Abstract
Drug resistance of cancer cells can be regulated by the dysregulated miRNAs, and sustained NFκB activation also plays an important role in tumor resistance to chemotherapy. Here, we sought to investigate whether there was a correlation between miR-20a and the NFκB pathway to clarify the effects that miR-20a exerted on gastric cancer (GC) chemoresistance. We found that miR-20a was significantly upregulated in GC plasma and tissue samples. In addition, it was upregulated in GC plasma and tissues from patients with cisplatin-resistant gastric cancer cell line SGC7901/cisplatin (DDP). And the upregulation of miR-20a was concurrent with the downregulation of NFKBIB (also known as IκBβ) as well as upregulation of p65, livin, and survivin. The luciferase activity suggested that NFKBIB was the direct target gene of miR-20a. Transfection of miR-20a inhibitor could increase NFKBIB level; downregulate the expression of p65, livin, and survivin; and lead to a higher proportion of apoptotic cells in SGC7901/DDP cells. Conversely, ectopic expression of miR-20a dramatically decreased the expression of NFKBIB; increased the expression of p65, livin, and survivin; and resulted in a decrease in the apoptosis induced by DDP in SGC7901 cells. Taken together, our findings suggested that miR-20a could promote activation of the NFκB pathway and downstream targets livin and survivin by targeting NFKBIB, which potentially contributed to GC chemoresistance.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Gastric cancer (GC) is one of the most common malignancies globally [1]. Currently, chemotherapy is still one of the major treatment methods for GC because of low early detection and diagnosis rate. However, the effectiveness of chemotherapy is limited by drug resistance. Earlier studies have revealed that multiple cytological mechanisms including DNA repair, cell cycle, apoptosis, and proliferation may play an important role in chemoresistance. Moreover, molecular mechanisms including increased rates of drug efflux, alterations in drug metabolism, and mutation of drug targets might also contribute to it [2, 3]. Further studies have found that the activation of some signaling pathways was also involved in drug resistance, such as PTEN/PI3K/AKT, NFκB/IκB, Ras/Raf/MEK, and P53/MDM2 [4–7].
MicroRNAs (miRNA) are a class of short noncoding RNAs with a length of 19–22 nucleotides and function as posttranscriptional regulators by directly cleaving target messenger RNA (mRNA) or translational repression [8]. Aberrant miRNAs has been found to be related to drug resistance in different types of tumors [9–12]. Increasing evidence has shown that miRNA regulates NFκB signaling [13], and NFκB activation plays an important role in tumor resistance to chemotherapy [14]. Therefore, we hypothesized that miRNA might contribute to chemoresistance through the activation of NFκB pathway.
In our study, we found that miR-20a was significantly upregulated in GC samples especially in those with chemoresistance. Moreover, it was upregulated in cisplatin-resistant GC cell line SGC7901/DDP and induced cisplatin resistance when overexpressed in GC cell line SGC7901. Furthermore, we demonstrated that miR-20a might activate the NFκB pathway and upregulate the downstream targets survivin and livin by repressing NFKBIB expression.
Materials and methods
Clinical samples collection
A total of 130 histopathologically confirmed GC patients and 60 healthy donors were obtained from the First Affiliated Hospital of Nanjing Medical University between 2012 and 2014. Thirty GC plasma samples and additional 30 formalin-fixed paraffin-embedded (FFPE) sections of GC were from preoperative patients who did not receive chemotherapy. Thirty GC plasma samples and additional 40 FFPE sections of GC were from advanced patients who underwent 2 cycles of platinum-based chemotherapy. All the procedures were approved by Institutional Review Boards of the First Affiliated Hospital of Nanjing Medical University, and the written informed consent was obtained from each participant.
Cell culture and transfection
The human gastric cancer cell line SGC7901 was obtained from the National Institute of Cells (Shanghai, China). Cisplatin-resistant variant SGC7901/DDP was purchased from KeyGEN Biotechnology Company (Nanjing, China). All the cells were cultured in RPMI-1640 medium supplemented with 10 % fetal calf serum (Gibco-BRL, Grand Island, NY) in a humidified atmosphere with 5 % CO2 at 37 °C as previously described [15, 16]. To maintain the cisplatin-resistant phenotype, cisplatin (DDP, with final concentration of 1 μg/mL) was added to the culture media for SGC7901/DDP cells.
MiRNA mimics and their appropriate negative control (NC) were purchased from Guangzhou RiboBio (RiboBio, Guangzhou, China). Transfection was performed using Lipofectamine 2000 reagent (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s protocol.
RNA isolation and quantitative real-time PCR analysis
Total RNA was isolated from cells using TRIzol reagent (Invitrogen, Carlsbad, CA, USA) from FFPE tissues using the High Pure FFPE RNA Micro Kit (Ambion, Austin, TX, USA) and from plasma using mirVana PARIS Kit (Ambion, Austin, TX, USA) following the manufacturer’s protocol. The quantification of miRNA was performed using the specific primers of reverse transcription (RT) and polymerase chain reaction (PCR) from Bulge-Loop™ miRNA qRT-PCR Primer Set (RiboBio, China) as previously described [17]. RT and PCR were performed according to the manufacturer’s protocol. PCR product amplification was determined by the level of fluorescence in emitted by SYBR Green (SYBR® Premix Ex Taq™ II, TaKaRa). U6 snRNA was used for normalizing cells and FFPE samples. The relative expression levels of target miRNAs were calculated using the comparative 2−ΔΔCt method as previously described [15, 16]. The amount of miRNA in plasma samples was determined based on a standard curve constructed with the use of synthetic miRNAs (micrON miRNA mimic, RiboBio, Guangzhou, China) as previously described [17].
In vitro drug sensitivity assay
SGC7901 cells and SGC7901/DDP cells were plated in 6-well plates (6 × 105 cells/well). The miR-20a mimic (100 nM) or mimic control (100 nM) were transfected in SGC7901 cells, while miR-20a inhibitor (100 nM) or inhibitor control (100 nM) were transfected in SGC7901/DDP cells. The miR-20a mimic, inhibitor, and relative controls were obtained from GenePharma Company (Shanghai, China) (sequence of each provided in Supplementary data 1).
Twenty-four hours after transfection, the cells were seeded in 96-well plates (5 × 103 cells/well). After cellular adhesion, freshly prepared DDP was added with the final concentrations of 10, 1, 0.1, and 0.01 times of the human peak plasma concentration for DDP (2.0 μg/mL). Forty-eight hours after drug addition, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-tetrazolium bromide (MTT) was added to each well to assess the viability of the cells, and the plate was incubated for 2 h in a humidified incubator. The absorbance at 490 nm of each well was measured on a spectrophotometer. The concentration of cisplatin that caused 50 % inhibition of growth (IC50) was calculated by the relative survival curve.
Dual-luciferase activity assay
The 3ʹ-untranslated regions (3ʹUTR) sequences of NFKBIB that contain the predicted target site of miR-20a were chemically synthesized by Guangzhou RiboBio Co., Ltd (sequence shown in Supplementary data 2). Human embryonic kidney cell line HEK293T cells were seeded in 24-well plates (1.5 × 105 cells/well) and 24 h later, 200 ng of pGL3-NFKBIB-3′-UTR, 60 pmol of the miR-20a mimic or miRNA mimic control, and 80 ngpRL-TK (Promega) were co-transfected. The Dual-Luciferase Reporter Assay System (Promega, Madison, WI, USA) was used to measure luciferase activity consecutively. The firefly luciferase activity was normalized to the renilla luciferase expression.
Immunohistochemistry
Thirty tissue samples were formalin-fixed and paraffin-embedded, cut to 4-μm thick, and stained using the avidin-biotin complex method. Tissue slides were subjected to antigen retrieval using microwave irradiation in 10 mM citrate buffer (pH 6.0), followed by incubation with primary antibodies at 4 °C overnight. The antibody for NFKBIB was purchased from Cell Signaling Technology (Danvers, MA, USA). Staining was repeated if the result was uncertain. The slides were scored independently by two observers blinded to clinicopathological characteristics. They evaluated the immunostaining of the slides under an optical microscope at a magnification of ×400. Discordant scores were reevaluated to reach consensus. Both staining intensity of NFKBIB expression (scores 0–3 for negative, weak, moderate, and strong expression, respectively) and percentage of positive cancer cells (0–100 %) were assessed. We multiplied the staining intensity by the proportion score of the percentage of positive cancer cells.
Western blot
Total protein was extracted from the cells using RIPA buffer in the presence of proteinase inhibitor. Nuclear proteins were prepared from the cells using a Nuclear Cytoplasmic Extraction Kit (Beyotime, China). Western blot was carried out as previously described [15, 16]. The primary antibodies for NFKBIB, P65, livin, and survivin were purchased from Cell Signaling Technology (Danvers, MA, USA). Total protein level was normalized to GAPDH and nuclear protein to H3. Fold changes were calculated.
Apoptosis assay
Cells transfected with miR-20a mimic or inhibitor were treated with DDP at a final concentration of 10 μg/mL. Forty-eight hours later, apoptosis was evaluated by counting annexin V-fluorescein isothiocyanate-positive and propidium iodide-negative cells using the flow cytometry, as described previously [15, 16].
Statistical analysis
Data are expressed as the mean ± SD from at least three separate experiments. And two-tailed Student’s t tests were used for comparisons. SPSS (version 15.0, SPSS Inc., Chicago, IL, USA) software was used to perform all statistical analysis. A p value <0.05 was considered as statistically significant.
Results
MiR-20a was upregulated in GC plasma samples and GC tissue
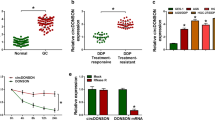
To reveal the expression of miR-20a in GC, we first detected the plasma levels of miR-20 in 30 cases of preoperative GC patients and 30 cases of healthy subjects by qRT-PCR. The expression of miR-20a was significantly increased in GC group compared with normal group (Fig. 1a). We further found that the expression of miR-20a was significantly higher in 30 cases of GC FFPE tissues compared with 30 cases of normal gastric tissue (Fig. 1b).
MiR-20a was upregulated in GC plasma samples and GC tissue. a Expression of miR-20a was detected in 30 normal plasma samples, 30 preoperative GC plasma samples by qRT-PCR. Expression levels of miR-20a in tumor group were higher than those in normal group. Y axis was presented as log10 (concentration; fmol/L). N normal, T tumor. b The expression levels of miR-20a were also upregulated in GC FFPE samples. Y axis was presented as relative expression (normalized to U6; 2−ΔΔCt). N normal, T tumor
MiR-20a was upregulated in GC patients with chemotherapeutic resistance
To further investigate the potential role of miR-20a in chemoresistance, 30 plasma samples and 40 FFPE sections from advanced GC who underwent 2 cycles of platinum-based chemotherapy were collected. Response to chemotherapy was evaluated by standard WHO criteria, defined as complete remission (CR), partial remission (PR), stable disease (SD), and progressive disease (PD). According to the criteria, 11 patients acquired PR, 9 patients acquired SD, 10 patients acquired PD, and no patients acquired CR in plasma samples. In tissue samples, 13 had PR, 12 had SD, 15 had PD, and none achieved CR. The expression of plasma miR-20a in the PD plus SD group was significantly higher than in the PR group (Fig. 2a). At the same time, upregulation of miR-20a in FFPE tissues was also found in the PD plus SD group compared with the PR group (Fig. 2b). QRT-PCR revealed that the expression of miR-20a was also significantly upregulated in SGC7901/DDP cells compared with the parental cell line SGC7901 (Fig. 2c), which was similar with the results of clinical GC samples. These results indicated that miR-20a was associated with sensitivity to chemotherapy in GC.
Expression of miR-20a was associated with chemoresistance in gastric cancer. a Plasma level of miR-20a is significantly higher in PD plus SD samples than in PR samples. b The expression of miR-20a in FFPE tissues was also upregulated in the PD plus SD group compared with the PR group. c The expression of miR-20a was also significantly upregulated in SGC7901/DDP cells compared with the parental cell line SGC7901. *p < 0.01
MiR-20a modulated cisplatin resistance of SGC7901/DDP cell line
Before the miRNA transfection, the significant resistance of SGC7901/DDP cells to cisplatin compared with the parental SGC7901 was verified (Supplementary data 3a). The levels of miR-20a were detected using qRT-PCR after the miRNA transfection (Supplementary data 3b, c). MTT assay revealed that sensitivity of SGC7901 cells transfected with miR-20a mimic to cisplatin was decreased compared with that treated with mimic control (Fig. 3a); however, miR-20a inhibitor significantly increased the sensitivity to cisplatin of SGC7901/DDP cells (Fig. 3b). These results suggested that miR-20a could regulate cisplatin resistance of SGC7901/DDP cells.
miR-20a modulated cisplatin resistance of SGC7901/DDP cell line. a MTT assay revealed that sensitivity of SGC7901 cells transfected with miR-20a mimic to cisplatin was decreased compared with that treated with mimic control. b In SGC7901/DDP cells, miR-20a inhibitor significantly increased the sensitivity to cisplatin. *p < 0.01
NFKBIB was a direct target of miR-20a
TargetScan predicted that NFKBIB is a potential direct target gene of miR-20a (Supplementary data 4). To assess the relevance of miR-20a/NFKBIB, we detected NFKBIB expression in 30 cases of GC tissues by IHC (Supplementary data 5). MiR-20a expression was also analyzed by qRT-PCR in the same samples. Among these 30 samples, using the median expression value of miR-20a as a cutoff point, the cohort was divided into miR-20a-high or miR-20a-low tumors. NFKBIB expression was significantly lower in the miR-20a-high group than in the miR-20a-low group (Fig. 4a). An inverse correlation (R 2 = 0.4033, p < 0.001) was observed between miR-20a and NFKBIB using Spearman’s correlation analysis (Fig. 4b). Moreover, we observed that NFKBIB was significantly decreased at protein level in SGC7901/DDP cells compared with the parental cell line SGC7901 (Fig. 4c).
miR-20a directly regulated NFKBIB expression. a NFKBIB and miR-20a expression levels were inversely correlated in GC tissues (n = 30). Y axis was presented as IHC scores. b A plot of the relative expression of miR-20a vs. NFKBIB showed an inverse correlation between the two. The correlation index R 2 was calculated using the Spearman’s rank test (R 2 = 0.4033, p < 0.001). Y axis was presented as IHC scores. X axis was presented as relative expression (normalized to U6; −ΔCt). c NFKBIB expression at protein level in SGC7901/DDP cells and the parental cell line SGC7901. d Luciferase assay showed that co-transfection of NFKBIB 3ʹUTR and miR-20a decreased relative luciferase activity in 293T cells. *p < 0.01
To further verify whether NFKBIB is a target for miR-20a, luciferase reporter assays were performed in human embryonic kidney cell line HEK293T. We found that luciferase activity was significantly decreased in cells transfected with miR-20a when the construct contained the 3ʹUTR of NFKBIB (Fig. 4d). These results showed that miR-20a might negatively regulate NFKBIB expression by directly targeting its 3ʹUTR.
MiR-20a could activate the NFκB pathway and upregulate the downstream targets survivin and livin by repressing NFKBIB expression
In our study, the increased expression of miR-20a in SGC7901/DDP cells was accompanied by the downregulation of the NFKBIB, compared with the parental SGC7901 cells. Interestingly, NFκB pathway-related proteins, p65, livin, and survivin were significantly higher in SGC7901/DDP cells than in the control group (Fig. 5a). Our study showed that NFKBIB is a direct target of miR-20a, and NFKBIB is a negative regulator of the NFκB pathway. Therefore, we hypothesized that miR-20a might activate the NFκB pathway by downregulating the expression of NFKBIB. To determine this, we transfected SGC7901/DDP cells and SGC7901 cells with the inhibitor or mimic of miR-20a, respectively, and Western blot was performed to detect the expression of these proteins. In SGC7901/DDP cells, at 72 h after transfection, the results revealed that a low level of miR-20a increased the expression of NFKBIB, while it decreased the expression of p65, survivin, and livin (Fig. 5b). In addition, the expression of NFKBIB in SGC7901 transfected with miR-20a mimic was downregulated; however, the expression levels of p65, survivin, and livin were upregulated (Fig. 5c). These results indicated that miR-20a repressed the expression of NFKBIB, thus leading to the activation of the NFκB pathway and the upregulation of the downstream targets livin and survivin.
miR-20a could activate the NFκB pathway and upregulate the downstream targets livin and survivin by repressing NFKBIB expression. a A Western blot analysis showed that p65, livin, and survivin was upregulated in SGC7901/DDP cells. The level of p65 in the nucleus was normalized to H3. b In SGC7901/DDP cells transfected with miR-20a inhibitor, at 72 h after transfection, the results revealed that low levels of miR-20a increased the expression of NFKBIB, while it decreased the expression of p65, livin, and survivin. c The expression of NFKBIB in SGC7901 transfected with miR-20a mimic was downregulated; however, the expression levels of p65, livin, and survivin were upregulated. NC mimic control, IC inhibitor control. *p < 0.01
Reduced miR-20a expression sensitized SGC7901/DDP cells to DDP-induced apoptosis
It was reported that defective drug-induced apoptosis caused by the overexpression of anti-apoptotic proteins such as livin, survivin, and inhibitors of apoptosis could be related to the development of drug resistance in numerous cancers [15, 16, 18]. Because miR-20a might enhance cisplatin resistance of GC cells at least in part through activating the NFκB pathway and its downstream targets livin and survivin. We hypothesized that miR-20a might also be involved in the development of cisplatin resistance by regulating the apoptosis of gastric cancer cells. To further verify this hypothesis, SGC7901/DDP and SGC7901 cells were transfected with the inhibitor or mimic of miR-20a, respectively, followed by analysis of DDP-induced apoptosis. The results showed that there was a higher proportion of apoptotic cells in SGC7901/DDP cells transfected with miR-20a inhibitor after DDP treatment compared with those transfected with inhibitor control (Fig. 6a). On the contrary, increased expression of miR-20a led to a decrease in the apoptosis induced by DDP in SGC7901 cells (Fig. 6b).
Reduced miR-20a expression sensitized SGC7901/DDP cells to DDP-induced apoptosis. a SGC7901/DDP was transfected with miR-20a inhibitor or control after DDP treatment. There were more cells undergoing apoptosis in the miR-20a downregulation group. b SGC7901 was transfected with miR-20a mimic or control. There were fewer cells undergoing apoptosis in the miR-20a overexpression group. Representative flow cytometry report was next to the graphs. *p < 0.01
Discussion
miR-20a, a member of miR-17∼92 cluster, functions as an oncogene. miR-20a was upregulated and could promote cancer progression in diverse cancer subtypes, including cervical cancer [19], ovarian cancer [20], osteosarcoma [21], anaplastic thyroid cancer [22], and nasopharyngeal carcinoma [23]. Li et al. reported that miR-20a was dramatically increased in GC tissues and cell lines and that ectopic expression of miR-20a promoted proliferation, migration, and invasion of GC cells [24]. Chai et al. revealed that miR-20a targeted BNIP2 and contributed to chemotherapeutic resistance in colorectal adenocarcinoma cell lines [25]. Our study confirmed that miR-20a showed pronounced upregulation in SGC7901/DDP cells and chemoresistance samples and modulated cisplatin resistance of SGC7901/DDP cell line.
The NFκB complex is a heterodimer (p65/p50 subunits) localized in cytoplasm with a family of inhibitory proteins called IκB proteins. Upon degradation of the IκB proteins, the NFκB complex translocate to the nucleus to activate target genes. NFKBIB is one of the NFκB inhibitor family [26]. Here, we found that miR-20a could directly target NFKBIB. Recent studies have shown that sustained NFκB activation and upregulation of drug resistance-related proteins (BCL-2, MCL-1, MDR1, and XIAP, etc.) play important roles in mediating resistance in various cancers [7, 27, 28]. Moreover, a previous study suggested that inhibition of the NFκB pathway enhanced cisplatin sensitivity in GC [29]. In this study, we sought to investigate whether there was a correlation between miR-20a and the NFκB pathway to clarify the effects that miR-20a exerted on GC chemoresistance. We found that increased expression of miR-20a in SGC7901/DDP cells was accompanied by the downregulation of the NFKBIB and the upregulation of p65. Moreover, the expression level of NFKBIB was upregulated with the downregulation of p65 when miR-20a was knocked down in SGC7901/DDP. In SGC7901, miR-20a overexpression decreased the expression of NFKBIB and increased the expression of p65. The aforementioned results indicated that miR-20a was essential at least in part for activating the NFκB pathway in GC.
Livin and survivin are members of the IAP family that function as anti-apoptotic factors and play important roles in inhibiting apoptosis [30]. The overexpression of IAPs is highly related to cancer chemoresistance. Our results showed that miR-20a could modulate the expression of livin and surviving through the NFκB pathway. Knockdown of miR-20a could sensitize SGC7901/DDP cells to DDP-induced apoptosis. We considered that miR-20a might also be associated with development of cisplatin resistance by regulating the apoptosis of GC cells.
However, the mechanism of the upregulation of miR-20a was still unknown. By using the UCSC database, we found that there are NFκB binding sites in the promoter regions of miR-20a, which indicated that NFκB might directly regulate miR-20a expression. The study of Zhou et al. supported our conjecture [31]. We hypothesized that the positive feedback loop, miR-20a/NFKBIB/NFκB/miR-20a, might play important roles in GC drug resistance. However, more research was required to clarify the mechanism.
In conclusion, the results presented here provided the first evidence that miR-20a could contribute to the development of cisplatin resistance in human GC cells, by targeting NFKBIB, leading to the activation of NFκB and upregulation of the downstream targets survivin and livin. However, it should be noted that our results were obtained from cell lines and did not necessarily reflect the actual surrogates for clinical tumors. Therefore, more research was needed to elucidate the function of miR-20a both in vivo and in clinical practice.
Abbreviations
- miRNAs:
-
microRNAs
- DDP:
-
Cisplatin
References
Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136(5):E359–86.
Holohan C, Van Schaeybroeck S, Longley DB, Johnston PG. Cancer drug resistance: an evolving paradigm. Nat Rev Cancer. 2013;13(10):714–26.
Longley DB, Johnston PG. Molecular mechanisms of drug resistance. J Pathol. 2005;205(2):275–92.
Nakagawa Y, Sedukhina AS, Okamoto N, Nagasawa S, Suzuki N, Ohta T, et al. NF-kappaB signaling mediates acquired resistance after PARP inhibition. Oncotarget. 2015;6(6):3825–39.
Halilovic E, She QB, Ye Q, Pagliarini R, Sellers WR, Solit DB, et al. PIK3CA mutation uncouples tumor growth and cyclin D1 regulation from MEK/ERK and mutant KRAS signaling. Cancer Res. 2010;70(17):6804–14.
Michaelis M, Rothweiler F, Barth S, Cinatl J, van Rikxoort M, Loschmann N, et al. Adaptation of cancer cells from different entities to the MDM2 inhibitor nutlin-3 results in the emergence of p53-mutated multi-drug-resistant cancer cells. Cell Death Dis. 2011;2:e243.
Lin X, Zhang X, Wang Q, Li J, Zhang P, Zhao M, et al. Perifosine downregulates MDR1 gene expression and reverses multidrug-resistant phenotype by inhibiting PI3K/Akt/NF-kappaB signaling pathway in a human breast cancer cell line. Neoplasma. 2012;59(3):248–56.
Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116(2):281–97.
Xiang Y, Ma N, Wang D, Zhang Y, Zhou J, Wu G, et al. MiR-152 and miR-185 co-contribute to ovarian cancer cells cisplatin sensitivity by targeting DNMT1 directly: a novel epigenetic therapy independent of decitabine. Oncogene. 2014;33(3):378–86.
Shang Y, Zhang Z, Liu Z, Feng B, Ren G, Li K, et al. miR-508-5p regulates multidrug resistance of gastric cancer by targeting ABCB1 and ZNRD1. Oncogene. 2014;33(25):3267–76.
Sui C, Meng F, Li Y, Jiang Y. miR-148b reverses cisplatin-resistance in non-small cell cancer cells via negatively regulating DNA (cytosine-5)-methyltransferase 1(DNMT1) expression. J Transl Med. 2015;13:132.
Fang L, Li H, Wang L, Hu J, Jin T, Wang J, et al. MicroRNA-17-5p promotes chemotherapeutic drug resistance and tumour metastasis of colorectal cancer by repressing PTEN expression. Oncotarget. 2014;5(10):2974–87.
van Jaarsveld MT, Helleman J, Boersma AW, van Kuijk PF, van Ijcken WF, Despierre E, et al. miR-141 regulates KEAP1 and modulates cisplatin sensitivity in ovarian cancer cells. Oncogene. 2013;32(36):4284–93.
Eberle KE, Sansing HA, Szaniszlo P, Resto VA, Berrier AL. Carcinoma matrix controls resistance to cisplatin through talin regulation of NF-kB. PLoS One. 2011;6(6):e21496.
Zhu W, Xu H, Zhu D, Zhi H, Wang T, Wang J, et al. miR-200bc/429 cluster modulates multidrug resistance of human cancer cell lines by targeting BCL2 and XIAP. Cancer Chemother Pharmacol. 2012;69(3):723–31.
Zhu W, Shan X, Wang T, Shu Y, Liu P. miR-181b modulates multidrug resistance by targeting BCL2 in human cancer cell lines. Int J Cancer. 2010;127(11):2520–9.
Zhao DS, Chen Y, Jiang H, Lu JP, Zhang G, Geng J, et al. Serum miR-210 and miR-30a expressions tend to revert to fetal levels in Chinese adult patients with chronic heart failure. Cardiovasc Pathol. 2013;22(6):444–50.
Zhu DX, Zhu W, Fang C, Fan L, Zou ZJ, Wang YH, et al. miR-181a/b significantly enhances drug sensitivity in chronic lymphocytic leukemia cells via targeting multiple anti-apoptosis genes. Carcinogenesis. 2012;33(7):1294–301.
Zhao S, Yao D, Chen J, Ding N, Ren F. MiR-20a promotes cervical cancer proliferation and metastasis in vitro and in vivo. PLoS One. 2015;10(3):e0120905.
Fan X, Liu Y, Jiang J, Ma Z, Wu H, Liu T, et al. miR-20a promotes proliferation and invasion by targeting APP in human ovarian cancer cells. Acta Biochim Biophys Sin. 2010;42(5):318–24.
Huang G, Nishimoto K, Zhou Z, Hughes D, Kleinerman ES. miR-20a encoded by the miR-17-92 cluster increases the metastatic potential of osteosarcoma cells by regulating Fas expression. Cancer Res. 2012;72(4):908–16.
Xiong Y, Zhang L, Kebebew E. MiR-20a is upregulated in anaplastic thyroid cancer and targets LIMK1. PLoS One. 2014;9(5):e96103.
Zeng X, Xiang J, Wu M, Xiong W, Tang H, Deng M, et al. Circulating miR-17, miR-20a, miR-29c, and miR-223 combined as non-invasive biomarkers in nasopharyngeal carcinoma. PLoS One. 2012;7(10):e46367.
Li X, Zhang Z, Yu M, Li L, Du G, Xiao W, et al. Involvement of miR-20a in promoting gastric cancer progression by targeting early growth response 2 (EGR2). Int J Mol Sci. 2013;14(8):16226–39.
Chai H, Liu M, Tian R, Li X, Tang H. miR-20a targets BNIP2 and contributes chemotherapeutic resistance in colorectal adenocarcinoma SW480 and SW620 cell lines. Acta Biochim Biophys Sin. 2011;43(3):217–25.
Hayden MS, Ghosh S. Shared principles in NF-kappaB signaling. Cell. 2008;132(3):344–62.
Wang ZH, Chen H, Guo HC, Tong HF, Liu JX, Wei WT, et al. Enhanced antitumor efficacy by the combination of emodin and gemcitabine against human pancreatic cancer cells via downregulation of the expression of XIAP in vitro and in vivo. Int J Oncol. 2011;39(5):1123–31.
Li F, Sethi G. Targeting transcription factor NF-kappaB to overcome chemoresistance and radioresistance in cancer therapy. Biochim Biophys Acta. 2010;1805(2):167–80.
Xia JT, Chen LZ, Jian WH, Wang KB, Yang YZ, He WL, et al. MicroRNA-362 induces cell proliferation and apoptosis resistance in gastric cancer by activation of NF-kappaB signaling. J Transl Med. 2014;12:33.
Vucic D, Fairbrother WJ. The inhibitor of apoptosis proteins as therapeutic targets in cancer. Clin Cancer Res. 2007;13(20):5995–6000.
Zhou R, Hu G, Gong AY, Chen XM. Binding of NF-kappaB p65 subunit to the promoter elements is involved in LPS-induced transactivation of miRNA genes in human biliary epithelial cells. Nucleic Acids Res. 2010;38(10):3222–32.
Acknowledgments
The authors are grateful to the fund support by the National Natural Science Foundation of China (grant number 81201705 and 81201796) and the Natural Science Foundation of Jiangsu Province (grant number BK2012442).
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding authors
Additional information
Yiping Du and Mingxia Zhu contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 285 kb)
Rights and permissions
About this article
Cite this article
Du, Y., Zhu, M., Zhou, X. et al. miR-20a enhances cisplatin resistance of human gastric cancer cell line by targeting NFKBIB. Tumor Biol. 37, 1261–1269 (2016). https://doi.org/10.1007/s13277-015-3921-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-3921-1