Abstract
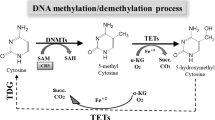
The zinc finger of the cerebellum (ZIC1) is a tumor suppressor gene that is silenced through promoter hypermethylation in colorectal cancer. This study was conducted to evaluate the efficacy of herbal medicines to demethylate the ZIC1 gene in colorectal cancer. We evaluated the effects of 30 herbal medicines on global DNA methylation, including the ZIC1 gene, in SW620 cells by a commercial DNA methylation assay. After the first round of screening, candidate herbal medicines were selected based on ZIC1 gene expression by reverse transcription-polymerase chain reaction and methylation-specific PCR analysis. The efficacy of the selected candidate medicines on DNA demethylation was then determined by methylationspecific PCR analysis and pyrosequencing. Of the extracts evaluated, Vitex rotundifolia was associated with ZIC1 re-expression in SW620 cells. Of the herbal medicines studied, V. rotundifolia is the best candidate for the demethylation of ZIC1. Additional studies should be conducted to determine whether V. rotundifolia and other herbal medicines represent novel therapeutic avenues that can be utilized for the prevention or treatment of colorectal cancer.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Jemal, A. et al. Cancer statistics, 2009. CA Cancer J Clin 59:225–249, doi:caac.20006 [pii] 10.3322/caac.20006 (2009).
Kim, M. S., Lee, J. & Sidransky, D. DNA methylation markers in colorectal cancer. Cancer Metastasis Rev 29:181–206, doi:10. 1007/s10555-010-9207-6 (2010).
Taylor, D. P., Burt, R. W., Williams, M. S., Haug, P. J. & Cannon-Albright, L. A. Population-based family history-specific risks for colorectal cancer: a constellation approach. Gastroenterology 138:877–885, doi: S0016-5085(09)02061-7 [pii] 10.1053/j.gastro.2009.11.044 (2010).
Kerber, R. A., Neklason, D. W., Samowitz, W. S. & Burt, R. W. Frequency of familial colon cancer and hereditary nonpolyposis colorectal cancer (Lynch syndrome) in a large population database. Fam Cancer 4:239–244, doi:10.1007/s10689-005-0657-x (2005).
Kinzler, K. W. & Vogelstein, B. Lessons from hereditary colorectal cancer. Cell 87:159–170, doi:S0092-8674(00)81333-1 [pii] (1996).
Jones, P. A. & Baylin, S. B. The fundamental role of epigenetic events in cancer. Nat Rev Genet 3:415–428, doi:10.1038/nrg816 nrg816 [pii] (2002).
Rennie, P. S. & Nelson, C. C. Epigenetic mechanisms for progression of prostate cancer. Cancer Metastasis Rev 17:401–409 (1998).
Esteller, M. et al. Cancer epigenetics and methylation. Science 297:1807–1808; discussion 1807–1808 (2002).
Baylin, S. B. Tying it all together: epigenetics, genetics, cell cycle, and cancer. Science 277:1948–1949 (1997).
Wolffe, A. P. & Matzke, M. A. Epigenetics: regulation through repression. Science 286:481–486, doi: 7899 [pii] (1999).
Holliday, R. & Grigg, G. W. DNA methylation and mutation. Mutat Res 285:61–67 (1993).
Bird, A. The essentials of DNA methylation. Cell 70:5–8 (1992).
Bird, A. P. CpG-rich islands and the function of DNA methylation. Nature 321:209–213 (1986).
Grinberg, I. & Millen, K. J. The ZIC gene family in development and disease. Clin Genet 67:290–296, doi:CGE418 [pii] 10.1111/j.1399-0004.2005.00418.x (2005).
Aruga, J. et al. A novel zinc finger protein, zic, is involved in neurogenesis, especially in the cell lineage of cerebellar granule cells. J Neurochem 63:1880–1890 (1994).
Merzdorf, C. S. Emerging roles for zic genes in early development. Dev Dyn 236:922–940, doi:10.1002/dvdy.21098 (2007).
Maurus, D. & Harris, W. A. Zic-associated holoprosencephaly: zebrafish Zic1 controls midline formation and forebrain patterning by regulating Nodal, Hedgehog, and retinoic acid signaling. Genes Dev 23:1461–1473, doi:23/12/1461 [pii] 10.1101/gad.517009 (2009).
Aruga, J. The role of Zic genes in neural development. Mol Cell Neurosci 26:205–221, doi:10.1016/j.mcn.2004.01.004 S1044743104000090 [pii] (2004).
Yokota, N. et al. Predominant expression of human zic in cerebellar granule cell lineage and medulloblastoma. Cancer Res 56:377–383 (1996).
Gan, L. et al. ZIC1 is downregulated through promoter hypermethylation, and functions as a tumor suppressor gene in colorectal cancer. PLoS One 6:e16916, doi:10.1371/journal.pone.0016916 (2011).
Colella, S., Shen, L., Baggerly, K. A., Issa, J. P. & Krahe, R. Sensitive and quantitative universal Pyrosequencing methylation analysis of CpG sites. Biotechniques 35:146–150 (2003).
The ENCODE (ENCyclopedia Of DNA Elements) Project. Science 306:636–640, doi:306/5696/636 [pii] 10.1126/science.1105136 (2004).
Jones, P. A. & Baylin, S. B. The epigenomics of cancer. Cell 128:683–692, doi:S0092-8674(07)00127-4 [pii] 10.1016/j.cell.2007.01.029 (2007).
Esteller, M. Epigenetics in cancer. N Engl J Med 358: 1148–1159, doi:358/11/1148 [pii] 10.1056/NEJMra 072067 (2008).
Grady, W. M. & Carethers, J. M. Genomic and epigenetic instability in colorectal cancer pathogenesis. Gastroenterology 135:1079–1099, doi:S0016-5085 (08)01451-0 [pii] 10.1053/j.gastro.2008.07.076 (2008).
Herman, J. G. et al. Inactivation of the CDKN2/p16/MTS1 gene is frequently associated with aberrant DNA methylation in all common human cancers. Cancer Res 55:4525–4530 (1995).
Lee, B. B. et al. Aberrant methylation of APC, MGMT, RASSF2A, and Wif-1 genes in plasma as a biomarker for early detection of colorectal cancer. Clin Cancer Res 15:6185–6191, doi:1078-0432.CCR-09-0111 [pii] 10.1158/1078-0432.CCR-09-0111 (2009).
Yu, J. et al. Epigenetic identification of ubiquitin carboxyl-terminal hydrolase L1 as a functional tumor suppressor and biomarker for hepatocellular carcinoma and other digestive tumors. Hepatology 48:508–518, doi:onc2010370 [pii] 10.1038/onc.2010.370 (2010).
Yu, J. et al. Epigenetic inactivation of T-box transcription factor 5, a novel tumor suppressor gene, is associated with colon cancer. Oncogene 29:6464–6474, doi:onc2010370 [pii] 10.1038/onc.2010.370 (2010).
Fearon, E. R. Molecular genetics of colorectal cancer. Annu Rev Pathol 6:479–507, doi:10.1146/annurevpathol-011110-130235 (2011).
Johnson, G. L. & Lapadat, R. Mitogen-activated protein kinase pathways mediated by ERK, JNK, and p38 protein kinases. Science 298:1911–1912, doi:10. 1126/science.1072682298/5600/1911 [pii] (2002).
Davis, R. J. Signal transduction by the JNK group of MAP kinases. Cell 103:239–252, doi:S0092-8674(00)00116-1 [pii] (2000).
Khor, T. O., Gul, Y. A., Ithnin, H. & Seow, H. F. Positive correlation between overexpression of phospho-BAD with phosphorylated Akt at serine 473 but not threonine 308 in colorectal carcinoma. Cancer Lett 210:139–150, doi:10.1016/j.canlet.2004.01.017S0304383504000771 [pii] (2004).
Chang, L. & Karin, M. Mammalian MAP kinase signalling cascades. Nature 410:37–40, doi:10.1038/35065000 35065000 [pii] (2001).
Datta, S. R., Brunet, A. & Greenberg, M. E. Cellular survival: a play in three Akts. Genes Dev 13:2905–2927 (1999).
Levine, B., Sinha, S. & Kroemer, G. Bcl-2 family members: dual regulators of apoptosis and autophagy. Autophagy 4:600–606, doi:6260 [pii] (2008).
Adams, J. M. & Cory, S. Life-or-death decisions by the Bcl-2 protein family. Trends Biochem Sci 26:61–66, doi:S0968-0004(00)01740-0 [pii] (2001).
Kim, J. et al. Inhibition effects of Moutan Cortex Radicis on secretion of eotaxin in A549 human epithelial cells and eosinophil migration. J Ethnopharmacol 114: 186–193 (2007).
Hu, Y. et al. Estrogen-like activity of volatile components from Vitex rotundifolia L. Indian J Med Res 126:68–72 (2007).
Laird, P. W. & Jaenisch, R. DNA methylation and cancer. Hum Mol Genet 3Spec No:1487–1495 (1994).
Baylin, S. B. et al. Aberrant patterns of DNA methylation, chromatin formation and gene expression in cancer. Hum Mol Genet 10:687–692 (2001).
Goelz, S. E., Vogelstein, B., Hamilton, S. R. & Feinberg, A. P. Hypomethylation of DNA from benign and malignant human colon neoplasms. Science 228: 187–190 (1985).
Rodriguez, J. et al. Chromosomal instability correlates with genome-wide DNA demethylation in human primary colorectal cancers. Cancer Res 66:8462–9468, doi:66/17/8462 [pii] 10.1158/0008-5472.CAN-06-0293 (2006).
Okuyama, E., Fujimori, S., Yamazaki, M. & Deyama, T. Pharmacologically active components of viticis fructus (Vitex rotundifolia). II. The components having analgesic effects. Chem Pharm Bull (Tokyo) 46:655–662 (1998).
Sohn, S. H. et al. Inhibition effects of Vitex rotundifolia on inflammatory gene expression in A549 human epithelial cells. Ann Allergy Asthma Immunol 103:152–159, doi:S1081-1206(10)60169-X [pii] 10.1016/S1081-1206(10)60169-X(2009).
Shin, T. Y. et al. Effect of Vitex rotundifolia on immediate-type allergic reaction. J Ethnopharmacol 72:443–450, doi:S0378874100002580 [pii] (2000).
Kumar, A. & Singh, A. Possible nitric oxide modulation in protective effect of (Curcuma longa, Zingiberaceae) against sleep deprivation-induced behavioral alterations and oxidative damage in mice. Phytomedicine 15:577–586 (2008).
Sun, D. I. et al. Bisacurone inhibits adhesion of inflammatory monocytes or cancer cells to endothelial cells through down-regulation of VCAM-1 expression. Int Immunopharmacol 8:1272–1281 (2008).
Everett, P. C., Meyers, J. A., Makkinje, A., Rabbi, M. & Lerner, A. Preclinical assessment of curcumin as a potential therapy for B-CLL. Am J Hematol 82:23–30, doi:10.1002/ajh.20757 (2007).
Jobin, C. et al. Curcumin blocks cytokine-mediated NF-kappa B activation and proinflammatory gene expression by inhibiting inhibitory factor I-kappa B kinase activity. J Immunol 163:3474–3483, doi:ji_v1 63n6p3474 [pii] (1999).
Subramanian, M., Sreejayan, Rao, M. N., Devasagayam, T. P. & Singh, B. B. Diminution of singlet oxygen-induced DNA damage by curcumin and related antioxidants. Mutat Res 311:249–255 (1994).
Lee, J. H. et al. Anti-inflammatory effect of the aqueous extract from Lonicera japonica flower is related to inhibition of NF-kappaB activation through reducing I-kappaBalpha degradation in rat liver. Int J Mol Med 7:79–83 (2001).
Kwon, S. H. et al. The neuroprotective effects of Lonicera japonica THUNB. against hydrogen peroxideinduced apoptosis via phosphorylation of MAPKs and PI3K/Akt in SH-SY5Y cells. Food Chem Toxicol 49: 1011–1019, doi:S0278-6915(11)00010-X [pii] 10.1016/j.fct.2011.01.008 (2011).
Suh, S. J. et al. The naturally occurring biflavonoid, ochnaflavone, inhibits LPS-induced iNOS expression, which is mediated by ERK1/2 via NF-kappaB regulation, in RAW264.7 cells. Arch Biochem Biophys 447: 136–146, doi:S0003-9861(06)00023-3 [pii] 10.1016/j.abb.2006.01.016 (2006).
Tang, D., Li, H. J., Chen, J., Guo, C. W. & Li, P. Rapid and simple method for screening of natural antioxidants from Chinese herb Flos Lonicerae Japonicae by DPPHHPLC-DAD-TOF/MS. J Sep Sci 31:3519–3526, doi: 10.1002/jssc.200800173 (2008).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sohn, SH., Cho, K. & Bae, H. Screening for herbal medicines that affect ZIC1 gene methylation in colorectal cancer. Mol. Cell. Toxicol. 9, 211–218 (2013). https://doi.org/10.1007/s13273-013-0027-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13273-013-0027-y