BP CML is supported by the self-renewal capacity of LSCs, and the process of self-renewal involves the Wnt/β-catenin signaling pathway. We next tested whether BCR-ABL/PI3K/AKT pathway inhibition had any effect on the colony-forming capability of leukemic cells. K562 cells were treated with IM, LY294002, AKTi IV, or DMSO and then cultured in methylcellulose medium. Inhibitor-treated K562 cells formed significantly lower numbers of colonies compared with DMSO-treated K562 cells (Fig. 2d). These results suggest that IM, LY294002, and AKTi IV suppressed β-catenin in leukemia cells and may potentially inhibit the survival and self-renewal of leukemia cells in vitro.
Inhibition of the BCR-ABL/PI3K/AKT pathway promotes degradation of β-catenin and reduces transcription of β-catenin target genes
We found that inhibition of the BCR-ABL/PI3K/AKT pathway reduced the protein expression of β-catenin. Next, to further explore the reason for the decrease in β-catenin, we examined whether inhibition of the BCR-ABL/PI3K/AKT pathway affected the mRNA level of β-catenin. Real-time RT-PCR analysis showed no significant change in the β-catenin mRNA level after treatments (Fig. 3a). We next tested whether the BCR-ABL/PI3K/AKT pathway affected the protein stability of β-catenin. K562 cells were treated with 10 μg/ml cycloheximide (CHX) to inhibit protein biosynthesis, and then, IM, LY294002, or AKTi IV was added. By blocking translation with CHX, we could specifically evaluate the role of post-translational events, including degradation, in regulating the protein level. We found that β-catenin degraded in a time-dependent manner and degraded faster in inhibitor-treated groups than in the untreated group (Fig. 3b).
Inhibition of the BCR-ABL/PI3K/AKT pathway promotes the degradation of β-catenin and reduces transcription of β-catenin target genes. a Real-time RT-PCR analysis of β-catenin expression in K562 cells treated with IM, LY294002, or AKTi IV. b Western blot analysis of β-catenin protein stability in K562 cells treated with the protein biosynthesis inhibitor CHX. c Western blot analysis of GSK-3β, p-GSK-3β, β-catenin, and p-β-catenin expression in K562 cells treated with IM, LY294002, or AKTi IV. d Western blot analysis of total β-catenin and p-β-catenin expression in K562 cells treated with IM, LY294002, or AKTi IV at various time points. e Western blot analysis of c-myc and cyclin D1 expression in K562 cells treated with IM, LY294002, or AKTi IV. f Real-time RT-PCR analysis of c-myc and cyclin D1 expression in K562 cells treated with IM, LY294002, or AKTi IV (*P < 0.05)
AKT inhibits GSK-3β activity through phosphorylation of Ser9, whereas active GSK-3β plays an important role in phosphorylating β-catenin for proteasome-mediated degradation [27]. Western blot analysis showed that the expression of p-GSK-3β deceased after treatments (Fig. 3c). However, p-β-catenin did not increase but decreased. Next, we determined whether p-β-catenin decreased after its increase because of degradation. We measured p-β-catenin (S33/S37/T41) at various time points after inhibitor treatments of K562 cells; as expected, p-β-catenin elevated before its decrease and total β-catenin increased before its decrease (Fig. 3d). These results demonstrated that BCR-ABL enhanced the protein stability of β-catenin through PI3K/AKT/GSK-3β in K562 cells.
β-Catenin has been verified to promote leukemic cell growth and contribute to the progression of leukemia through its role in activating the transcription of T cell factor/lymphoid enhancer factor (TCF/LEF) family genes. We next determined the effects of IM, LY294002, and AKTi IV on the downstream target genes of the Wnt/β-catenin pathway. Western blot analysis showed that blockade of the BCR-ABL/PI3K/AKT pathway resulted in decreased expression of c-myc and cyclin D1 (Fig. 3e). Subsequently, RT-PCR analysis was performed to evaluate the mRNA levels of c-myc and cyclin D1. The results suggested that c-myc and cyclin D1 mRNAs were also significantly downregulated after drug treatments (Fig. 3f).
Inhibition of the BCR-ABL/PI3K/AKT pathway delays CML development
We found that inhibition of the BCR-ABL/PI3K/AKT pathway reduced expression of β-catenin and its target oncogenes in K562 cells. Therefore, we tested whether the BCR-ABL/PI3K/AKT pathway played an important role in the development of CML in mice. To induce CML in mice, K562 cells treated with IM, LY294002, AKTi IV, or DMSO were injected into lethally irradiated recipient NOD/SCID mice. Mice were grouped as IM, LY294002, AKTi, and control. The mice were continually treated with IM, LY294002, AKTi IV, or normal saline beginning at day 8 post-K562 cell injection. Mice began to develop leukemic symptoms at around 4 weeks after injection of tumor cells, including weight loss, hindlimb paralysis, reduced activity, and fluffy hair. Compared with the control group, mice in the three inhibitor-treated groups had significantly less weight loss (Fig. 4a) and fewer leukocytes (P < 0.05) (Fig. 4b). The mice that received DMSO-treated K562 cells died faster than those that received K562 cells treated with IM, LY294002, or AKTi IV (Fig. 4c), which correlated with more myeloid cells in peripheral blood (Fig. 4d). In the peripheral blood of the control group, leukemia cells were found at about 4 weeks after injection, which was earlier than in the three inhibitor-treated groups. Analysis of CD45+ cells in peripheral blood by flow cytometry measured the percentages of leukemia cells at 2 and 4 weeks after K562 cell injection. The data showed the development of typical CML and more CD45+ leukemia cells in the control group than in the three inhibitor-treated groups at 4 weeks after injection (Fig. 4e), which was consistent with more severe infiltration of myeloid leukemia cells into the lungs and spleen (Fig. 4f). Similar pathological changes were observed in the liver (Fig. 4g). We observed hepatosplenomegaly in almost all morbid mice with more severe hepatosplenomegaly in the control group than in the three inhibitor-treated groups (Fig. 4h, i). Specifically, inhibitor treatments significantly reduced splenomegaly in K562 cell-transplanted mice (P < 0.05) (spleen weights 0.39 ± 0.07 g in the control group, 0.14 ± 0.04 g in the IM group, 0.21 ± 0.07 g in the LY294002 group, and 0.20 ± 0.04 g in the AKTi group). Hepatomegaly was observed in some mice (liver weights 1.06 ± 0.23 g in the control group, 0.55 ± 0.10 g in the IM group, 0.0.67 ± 0.0.12 g in the LY294002 group, and 0.72 ± 0.16 g in the AKTi group). In addition, three mice from the control group had abdominal and thigh tumors (Fig. 4j). Taken together, these results demonstrate that the BCR-ABL/PI3K/AKT pathway plays an important role in the development of CML induced by K562 cells.
Inhibition of the BCR-ABL/PI3K/AKT pathway delays CML development. a Mouse weights at various time points after K562 cell injection. b White blood cell counts in peripheral blood from the four mouse groups (*P < 0.05 versus other groups). c Kaplan-Meier survival curves of mice (six mice per group). d Peripheral blood smears indicating the accumulation of neutrophils and high white blood cell counts in CML mice. e Flow cytometric analysis of the percentage of CD45+ leukemia cells in peripheral blood from the four mouse groups (*P < 0.05 versus the control group). f Photomicrographs of hematoxylin-eosin-stained lung and spleen sections from the four mouse groups. g Photomicrographs of hematoxylin-eosin-stained liver sections from the four mouse groups. h Comparison of general morphology of livers and spleens from the four mouse groups. i Liver and spleen weights. j Images of abdominal and thigh tumors
Inhibition of the BCR-ABL/PI3K/AKT pathway reduces β-catenin expression in CML mice
Because inhibition of BCR/ABL, PI3K, and AKT kinase activity delays CML development in mice, we investigated whether this effect is mediated through suppressing β-catenin expression in mice. We analyzed the expression of β-catenin in CD45+ cells isolated from mouse peripheral blood. We observed a significant difference in the mean fluorescence intensity of β-catenin between the control group and the three inhibitor-treated groups (Fig. 5a). Next, we performed an immunofluorescence assay in bone marrow cells to detect β-catenin. As expected, β-catenin expression in control group mice was higher than that in inhibitor-treated mice (Fig. 5b). Finally, we performed histochemical staining to examine the expression of β-catenin in mouse lungs and spleen. Compared with inhibitor-treated mice, β-catenin expression was higher in the lungs and spleen of control mice (Fig. 5c). These results further support the role of β-catenin in CML development.
Inhibition of the BCR-ABL/PI3K/AKT pathway reduces β-catenin expression in CML mice. a Flow cytometric analysis of β-catenin expression (mean fluorescence intensity) in mouse peripheral blood CD45+ cells. b Immunofluorescence of β-catenin expression in mouse bone marrow cells. c Immunohistochemical staining of β-catenin (brown) in mouse lungs and spleen
Inhibition of the BCR-ABL/PI3K/AKT pathway reduces the tumor-initiating ability of CML cells in secondary transplantation
To investigate the effect of the BCR-ABL/PI3K/AKT pathway on the tumor-forming capacity of CML cells, we performed secondary transplantations. K562-NOD/SCID CML mice were again induced by cell injection. Flow cytometric analysis of CD45+ cells in the peripheral blood of K562-NOD/SCID CML mice showed that the percentage of leukemia cells reached 5–12 % at about 4 weeks after K562 cell injection (Fig. 6a). Therefore, at week 5, equal numbers of CD45+ bone marrow cells from primary K562-NOD/SCID CML mice were transplanted into lethally irradiated recipient NOD/SCID mice. The mice injected with bone marrow cells from IM, LY294002, AKTi IV, or control group mice were grouped as IM-2nd, LY294002-2nd, AKTi-2nd, and control-2nd, respectively.
Inhibition of the BCR-ABL/PI3K/AKT pathway reduces the tumor-initiating ability of CML cells in secondary transplantation. a Flow cytometric analysis of the percentage of CD45+ leukemia cells in peripheral blood at 4 weeks post-K562 cell injection. b White blood cell counts in mouse peripheral blood after bone marrow cell transplantation (*P < 0.05 versus control-2nd group). c Flow cytometric analysis of the percentage of CD45+ leukemia cells in peripheral blood at 7 weeks post-bone marrow cell transplantation (*P < 0.05 versus control-2nd group). d Flow cytometric analysis of the percentage of CD45+ leukemia cells in mouse bone marrow cells at 7 weeks post-bone marrow cell transplantation. e Kaplan-Meier survival curves of secondary transplantation mice (five mice per group)
Mice began to develop leukemic symptoms at around 3 weeks after secondary transplantation, including weight loss, hindlimb paralysis, reduced activity, and fluffy hair. At about 4 weeks after transplantation, peripheral white blood cells in mice began to increase. Compared with the control-2nd group, mice in the other three groups had significantly fewer leukocytes (P < 0.05) (Fig. 6b). Flow cytometric analysis was performed to observe CD45+ leukemia cells in peripheral blood and bone marrow. The flow cytometry data showed a higher percentage of leukemia cells in control-2nd group mice at 7 weeks after secondary transplantation than in the other three groups (Fig. 6c, d). We also found that the mice in the control-2nd group died faster than those in the other three groups (Fig. 6e). Secondary transplantation experiments indicated that inhibition of the BCR-ABL/PI3K/AKT pathway reduced the tumor-initiating ability of K562 cells, decreased the cell infiltration of peripheral blood and bone marrow in the CML secondary transplantation mouse model, and prolonged the survival of mice.
Discussion
Blastic transformation of CML makes patients become unresponsive to TKIs and results in a poor prognosis. The basic reason for this transformation is BCR-ABL amplification and LSCs [28]. It has been previously reported that Wnt/β-catenin is crucial for the self-renewal and proliferation of hematopoietic stem cells and CML LSCs [17–19]. Recently, increasing evidence has indicated the involvement of the Wnt/β-catenin signaling pathway in the TKI resistance of CML-BP patients [16, 23, 29].
In the Wnt pathway, the central player is β-catenin, a very important transcription cofactor with TCF/LEF. β-Catenin is targeted for ubiquitination and degradation by phosphorylation through GSK-3β, casein kinase I, axin protein, and adenomatous polyposis coli protein. Activation of the Wnt/β-catenin pathway causes β-catenin accumulation in a complex with the transcription factor TCF/LEF, which then promotes expression of target genes such as c-myc and cyclin D1 [30]. In the absence of Wnt signaling, the level of β-catenin is maintained at a low level through phosphorylation by GSK-3β and proteasome degradation of cytoplasmic β-catenin. In the presence of Wnt signaling, the β-catenin destruction complex is disrupted, and β-catenin is stabilized in the cytosol [31]. Then, the accumulated cytoplasmic signaling β-catenin binds to transcription factors TCF/LEF and induces target gene expression. Therefore, stabilization and accumulation of β-catenin in the cytoplasm are the key factors.
PI3K/AKT is one of the most important signaling pathways of BCR-ABL downstream in CML. Recent studies have indicated the relationship between Wnt/β-catenin and PI3K/AKT in other cancers [32, 33]. The link between Wnt/β-catenin and PI3K/AKT is GSK-3β. GSK-3β is phosphorylated by activation of PI3K/AKT, resulting in the inhibition of GSK-3β kinase activity [34–36], which inhibits degradation of β-catenin and promotes β-catenin stabilization. The synergistic cytotoxic effect of a specific PI3K inhibitor, LY294002, and tamoxifen is achieved by inhibition of the GSK-3β/β-catenin signaling pathway in malignant gliomas [37]. Therefore, the phosphorylation and degradation of β-catenin through activation of GSK-3β exert the anti-cancer effect of PI3K/AKT inhibitors. However, the relationship between PI3K/AKT and Wnt/β-catenin pathways in CML blastic transformation remains unknown. It is important to understand the relationship between BCR-ABL, PI3K/AKT, and β-catenin in CML-BP patients.
In this study, we demonstrated markedly increased BCR-ABL expression in CML-BP patients. Elevated BCR-ABL expression is always accompanied by increased β-catenin. Compared with normal cell lines, the expressions of β-catenin and p-AKT were both increased in cell lines (BP210 and 32DP) transfected with the BCR-ABL fusion gene (P210). To further understand the mechanism of BCR-ABL activating Wnt/β-catenin signaling pathways in vitro, three inhibitors (IM, LY294002, and AKTi IV) were used to inhibit the BCR-ABL/PI3K/AKT pathway in K562 cells. We found that inhibition of BCR-ABL, PI3K, or AKT kinase activities resulted in decreased β-catenin protein levels and suppressed proliferation in CML-BP cells but no significant influence on the mRNA level of β-catenin. Moreover, the inhibitors reduced the phosphorylation of GSK-3β, the component of the proteasome complex whose dephosphorylated form promotes β-catenin degradation, and subsequently decreased the expression of β-catenin downstream targets including cyclin D1 and c-myc. Interestingly, p-β-catenin did not increase but decreased. In the Wnt/β-catenin pathway, phosphorylation of β-catenin (S33/S37/T41) is essential for GSK-3β targeting β-catenin for ubiquitin-proteasome pathway degradation [38]. Reduced p-GSK-3β (serine-9) may result in an increase of p-β-catenin (S33/S37/T41). In contrast, we found decreased p-β-catenin and decreased total β-catenin. The phosphorylation of β-catenin was sequential and transient, and p-β-catenin was degraded after the increase [39]. To confirm our assumption, we measured p-β-catenin (S33/S37/T41) at various time points after inhibitor treatments of K562 cells. As expected, p-β-catenin elevated before it decreased. We also found that total β-catenin increased before the decrease. Taken together, we propose that BCR-ABL enhances the protein stability of β-catenin through PI3K/AKT/GSK-3β in K562 cells.
To examine whether the BCR-ABL/PI3K/AKT pathway also plays an important role in vivo, we established CML mice by K562 cell injection into lethally irradiated recipient NOD/SCID mice [40, 41]. We found that inhibition of BCR-ABL/PI3K/AKT partially prevented the development of CML in mice and promoted their survival. These results suggested that the level of β-catenin may act as a key player in mediating the transformation of CML from CP to BP. In addition, secondary transplantation experiments indicated that inhibition of the BCR-ABL/PI3K/AKT pathway reduced the tumor-initiating ability of K562 cells, decreased the cell infiltration into peripheral blood and bone marrow in the CML secondary transplantation mouse model, and prolonged the survival of mice.
Based on our results, we propose a model for the BCR-ABL/PI3K/AKT/GSK-3β/β-catenin signaling pathway (Fig. 7). In summary, our study confirmed the crucial role of β-catenin in CML development, demonstrated the mechanism in which BCR-ABL/PI3K/AKT regulates β-catenin through phosphorylation of GSK-3β, and then accelerates the progression of the disease and results in progression to BP CML. Blastic transformation of CML is affected by many factors and pathways. Our study may provide a potential clinical theory and strategy for the prevention and treatment of CML-BP patients.
References
Becker MW, Jordan CT. Leukemia stem cells in 2010: current understanding and future directions. Blood Rev. 2011;25(2):75–81.
Rice KN, Jamieson CH. Molecular pathways to CML stem cells. Int J Hematol. 2010;91(5):748–52.
Gerber JM, Qin L, Kowalski J, et al. Characterization of chronic myeloid leukemia stem cells. Am J Hemato. 2011;86(1):31–7.
Quintás-Cardama A, Cortes J. Molecular biology of bcr-abl1-positive chronic myeloid leukemia. Blood. 2009;113:1619–30.
Melo JV, Barnes DJ. Chronic myeloid leukaemia as a model of disease evolution in human cancer. Nat Rev Cancer. 2007;7(6):441–53.
Perrotti D, Jamieson C, Goldman J, et al. Chronic myeloid leukemia: mechanisms of blastic transformation [J]. J Clin Invest. 2010;120(7):2254.
Sawyers CL. Chronic myeloid leukemia. N Engl J Med. 1999;341(10):765.
Druker BJ, Sawyers CL, Kantarjian H, et al. Activity of a specific inhibitor of the BCR-ABL tyrosine kinase in the blast crisis of chronic myeloid leukemia and acute lymphoblastic leukemia with the Philadelphia chromosome. N Engl J Med. 2001;344(14):1038–42.
Graham SM, Jørgensen HG, Allan E, et al. Primitive, quiescent, Philadelphia-positive stem cells from patients with chronic myeloid leukemia are insensitive to STI571 in vitro. Blood. 2002;99(1):319–25.
Hu Y, Swerdlow S, Duffy TM, et al. Targeting multiple kinase pathways in leukemic progenitors and stem cells is essential for improved treatment of Ph+ leukemia in mice. Proc Natl Acad Sci U S A. 2006;103(45):16870–5.
Eiring AM, Khorashad JS, Morley K, et al. Advances in the treatment of chronic myeloid leukemia. BMC Med. 2011;9:99.
Kantarjian H, Sawyers C, Hochhaus A, et al. Hematologic and cytogenetic responses to imatinib mesylate in chronic myelogenous leukemia. International STI571 CML Study Group. N Engl J Med. 2002;346(9):645–52.
Kinstrie R, Copland M. Targeting chronic myeloid leukemia stem cells. Curr Hematol Malig Rep. 2013;8(1):14–21.
Malhotra S, Kincade PW. Wnt-related molecules and signaling pathway equilibrium in hematopoiesis. Cell Stem Cell. 2009;4:27–33.
Abrahamsson AE, Geron I, Gotlib J, et al. Aberrant regulation of Wnt/beta-catenin pathway mediators in chronic myelogenous leukemia stem cells. Blood (ASH Ann Meet Abstr). 2006;108:2135.
Zhao C, Blum J, Chen A, et al. Loss of beta-catenin impairs the renewal of normal and CML stem cell in vivo. Cancer Cell. 2007;12(4):528–41.
Staal FJ, Clevers HC. Wnt signaling and hematopoiesis: a WNT-WNT situation. Nat Rev Immunol. 2005;5(1):21–30.
Huntly BJ, Gilliland DG. Leukaemia stem cells and the evolution of cancer-stem-cell research. Nat Rev Cancer. 2005;5(4):311–21.
de Sousa EM, Vermeulen L, Richel D, et al. Targeting Wnt signaling in colon cancer stem cells. Clin Cancer Res. 2011;17(4):647–53.
Dodge ME, Lum L. Drugging the cancer stem cell compartment: lessons learned from the hedgehog and Wnt signal transduction pathways. Annu Rev Pharmacol Toxicol. 2011;51:289–310.
Jamieson CH, Ailles LE, Dylla SJ, et al. Granulocyte–macrophage progenitors as candidate leukemic stem cells in blast-crisis CML. N Engl J Med. 2004;351(7):657–67.
Gunsilius E. Evidence from a leukemia model for maintenance of vascular endothelium by bone marrow-derived endothelial cells. Adv Exp Med Biol. 2003;522:17–24.
Coluccia AM, Vacca A, Duñach M, et al. Bcr-Abl stabilizes β-catenin in chronic myeloid leukemia through its tyrosine phosphorylation. EMBO J. 2007;26:1456–66.
He XC, Yin T, Grindley JC, et al. PTEN-deficient intestinal stem cells initiate intestinal polyposis. Nat Genet. 2007;39(2):189–98.
Guo W, Lasky 3rd JL, Wu H. Cancer stem cells. Pediatr Res. 2006;59(4 Pt 2):59R–64R.
Pool CR. Hematoxylin-eosin staining of OsO4-fixed epon-embedded tissue; prestaining oxidation by acidified H2O2. Stain Technol. 1969;44(2):75–9.
McCubrey JA, Steelman LS, Bertrand FE, et al. Multifaceted roles of GSK-3 and Wnt/beta-catenin in hematopoiesis and leukemogenesis: opportunities for therapeutic intervention. Leukemia. 2014;28(1):15–33.
AS Corbin A, Agarwal M. Loriaux, et al. human chronic myeloid leukemia stem cells are insensitive to imatinib despite inhibition of BCR-ABL activity. J Clin Invest. 2011;121(1):396–409.
Calabretta B, Perrotti D. The biology of CML blast crisis. Blood. 2004;103(11):4010–22.
Nelson WJ, Nusse R. Convergence of Wnt, beta-catenin, and cadherin pathways. Science. 2004;303(5663):1483–7.
Kolligs FT, Bommer G, Göke B. Wnt/beta-catenin/tcf signaling: a critical pathway in gastrointestinal tumorigenesis. Digestion. 2002;66(3):131–44.
Regmi SC, Park SY, Kim SJ, et al. The anti-tumor activity of succinyl macrolactin A is mediated through the β-catenin destruction complex via the suppression of tankyrase and PI3K/Akt. PLoS One. 2015;10(11):e0141753.
Meng F, Li H, Shi H, et al. MACC1 down-regulation inhibits proliferation and tumourigenicity of nasopharyngeal carcinoma cells through Akt/β-catenin signaling pathway. PLoS One. 2013;8(4):e60821.
Cross DA, Alessi DR, Cohen P, et al. Inhibition of glycogen synthase kinase-3 by insulin mediated by protein kinase B. Nature. 1995;378(6559):785–9.
Ishibe S, Haydu JE, Togawa A, et al. Cell confluence regulates hepatocyte growth factor-stimulated cell morphogenesis in a beta-catenin-dependent manner. Mol Cell Biol. 2006;26(24):9232–43.
Voskas D, Ling LS, Woodgett JR. Does GSK-3 provide a shortcut for PI3K activation of Wnt signalling? F1000 Biol Rep. 2010;2:82.
Li C, Zhou C, Wang S, et al. Sensitization of glioma cells to tamoxifen-induced apoptosis by Pl3-kinase inhibitor through the GSK-3β/β-catenin signaling pathway. PLoS One. 2011;6(10):e27053.
Liu C, Li Y, Semenov M, et al. Control of β-catenin phosphorylation/degradation by a dual-kinase mechanism. Cell. 2002;108(6):837–47.
Wang PS, Chou FS, Bloomston M, et al. Thiazolidinediones downregulate Wnt/β-catenin signaling via multiple mechanisms in breast cancer cells. J Surg Res. 2009;153(2):210–6.
Shirasaki R, Tashiro H, Oka Y, et al. Chronic myelogenous leukemia cells contribute to the stromal myofibroblasts in leukemic NOD/SCID mouse in vivo. J Oncol. 2012;2012:901783.
Li L, Wang L, Li L, et al. Activation of p53 by SIRT1 inhibition enhances elimination of CML leukemia stem cells in combination with imatinib. Cancer Cell. 2012;21(2):266–81.
Acknowledgments
This work was funded by the Natural Science Foundation of CQ CSTC (cstc2012jjA10013) to Jing Hu and the Special Fund of Chongqing Key Laboratory (CSTC) (2012-2015) to Jing Hu.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This study was authorized by the Human Ethics Committee of Chongqing Medical University. All participants provided written informed consent.
The ethical standards includes:
1. Whether the project should be carried out with experimental animal experiment. Whether it could be done with computer simulation, cell culture or lower animals instead of higher animal.
2. Whether the animals used in this experiment are suitable. Whether the number of animals could be reduced through improved design or high quality animals.
3. Whether animals are treated ethically through improving experiment method, adjusting experimental observation index, optimizing the experiment scheme.
4. Whether animal welfare measures are implemented.
Ethical approval
All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Conflicts of interest
None
Rights and permissions
About this article
Cite this article
Hu, J., Feng, M., Liu, ZL. et al. Potential role of Wnt/β-catenin signaling in blastic transformation of chronic myeloid leukemia: cross talk between β-catenin and BCR-ABL. Tumor Biol. 37, 15859–15872 (2016). https://doi.org/10.1007/s13277-016-5413-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-016-5413-3