Abstract
The current study aims to investigate the fuctional role of miRNA-25 in non-small cell lung cancer (NSCLC) cells. Quantitative real-time polymerase chain reaction (qRT-PCR) was used to detect the expression of miR-25 in NSCLC cell lines and 11 pairs of human NSCLC and non-cancerous tissues. The inhibitor of miR-25 was stably transfected into NSCLC cell line A549 cells. Then the effects of downregulating miR-25 on cancer cell proliferation, cell cycle arrest, chemosensitivity to cisplatin, and growth of in vivo xenograft were investigated. Direct regulation of miR-25 on its target gene, cell division cycle 42 (CDC42), was examined by luciferase reporter assay, qRT-PCR and western blot. CDC42 was then upregulated in A549 cells to investigate its effect on miR-25-mediated NSCLC cell proliferation and cell cycle arrest. The expression of miR-25 in NSCLC cells or human tissues was significantly higher than that in normal lung cells or adjacent non-cancerous tissues, respectively. Downregulation of miR-25 markedly inhibited A549 cell proliferation, induced G1 cell cycle arrest, increased cisplatin sensitivity, and suppressed the growth of caner cell xenograft in vivo. CDC42 was confirmed to be the directly regulated by miR-25 in A549 cells. Upregulation of CDC42 in A549 cells rescued the inhibitory effect on proliferation and the G1 cell cycle arrest induced by miR-25 downregulation. Our study demonstrates miR-25, by targeting CDC42, is an important regulator in NSCLC.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Lung cancer is one of the leading causes of tumor-related deaths in adults. Around the world, including both men and women, more than 22,000 new cases of lung cancer emerge every year with very high mortality rate [1]. Among all forms of lung cancer, non-small cell lung cancer (NSCLC) accounts for more than 80 % of the cases [2]. Although great progress has been made in early diagnosis and treatment strategies in recent decades, patients with NSCLC are normally associated with poor prognosis, with an estimated 5-year survival rate of no more than 20 % [1, 2]. Thus, it is of great meaning to further our understanding on the underlying molecular and cellular mechanisms of NSCLC to develop better diagnostic and treatment methods.
MicroRNAs (miRNAs) are groups of small length, non-coding RNAs that play important roles in cell regulation by binding the 3′-untreanslated region (3′-UTR) of targeted genes and regulating gene expression posttranscriptionally [3, 4]. In cancers, miRNA can act on both tumor oncogenes and tumor-suppressing genes to exert wide range of regulations on tumorigenesis [5–7], cancer cell proliferation and development [7, 8], cancer cell apoptosis [9, 10], and cancer cell chemosensitivity [11–14]. Recently, miR-25 has been identified as a key factor in regulating molecular functions in various cancer forms, including apoptosis in ovarian cancer [15], tumor initiation in breast cancer [16], and tumor proliferation in hepatocellular carcinoma [17, 18]. In lung cancer, miR-25 was found to be highly expressed in cancerous tissues [19, 20], but its exact role in lung cancer remains elusive.
Cell division cycle 42 (CDC42), a member of Rho GTPase family, is involved in various cancer functions, including cancer proliferation, cancer invasion, and cancer metastasis [21, 22]. In lung cancer, CDC42 was found to be highly associated with poor clinical outcomes in patients with NSCLC [23], and inhibition on CDC42 or GTPase signaling pathway could suppress cancer cell metastasis [24].
In the present study, we firstly examined the expression level of miR-25 in both NSCLC cancer cell lines and cancer patients. Then we modified the endogenous expression level of miR-25 in NSCLC cell line, A549 cells to investigate the functional role of miR-25 in regulating NSCLC proliferation, cell cycle arrest, chemosensitivity to cisplatin and tumor growth of xenograft in vivo. Furthermore, we explored the possibility of miR-25 modulating NSCLC through CDC42 gene. The results here would help to improve our understanding on the underlying mechanisms of miRNA regulation in NSCLC.
Materials and methods
Cell lines and culture
The NSCLC cell lines, including H520, H460, A549, and the control fetal lung fibroblast cell line MRC5, were all purchased from Shanghai Institute of Cell Biology, China Academy of Sciences in Shanghai, China. All cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM; Invitrogen, USA) supplemented with 10 % fetal bovine serum (FBS; Invitrogen, USA), 100 μg/mL streptomycin, and 100 U/mL penicillin, in a tissue culture incubator under the atmosphere of 5 % CO2 and 37 C.
Patients and clinical samples
There were a total of 11 pairs of matched NSCLC tissues (T) and adjacent non-cancerous tissues (ANT) extracted from patients in the Department of Respiratory Medicine at the First Affiliated Hospital, College of Medicine, Xi’an Jiaotong University in Xi’an, China, between October 2013 and June 2014. All patients gave written consent forms. The study was reviewed and approved by the ethics committee of the First Affiliated Hospital. None of the patients received chemotherapy or radiotherapy before surgery. Clinical samples were snap frozen in liquid nitrogen and stored at −80 C until further use.
Quantitative real-time polymerase chain reaction
Total RNA was isolated from cancer cell lines or clinical samples using TRIzol reagent per manufacturer’s protocol (Invitrogen, US). Reverse-transcribed complementary DNA (cDNA) was synthesized with the Prime-script RT reagent kit per manufacturer’s protocol (TaKaRa, China). Quantitative RT-PCR assay was performed with SYBR Premix Ex Taq per manufacturer’s protocol (TaKaRa, China). The primers for miR-25 were forward 5′-TCTGGTCTCCCTCACAGGAC-3′ and reverse 5′-CATGGGTCGCCTACTCAC-3′. The primers form CDC42 were forward 5-ACGACCGCTGAGTTATCCAC-3′ and reverse 5′-TATGGGCCTTGTCTCACACG-3′. MiRNA was detected by the TaqMan miRNA Assay per manufacturer’s protocol (Applied Biosystems). The amplification conditions were 35 cycles of 12 s at 95 °C and 1 min at 60 °C. The expression levels of miR-25 and CDC42 were normalized by the expression level of U6 small nuclear RNA and GAPDH, respectively.
MiRNA transfection
The miR-25 inhibitor (miR-25-I) and its non-specific control miRNA (miR-NC) were synthesized by Shanghai GenePharma (Shanghai, China). For cell transfection, A549 cells were seeded in six-well plates (3 × 105/well) and transfected with 100 nM miR-25-I or miR-NC using Lifofectamine 2000 per manufacturer’s protocol (Invitrogen, USA).
Cell proliferation assay
Lung cancer A549 cells were seeded in 96-well plates (3 × 105/well) and transfected with 100 nM miR-25-I or miR-NC. One day after transfection, viable cells were transferred to 96-well plates (3 × 105/well) and maintained at 37 C and 5 % CO2 for additional 4 days. Cell proliferation was estimated by a 3-(4,5-dimethylthazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay and determined by measuring the optical density (O.D.) at 490 nm using a CytoFluor II multiwell plate reader (PerSeptive Biosystems, USA).
Cell cycle assay
Lung cancer A549 cells were seeded in 96-well plates (3 × 105/well) and transfected with 100 nM miR-25-I or miR-NC. Three days after transfection, cells were collected by trypsinization and centrifugation. The cell pellets were fixed in ice-cold 70 % ethanol for 48 h, washed by ice-cold PBS (2 × 10 min), and resuspended in 1 mL of PBS containing 1 mg/mL RNase and 50 μg/mL propidium iodide at room temperature. It was then incubated in a dark box for 30 min, followed by fluorescence-activated cell sorter scan (FACS; Becton Dickinson, USA). A total number of 10,000 cells was estimated for each data set and analyzed using the ELITE software (Phoenix Flow Systems, USA).
Cisplatin assay
A549 cells were seeded in 96-well plates (3 × 105/well) and transfected with 100 nM miR-25-I or miR-NC. Twenty-four hours after transfection, fresh media containing various concentrations of cisplatin (0, 5, 10, 15, 20, and 25 μg/mL) was added into culture. Twelve hours later, cell viability was determined by MTT assay. For time-dependent study, 5 μg/mL cisplatin was used and MTT assay was performed in 0, 12, 24, 36, and 48 h.
Lung cancer xenograft
A549 cells were transfected with 250 nM miR-25-I or miR-NC for 6 h. Cells were collected and subcutaneously injected into nude BALB/c mice (SLAC laboratory, Shanghai, China) with a sterile 27 1/2-gauge needle with 0.2 mL cell suspension of 3.5 × 106 cells. Tumor volumes were calculated using the formula: tumor volume = length × width2/2. Tumors were monitored weekly and extracted 35 days after inoculation.
Luciferase reporter assay
The cDNA library of A549 cells was used to conduct conventional PCR to amplify the putative miR-25 binding site in CDC42 3′-UTR and its mutated binding site. The wild-type 3′-UTR and the mutated 3′-UTR sequences of CDC42 were inserted into a luciferase reporter vector (pmiR-REPORT, Ambion, USA) to make constructs of wild-type 3′-UTR segment of CDC42 (Luc-CDC42-W) and mutated 3′-UTR segment of CDC42 (Luc-CDC42-m), respectively. The sequences were verified by DNA sequencing. HA549 cells were seeded in 24-well plates (3 × 105/well). One day later, Luc-CDC42-W, Luc-CDC42-m, and pmiR-REPORT control vector (Luc-C) were co-transfected with miR-25-I and β-galactosidase in A549 cells using Lipofectamine 2000 per manufacturer’s protocol. The luciferase activity was examined using a dual luciferase reporter assay system (Promega, USA) 48 h after transfection. The luciferase activities were normalized to the β-galactosidase activity of Luc-C.
Western blot assay
Two days after transfection, A549 cell lysis was collected. The proteins were run on 9 % sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SS-PAGE) at 70 V for 3 h, then transferred to polyvinylidene difuoride membranes and incubated with primary antibody of CDC42 (1:200, Santa Cruz Technology, USA). After washing, the blot were incubated with horseradish peroxidase-conjugated secondary antibodies and visualized with an X-ray film. β-actin was set as internal control.
CDC42 overexpression
The recombinant plasmid eukaryotic expression vector pcDNA3.1-CDC42 and non-specific control vector (pcDNA3.1-NC) were purchased from Cell Signaling Technology, Inc (Cell Signaling Technology, USA). A549 cells were seeded in a six-well plate (3 × 105/well), and pcDNA3.1-CDC42 or pcDNA3.1-NC were then transfected into cells using Lipofectamine 2000 per manufacturer’s protocol for 48 h before next experiments.
Statistical analysis
The results were representative of at least three independent experiments and presented as the mean ± standard errors. Comparison of time-dependent cell proliferation and xenograft tumor volumes were conducted using one-way ANOVA, and other comparisons were made with Student’s t test. Statistical significance was defined as P < 0.05.
Results
MiR-25 is upregulated in NSCLC cell lines and NSCLC patients
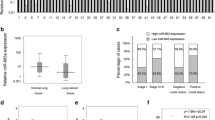
We used quantitative real-time polymerase chain reaction (qRT-PCR) to measure miR-25 expression in three NSCLC cell lines, H520, H460, and A549 cells, and compare them with the miR-25 expression in a normal fetal lung fibroblast cell line MRC5 cells. The data showed that compared with MRC5, all NSCLC cells had significantly high expression levels of miR-25 (Fig. 1a, *P < 0.05, as compared with MRC5), suggesting miR-25 might play a functional role in NSCLC.
MiR-25 expression levels in NSCLC cell lines and patients. a MiR-25 expression levels were examined by qRT-PCR in H520, H460, and A549 cells and normalized to the expression level in MRC5 cells (*P < 0.05, as compared with MRC5). b MiR-25 expression levels in 11 pairs of cancer tissues (T) and adjacent non-cancerous tissues (ANT) were compared by qRT-PCR (*P < 0.05, as compared with ANT of each case)
Similar results were obtained from NSCLC patients. We compared the miR-25 expressions in paired NSCLC cancer T and ANT in 11 patients (Fig. 1b). We found that miR-25 expression in cancer tissues was much higher than the expression in adjacent non-cancerous tissues (Fig. 1b, *P < 0.05).
Downregulation of miR-25 inhibits NSCLC cell proliferation, induces cell cycle arrest, and increases chemosensitivity to cisplatin
We then asked what the functional role would be for miR-25 in regulating NSCLC. We transfected NSCLC cell line A549 cells with either miR-25-I (100 nM) or miR-NC. The efficiency and specificity of miR-25-I in downregulating endogenous miR-25 in A549 cells were confirmed by qPCR (Fig. 2a, *P < 0.05). The effect of downregulating miR-25 on cell growth was examined by a proliferation assay in A549 cells. The result showed that miR-25 downregulation significantly inhibited A549 cell proliferation from 48 to 96 h after transfection (Fig. 2b, *P < 0.05, one-way ANOVA).
MiR-25 downregulation had antitumor effect on NSCLC cells. A549 cells were transfected with miR-25 inhibitor (miR-25-I; 100 nM) and a non-specific control miRNA (miR-NC; 100 nM). a The efficiency of miR-25-I was examined by qPCR. b A cell proliferation assay was performed at 24, 48, 72, and 96 h after transfection. Cell proliferation in A549 cells was significantly inhibited by downregulating miR-25 (*P < 0.05, one-way ANOVA). c A cell cycle assay was performed 72 h after transfection. MiR-25 downregulation significantly induced cell cycle G1 arrest in A549 cells (*P < 0.05). d Twenty-four hours after transfection, various concentrations of cisplatin (0, 5, 10, 15, 20, 25 μg/ml) were added into the culture of A549 cells. Cell proliferation was compared between A549 cells transfected with miR-25-I and the cells transfected with miR-NC, by using a MTT assay 72 h after cisplatin treatment (*P < 0.05, as compared with miR-NC). e With the application of 5 μg/ml cisplatin, cell proliferation was also compared between A549 cells transfected with miR-25-I and the cells transfected with miR-NC, in a time-dependent manner at 12, 24, 36, and 48 h after cisplatin treatment (*P < 0.05, as compared with miR-NC)
We then analyzed the effect of downregulating miR-25 on NSCLC cell cycle. The result showed that a significant reduction in the number of cells in the S-phase of the cell cycle, as well as a marked increase in the number of cells in G0/G1 in A549 cells transfected with miR-25-I (Fig. 2c, *P < 0.05). On the other hand, no significant difference in G2/M phases was detected between A549 cells transfected with miR-25-I and A549 cells transfected with miR-NC. Thus, our results suggest that miR-25 downregulation induces G1-phase arrest in NSCLC cells.
We also examined the effect of downregulating miR-25 on NSCLC cell’s chemoresistance to cisplatin. A549 cells were transfected with miR-25-I or miR-NC (100 nM). Twenty-four hours after transfection, A549 cells were treated with various concentrations (0, 5, 10, 15, 20, 25 μg/mL) of cisplatin for 12 h, or 5 μg/mL of cisplatin for 0, 12, 24, 36, and 48 h. The results of MTT proliferation assay demonstrated that, 12 h after cisplatin treatment, downregulation of miR-25 significantly increased chemosensitivity of A549 cells at concentrations of 10, 15, 20, and 25 μg/mL of cisplatin (Fig. 2d, *P < 0.05). The results also showed that with 5 μg/mL of cisplatin, miR-25 downregulation dramatically increased chemosensitivity of A549 cells 24, 36, and 48 h after treatment (Fig. 2e, *P < 0.05). Thus, our results suggest that downregulation of miR-25 increased cisplatin chemosensitivity in both dose- and time-dependent manners in NSCLC cells.
Downregulation of miR-25 inhibits tumor growth of NSCLC cells in vivo
As we showed downregulating miR-25 had functional role in regulating NSCLC cells in vitro, we further investigated whether suppressing miR-25 would have similar antitumor effect to inhibit NSCLC tumor growth in vivo. A549 cells were transfected with either miR-25-I (250 nM) or miR-NC (250 nM). Six hours later, cells were subcutaneously inoculated into female null mice. The tumors were extracted 5 weeks after injection. The results showed that tumors were significantly smaller while miR-25 was downregulated (Fig. 3a). Quantification of tumor sizes confirmed that suppressing miR-25 markedly inhibited the ability of A549 cells to form tumors in vivo (Fig. 3b, *P < 0.05, one-way ANOVA).
MiR-25 downregulation inhibited tumor growth in vivo. A549 cells were transfected with miR-25 inhibitor (miR-25-I; 250 nM) and a non-specific control miRNA (miR-NC; 250 nM). Cells were then subcutaneously injected into female nude mice. a A photograph was shown for the tumors extracted 5 weeks after injection. b Tumor sizes were calculated by a formula: (length × width2)/2 (*P < 0.05, one-way ANOVA)
CDC42 is the target of miR-25 in NSCLC cells
As we showed downregulating miR-25 inhibited A549 cells growth both in vitro and in vivo, we then investigated the downstreaming targets of miR-25. By searching the target genes of miR-25 through the algorithm of miRana and Target Scan, we found CDC42, a cell proliferation regulator, had putative miR-25 binding sites within its 3′-UTR in the online database of Target Scan (Fig. 4a, top panel). Thus, we used a luciferase report assay to examine whether miR-25 directly suppresses CDC42 production in A549 cells. We cloned the speculated miR-25 binding site on 3′-UTR of wild-type CDC42 into luciferase vector Luc-CDC42-W and co-transfected it with β-galactosidase and miR-25-I into A549 cells. The luciferase vectors containing Luc-CDC42-m, or a non-specific control luciferase vector (Luc-C) were also constructed and transfected into A549 cells, with β-galactosidase and miR-25-I. The result showed that the luciferase activity of Luc-CDC42-W was significantly lower than the luciferase activities of Luc-C or Luc-CDC42, suggesting that miR-25 directly regulated CDC42 production in A549 cells (Fig. 4a, bottom panel, *P < 0.05).
MiR-25 downregulation targeted CDC42. a The speculated miR-25 binding site on CDC42 3′-UTR was inserted into luciferase reporter vector pmiR-REPORT to make the wild-type fragment vector Luc-CDC42-W. The luciferase vector containing a mutated 3′-UTR segment of CDC42 (Luc-CDC42-m), as well as a blank control luciferase vector (Luc-C) were also constructed and verified by sequencing. A549 cells were then co-transfected with β-galactosidase, miR-25-I, and either of the three luciferase vectors, Luc-CDC42-W, Luc-CDC42-m, or Luc-C for 24 h. The relative luciferase activities were analyzed and normalized to the luciferase activity of Luc-C vector (*P < 0.05). b A549 cells were transfected with either miR-25-I (100 nM) or miR-NC (100 nM). Twenty-four hours after transfection, the endogenous mRNA level of CDC42 was examined by qRT-PCR (*P < 0.05). c Forty-eight hours after transfection, CDC42 protein levels were assessed by western blotting using β-actin as internal control
To further examine whether miR-25 was directly regulating CDC42 in NSCLC cells, we transfected A549 cells with either miR-25-I (100 nM) or miR-NC (100 nM). Twenty-four hours halter, qRT-PCR showed that CDC42 mRNA was significantly downregulated by miR-25-I (Fig. 4b). Forty-eight hours later, western blots demonstrated that CDC42 protein was also markedly downregulated by miR-25-I (Fig. 4c).
Upregulation of CDC42 rescued the reduced proliferation and G1 cell cycle arrest induced by miR-25 downregulation in NSCLC cells
Finally, we investigated whether miR-25 regulation on NSCLC could be affected by CDC42. We inserted CDC42 sequence into recombinant plasmid eukaryotic expression vector pcDNA3.1 (pcDNA3.1-CDC42) to ectopically overexpress CDC42 in A549 cells. The result of qRT-PCR demonstrated that the endogenous CDC42 mRNA was upregulated in A549 cells transfected with pcDNA3.1-CDC42, as compared with the cells transfected with control vector of pcDNA3.1-NC (Fig. 5a). Further western blotting analysis confirmed that the protein level of CDC42 was also upregulated (Fig. 5b).
Overexpressing CDC42 reversed the tumor-suppressing effect by miR-25 downregulation. A549 cells were transfected with recombinant plasmid eukaryotic expression vector pcDNA3.1-CDC42 to overexpress CDC42. Control A549 cells were transfected with a non-specific control vector (pcDNA3.1-NC). Forty-eight hours after transfection, qRT-PCR (a) and western blot (b) were used to examine the mRNA and protein expression levels of CDC42 in A549 cells (*P < 0.05). c A549 cells were initially transfected miR-25-I for 24 h, then transfected with either pcDNA3.1-CDC42 or pcDNA3.1-NC. Cell proliferation was monitored by a MTT assay for five more consecutive days (*P < 0.05, one-way ANOVA). d Seventy-two hours after second transfection, a cell cycle assay was used to examined the arrested cell cycle progression (*P < 0.05)
We then conducted double transfection in A549 cells, as they were initially transfected with miR-25-I for 24 h, then with either pcDNA3.1-CDC42 or pcDNA3.1-NC. A MTT assay showed that ectopic expression of CDC42 rescued the impaired proliferation induced by miR-25 downregulation in A549 cells (Fig. 5c, *P < 0.05, one-way ANOVA). A cell cycle assay also demonstrated that ectopic expression of CDC42 reversed the G1 cell cycle arrest induced by miR-25 downregulation in A549 cells (Fig. 5d, *P < 0.05).
Discussions
Strong evidence demonstrates that the expression level of miRNA differs in carcinoma and non-carcinoma tissues with great molecular and clinical implications. In the present study, we investigated the expression levels of miR-25 in three NSCLC cell lines, as well as cancer samples from NSCLC patients. We demonstrated that miR-25 was generally highly expressed in NSCLC cell lines and cancer tissues from patients, as compared with normal lung fibroblast cell line MRC5 and adjacent non-cancerous tissues from patients, respectively. These results are consistent with previous study showing that miR-25 was highly expressed in the serum samples from NSCLC patients [19], suggesting that miR-25 might exert oncogenic effect on NSCLC development. Interestingly, a recent study focusing on the subtypes of NSCLC discovered that, miR-25 was more likely to predict the survival of patients with squamous cell carcinomas, one subtype of NSCLC, than the survival of patients with adenocarcinoma, another subtype of NSCLC. Thus, further study to compare the expression and function of miR-25 among different subtypes of NSCLC would help to define the specificity of miR-25 as a possible biomarker in NSCLC.
Not only did we investigate the expression of miR-25 but also we explored the functional role of miR-25 in regulating NSCLC, the first ever attempt to address the regulatory mechanism of miR-25 in NSCLC. Based on our results showing high expression level of miR-25 in NSCLC, we downregulated endogenous miR-25 in A549 cell. We found that downregulation of miR-25 could significantly inhibit A549 cell proliferation, induce G1 cell cycle arrest, and increase cisplatin chemosensitivity in vitro. We also found that miR-25 downregulated A549 cells had significantly reduced growth rate in tumor xenograft in vivo. These data are in line with the functional role of miR-25 in other forms of the cancer, such as initiating tumorigenesis in breast cancer through TGF-β signaling pathway [16] or facilitating tumor growth in hepatocellular carcinoma through the activation of BH3-only protein Bim by miR-106b-25 cluster [17]. Thus, our results, along with others, confirm that miR-25 is indeed acting as a tumor oncogenic factor in NSCLC.
CDC42 was shown to be overexpressed in NSCLC [23] and has been demonstrated to regulate cancer cell proliferation, invasion, and G1/S-phase transition of cell cycle [22, 25]. In the present study, luciferase reporter assay showed miR-25 bound on CDC42, qRT-PCR and western blots showed CDC42 was downregulated while miR-25 was inhibited in A549 cells. More importantly, re-expressing CDC42 in A549 cells reversed the inhibition on cancer cell proliferation and G1 cell cycle arrest induced by miR-25 downregulation. These functional and signaling pathway findings strongly suggest that miR-25 regulated NSCLC behaviors by downregulating CDC42.
In conclusion, downregulating miR-25 could inhibit NSCLC proliferation, induce G1 cell cycle arrest, increase the sensitivity to cisplatin, and reduce the growth of tumor xenograft in vivo, possibly through downregulation on CDC42. Thus, inhibiting miR-25 and its downstreaming signaling pathways might be a potential therapeutic strategy for treating patients with NSCLC in the future.
References
Siegel R, Naishadham D, Jemal A. Cancer statistics, 2012. CA: A Cancer J Clin. 2012;62:10–29.
Rivera MP. Multimodality therapy in the treatment of lung cancer. Semin Respir Crit Care Med. 2004;25 Suppl 1:3–10.
Lewis BP, Burge CB, Bartel DP. Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell. 2005;120:15–20.
Pillai RS. MicroRNA function: multiple mechanisms for a tiny RNA? RNA. 2005;11:1753–61.
Kumar MS, Lu J, Mercer KL, Golub TR, Jacks T. Impaired microRNA processing enhances cellular transformation and tumorigenesis. Nat Genet. 2007;39:673–7.
He L, Thomson JM, Hemann MT, Hernando-Monge E, Mu D, Goodson S, et al. A microRNA polycistron as a potential human oncogene. Nature. 2005;435:828–33.
Hwang HW, Mendell JT. MicroRNAs in cell proliferation, cell death, and tumorigenesis. Br J Cancer. 2006;94:776–80.
Garzon R, Calin GA, Croce CM. MicroRNAs in cancer. Annu Rev Med. 2009;60:167–79.
Lynam-Lennon N, Maher SG, Reynolds JV. The roles of microRNA in cancer and apoptosis. Biol Rev Camb Philos Soc. 2009;84:55–71.
Wang Y, Lee CG. MicroRNA and cancer—focus on apoptosis. J Cell Mol Med. 2009;13:12–23.
Ma J, Dong C, Ji C. MicroRNA and drug resistance. Cancer Gene Ther. 2010;17:523–31.
Nordentoft I, Birkenkamp-Demtroder K, Agerbaek M, Theodorescu D, Ostenfeld MS, Hartmann A, et al. MiRNAs associated with chemo-sensitivity in cell lines and in advanced bladder cancer. BMC Med Genet. 2012;5:40.
Singh S, Chitkara D, Kumar V, Behrman SW, Mahato RI. MiRNA profiling in pancreatic cancer and restoration of chemosensitivity. Cancer Lett. 2013;334:211–20.
Kim CH, Kim HK, Rettig RL, Kim J, Lee ET, Aprelikova O, et al. MiRNA signature associated with outcome of gastric cancer patients following chemotherapy. BMC Med Genet. 2011;4:79.
Zhang H, Zuo Z, Lu X, Wang L, Wang H, Zhu Z. MiR-25 regulates apoptosis by targeting Bim in human ovarian cancer. Oncol Rep. 2012;27:594–8.
Smith AL, Iwanaga R, Drasin DJ, Micalizzi DS, Vartuli RL, Tan AC, et al. The miR-106b-25 cluster targets Smad7, activates TGF-beta signaling, and induces EMT and tumor initiating cell characteristics downstream of Six1 in human breast cancer. Oncogene. 2012;31:5162–71.
Li Y, Tan W, Neo TW, Aung MO, Wasser S, Lim SG, et al. Role of the miR-106b-25 microRNA cluster in hepatocellular carcinoma. Cancer Sci. 2009;100:1234–42.
Tan W, Li Y, Lim SG, Tan TM. MiR-106b-25/miR-17-92 clusters: polycistrons with oncogenic roles in hepatocellular carcinoma. World J Gastroenterol: WJG. 2014;20:5962–72.
Chen X, Ba Y, Ma L, Cai X, Yin Y, Wang K, et al. Characterization of microRNAs in serum: a novel class of biomarkers for diagnosis of cancer and other diseases. Cell Res. 2008;18:997–1006.
Landi MT, Zhao Y, Rotunno M, Koshiol J, Liu H, Bergen AW, et al. MicroRNA expression differentiates histology and predicts survival of lung cancer. Clin Cancer Res: off J Am Assoc Cancer Res. 2010;16:430–41.
Arias-Romero LE, Chernoff J. Targeting Cdc42 in cancer. Expert Opin Ther Targets. 2013;17:1263–73.
Stengel K, Zheng Y. Cdc42 in oncogenic transformation, invasion, and tumorigenesis. Cell Signal. 2011;23:1415–23.
Zhang JY, Zhang D, Wang EH. Overexpression of small GTPases directly correlates with expression of delta-catenin and their coexpression predicts a poor clinical outcome in nonsmall cell lung cancer. Mol Carcinog. 2013;52:338–47.
Hua KT, Tan CT, Johansson G, Lee JM, Yang PW, Lu HY, et al. N-alpha-acetyltransferase 10 protein suppresses cancer cell metastasis by binding PIX proteins and inhibiting Cdc42/Rac1 activity. Cancer Cell. 2011;19:218–31.
Olson MF, Ashworth A, Hall A. An essential role for Rho, Rac, and Cdc42 GTPases in cell cycle progression through G1. Science. 1995;269:1270–2.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, T., Chen, T., Li, Y. et al. Downregulation of miR-25 modulates non-small cell lung cancer cells by targeting CDC42. Tumor Biol. 36, 1903–1911 (2015). https://doi.org/10.1007/s13277-014-2793-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-014-2793-0