Abstract
The purpose of this research was to study the roles of chloride intracellular channel protein 1 (CLIC1) and heat shock protein 27 (HSP27) in the clinical pathology of lung adenocarcinoma and to explore whether the expression of CLIC1 and HSP27 can be used as independent factors for the prediction of recurrence and prognosis after radical resection of lung adenocarcinoma. One hundred and three paraffin sections of lung adenocarcinoma tissues were collected, and the expression of CLIC1 and HSP27 was detected in these tumors using immunohistochemistry. The correlation of the expression of these two proteins with clinicopathological parameters and prognosis was statistically analyzed. In the 103 samples, the expression of HSP27 and CLIC1 was strongly positive in 61 (59.2%) and 49 cases (47.6%), respectively. Statistical analysis showed that the expression level of HSP27 did not significantly correlate with the patient’s age, sex, degree of tumor differentiation, T staging of tumors, and TNM staging of tumors (p > 0.05), whereas the expression of CLIC1 did significantly correlate with T staging of tumors (p = 0.029). Univariate analysis indicated that the patient’s ECOG score, T staging, N staging, TNM staging, and CLIC1 expression correlated with prognosis (p = 0.031, 0.001, 0.011, 0.013, and <0.001, respectively). Multivariate statistical analysis showed that age, T staging, and CLIC1 expression were independent associated factors for predicting the 5-year survival rate of patients (p = 0.026, 0.004, and <0.001, respectively). Age, T staging, and CLIC1 expression significantly correlated with the overall survival of post-operative lung adenocarcinoma patients. CLIC1 may be closely associated with the occurrence and development of lung adenocarcinoma and may be used as an effective marker for predicting the prognosis of this disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Lung cancer is a severe disease that affects human health and life. Recently, the morbidity and mortality of this disease has shown an increasing trend worldwide. The morbidity and mortality rates associated with lung cancer have jumped, and it is now the leading cause of cancer-related morbidity and mortality worldwide, including both developed and developing countries [1]. Lung adenocarcinoma is one of the common types of non-small cell lung carcinoma (NSCLC) and is the pathological type with the highest incidence among non-smoking patients. The incidence rate of lung adenocarcinoma has recently undergone a significant increase. Because lung adenocarcinoma is associated with early blood metastasis and recurrence, this disease has a relatively poor prognosis [2]. Most NSCLC patients are already beyond the early stage of tumor development at the time of diagnosis; the surgical resection rates for these patients are between 10% and 28% [3]. Furthermore, even if lung cancer patients at the early stage receive standardized treatment, approximately 40% of early stage patients will still develop recurrence and metastasis in a short time [4].
The recurrence and metastasis of lung cancer may be associated with micro-metastasis of tumor cells; these micro-metastases may occur before the surgery [5]. Therefore, if related clinical markers can accurately predict tumor recurrence, metastasis, and prognosis, clinicians can better choose appropriate treatment regimens. Recently, some scholars have discovered that mRNAs and proteins of some genes can be used to predict the prognosis of lung adenocarcinoma [6–9]. However, these detection markers cannot be used to accurately predict lung cancer recurrence and metastasis. Therefore, many scholars are currently focusing on discovering related detection markers to accurately predict lung cancer recurrence and metastasis.
Heat shock protein 27 (HSP27) is one of the important members of the small-molecular weight heat shock protein family and is a highly conserved protein that is expressed widely in the biosphere. Stress stimulation will increase the expression of HSP27 to exert its anti-oxidative damage activities [10]. The function of HSP27 is associated with the protection of cells from damage induced by a variety of stress factors, cell proliferation and differentiation, and the regulation of apoptotic signal transduction [11]. Previous reports have shown that HSP27 is expressed in many tumors and is associated with tumor malignancy and clinical prognosis [12–17]. In lung cancers, HSP27 has been shown to be associated with lung cancer occurrence, development, and prognosis [18]; however, the association between HSP27 expression and the prognosis of lung adenocarcinoma patients is rarely reported.
Chloride intracellular channel 1 (CLIC1) is a newly discovered member of a chloride channel protein family. This protein family has many members, including CLIC1, CLIC2, CLIC3, CLIC4, CLIC5, CLIC6, p64, and Parchorin [19]. CLIC1 is expressed ubiquitously in human tissues and plays important roles in many physiological processes, such as the regulation of cell volume, acidification of organelles, transepithelial transport, regulation of electrical excitability [20, 21]. CLIC1 has been shown to interacts with Sedlin. Sedlin which as a components of TRAPP complex, has been reported to be involved in a defect in cartilage transport from endoplasmic reticulum to the Golgi complex [22].CLIC1 is also involved in cell cycle regulation [23] and in the regulation of cell proliferation and differentiation [24, 25]. Recently, several studies have shown that CLIC1 is highly expressed in malignant tumors, such as gastric [26], colon [27], and liver [28, 29] cancers. This protein may have important functions in the malignant biology of tumor cells, such as roles in proliferation, invasiveness, and metastasis. However, related studies in lung cancer, especially in lung adenocarcinoma, have not been reported.
Accordingly, this study examined the expression of HSP27 and CLIC1 in surgically resected lung adenocarcinoma specimens to analyze whether the expression of these proteins correlates with the survival time and tumor recurrence of lung adenocarcinoma patients after radical resection.
Materials and methods
Patient
One hundred and three paraffin specimens from pathologically confirmed primary lung adenocarcinoma patients after thoracic surgery were collected in the First Affiliated Hospital of Guangzhou Medical College from 2003 to 2004. None of the patients had distal metastases and none received chemotherapy or radiotherapy before surgery. All of the cases received standard video-assisted thoracic surgery and hilar and mediastinal lymph node dissection. The patients included 66 males and 37 females with an average age of 60.46 ± 11.07. According to the 7th edition of UICC, TNM staging of the tumors, pathological staging, and clinical classification were performed on all patient tumors [30]. Among these tumors, 43 cases were stage I, 14 cases were stage II, and 46 cases were stage III. Pathological confirmation and classification were performed by two experienced pathologists according to the “Histological Typing for Lung Tumors” by WHO [31]. The tumors were classified as 72 cases of moderate differentiation and 31 cases of poor differentiation. In these 103 patients, stage II/III patients received three to four cycles adjuvant chemotherapy. The regimen is third platinum-based chemotherapy including gemcitabine, Navelbine, and taxol. This study was approved by the Ethics Committee of the First Affiliated Hospital of Guangzhou Medical College.
Immunohistochemistry
All of the paraffin sections of tumors in this study were first stained with hematoxylin and eosin, and the pathological types were confirmed by two experienced pathologists. Immunohistochemistry was performed using the streptavidin-peroxidase method. After deparaffinization using an automatic dewaxing machine (Leica TP1020, Leica), the sections were boiled for 5 min in citrate buffer (pH 6.0) using a microwave oven. After the sections cooled naturally, 50 μl of 3% H2O2 was added onto each tissue for 15 min at room temperature to inactivate endogenous peroxidase. The tissues were washed three times (3 min each) with phosphate-buffered saline (PBS) and then blocked with 50-μl goat serum working solution for 15 min at room temperature. After the serum was discarded, the sections were incubated with 50 μl HSP27 (1:200 dilution, Abcam) or CLIC1 (1:175 dilution, Abcam) monoclonal mouse anti-human antibodies in a humid chamber at 4°C for overnight. After 14 h, the sections were removed from the 4°C refrigerator and placed at room temperature for 30 min before being washed three times (5 min each) with PBS. The sections were then incubated with biotinylated secondary antibodies (1:100, DAKO) for 15 min at room temperature. After being washed three times (3 min each) with PBS, the sections were incubated with horseradish peroxidase-labeled streptavidin working solution at room temperature for 15 min. After being washed three times (3 min each) with PBS, the sections were developed using diaminobenzidine (DAKO) until the tissues became yellow under a microscope. The sections were quickly washed with distilled water and counter-stained with hematoxylin. The sections were then dehydrated and mounted.
Evaluation of immunostaining results
The intensity of immunostaining was judged separately by two pathologists without knowledge of the clinical condition of the patients. For HSP27, staining in the nucleus of tumors cells was considered to be positive, whereas staining in the cytoplasm of tumor cells was regarded as positive for CLIC1. Five representative fields at ×400 magnification under a microscope were observed; each field contained at least 200 tumor cells. The semi-quantitative scoring of the expression intensity in each sample was performed according to a previous report and was based on the staining intensity and area [32]. The standard for the immuostaining intensity score was as follows: no staining (0 points), mild staining (1 point), moderate staining (2 points), and strong staining (3 points). The score for the staining area was based on the percentage of positive cells: <5% (0 points), 6–25% (1 point), 26–50% (2 points), 51–75% (3 points), and >75% (4 points). The final score for the immunohistochemistry intensity of each case was the multiplication of the two scores for the immunostaining intensity and immunostaining area; 0 represented negative expression (−), 1–4 points represented weak expression (+), 6–8 points was moderate expression (++), and 9–12 points was strong expression (+++). In this study, we grouped all of the samples into the high expression group (++ or +++) and the low expression group (− or +) according to the protein expression.
Statistical analysis
Measurement data are presented as the mean ± standard deviation (X ± SD). The χ 2 of the correlation between the immunostaining intensity and clinical data of the patients was calculated from a contingency table. The correlation between the immunohistochemistry results and the post-operative survival time of the patients was analyzed using the Kaplan–Meier method. Follow-up by telephone, letter, and clinical service were carried out to obtain the information of patients’ outcomes. The follow-up period lasted up to 60 months. Overall survival was calculated from the time of surgery to the time of death. The variables that demonstrated values of p ≤ 0.20 in the univariate analysis were tested using the Cox regression model. Statistical analysis was performed using the SPSS 13.0 software package; p < 0.05 was considered statistically significant.
Results
The correlation between HSP27 and CLIC1 expression and clinicopathological features
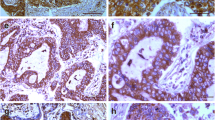
In lung adenocarcinoma cells, HSP27 and CLIC1 were expressed in the nucleus (Fig. 1(A–C)) and cytoplasm (Fig. 1(E, F)), respectively. In the 103 samples, 61 cases (59.2%) demonstrated high HSP27 expression and 49 cases (47.6%) demonstrated high CLIC1 expression. The correlation of HSP27 and CLIC1 expression with clinicopathological features is shown in Table 1. Statistical analysis results showed that HSP27 expression did not significantly correlate with patient’s age, sex, degree of tumor differentiation, T staging of tumors, or TNM staging of tumors (p > 0.05). In the CLIC1 group, the expression of CLIC1 significantly correlated with the T staging of tumors (p = 0.029).
The correlation between HSP27 and CLIC1 expression and the prognosis of adenocarcinoma patients
Univariate analysis using a Log-rank test showed that patient’s ECOG score, T staging, N staging, TNM staging, and CLIC1 expression correlated with prognosis (p = 0.031, 0.001, 0.011, 0.013, and <0.001, respectively) (Table 2). The 5-year survival rates of the high HSP27 expression group and the low HSP27 expression group were 41.2% and 44.4%, respectively (p = 0.293), whereas the 5-year survival rates of the high CLIC1 expression and the low CLIC1 expression groups were 33.7% and 50.6%, respectively (p < 0.001). All of the variables in the univariate analysis that showed significant effects on survival were tested using the Cox regression model; the results showed that the patient’s age, T staging of tumor, and CLIC1 expression were independently associated factors for predicting the 5-year survival rate of patients (p = 0.026, 0.004, and <0.001, respectively) (Table 3).
Discussion
Patients with early lung cancer have better 5-year survival rates after surgical resection; these survival rates can reach 70% [1]. Therefore, early detection, early diagnosis, and early treatment have been recognized as the keys to treating lung cancer. Currently, the research focus of clinicians and researchers is to accurately predict the occurrence, recurrence, and metastasis of lung cancer.
Immunohistochemistry is a widely used technology in histopathology analysis. This technique primarily uses the principle of specific binding between an antigen and antibody, which can be used to localize an antigen and therefore perform semi-quantitative analysis of an antigen in tissues. Because of the strong specificity, high sensitivity, simple sample processing requirement, and rapid analysis, this technology is very suitable for clinical work. Therefore, immunohistochemistry is widely used for the discovery of markers to predict the prognosis of lung cancer patients after surgery [33]. In this study, we also used immunohistochemistry technology to explore whether HSP27 and CLIC1 are associated with the prognosis of lung adenocarcinoma patients after surgery.
HSP27 is an important member of the HSP family and has very broad biological functions. HSP27 is overexpressed in many tumors and has a certain correlation with prognosis. Studies have shown that overexpression of HSP27 is associated with poor prognosis of some malignant tumors, such as liver [14], prostate [15], and colon [16] cancers. In contrast, cervical cancer [12] and oral squamous cell carcinoma [13] patients have better prognosis when HSP27 is overexpressed. However, the correlation between HSP27 and the prognosis of patients is still controversial for some types of tumors. Kapranos et al. have found that gastric cancer patients with HSP27 overexpression have a poor prognosis [17], whereas in 2009, Giaginis et al. showed that HSP27 expression had no significant correlation with the prognosis of gastric cancer [34].
HSP27 is also highly expressed in lung cancer. Thus far, a report has shown that HSP27 expression correlates with lung cancer prognosis. Malusecka et al. [18] have performed immunohistochemistry and statistical analysis on 103 NSCLC specimens and found that HSP27 is significantly overexpressed in tumor tissues. When examined in relation to the survival condition of these patients, they also found that HSP27 expression significantly correlated with prognosis. Patients with HSP27 overexpression had better prognosis, whereas patients without HSP27 expression had poorer prognosis. In our study, we measured the expression of HSP27 in 103 lung adenocarcinoma samples after surgical resection. Our results differed from previous studies in NSCLC [18]. We found that the expression of HSP27 was significantly higher in lung adenocarcinoma tissues, which was consistent with the results in most of the literature [12–17]. However, we also found that HSP27 expression did not significantly correlate with the clinicopathological features of lung adenocarcinoma patients. The post-operative 5-year survival rate of these patients did not significantly correlate with the expression level of HSP27. Possible explanations for the difference between our results and those of previous reports include the following: (1) In different pathological types of tumors, HSP27 expression may be different from the clinical features of patients. The study by Malusecka has also shown that HSP27 expression is an important predictor of survival in lung squamous cell carcinoma patients, but its role in lung adenocarcinoma patients was not significant [18]; this result was consistent with our results. (2) The differences may be associated with the number of lung adenocarcinoma cases. The study by Malusecka focused on NSCLC, and only 31 cases of lung adenocarcinoma were included. Our study concentrated on 103 cases of lung adenocarcinoma; with the increase in sample size, the results may be different.
CLIC1 plays important roles in many physiological processes, such as the regulation of cell volume, acidification of organelles, transepithelial transport, regulation of electrical excitability [20, 21]. Studies of CLIC1 in tumors are gradually being reported. In the literature, CLIC1 has been shown to have different high levels of expression in nasopharyngeal carcinoma [20], gastric cancer [26], liver cancer [28, 29], breast cancer [35], and colon cancer [27, 36]. In a previous study, we used differential proteomics to screen differential proteins in lung adenocarcinoma and adjacent normal lung tissues; CLIC1 was one of the highly expressed proteins in the lung adenocarcinoma tissues. This result was consistent with the results reported in other studies in the literature.
Chen et al. [26] have performed differential proteomics studies in gastric cancer tissues and adjacent normal tissues and have shown that CLIC1 is highly expressed in gastric cancer. This result was confirmed by Q-PCR and western blot analysis [26]. The authors performed immunohistochemistry examination of 56 paraffin samples of gastric cancer and found that CLIC1 protein was up-regulated in 67.9% of gastric cancer samples. High levels of CLIC1 expression significantly correlated with lymph node metastasis, lymphatic vessels and surrounding tissues infiltration, pathological staging, and survival time of patients. CLIC1 expression did not have a significant correlation with the patient’s age, sex, tumor location, tumor size, pathological staging, invasion depth, distal metastasis, and vascular invasion. Chang et al. have performed immunohistochemistry on 40 paraffin specimens of nasopharyngeal carcinoma and ELISA on 74 blood samples from nasopharyngeal carcinoma patients and have shown that nasopharyngeal carcinoma tissues and blood have significantly high levels of CLIC1 expression, but the high levels of CLIC1 expression did not correlate with tumor size, lymph node metastasis, or TNM staging [20]. The authors believed that, although CLIC1 expression did not correlate with tumor size, lymph node metastasis, and TNM staging, the expression of CLIC1 in early nasopharyngeal carcinoma plasma was significantly higher than in plasma from normal individuals, suggesting that this protein can be used as a potential hematological predictor for early nasopharyngeal carcinoma.
In this study, we also used immunohistochemistry technology to detect CLIC1 expression in 103 paraffin sections of lung adenocarcinoma patients with complete clinical and follow-up data. We also analyzed the correlation between CLIC1 expression and clinicopathological features of these cases and the correlation between CLIC1 expression and prognosis. Our results showed that the expression rate of CLIC1 in the 103 tumor tissues was 47.6%. Further statistical analysis showed that CLIC1 expression did not significantly correlate with the patient’s sex, age, smoking condition, ECOG score, serum CEA content, tumor location, degree of tumor differentiation, lymph node metastasis, and TNM staging (p > 0.05). CLIC1 expression had a significant positive correlation with the T staging of the tumor (p < 0.05). Kaplan–Meier survival analysis results showed that lung adenocarcinoma patients in the CLIC1 high expression group had shorter post-operative survival time than did patients in the low expression group (Fig. 2); this difference was statistically significant (p < 0.05). This result was consistent with the results of Chen [26] in gastric cancer. In this study, after Cox regression model analysis, we found that patient’s age, T staging of tumors, and CLIC1 expression were independently associated factors for the prediction of the 5-year survival rate of lung adenocarcinoma patients (p = 0.026, 0.004, and <0.001, respectively).
Some data have indicated that lung cancer is most prevalent among the elderly population. Patients over 65 years of age account for more than 50% of patients, and patients over 70 years old account for 30% to 40% [37]. Elderly patients often suffer from additional diseases, such as hypertension and diabetes. In particular, elderly patients with smoking habits are more likely to have the complications of chronic obstructive pulmonary disease and cardiovascular disease. These accompanying diseases may directly or indirectly affect the treatment and prognosis of these patients. Janssen-Heijnen et al. have mentioned that age is an independent prognostic factor affecting survival for stage I and II NSCLC patients; however, age does not significantly affect the survival time of stage III and IV patients [38]. Li et al. have found that, in stage I lung cancer patients, the 5-year survival rate of patients younger than 65 years is significantly higher than that of patients older than 65 years of age [39]. T staging of tumors is an important clinical factor to determine the survival rate of patients. Li et al. have also found that with the increase of tumor size, the 5-year patient survival rate decreases and the tumor recurrence rate significantly increases [39]. Chabowski et al. have analyzed the post-operative prognosis of 431 lung cancer patients and found that T staging of the tumor significantly correlates with the 5-year survival rate; with the increase in T staging, the patient’s survival time shortened [40]. In our study, multivariate Cox regression analysis results showed that patient’s age and T staging were independent associated factors for predicting the 5-year survival rate of lung adenocarcinoma patients after surgery (p = 0.026 and 0.004, respectively), these data are consistent with previous reports.
This study was a retrospective analysis and included many limitations. The sample size of this study was relatively small, with only 103 cases, and we analyzed only the correlation between the expression of HSP27 and CLIC1 and the 5-year total survival rate of lung adenocarcinoma patients. We neither observed disease progression time of patients nor included the effects of follow-up treatment after surgery on the 5-year survival rate of the patients. To further understand the specific role of CLIC1 in the prognosis of lung adenocarcinoma, additional comprehensive prospective studies must be performed.
In summary, the results in this study showed that age, T staging, and CLIC1 expression significantly correlate with the total post-operative survival of lung adenocarcinoma patients. CLIC1 may be closely associated with the occurrence and development of lung adenocarcinoma, and CLIC1 expression may be used as an effective predictor for the prognosis of this disease.
References
Jemal A, Bray F, Center MM, et al. Global cancer statistics. CA Cancer J Clin. 2011;61:69–90.
Li HL, Qiu Y, Zhang X, et al. Establishment of a lung cancer biobank of a southern Chinese population. J Thorac Dis. 2009;1:17–22.
Bethune G, Bethune D, Ridgway N, et al. Epidermal growth factor receptor (EGFR) in lung cancer: an overview and update. J Thorac Dis. 2010;2:48–51.
Rena O, Carsana L, Cristina S, et al. Lymph node isolated tumor cells and micrometastases in pathological stage I non-small cell lung cancer: prognostic significance. Eur J Cardiothorac Surg. 2007;32:863–7.
Ruffato A, Mattioli S, Pileri S, et al. Do bone marrow isolated tumor cells influence long-term survival of non-small cell lung cancer? Eur J Cardiothorac Surg. 2009;35:463–8.
Shao WL, Wang DY, He JX. The role of gene expression profiling in early-stage non-small cell lung cancer. J Thorac Dis. 2010;2:89–99.
Nakamura N, Kobayashi K, Nakamoto M, et al. Identification of tumor markers and differentiation markers for molecular diagnosis of lung adenocarcinoma. Oncogene. 2006;25:4245–55.
Méndez M, Custodio A, Provencio M. New molecular targeted therapies for advanced non-small-cell lung cancer. J Thorac Dis. 2011;3:30–56.
Liu YF, Xiao ZQ, Li MX, et al. Quantitative proteome analysis reveals annexin A3 as a novel biomarker in lung adenocarcinoma. J Pathol. 2009;217:54–64.
Hollander JM, Martin JL, Belke DD. Overexpression of wild- type heat shock protein 27 and a nonphosphorylatable heat shock protein 27 mutant protects against ischemia/reperfusion injury in a transgenic mouse model. Circulation. 2004;110:3544–52.
Fincham VJ, James M, Frame MC, et al. Active ERK/MAPkinase15 targeted to newly forming cell-matrix adhesions by integrin engagement and v-Src. EMBOJ. 2000;19:2911–23.
Lomnytska MI, Becker S, Bodin I, et al. Differential expression of ANXA6, HSP27, PRDX2, NCF2, and TPM4 during uterine cervix carcinogenesis: diagnostic and prognostic value. Br J Canc. 2011;104:110–9.
Lo Muzio L, Leonardi R, Mariggio MA, et al. HSP 27 as possible prognostic factor in patients with oral squamous cell carcinoma. Histol Histopathol. 2004;9:119–28.
King KL, Li AF, Chau GY, et al. Prognostic significance of heat shock protein-27 expression in hepatocellular carcinoma and its relation to histologic grading and survival. Cancer. 2000;88:2464–70.
Glaessgen A, Jonmarker S, Lindberg A, et al. Heat shock proteins 27, 60 and 70 as prognostic markers of prostate cancer. APMIS. 2008;116:888–95.
Tweedle EM, Khattak I, Ang CW, et al. Low molecular weight heat shock protein HSP27 is a prognostic indicator in rectal cancer but not colon cancer. Gut. 2010;59:1501–10.
Romani AA, Crafa P, Desenzani S, et al. The expression of HSP27 is associated with poor clinical outcome in intrahepatic cholangiocarcinoma. BMC Cancer. 2007;7:232.
Malusecka E, Krzyzowska-Gruca S, Gawrychowski J, et al. Stress proteins HSP27 and HSP70i predict survival in non-small cell lung carcinoma. Anticancer Res. 2008;28:501–6.
Tulk BM, Kapadia S, Edwards JC. CLIC1 inserts from the aqueous phase into phospholipid membranes, where it functions as an anion channel. Am J Physiol Cell Physiol. 2002;282:1103–12.
Chang YH, Wu CC, Chang KP, et al. Cell secretome analysis using hollow fiber culture system leads to the discovery of CLIC1 protein as a novel plasma marker for nasopharyngeal carcinoma. J Proteome Res. 2009;8:5465–74.
Shi ZH, Zhao C, Wu H, et al. CLIC1 protein: a candidate prognostic biomarker for malignant-transformed hydatidiform moles. Int J Gynecol Cancer. 2011;21:153–60.
Liu XY, Wang YF, Zhu HR. Interaction of Sedlin with PAM14. J Cell Biochem. 2010;109:1129–33.
Valenzuela SM, Mazzanti M, Tonini R, et al. The nuclear chloride ion channel NCC27 is involved in regulation of the cell cycle. J Physiol. 2000;529:541–52.
Sun HJ, Bahk YY, Choi YR, et al. A proteomic analysis during serial subculture and osteogenic differentiation of human mesenchymal stem cell. J Orthop Res. 2006;24:2059–71.
Rahman A, Kumar SG, Kim SW, et al. Proteomic analysis for inhibitory effect of chitosan oligosaccharides on 3T3-L1 adipocyte differentiation. Proteomics. 2008;8:569–81.
Chen CD, Wang CS, Huang YH, et al. Overexpression of CLIC1 in human gastric carcinoma and its clinicopathological significance. Proteomics. 2007;7:155–67.
Petrova DT, Asif AR, Armstrong VW, et al. Expression of chloride intracellular channel protein 1 (CLIC1) and tumor protein D52 (TPD52) as potential biomarkers for colorectal cancer. Clin Biochem. 2008;41:1224–36.
Huang JS, Chao CC, Su TL, et al. Diverse cellular transformation capability of overexpressed genes in human hepatocellular carcinoma. Biochem Biophys Res Commun. 2004;315:4950–8.
Kim W, Oe Lim S, Kim JS, et al. Comparison of proteome between hepatitis B virus- and hepatitis C virus-associated hepatocellular carcinoma. Clin Cancer Res. 2003;9:5493–500.
Goldstraw P, Crowley J, Chansky K, et al. The IASLC Lung Cancer Staging Project: proposals for the revision of the TNM stage groupings in the forthcoming (seventh) edition of the TNM Classification of malignant tumours. J Thorac Oncol. 2007;2:706–14.
Travis WD. World Health Organization, International Agency for Research on Cancer, International Academy of Pathology and International Association for the Study of Lung Cancer. Pathology and genetics of tumours of the lung, pleura, thymus, and heart. Lyon: IARC Press;2004.
Li Z, Zhao X, Bai S, et al. Proteomics identification of cyclophilin a as a potential prognostic factor and therapeutic target in endometrial carcinoma. Mol Cell Proteomics. 2008;7:1810–23.
Zhu CQ, Shih W, Ling CH, et al. Immunohistochemical markers of prognosis in non-small cell lung cancer: a review and proposal for a multiphase approach to marker evaluation. J Clin Pathol. 2006;59:790–800.
Kapranos N, Kominea A, Konstantinopoulos PA, et al. Expression of the 27-kDa heat shock protein (HSP27) in gastric carcinomas and adjacent normal, metaplastic, and dysplastic gastric mucosa, and its prognostic significance. J Cancer Res Clin Oncol. 2002;128:426–32.
Selicharová I, Smutná K, Sanda M, et al. 2-DE analysis of a new human cell line EM-G3 derived from breast cancer progenitor cells and comparison with normal mammary epithelial cells. Proteomics. 2007;7:1549–59.
Tomonaga T, Matsushita K, Yamaguchi S, et al. Identification of altered protein expression and post-translational modifications in primary colorectal cancer by using agarose two-dimensional gel electrophoresis. Clin Cancer Res. 2004;10:2007–14.
Yancik R. Cancer burden in the aged: an epidemiologic and demographic overview. Cancer. 1997;80:1273–83.
Janssen-Heijnen ML, Smulders S, Lemmens VE, et al. Effect of comorbidity on the treatment and p rognosis of elderly patientswith non-small cell lung cancer. Thorax. 2004;59:602–7.
Li Z, Yu Y, Lu J, et al. Analysis of the T descriptors and other prognosis factors in pathologic stage I non-small cell lung cancer in China. J Thorac Oncol. 2009;4:702–9.
Chabowski M, Orłowski TM, Rabczenko D. Analysis of prognostic factors and efficacy of surgical treatment for non-small cell lung cancer: department of surgery NTLDRI (1998–1999). Pneumonol Alergol Pol. 2008;76:1–10.
Conflicts of interest
None.
Funding
This study was supported by Science and Technology Planning Project of Guangdong Province, China (Nos: 2007B031515017 and 2008A030201024). This work was also supported by Science and Technology Planning Project of Guangzhou, China (Nos: 2007Z1-E0111 and 2007Z3-E0261).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Wei Wang and Xin Xu have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Wang, W., Xu, X., Wang, W. et al. The expression and clinical significance of CLIC1 and HSP27 in lung adenocarcinoma. Tumor Biol. 32, 1199–1208 (2011). https://doi.org/10.1007/s13277-011-0223-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-011-0223-0