Abstract
Cytochrome P450 2E1 (CYP2E1) is a natural enzyme involved in the metabolic activation of many carcinogens, and the functional polymorphisms in the CYP2E1 gene might have impacts on colorectal cancer risk. Many studies were published to assess the associations of CYP2E1 rs2031920 and rs3813867 polymorphisms with colorectal cancer risk, but no consistent findings were reported. A systemic review and meta-analysis of eligible studies was performed to comprehensively assess the associations above. Odds ratios (ORs) with 95 % confidence interval (95 % CIs) were used to assess the strength of the associations. Seventeen studies from 15 publications with 17,082 individuals were finally included into this meta-analysis. Meta-analysis of the 13 studies on CYP2E1 rs2031920 polymorphism showed that there was a significant association between CYP2E1 rs2031920 polymorphism and colorectal cancer risk under two genetic models (c2 versus c1: OR = 1.19, 95 % CI 1.03–1.37, P = 0.022; c2c2/c2c1 versus c1c1: OR = 1.16, 95 % CI 1.00–1.35, P = 0.046). Meta-analysis of those four case–control studies on CYP2E1 rs3813867 polymorphism showed that there was no significant association between CYP2E1 rs3813867 polymorphism and colorectal cancer risk under all contrast models (c2 versus c1: OR = 0.96, 95 % CI 0.80–1.16, P = 0.672; c2c2 versus c1c1: OR = 1.26, 95 % CI 0.43–3.67, P = 0.672; c2c2/c1c2 versus c1c1: OR = 0.95, 95 % CI 0.78–1.16, P = 0.114; and c2c2 versus c1c2/c1c1: OR = 1.17, 95 % CI 0.41–3.36, P = 0.775). Therefore, the findings from this meta-analysis suggest that CYP2E1 rs2031920 polymorphism is associated with colorectal cancer risk, but CYP2E1 rs3813867 polymorphism is not associated with colorectal cancer risk. In addition, more well-designed studies with large sample size are needed to provide a more precise evaluation on the associations above.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Colorectal cancer is the second most commonly diagnosed cancer with over 1.2 million new cases and 608,700 deaths in 2008 [1, 2]. The highest incidence rate of colorectal cancer is found in Australia, Europe, and North America [2]. In addition, the incidence rate of colorectal cancer is rapidly increasing in a number of countries within Eastern Asia, such as China [2]. Though the definite mechanism of colorectal cancer is still unknown, it has been well accepted that smoking, obesity, red meat consumption, and excessive alcohol consumption are risk factors for colorectal cancer [3, 4]. However, only a small part of individuals exposing to these risk factors develop colorectal cancer, suggesting that genetic susceptibility also plays an important role in the development of colorectal cancer [3–5]. N-nitrosamines present in tobacco and red meat are well-recognized carcinogens involved in the development of colorectal cancer [6, 7]. Cytochrome P4502E1 (CYP2E1) is a member of the cytochrome P450 superfamily and is involved in the metabolic activation of many carcinogens such as N-nitrosamines and aniline [8–10]. There are several polymorphisms found in the CYP2E1 gene which may have some impacts on susceptibility to colorectal cancer [11, 12]. The CYP2E1 rs2031920 (RsaI) and rs3813867 (PstI) polymorphisms in the 5′-flanking promoter region of the CYP2E1 gene are reported to affect the transcriptional activity of CYP2E1 gene [11, 12]. For CYP2E1 rs2031920 and rs3813867 polymorphisms, the three different genotypes are named the homozygous wild-type genotype (c1c1), heterozygous genotype (c1c2), and homozygous rare genotype (c2c2), respectively. Currently, many studies were published to assess the associations of CYP2E1 rs2031920 and rs3813867 polymorphisms with colorectal cancer risk, but no consistent findings were reported [13–24]. A systemic review and meta-analysis of eligible studies was performed to comprehensively assess the associations between CYP2E1 rs2031920 and rs3813867 polymorphisms and colorectal cancer risk.
Materials and methods
Publication search and eligible criteria
PubMed, Embase, Web of Science, and China National Knowledge Infrastructure (CNKI) were searched for case–control studies on the associations between CYP2E1 rs2031920 and rs3813867 polymorphisms and colorectal cancer risk. The following keywords were used in the literature search: (“colorectal cancer”’ or “colon cancer” or “rectal cancer”) and (“CYP2E1” or “CYP4502E1” or “rs2031920” or “rs3813867”). All studies included into this meta-analysis must meet the following eligibility criteria: (1) case–control study; (2) assessing the associations between CYP2E1 rs2031920 and rs3813867 polymorphisms and colorectal cancer risk; and (3) providing sufficient data on the genotype frequencies of CYP2E1 rs2031920 and rs3813867 polymorphisms to estimate the odds ratio (OR) with 95 % confidence interval (95 % CI). When two or more publications were performed on the same population, only the study with the largest or the most recent data was included into this meta-analysis.
Data extraction
The following information was extracted from those included studies: the first author’s name, publication year, country of the studied population, ethnicity of the studied population, genotyping methods, and genotype frequencies of CYP2E1 rs2031920 and rs3813867 polymorphisms in both cases and controls. The ethnicities of the studied population were categorized as Caucasians, Asians, and others.
Statistical analysis
The pooled OR with 95 % CI was calculated to assess the associations of CYP2E1 rs2031920 and rs3813867 polymorphisms with colorectal cancer risk. For both rs2031920 and rs3813867 polymorphisms, the ORs with their 95 % CIs were calculated for the allele model (c2 versus c1), the codominant model (c2c2 versus c1c1), the dominant model (c2c2/c1c2 versus c1c1), and the recessive model (c2c2 versus c1c2/c1c1), respectively. The statistical heterogeneity among studies was assessed with the I 2 test, and a I 2 value ≥50 % was considered to represent significant heterogeneity [25]. When there was obvious between-study heterogeneity, the random effect model (DerSimonian–Laird method) was used to calculate the pooled OR [26]. When there was no obvious between-study heterogeneity, the fixed effect model (Mantel–Haenszel method) was used to calculate the pooled OR [27]. Subgroup analysis based on ethnicity was also performed. Publication bias was tested by a funnel plot. All statistical analyses were undertaken using Stata version 12.0 (StataCorp, College Station, TX, USA). All the P values in this meta-analysis were two-sided, and a P value less than 0.05 was defined statistically significant.
Results
Study characteristics
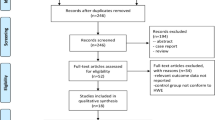
There were a total of 20 studies preliminarily identified by searching the PubMed, Embase, Web of Science, and CNKI databases [13–24, 28–35]. According to the eligible criteria, five studies were further excluded including three studies for other CYP2E1 polymorphism, one for case-only studies and the other one for the lack of useful data [31–35]. At last, 17 studies from 15 publications with 17,082 individuals were finally included into this meta-analysis [13–24, 28–30]. There were 14 studies with a total of 5,291 cases and 6,477 controls on the association between CYP2E1 rs2031920 polymorphism and colorectal cancer risk. Besides, there were four studies with 2,312 cases and 3,002 controls on the association between CYP2E1 rs3813867 polymorphism and colorectal cancer risk. Among those 15 studies, the polymerase chain reaction restriction fragment length polymorphism was the most common method used to analyze the genotype frequencies of those two polymorphisms.
Meta-analysis outcomes
Meta-analysis of the 13 studies on CYP2E1 rs2031920 polymorphism showed that there was a significant association between CYP2E1 rs2031920 polymorphism and colorectal cancer risk under two genetic models (c2 versus c1: OR = 1.19, 95 % CI 1.03–1.37, P = 0.022; c2c2/c2c1 versus c1c1: OR = 1.16, 95 % CI 1.00–1.35, P = 0.046) (Table 1, Figs. 1 and 2). However, subgroup analysis by ethnicity showed that there was no obvious association between CYP2E1 rs2031920 polymorphism and colorectal cancer risk in both Caucasians and Asians (Table 1).
Meta-analysis of those four case–control studies on CYP2E1 rs3813867 polymorphism showed that there was no significant association between CYP2E1 rs3813867 polymorphism and colorectal cancer risk under all contrast models (c2 versus c1: OR = 0.96, 95 % CI 0.80–1.16, P = 0.672; c2c2 versus c1c1: OR = 1.26, 95 % CI 0.43–3.67, P = 0.672; c2c2/c1c2 versus c1c1: OR = 0.95, 95 % CI 0.78–1.16, P = 0.114; and c2c2 versus c1c2/c1c1: OR = 1.17, 95 % CI 0.41–3.36, P = 0.775) (Table 2). In addition, subgroup analysis by ethnicity suggested there was no obvious association between the CYP2E1 rs3813867 polymorphism and colorectal cancer risk in both Caucasians and Asians (Table 2).
A funnel plot was performed to estimate the publication bias of literatures in this meta-analysis. The shapes of funnel plots for all contrast models in the meta-analysis of the association between the CYP2E1 rs2031920 polymorphism and colorectal cancer risk revealed obvious evidence of asymmetry (Fig. 3). However, the shapes of funnel plots for all contrast models in the meta-analysis of the association between the CYP2E1 rs3813867 polymorphism and colorectal cancer risk also did not reveal evidence of asymmetry. Therefore, there was risk of publication bias in the meta-analysis of the association between the CYP2E1 rs2031920 polymorphism and colorectal cancer risk.
Discussion
In the present meta-analysis, we provided the most comprehensive assessment of the associations of CYP2E1 rs2031920 and rs3813867 polymorphisms with colorectal cancer risk. Seventeen studies from 15 publications with a total of 17,082 individuals were finally included into this meta-analysis [13–24, 28–30]. Meta-analysis of the 13 studies on CYP2E1 rs2031920 polymorphism showed that there was a significant association between CYP2E1 rs2031920 polymorphism and colorectal cancer risk under two genetic models (Table 1). Meta-analysis of those four case–control studies on CYP2E1 rs3813867 polymorphism showed that there was no significant association between CYP2E1 rs3813867 polymorphism and colorectal cancer risk under all contrast models (Table 2). Therefore, the findings from this meta-analysis suggest that CYP2E1 rs2031920 polymorphism is associated with colorectal cancer risk, but CYP2E1 rs3813867 polymorphism is not associated with colorectal cancer risk.
Currently, though smoking, obesity, red meat consumption, and excessive alcohol consumption have been identified as risk factors of colorectal cancer, the definite mechanism of colorectal cancer is still unclear [36, 37]. However, there is no doubt that genetic susceptibility also plays an important role in the development of colorectal cancer [38–40]. There are many genetic polymorphisms which have been identified as risk factors of colorectal cancer, and many of them are involved in the metabolism of carcinogens, such as the glutathione S-transferase superfamily [38, 41]. N-nitrosamines present in tobacco and red meat are well-recognized carcinogens involved in the development of colorectal cancer. As a member of the cytochrome P450 superfamily, CYP2E1 is involved in the metabolic activation of many carcinogens such as N-nitrosamines and aniline, and it has important roles in the host’s susceptibility to colorectal cancer [40].
There are a number of polymorphisms found in the CYP2E1 gene which may have some impacts on susceptibility to colorectal cancer. The CYP2E1 rs2031920 (RsaI) and rs3813867 (PstI) polymorphisms in the 5′-flanking promoter region of the CYP2E1 gene are reported to affect the transcriptional activity of CYP2E1 gene. These two polymorphisms have some impacts on the activity of CYP2E1 enzyme and may further result in some inevitable influences on the host’s ability to metabolizing the carcinogens. Therefore, there are some possibilities for the obvious associations between CYP2E1 rs2031920 and rs3813867 polymorphisms and colorectal cancer risk. Many studies were published to assess the associations of CYP2E1 rs2031920 and rs3813867 polymorphisms with colorectal cancer risk, but no consistent findings were reported. So we performed this systemic review and meta-analysis of eligible studies to comprehensively assess the associations between CYP2E1 rs2031920 and rs3813867 polymorphisms and colorectal cancer risk. At last, the findings from this meta-analysis suggest that CYP2E1 rs2031920 polymorphism is associated with colorectal cancer risk, but CYP2E1 rs3813867 polymorphism is not associated with colorectal cancer risk.
There are three limitations when interpreting the findings from the meta-analysis. Firstly, there were only four studies with 2,312 cases and 3,002 controls on the association between CYP2E1 rs3813867 polymorphism and colorectal cancer risk. The limited number of included studies on this association may increase the risk of bias in the meta-analysis. To get a more precise estimation on the association between CYP2E1 rs3813867 polymorphism and colorectal cancer risk, more studies with large number of participants are needed. Secondly, this present meta-analysis was based on unadjusted estimates. There is no doubt that the real estimation between CYP2E1 rs3813867 polymorphism and colorectal cancer risk could be biased by the confounding factors. To obtain a more precise estimation on the associations between CYP2E1 rs2031920 and rs3813867 polymorphisms and colorectal cancer risk, we need carry out more studies with the consideration of confounding factors, such as age, tobacco smoking, and other environment factors. Finally, the shapes of funnel plots for all contrast models in the meta-analysis of the association between the CYP2E1 rs2031920 polymorphism and colorectal cancer risk revealed obvious evidence of asymmetry (Fig. 3). As was shown in Fig. 3, the studies with large sample size tend to show negative findings on the association between the CYP2E1 rs2031920 polymorphism and colorectal cancer risk, while the studies with relative small sample size tend to show obvious findings on the association above. Therefore, to get a more precise estimation on the association between CYP2E1 rs2031920 polymorphism and colorectal cancer risk, more well-designed studies with large sample size are needed to provide a more precise evaluation on the association above.
In conclusion, the findings from this meta-analysis suggest that CYP2E1 rs2031920 polymorphism is associated with colorectal cancer risk, but CYP2E1 rs3813867 polymorphism is not associated with colorectal cancer risk. In addition, more well-designed studies with and large sample size are needed to provide a more precise evaluation on the associations above.
References
Akin O, Brennan SB, Dershaw DD, Ginsberg MS, Gollub MJ, Schoder H, et al. Advances in oncologic imaging: update on 5 common cancers. CA Cancer J Clin. 2012;62:364–93.
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61:69–90.
Theodoratou E, Montazeri Z, Hawken S, Allum GC, Gong J, Tait V, et al. Systematic meta-analyses and field synopsis of genetic association studies in colorectal cancer. J Natl Cancer Inst. 2012;104:1433–57.
Burn J, Mathers J, Bishop DT. Genetics, inheritance and strategies for prevention in populations at high risk of colorectal cancer (crc). Recent Results Cancer Res. 2013;191:157–83.
Liu F, Yuan D, Wei Y, Wang W, Yan L, Wen T, et al. Systematic review and meta-analysis of the relationship between ephx1 polymorphisms and colorectal cancer risk. PLoS One. 2012;7:e43821.
Magalhaes B, Peleteiro B, Lunet N. Dietary patterns and colorectal cancer: systematic review and meta-analysis. Eur J Cancer Prev. 2012;21:15–23.
Vargas AJ, Thompson PA. Diet and nutrient factors in colorectal cancer risk. Nutr Clin Pract. 2012;27:613–23.
Knockaert L, Fromenty B, Robin MA. Mechanisms of mitochondrial targeting of cytochrome p450 2e1: physiopathological role in liver injury and obesity. FEBS J. 2011;278:4252–60.
Cederbaum AI. Cyp2e1 potentiates toxicity in obesity and after chronic ethanol treatment. Drug Metabol Drug Interact. 2012;27:125–44.
Zadoyan G, Fuhr U. Phenotyping studies to assess the effects of phytopharmaceuticals on in vivo activity of main human cytochrome p450 enzymes. Planta Med. 2012;78:1428–57.
Neafsey P, Ginsberg G, Hattis D, Johns DO, Guyton KZ, Sonawane B. Genetic polymorphism in cyp2e1: population distribution of cyp2e1 activity. J Toxicol Environ Health B Crit Rev. 2009;12:362–88.
Pohl HR, Scinicariello F. The impact of cyp2e1 genetic variability on risk assessment of voc mixtures. Regul Toxicol Pharmacol. 2011;59:364–74.
Kiss I, Sandor J, Pajkos G, Bogner B, Hegedus G, Ember I. Colorectal cancer risk in relation to genetic polymorphism of cytochrome p450 1a1, 2e1, and glutathione-s-transferase m1 enzymes. Anticancer Res. 2000;20:519–22.
Le Marchand L, Donlon T, Seifried A, Wilkens LR. Red meat intake, cyp2e1 genetic polymorphisms, and colorectal cancer risk. Cancer Epidemiol Biomarkers Prev. 2002;11:1019–24.
Landi S, Gemignani F, Moreno V, Gioia-Patricola L, Chabrier A, Guino E, et al. A comprehensive analysis of phase I and phase II metabolism gene polymorphisms and risk of colorectal cancer. Pharmacogenet Genomics. 2005;15:535–46.
van der Logt EM, Bergevoet SM, Roelofs HM, Te Morsche RH, Dijk Y, Wobbes T, et al. Role of epoxide hydrolase, nad(p)h:Quinone oxidoreductase, cytochrome p450 2e1 or alcohol dehydrogenase genotypes in susceptibility to colorectal cancer. Mutat Res. 2006;593:39–49.
Gao CM, Takezaki T, Wu JZ, Chen MB, Liu YT, Ding JH, et al. Cyp2e1 Rsa I polymorphism impacts on risk of colorectal cancer association with smoking and alcohol drinking. World J Gastroenterol. 2007;13:5725–30.
Kiss I, Orsos Z, Gombos K, Bogner B, Csejtei A, Tibold A, et al. Association between allelic polymorphisms of metabolizing enzymes (cyp 1a1, cyp 1a2, cyp 2e1, meh) and occurrence of colorectal cancer in Hungary. Anticancer Res. 2007;27:2931–7.
Kury S, Buecher B, Robiou-du-Pont S, Scoul C, Sebille V, Colman H, et al. Combinations of cytochrome p450 gene polymorphisms enhancing the risk for sporadic colorectal cancer related to red meat consumption. Cancer Epidemiol Biomarkers Prev. 2007;16:1460–7.
Cotterchio M, Boucher BA, Manno M, Gallinger S, Okey AB, Harper PA. Red meat intake, doneness, polymorphisms in genes that encode carcinogen-metabolizing enzymes, and colorectal cancer risk. Cancer Epidemiol Biomarkers Prev. 2008;17:3098–107.
Morita M, Le Marchand L, Kono S, Yin G, Toyomura K, Nagano J, et al. Genetic polymorphisms of cyp2e1 and risk of colorectal cancer: the Fukuoka Colorectal Cancer Study. Cancer Epidemiol Biomarkers Prev. 2009;18:235–41.
Sameer AS, Nissar S, Qadri Q, Alam S, Baba SM, Siddiqi MA. Role of cyp2e1 genotypes in susceptibility to colorectal cancer in the Kashmiri population. Hum Genomics. 2011;5:530–7.
Silva TD, Felipe AV, Pimenta CA, Barao K, Forones NM. Cyp2e1 RsaI and 96-bp insertion genetic polymorphisms associated with risk for colorectal cancer. Genet Mol Res. 2012;11:3138–45.
Wang J, Joshi AD, Corral R, Siegmund KD, Marchand LL, Martinez ME, et al. Carcinogen metabolism genes, red meat and poultry intake, and colorectal cancer risk. Int J Cancer. 2012;130:1898–907.
Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327:557–60.
DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7:177–88.
Mantel N, Haenszel W. Statistical aspects of the analysis of data from retrospective studies of disease. J Natl Cancer Inst. 1959;22:719–48.
Yu WP, Chen K, Ma XY, Yao KY, Jiang QT, Zou Y, et al. Genetic polymorphism in cytochrome p450 2e1, salted food and colorectal cancer susceptibility: a case-control study. Zhonghua Yu Fang Yi Xue Za Zhi. 2004;38:162–6.
Chen K, Jin MJ, Fan CH, Song L, Jiang QT, Yu WP, et al. A case-control study on the association between genetic polymorphisms of metabolic enzymes and the risk of colorectal cancer. Zhonghua Liu Xing Bing Xue Za Zhi. 2005;26:659–64.
Yang H, Cao J. Genetic association study between polymorphisms of major alcohol metabolizing and jwa genes and colorectal cancer. Wanfang Doctoral Dissertation 2009; Third Military Medical University
Butler WJ, Ryan P, Roberts-Thomson IC. Metabolic genotypes and risk for colorectal cancer. J Gastroenterol Hepatol. 2001;16:631–5.
Fan C, Jin M, Chen K, Zhang Y, Zhang S, Liu B. Case-only study of interactions between metabolic enzymes and smoking in colorectal cancer. BMC Cancer. 2007;7:115.
Cleary SP, Cotterchio M, Shi E, Gallinger S, Harper P. Cigarette smoking, genetic variants in carcinogen-metabolizing enzymes, and colorectal cancer risk. Am J Epidemiol. 2010;172:1000–14.
Darazy M, Balbaa M, Mugharbil A, Saeed H, Sidani H, Abdel-Razzak Z. Cyp1a1, cyp2e1, and gstm1 gene polymorphisms and susceptibility to colorectal and gastric cancer among Lebanese. Genet Test Mol Biomarkers. 2011;15:423–9.
Ebrahimkhani S, Asgharian AM, Nourinaier B, Ebrahimkhani K, Vali N, Abbasi F, et al. Association of GSTM1, GSTT1, GSTP1 and CYP2E1 single nucleotide polymorphisms with colorectal cancer in Iran. Pathol Oncol Res. 2012;18:651–6.
Morikawa T, Kuchiba A, Lochhead P, Nishihara R, Yamauchi M, Imamura Y, et al. Prospective analysis of body mass index, physical activity, and colorectal cancer risk associated with beta-catenin (ctnnb1) status. Cancer Res. 2013;73:1600–10.
Yarnall JM, Crouch DJ, Lewis CM. Incorporating non-genetic risk factors and behavioural modifications into risk prediction models for colorectal cancer. Cancer Epidemiol. 2013. doi:10.1016/j.canep.2012.12.008.
Zhao ZQ, Guan QK, Yang FY, Zhao P, Zhou B, Chen ZJ. System review and metaanalysis of the relationships between five metabolic gene polymorphisms and colorectal adenoma risk. Tumour Biol. 2012;33:523–35.
Andersen V, Holst R, Vogel U. Systematic review: diet-gene interactions and the risk of colorectal cancer. Aliment Pharmacol Ther. 2013;37:383–91.
Jiang O, Zhou R, Wu D, Liu Y, Wu W, Cheng N. Cyp2e1 polymorphisms and colorectal cancer risk: a huge systematic review and meta-analysis. Tumour Biol. 2013;34:1215–24.
Zeng FR, Ling Y, Yang J, Tian XC, Yang X, Luo RC. X-ray repair cross-complementing group 1 arg399gln gene polymorphism and susceptibility to colorectal cancer: a meta-analysis. Tumour Biol. 2013;34:555–63.
Acknowledgments
This work was financially supported by research grants from the Guangdong Natural Science Foundation of China (No. S2012010009082).
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Additional information
Hui Peng and Shang-Kui Xie contributed equally to this work.
Rights and permissions
About this article
Cite this article
Peng, H., Xie, SK., Huang, MJ. et al. Associations of CYP2E1 rs2031920 and rs3813867 polymorphisms with colorectal cancer risk: a systemic review and meta-analysis. Tumor Biol. 34, 2389–2395 (2013). https://doi.org/10.1007/s13277-013-0788-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-013-0788-x