Abstract
A vascular access (VA) is used to facilitate hemodialysis in patients that suffer from end-stage renal disease. However, they suffer from high failure rates due to non-maturation and venous stenosis, with intimal hyperplasia (IH) the underlying cause of both conditions. Abnormal hemodynamic profiles, which arise following VA creation, are believed to lead to the development of IH. However, the exact physiological response that initiates this process is unknown. This review evaluates the different hemodynamic parameters that are hypothesised to correlate with the development of IH. Review studies that examine the correlation between hemodynamic parameters and the onset of IH using computational fluid dynamics. These studies are divided into groups depending on the type of analysis conducted; longitudinal studies, patient specific arteriovenous fistula (AVF) studies, arteriovenous graft studies, idealised AVF studies and studies that analyse the bulk flow. Studies that conduct longitudinal analysis identify an overall reduction in wall shear stress (WSS) as the VA matures. This is further associated with outward remodelling and the successful maturation of the VA. The majority of studies that conduct a transversal analysis find that low/oscillating shear is associated with the development of IH. However, a number of studies find a link between high shear and high spatial and temporal WSS gradients and the onset of IH. This review highlights the lack of unanimity between studies and emphasises the fact that the exact physiological response that leads to the development of IH remains unknown. This accentuates the need for a single, precise hypothesis capable of accurately predicting the onset of IH. If computational modelling is to assist in this process, the number of longitudinal studies conducted must increase. This will provide a better understanding of the effect that hemodynamic parameters have on the remodelling process and potentially identify a single/group of parameter/s that can accurately predict the onset of IH.
Similar content being viewed by others

Avoid common mistakes on your manuscript.
Introduction
Patients suffering from end-stage renal disease (ESRD) are often referred to hemodialysis to replace the lost function of their kidneys. In order for patients to receive effective dialysis, a vascular access (VA) capable of supplying high flow rates is required. The most common types of VA are an arteriovenous fistula (AVF) and an arteriovenous graft (AVG). Both access types suffer from high failure rates ranging from 18 to 28% for AVFs and higher again for AVGs.1 While both access types are considerably different they suffer from the same primary mode of failure, venous stenosis due to intimal hyperplasia (IH).63, 64, 68, 75
IH can be characterised as the thickening of the vascular wall due to the migration of smooth muscle cells (SMCs) from the media to the intima which then proliferate into the subintimal layer. Endothelial cells (ECs) play a fundamental role in this process. Once activated, ECs increase expression of growth factors that promote SMC migration from the media to the intima where they then proliferate. The proliferation of SMCs in the intima is associated with the deposition of extracellular matrix, in a process similar to the formation of scar tissue. This process results in the rapid formation of a neointimal layer over the activated ECs.12, 31, 49
It is well documented that IH can occur due to vascular injury caused by surgery, compliance mismatch between the graft and native vessel, dialysis needle insertion, inflammatory response to sutures and hemodynamic stresses.42, 65 – 67 This review will focus on the impact hemodynamic stresses have on the development of IH and the current hemodynamic parameters utilised to characterise these stresses using computational modelling.
Hemodynamic stresses are detected by the endothelial cells (EC), which act as mechanotransductors converting stimuli into specific biochemical signals depending on the type of flow they are subjected to. There are 3 predominant types of flow EC experience within a VA; unidirectional flow, flow reversal and multidirectional flow, Fig. 1. It is well documented that unidirectional flow which results in uniform wall shear stress (WSS) triggers endothelial quiescence and alignment in the predominant direction of the flow, resulting in the secretion of anti-inflammatory and anti-coagulant substances. These flow conditions tend to prevent the onset and progression of IH, while nonuniform WSS causes EC activation which in turn causes the expression of pro-inflammatory and pro-coagulant stimulants that predispose to the onset of IH.23, 28, 42, 54
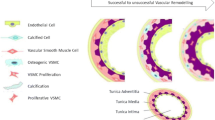
AVF maturation failure can be characterised by impaired outward remodelling or by the presence of a clinically significant stenosis in the venous segment of the AVF, both of which prevent the AVF from ever being used for dialysis. Hemodynamic stresses are believed to play a role in this process through the activation of endothelial cells which leads to a reduction in nitric oxide (NO). The low levels of NO can result in IH as NO blocks smooth muscle cell migration and proliferation. This can also result in impaired outward remodelling as NO is a potent vasodilator, Fig. 2.61
A typical configuration of an AVF, highlighting the cross section of a healthy artery, a healthy and chronic kidney disease (CKD) vein as well as the vascular remodelling process, displaying the different remodelling responses following fistula creation. IH, intimal hyperplasia; CKD, chronic kidney disease; VSMC, vascular smooth muscle cells.
It has been previously shown that stenoses develop at specific sites within an AVF; the anastomosis floor, the inner wall of the swing segment and the inner wall of the proximal vein where the vein straightens out, Fig. 3.4, 73 This is also true for AVG, where stenoses primarily develop at the venous anastomosis and in the draining vein.80
The sites within an AVF that are susceptible to the development of IH due to the presence of low/oscillating shear caused by areas of disturbed flow. (a) An AVF with antegrade flow in the DA. (b) An AVF with retrograde flow in the DA. The thick black arrows represent the predominant direction of flow within the AVF, while the thin black arrows represent the locations where recirculating vortexes and flow stagnation occur due to disturbed flow and the dashed lines represent the flow streamlines. AVF, arteriovenous fistula; PA, proximal artery; DA, distal artery; PV, proximal vein.
The geometric configuration of an AVF has been shown to strongly influence the hemodynamics. Minor changes in geometry can result in a drastic change in shear stress which is an important mediator of vasodilation and vessel remodelling. Ene-Iordache et al.18 investigated the affect that the anastomosis angle has on producing disturbed flow in a parametrised model of an AVF and found that low/oscillating shear was prominent in the locations predisposed to the development of stenoses, Fig. 3.4, 18, 73 They also found that acute anastomosis angles (~30°) are the most efficient at minimising areas exposed to low/oscillating shear, which can lead to the development of IH.18 A study conducted by Bharat et al. tested this hypothesis in a clinical setting and found that compared to traditional AVF configurations, fistulas with smaller anastomosis angles had fewer stenoses.5
Computational fluid dynamics (CFD) can approximate analytically complex flow fields, such as those that occur within VA following creation. CFD can also calculate hemodynamic parameters derived from the flow fields such as WSS. Computational modelling is currently utilised to retrospectively analyse VA to determine the cause of failure by attempting to correlate different hemodynamic parameters to the development of IH.
There is a significant amount of evidence to suggest that hemodynamic factors, specifically WSS, are involved in the onset and progression of different disease pathologies within the vasculature, a phenomenon Morbiducci et al. described as the ‘hemodynamic hypothesis’.52 To date, WSS based parameters have been the predominant indicators of the development of IH by identifying areas that experience disturbed shear.41 Disturbed shear is characterised as the exposure of the vasculature to adverse, non-physiological shear stress conditions. However, there are different hypotheses regarding this definition, some researchers define disturbed shear as exposure to low/oscillating shear,6, 15, 20, 35, 59, 76, 79, 80 while others identify it as exposure to high shear.8, 10, 11, 38, 45, 81 Both classifications of disturbed shear are hypothesised to lead to the development of IH. The majority of relevant research supports the former hypothesis. However, there is not a general consensus on the matter and there is a considerable amount of research to support the latter.
Research on the development of IH has primarily focused on the progression of the disease in the arterial system. However, the development of IH in VAs largely occurs in the venous segment of the access. It is well documented that the remodelling process of arteries and veins is considerably different, especially in the veins of chronic kidney disease (CKD) patients.62 A characteristic of CKD is the presence of calcifications within the vessel wall resulting in vessel stiffness. In arteries this impairs their ability to expand, limiting remodelling. While there has been limited research on the effect calcifications have in veins, it is likely to reduce venous compliance, also limiting the veins ability to expand during remodelling.43 This could be detrimental to fistula maturation by inhibiting outward remodelling and ultimately leading to fistula failure.
Retrospectively using WSS parameters to identify areas of disturbed shear within VA is of little clinical benefit to the patient as it will have no impact on the survival of their VA. If new therapeutic strategies that utilise WSS parameters are to have an impact on VA survival, then these parameters need to be quantified in vivo so that increased surveillance can take place to ensure that areas of disturbed shear do not transition into something more sinister such as a stenosis, which could result in access failure. A major hindrance to the implementation of WSS parameters in vivo is the requirement of expensive clinical imaging techniques such as phase-contrast magnetic resonance imaging (PC-MRI). While PC MRI is widely used in the acquisition of medical images for research purposes, its implementation into the routine surveillance of VA is impractical due to the additional cost per patient and lengthy analysis times.
More recently bulk flow parameters, specifically helicity based parameters, have been shown to correlate with areas of disturbed shear in the carotid bifurcation.26 If the same correlation exists in VA such parameters could be utilised to identify areas of disturbed shear within the VA in vivo, as helicity based parameters are much easier to quantify in vivo through the use of ultrasound imaging techniques.57
The aim of this review is to conduct an extensive literature review to determine the different hypotheses that surround the development of IH within VAs in an effort to identify the most widely adopted hypothesis.
WSS Parameters
The following section details studies that conduct a computational analysis of VA and attempt to correlate WSS based parameters with the development of IH. This section is divided into 4 subsections related to the type of VA and the analysis performed.
Longitudinal Studies
This section focuses on the computational analysis of realistic AVF that incorporate longitudinal data which is classified as data from two or more time points of the same VA. Such studies allow researchers to monitor the hemodynamics over time and attempt to correlate certain hemodynamic parameters to the development of vascular pathology. The VA analysed in this section are from both human and non-human subjects. Table 1 highlights the studies included in this section in addition to the WSS parameters utilised in their analysis as well as the findings of each study.
Human Models
The most significant longitudinal study conducted to date was carried out by Sigovan et al.71 who analysed the temporal and morphological changes of 3 patient specific AVFs at 5 days, 1 month and 3 months post-surgery. Sigovan et al.71 attempted to correlate certain hemodynamic parameters to vascular remodelling and discovered that disturbed flow (recirculation zones) present in the outflow vein of the AVF was associated with the non-uniform remodelling of the vessel. Out of the 3 AVFs analysed in this study only 1 of them was deemed mature and used for dialysis. However, the other 2 fistulas failed after 3 months and as a result no correlation could be established between the hemodynamics and the ultimate cause of fistula failure.
He et al.32 conducted a similar analysis as Sigovan et al.71 and analysed the temporal and morphological changes of a single AVF at 4, 5 and 7 months post-surgery. The aim of this study was to develop a procedure for the large scale assessment of hemodynamic parameters and lumen cross sectional area changes that occur following fistula creation and therefore this study does not formulate a hypothesis in relation to the development of IH. It was noticed that the AVF analysed in this study underwent an overall reduction in WSS between the 4 and 7 month scans which correlated with an increase in vein diameter. This is in agreement with the results of Rajabi-Jagahrgh et al.58, 59 who also found that an overall reduction in WSS over time was associated with outward remodelling and fistula maturation thus adding further merit to this hypothesis.
Boghosian et al.6 analysed a single brachiocephalic fistula for 1 year following its creation. They scanned the fistula at 3 months and 1 year and found that the location where a stenosis developed was subject to low shear. Boghosian et al.6 therefore hypothesises that low shear is associated with the development of IH and ultimately fistula failure. They also propose a secondary hypothesis whereby high shear may cause endothelial damage which could also lead to neointimal hyperplasia (NIH).
Animal Models
Jia et al.35 analysed the effect that WSS has on the development of NIH in 20 canine models. The histological results from this study revealed that NIH correlated with the areas exposed to low/disturbed shear at both 7 and 28 days as quantified from computational models that mimic the in vivo conditions of the canine models.
A similar study conducted by Rajabi-Jagahrgh et al.60 attempted to determine the longitudinal effects of different hemodynamic parameters on intimal medial thickening (IMT) in a single porcine model. A radiopaque marker was sutured to the outside of the AVF during its creation, acting as a reference for in vivo and ex vivo analysis of the AVF. Computational models were created from scans taken at 2 and 28 days from which changes in WSS over time were monitored. The histological results revealed that areas of the AVF that suffered from the most IMT were also exposed to recirculation zones at both 2 and 28 days. While not specifically outlined in this study recirculation zones predominantly correlate with areas of low/oscillating shear.
Rajabi-Jagahrgh et al.58, 59 also analysed the temporal changes in 6 AVF created in 3 porcine models. Results revealed that an overall reduction in WSS from the time of fistula creation was associated with outward remodelling and ultimately successful maturation. Conversely, an overall increase in WSS was found to be detrimental to AVF maturation.
The significance of these results is emphasised by the fact that 4 out of the 7 studies reviewed in this section compared histological results from porcine models sacrificed at 7 and 28 days with WSS profiles obtained through CFD simulations and all came to the same conclusion.
The main conclusions drawn from the longitudinal studies are as follows:
-
1.
Low shear that occurs as a result of recirculating flow is prone to the development of IH.
-
2.
A reduction in overall WSS as the fistula matures is associated with outward remodelling and the successful maturation of the fistula.
-
3.
An increase in overall WSS as the fistula matures is associated with inward remodelling and fistula non-maturation.
Realistic AVF Studies
The results of the realistic AVF studies are more diverse than the longitudinal studies; with only 5 out of the 10 studies supporting the hypothesis that exposure to low/oscillating shear can lead to development of IH, Table 2.
Hammes et al.30 conducted the most recent study in this area and analysed the WSS profile of 22 brachiocephalic AVF between 8 and 32 weeks post-surgery. Results revealed a significant correlation between low shear and the development of NIH. They do state however, that further studies would be required to conclude if low shear is actually associated with the onset of NIH.
Decorato et al.16 support this hypothesis and conducted a fluid structure interaction (FSI) simulation on a patient specific AVF and found that low/oscillating shear correlated with areas that are prone to lesion development as outlined in literature. Previous work conducted by Ene-Iordache et al.20, 80 found that low/oscillating shear occurred at locations that correspond closely to sites of future stenosis in idealised models of AVFs. In their most recent study they found that both multidirectional flow (TransWSS) and reciprocating disturbed flow (OSI) also occurred at locations predisposed to IH development.21
Kharboutly et al.38 suggest a potential link between exposure to high TWSSG and regions of calcification. Similarly, Carroll et al.11 also found that high WSS, high WSSG and flow reversal persist throughout a mature AVF. They propose that high shear, elevated WSSG and flow reversal result in venous neointimal hyperplasia (VNH) development. Carroll et al.10 tested this hypothesis by applying the elevated WSS waveforms obtained from the computational models to human umbilical vein endothelial cells (HUVEC) using a cone and plate bioreactor. The elevated temporal WSS waveforms induced an early induction of MMP-2 and delayed transcriptional upregulation of MCP-1, suggesting a link between elevated WSS and temporal WSSG and subsequent VNH development. MMP-2 and MCP-1 are inflammatory regulators that play a role in the initiation/contribution to the development of VNH.
Franzoni et al.24 conducted a similar study in which they exposed human umbilical vein ECs (HUVECs) to both unidirectional and reciprocating flows using a cone and plate device. They found that there was a significant increase in the expression of KLF2 in cells exposed to unidirectional flow. Simultaneously this flow condition induced a decrease in the expression of PLD1, ITGA4 and RASA1. In contrast to this, cells exposed to reciprocating flow did not increase KLF2 expression but did increase in the expression of PLD1, ITGA4 and RASA1. The expression of KLF2 has been previously shown to induce the expression of nitric oxide (NO), which has anti-inflammatory and anti-coagulant properties.14, 17, 22 These results suggest that unidirectional flow is necessary to maintain KLF2 related protective function of ECs and that this protective effect is lost in the presence of reciprocating flow where KLF2 expression is not increased.
In contrast to this McGah et al.46, 47 found high shear in mature fistulas ranging from 2 to 20 years. This suggests that high shear stress alone may not result in the development of IH or that there may be a threshold value for high shear above which initiation of IH occurs.37, 39, 47 While different studies suggest different threshold values for the onset of IH, Tricht et al.81 propose that WSS above 3 Pa could initiate the disease process. Hellums and Hardwick33 state that WSS values over 7.5 Pa activate leukocytes adhesion, which can lead to the development of IH while values over 10 Pa can cause the growth of platelet aggregation and thrombosis. In addition, WSS over 35 Pa can cause physical damage to the EC resulting in platelet adhesion.25
Other relevant theories include a study conducted by Bozzetto et al.7 who analysed the hemodynamics in 4 patient-specific AVF and observed the presence of transitional laminar to turbulent like flow developing in the juxta-anastomotic vein and dissipating towards the venous outlet. This resulted in high frequency fluctuations of the velocity vector, which in turn induced similar fluctuations of the WSS vector in these specific areas. This led them to hypothesis that these fluctuations may impair the physiological response of ECs and could be responsible for the development of IH in AVF.
The findings of these studies carry less significance compared to the findings of the longitudinal studies as there is no way of determining the longitudinal effect that the hemodynamics have on the VA. Therefore, it is difficult to link different hemodynamic parameters to disease progression.
AVG Studies
The most impactful study detailed in this section was carried out by Manos et al.45 who conducted a computational simulation of a realistic AVG model acquired from a porcine model 1 month following graft placement. The results from the computational model were compared to histological sections. Comparison revealed that high WSS and high WSSG correlate with areas of IH and increased vessel wall stiffness.
Two further studies utilised patient specific geometries of AVG in their analysis. However, neither of these studies focused on determining the cause of IH but rather applied hypotheses proposed by other researchers on the development of IH to the analysis of their results. Sung et al.76 followed the hypothesis that low/oscillating shear leads to the development of IH, while Choi et al.13 states that both low shear (less than 0.3 Pa) and high shear (greater than 3 Pa) is associated with the development of IH.
The remainder of the studies included in this section were all conducted on idealised AVG models and utilise hypotheses formulated by other researchers to analyse their results, Table 3. Among these studies there was a wide range of hypotheses adopted.40, 44, 69, 77, 81
There is little coherence between the studies and the hypotheses they supported in this section, with 3 out of these 8 studies stating that both high and low shear are associated with IH development. Only one of these studies solely supports the hypothesis that low/oscillating shear leads to the development of IH. These findings are considerably different to the findings of both the longitudinal and realistic AVF studies where the majority of researchers agreed with the low/oscillating shear hypothesis.
Idealised Studies
Each of the studies detailed in this section analyse idealised models of AVF, Table 4. Such studies are generally not able to make substantial claims based on their results but rather utilise hypotheses developed by other researchers to analyse their results. This is highlighted here as 5 out of the 6 studies support the hypothesis that low/oscillating shear is associated with the development of IH.19, 20, 34, 72, 79 The most impactful of these studies were conducted by Ene-Iordache et al.20, 80 who compared their results to literature and established that areas exposed to low/oscillating shear correlate with sites of luminal reduction. Browne et al.8 utilised CFD to acquire different WSS profiles that are likely to lead to the development of IH. These profiles were then applied to endothelial cells (EC) in a cone and plate bioreactor to monitor EC proliferation which was used as an indicator of IH development. This revealed that the maximum absolute TWSSG had the most significant correlation to EC proliferation.
Bulk Flow Parameters
The inclusion criteria for the studies detailed in this section is less stringent than previous sections as limited research has been conducted on the use of helicity based parameters to characterise the bulk flow. As a result, this section includes studies that conducted computational analyses of VAs as well as the carotid bifurcation, Table 5. The helicity based parameters utilised by each study varied between a quantitative and a qualitative analysis of the bulk flow, with the majority of studies adopting a purely qualitative analysis using the parameter the localised normalised helicity (LNH), which is a useful quantity for the visualisation of complex flow patterns within cardiovascular flows.70 Some studies do offer a more detailed analysis by quantifying the helical content of the bulk flow, which will be discussed in detail in the following section.
Morbiducci et al.51 highlights that most of the research conducted on the development of vascular pathology focuses primarily on WSS based parameters. They add that the bulk flow also plays a significant role in the progression vascular pathology. They state that the development of vascular pathology involves not only fluid related forces exerted on the wall (WSS) but also transport phenomenon within the blood. They suggest that the reason for this singular approach is partly due to the lack of parameters capable of characterising the bulk flow. In this study Morbiducci et al.51 develop new metrics capable of characterising the helical content of the bulk flow and highlight the possibility of these new metrics being utilised to relate certain aspects of the bulk flow to vascular physiopathology.
Gallo et al.26 elaborated on the work conducted by Morbiducci et al.51 by characterising the relationship between WSS based parameters, specifically disturbed shear (low/oscillating shear) and the bulk flow using helicity based descriptors in the carotid bifurcation. For this study Gallo et al.26 defined new metrics which included the helicity based bulk flow descriptors h1, h2, h3, h4, h5 and h6. These descriptors quantify the helical content of the flow and also the balance between counter rotating helical structures. The LNH was also used to visualise the flow features present in the bulk flow. The study revealed a significant correlation between exposure to disturbed shear and their helicity based descriptors suggesting the possibility of utilising these parameters in large scale in vivo studies to identify areas of the vasculature susceptible to the development of vascular pathologies. However, it remains unclear if these parameters possess the same diagnostic capabilities when applied to VAs. Two other studies conducted by this group utilise these parameters to quantify the helicity content of the bulk flow. However, they were not employed to determine areas of the vasculature that are susceptible to disease progression but rather to quantitatively compare the results between different computational models.27, 53
Bozzetto et al.7 and Browne et al.9 are the only two groups to include a detailed analysis of the bulk flow in their investigation of the hemodynamics within AVFs. Both groups evaluate the velocity within their respective models and observed high frequency fluctuations, which are synonymous with transitional laminar to turbulent like flow in the venous segments of their models.79 Both groups state that the presence of these fluctuations may impact EC function, which could result in the cascade of events that leads to the development of IH. They also include a qualitative assessment of the bulk flow through the application of the parameter LNH.
Van Canneyt et al.78 investigated the effect that varying degrees of helicity have on the suppression of disturbed shear (low/oscillating shear). They found that the presence of centralised single vertical structures stabilise the flow field by avoiding the breakdown in multiple vortex patterns and fostering the mitigation of transitional effects downstream of the anastomosis. Similarly, to the other studies detailed in this section Van Canneyt et al.78 also provided a qualitative assessment of the bulk flow using the parameter LNH.
Discussion
VAs suffer from high failure rates ranging from approximately 18–28% for AVF and even higher for AVG.1 The main cause of these high failure rates is stenosis due to IH.63, 64, 68, 75 Computational modelling is utilised to retrospectively determine the cause of VA failure by attempting to correlate hemodynamic parameters with the development of IH. The effectiveness of this method is limited as the exact physiological response that leads to IH within VAs remains unknown. This is highlighted by the numerous hypotheses surrounding the development of IH in VAs, exposing the lack of unanimity between studies, Table 6. This review focuses on the hemodynamic parameters utilised by each study to predict the onset of IH. The main hypotheses proposed by the studies included in this review are:
-
1.
An overall reduction in WSS is observed as the AVF remodels. This is associated with outward remodelling and fistula maturation.32,58,60,71
-
2.
Exposure to low/oscillating shear can lead to the development of IH.19,27,29,30*,34,36,37*,39,40,48,49*,50*,51,52*,56,57,59,61
-
3.
Exposure to high shear can lead to the development of IH.30*,37*,41,42,43**,49*,50*,52*,53,55,58**
*Studies that support more than one hypothesis.
**Studies that reported a link between high TWSSG and the onset of IH.
The majority of studies support the hypothesis that low/oscillating shear can lead to the development of IH. However, a significant number of studies support the opposite and state that high shear can lead to the development of IH. In addition, a number of studies do not adhere to a single hypothesis and state that both low/oscillating shear and high shear can be detrimental to the survival of the VA.
There are numerous alternate hypotheses proposed by the studies in this review, most notably that the occurrence of high frequency fluctuations synonymous with transitional laminar to turbulent like flow may impact endothelial cell function and contribute to the physiological response that leads to the development of IH. This hypothesis was first proposed by Browne et al.9 in an idealised model of an AVF and later by Bozzetto et al.7 in a patient specific study of AVFs. However, this hypothesis requires further investigation in future studies to determine the full extent of its effect on endothelial cell function and the development of IH.
The most substantial hypothesis is proposed by the studies that conduct longitudinal analyses. These studies find that an overall reduction in WSS (regardless of initial value) as the AVF remodels is associated with outward remodelling and fistula maturation. This emphasises the increased importance of longitudinal studies over single time point studies as it’s not possible to determine the longitudinal effect of hemodynamic parameters on the remodelling process from a single time point study.
This review highlights the lack of longitudinal studies to date. A possible reason for this could be due to the complicated process involved in acquiring longitudinal data. This process requires that patients have their VA scanned in an MRI machine. A computational model must then be created of the VA from this scan. CFD simulations can then be conducted to analyse the hemodynamics within the VA. This process is then repeated at several time points to capture the hemodynamic and morphological changes that occur in the VA as it remodels. The typical longitudinal study contains data from three different time points of the same VA, and requires the patient to have their VA scanned on three separate occasions. This process can be overly time restricting for patients who are already undergoing hemodialysis several times a week.
Until recently it was believed that areas exposed to low/oscillating shear are susceptible to the development of IH. However, a review conducted by Peiffer et al.55 investigating the hypothesis that low/oscillating shear correlates with the development of atherosclerosis, found that studies that perform a rigorous point-by-point comparison between WSS and IMT do not find significant correlations, whereas studies that report significant correlations conduct less rigorous analyses.3, 29, 36, 74, 82 Studies that fail to find a correlation are all conducted on human vessels, while a significant number of the studies that find significant correlations are conducted on hypercholesterolaemic animal models.
It should be noted that the studies included in the review by Peiffer et al.55 were conducted on the biological response of arteries and the development of atherosclerosis. This review focuses on the progression of IH in VAs, which predominantly occurs in the venous segment of the access. As discussed previously, the remodelling response of arteries is considerably different to that of veins, particularly the veins of CKD patients. That is why the conclusions drawn by the longitudinal studies discussed in this review are more relevant than the conclusions drawn in the review conducted by Peiffer et al.55
IH is a complex process that can be trigged by multiple aspects of WSS. It is therefore difficult to ascertain a single WSS metric that is capable of quantitatively identifying areas of the vasculature susceptible to IH development. Peiffer et al.56 propose a relatively new metric called transverse wall shear stress which is a measure of the multidirectionality of disturbed flow. It is characterised as the average magnitude of WSS components acting transversely to the mean WSS vector. This metric could potentially provide a more accurate prediction of the locations of IH. Preliminary studies conducted on animal models have revealed that lesion prevalence correlates strongly with transWSS while other shear metrics correlate less significantly.50 While this study was not conducted on a VA model, the results are promising for the use of transWSS as a new metric for the detection of lesion formation/development of IH. Additionally, a more recent study conducted by Anderson et al.2 who analysed the effectiveness of the TransWSS metric in a model of a human aorta, found that in regions of transitional to turbulent flow, similar to the flow found in a VA, TransWSS correlated with areas of low/oscillating shear. The study concludes by saying that the TransWSS metric demonstrated a more multi-featured picture of WSS distribution when exposed to different types of flow regimes. However, this metric must be included in longitudinal studies before any significant conclusions can be drawn about its ability to identify areas of IH in VA.
Conventionally, WSS parameters have been utilised to predict the onset of IH within VA. However, bulk flow parameters have been introduced to provide a more detailed analysis of the flow as it is hypothesised to play a significant role in the physiological response that leads to the development of IH. A limited amount of research has been conducted on the use of bulk flow parameters in predicting VA dysfunction. However, its potential, as highlighted by Gallo et al.26 as a surrogate marker of exposure to disturbed shear in the carotid bifurcation warrants further research in a VA setting. This will determine if bulk flow parameters possess the same diagnostic capabilities in VAs.
Conclusion
The diversity of hypotheses detailed in this review reveals that the exact physiological response that leads to the development of IH remains unknown. This emphasises the need for a single, precise hypothesis capable of predicting the onset of IH. The discord among studies weakens the impact of computational modelling and more longitudinal studies must be conducted to assist in predicting the onset of IH. While there has been numerous studies conducted on the hemodynamics within patient specific models of VAs, the number of longitudinal studies that attempt to monitor the remodelling process over time is limited. Single time point studies are useful at accessing the hemodynamics occurring at a particular point in time but there is no way of determining the longitudinal effect they have on VA remodelling process. The future of computational modelling in this field must therefore focus on longitudinal studies with large cohorts of patients. Longitudinal studies will allow for the remodelling process of the newly created VA to be monitored over time, which will provide a greater understanding of the hemodynamic changes that take place during this complex remodelling process. This will also provide the opportunity to investigate different hemodynamic parameters and their ability to predict the onset of IH.
References
Al-Jaishi, A. A., M. J. Oliver, S. M. Thomas, C. E. Lok, J. C. Zhang, A. X. Garg, and S. D. Kosa. Patency rates of the arteriovenous fistula for hemodialysis: A systematic review and meta-analysis. Am. J. Kidney Dis. 63(3):464–478, 2014.
Andersson, M., J. Lantz, T. Ebbers, and M. Karlsson. Multidirectional WSS disturbances in stenotic turbulent flows: A pre-and post-intervention study in an aortic coarctation. J. Biomech. 51:8–16, 2017.
Augst, A. D., and B. Ariff. Analysis of complex flow and the relationship between blood pressure, wall shear stress, and intima-media thickness in the human carotid artery. Am. J. Physiol.-Heart Circ. Physiol. 62(2):1031–1037, 2007.
Badero, O. J., M. O. Salifu, H. Wasse, and J. Work. Frequency of swing-segment stenosis in referred dialysis patients with angiographically documented lesions. Am. J. Kidney Dis. 51(1):93–98, 2008.
Bharat, A., M. Jaenicke, and S. Shenoy. A novel technique of vascular anastomosis to prevent juxta-anastomotic stenosis following arteriovenous fistula creation. J. Vasc. Surg. 55(1):274–280, 2012.
Boghosian, M., K. Cassel, M. Hammes, B. Funaki, S. Kim, X. Qian, X. Wang, P. Dhar, and J. Hines. Hemodynamics in the cephalic arch of a brachiocephalic fistula. Med. Eng. Phys. 36(7):822–830, 2014.
Bozzetto, M., B. Ene-Iordache, and A. Remuzzi. Transitional flow in the venous side of patient-specific arteriovenous fistulae for hemodialysis. Ann. Biomed. Eng. 44(8):2388–2401, 2015.
Browne, L. D., S. O’Callaghan, D. A. Hoey, P. Griffin, T. M. McGloughlin, and M. T. Walsh. Correlation of hemodynamic parameters to endothelial cell proliferation in an end to side anastomosis. Cardiovasc. Eng. Technol. 5(1):110–118, 2014.
Browne, L. D., M. T. Walsh, and P. Griffin. Experimental and numerical analysis of the bulk flow parameters within an arteriovenous fistula. Cardiovasc. Eng. Technol. 6(4):450–462, 2015.
Carroll, G. T., T. M. McGloughlin, P. E. Burke, M. Egan, F. Wallis, and M. T. Walsh. Wall shear stresses remain elevated in mature arteriovenous fistulas: a case study. J. Biomech. Eng. 133(2):p21003, 2011.
Carroll, G. T., T. M. McGloughlin, L. M. O’Keeffe, A. Callanan, and M. T. Walsh. Realistic temporal variations of shear stress modulate MMP-2 and MCP-1 expression in arteriovenous vascular access. Cell. Mol. Bioeng. 2(4):591–605, 2009.
Chiu, J. J., and S. Chien. Effects of disturbed flow on vascular endothelium: pathophysiological basis and clinical perspectives. Physiol. Rev. 91:327–387, 2011.
Choi, Y. K., H. J. Shin, J. T. Kim, and H. S. Ryou. Investigation on the effect of hematocrit on unsteady hemodynamic characteristics in arteriovenous graft using the multiphase blood model. J. Mech. Sci. Technol. 29(6):2565–2571, 2015.
Das, H., A. Kumar, Z. Lin, W. D. Patino, P. M. Hwang, M. W. Feinberg, P. K. Majumder, and M. K. Jain. Kruppel-like factor 2 (KLF2) regulates proinflammatory activation of monocytes. Proc. Natl. Acad. Sci. USA 103:6653–6658, 2006.
Decorato, I., Z. Kharboutly, C. Legallais, and A. V. Salsac. Numerical study of the influence of wall compliance on the haemodynamics in a patient-specific arteriovenous fistula. Comput. Methods Biomech. Biomed. Eng. 14:121–123, 2011.
Decorato, I., Z. Kharboutly, T. Vassallo, J. Penrose, C. Legallais, and A. Salsac. Numerical simulation of the fluid structure interactions in a compliant patient-specific arteriovenous fistula. Int. J. Numer. Methods Biomed. Eng. 30(2):143–159, 2014.
Dekker, R. J., J. V. van Thienen, J. Rohlena, S. C. de Jager, Y. W. Elderkamp, J. Seppen, C. J. de Vries, E. A. Biessen, T. J. van Berkel, H. Pannekoek, and A. J. Horrevoets. Endothelial KLF2 links local arterial shear stress levels to the expression of vascular tone-regulating genes. Am. J. Pathol. 167:609–618, 2005.
Ene-Iordache, B., L. Cattaneo, G. Dubini, and A. Remuzzi. Effect of anastomosis angle on the localization of disturbed flow in “side-to-end” fistulae for haemodialysis access. Nephrol. Dial. Transplant. 28(4):997–1005, 2013.
Ene-Iordache, B., L. Cattaneo, G. Dubini, and A. Remuzzi. Effect of anastomosis angle on the localization of disturbed flow in “side-to-end” fistulae for haemodialysis access. Nephrol. Dial. Transplant. 28(4):997–1005, 2013.
Ene-Iordache, B., and A. Remuzzi. Disturbed flow in radial-cephalic arteriovenous fistulae for haemodialysis: Low and oscillating shear stress locates the sites of stenosis. Nephrol. Dial. Transplant. 27(1):358–368, 2012.
Ene-Iordache, B., C. Semperboni, G. Dubini, and A. Remuzzi. Disturbed flow in a patient-specific arteriovenous fistula for hemodialysis: Multidirectional and reciprocating near-wall flow patterns. J. Biomech. 48(10):2195–2200, 2015.
Fledderus, J. O., R. A. Boon, O. L. Volger, H. Hurttila, S. Yla-Herttuala, H. Pannekoek, A. L. Levonen, and A. J. Horrevoets. KLF2 primes the antioxidant transcription factor Nrf2 for activation in endothelial cells. Arterioscler. Thromb. Vasc. Biol. 28:1339–1346, 2008.
Frangos, J. A., S. G. Eskin, L. V. McIntire, and C. L. Ives. Flow effects on prostacyclin production by cultured human endothelial cells. Science 227:1477–1480, 1985.
Franzoni, M., I. Cattaneo, L. Longaretti, M. Figliuzzi, B. Ene-Iordache, and A. Remuzzi. Endothelial cell activation by hemodynamic shear stress derived from arteriovenous fistula for hemodialysis access. Am. J. Physiol.-Heart Circ. Physiol. 310(1):H49–59, 2015. doi:10.1152/ajpheart.00098.
Fry, D. L. Certain histological and chemical responses of the vascular interface to acutely induced mechanical stress in the aorta of the dog. Circ. Res. 24(1):93–108, 1969.
Gallo, D., D. A. Steinman, P. B. Bijari, and U. Morbiducci. Helical flow in carotid bifurcation as surrogate marker of exposure to disturbed shear. J. Biomech. 45(14):p2398–2404, 2012.
Gallo, D., D. A. Steinman, and U. Morbiducci. An insight into the mechanistic role of the common carotid artery on the hemodynamics at the carotid bifurcation. Ann. Biomed. Eng. 43(1):68–81, 2014.
Garanich, J. S., M. Pahakis, J. M. Tarbell, S. Jeffrey, M. Pahakis, and J. M. Tarbell. Shear stress inhibits smooth muscle cell migration via nitric oxide-mediated downregulation of matrix metalloproteinase-2 activity. Am. J. Physiol.-Heart Circ. Physiol. 288(5):2244–2252, 2005.
Gijsen, F. J. H., J. J. Wentzel, A. Thury, B. Lamers, J. C. H. Schuurbiers, P. W. Serruys, and A. F. van der Steen. A new imaging technique to study 3-D plaque and shear stress distribution in human coronary artery bifurcations in vivo. J. Biomech. 40(11):2349–2357, 2007.
Hammes, M., M. Boghosian, K. Cassel, S. Watson, B. Funaki, Taral Doshi, S. M. Javid Mahmoudzadeh Akherat, J. Hines, and F. Coe. Increased inlet blood flow velocity predicts low wall shear stress in the cephalic arch of patients with brachiocephalic fistula access. PLoS ONE 11(4):1–18, 2016.
Haruguchi, H., and S. Teraoka. Intimal hyperplasia and hemodynamic factors in arterial bypass and arteriovenous grafts: A review. J. Artif. Organs 6:227–235, 2003.
He, Y., C. M. Terry, C. Nguyen, S. A. Berceli, Y. T. E. Shiu, and A. K. Cheung. Serial analysis of lumen geometry and hemodynamics in human arteriovenous fistula for hemodialysis using magnetic resonance imaging and computational fluid dynamics. J. Biomech. 46(1):165–169, 2013.
Hellums, J. D., and R. A. Hardwick. Response of Platelets to Shear Stress-A Review. The Rheology of Blood, Blood Vessels and Associated Tissues. Leiden: Sijthoff and Noordhoff, pp. 160–183, 1981.
Iori, F., L. Grechy, R. W. Corbett, W. Gedroyc, N. Duncan, C. G. Caro, and P. E. Vincent. The effect of in-plane arterial curvature on blood flow and oxygen transport in arterio-venous fistulae. Phys. Fluids 27(3):p031903, 2015.
Jia, L., L. Wang, F. Wei, H. Yu, H. Dong, B. Wang, Z. Lu, G. Sun, H. Chen, J. Meng, and B. Li. Effects of wall shear stress in venous neointimal hyperplasia of arteriovenous fistulae. Nephrology 20(5):335–342, 2015.
Joshi, K., R. L. Leask, J. G. Myers, M. Ojha, J. Butany, and C. R. Ethier. Intimal thickness is not associated with wall shear stress patterns in the human right coronary artery. Arterioscler. Thromb. Vasc. Biol. 24(12):2408–2413, 2004.
Kharboutly, Z., V. Deplano, E. Bertrand, and C. Legallais. Numerical and experimental study of blood flow through a patient-specific arteriovenous fistula used for hemodialysis. Med. Eng. Phys. 32(2):111–118, 2010.
Kharboutly, Z., M. Fenech, J. M. Treutenaere, I. Claude, and C. Legallais. Investigations into the relationship between hemodynamics and vascular alterations in an established arteriovenous fistula. Med. Eng. Phys. 29(9):999–1007, 2007.
Kharboutly, Z., M. Fenech, J. M. Treutenaere, I. Claude, and C. Legallais. Investigations into the relationship between hemodynamics and vascular alterations in an established arteriovenous fistula. Med. Eng. Phys. 29(9):999–1007, 2007.
Kim, H.-H., Y. H. Choi, S.-H. Suh, J. S. Lee, Y. H. Jung, and Y. H. So. Arteriovenous graft modeling and hemodynamic interpretation. Open J. Fluid Dyn. 2(4):324–330, 2012.
Ku, D. N., D. P. Giddens, C. K. Zarins, and S. Glagov. Pulsatile flow and atherosclerosis in the human carotid bifurcation. Positive correlation between plaque location and low oscillating shear stress. Arterioscler. Thromb. Vasc. Biol. 5(3):293–302, 1985.
Lee, T., and P. Roy-Chaudhury. Advances and new frontiers in the pathophysiology of venous neointimal hyperplasia and dialysis access stenosis. Adv. Chronic Kidney Dis. 16(5):329–338, 2009.
Lee, T., N. Safdar, M. J. Mistry, Y. Wang, V. Chauhan, B. Campos, R. Munda, V. Cornea, and P. Roy-Chaudhury. Preexisting venous calcification prior to dialysis vascular access surgery. Semin. Dial. 25(5):592–595, 2012.
Lee, S. W., D. S. Smith, F. Loth, P. F. Fischer, and H. S. Bassiouny. Importance of flow division on transition to turbulence within an arteriovenous graft. J. Biomech. 40(5):981–992, 2007.
Manos, T. A., D. P. Sokolis, A. T. Giagini, C. H. Davos, J. D. Kakisis, E. P. Kritharis, N. Stergiopulos, P. E. Karayannacos, and S. Tsangaris. Local hemodynamics and intimal hyperplasia at the venous side of a porcine arteriovenous shunt. IEEE Trans. Inf. Technol. Biomed. 14(3):681–690, 2010.
McGah, P. M., D. F. Leotta, K. W. Beach, and A. Aliseda. Effects of wall distensibility in hemodynamic simulations of an arteriovenous fistula. Biomech. Modeling Mechanobiol. 13(3):679–695, 2013.
McGah, P. M., D. F. Leotta, K. W. Beach, R. Eugene Zierler, and A. Aliseda. Incomplete restoration of homeostatic shear stress within arteriovenous fistulae. J. Biomech. Eng. 135(1):p11005, 2013.
McGah, P. M., D. F. Leotta, K. W. Beach, J. J. Riley, and A. Aliseda. A longitudinal study of remodeling in a revised peripheral artery bypass graft using 3D ultrasound imaging and computational hemodynamics. J. Biomech. Eng. 133(4):041008, 2011.
Mitra, A. K., D. M. Gangahar, and D. K. Agrawal. Cellular, molecular and immunological mechanisms in the pathophysiology of vein graft intimal hyperplasia. Immunol. Cell Biol. 84:115–124, 2006.
Mohamied, Y., E. M. Rowland, E. L. Bailey, S. J. Sherwin, M. A. Schwartz, and P. D. Weinberg. Change of direction in the biomechanics of atherosclerosis. Ann. Biomed. Eng. 43(1):16–25, 2014.
Morbiducci, U., G. Diego, R. Ponzini, M. Diana, L. Antiga, F. M. Montevecchi, and A. Redaelli. Quantitative analysis of bulk flow in image-based hemodynamic models of the carotid bifurcation: The influence of outflow conditions as test case. Ann. Biomed. Eng. 38(12):3688–3705, 2010.
Morbiducci, U., A. M. Kok, B. R. Kwak, P. H. Stone, D. A. Steinman, and J. J. Wentzel. Atherosclerosis at arterial bifurcations: evidence for the role of haemodynamics and geometry. Thromb. Haemost. 115(3):484–492, 2016.
Morbiducci, U., R. Ponzini, D. Gallo, C. Bignardi, and G. Rizzo. Inflow boundary conditions for image-based computational hemodynamics: Impact of idealized versus measured velocity profiles in the human aorta. J. Biomech. 46(1):102–109, 2013.
Owens, C. D. Adaptive changes in autogenous vein grafts for arterial reconstruction: Clinical implications. J. Vasc. Surg. 51(3):736–746, 2010.
Peiffer, V., S. J. Sherwin, and P. D. Weinberg. Does low and oscillatory wall shear stress correlate spatially with early atherosclerosis? A systematic review. Cardiovasc. Res. 99(2):242–250, 2013.
Peiffer, V., S. J. Sherwin, and P. D. Weinberg. Computation in the rabbit aorta of a new metric—the transverse wall shear stress—to quantify the multidirectional character of disturbed blood flow. J. Biomech. 46(15):2651–2658, 2013.
Poelma, C., R. M. E. Van Der Mijle, J. M. Mari, M. X. Tang, P. D. Weinberg, and J. Westerweel. Ultrasound imaging velocimetry: Toward reliable wall shear stress measurements. Eur. J. Mech.-B/Fluids 35:70–75, 2012.
Rajabi-Jagahrgh, E., M. K. Krishnamoorthy, P. Roy-Chaudhury, P. Succop, Y. Wang, A. Choe, and R. K. Banerjee. Longitudinal assessment of hemodynamic endpoints in predicting arteriovenous fistula maturation. Semin. Dial. 26(2):208–215, 2012.
Rajabi-Jagahrgh, E., M. K. Krishnamoorthy, Y. Wang, A. Choe, P. Roy-Chaudhury, and R. K. Banerjee. Influence of temporal variation in wall shear stress on intima-media thickening in arteriovenous fistulae. Semin. Dial. 26(4):511–519, 2013.
Rajabi-Jagahrgh, E., P. Roy-Chaudhury, Y. Wang, M. Al-Rjoub, B. Campos-Naciff, A. Choe, C. Dumoulin, and R. K. Banerjee. New techniques for determining the longitudinal effects of local hemodynamics on the intima-media thickness in arteriovenous fistulae in an animal model. Semin. Dial. 27(4):424–435, 2014.
Riella, M. C., and P. Roy-Chaudhury. Vascular access in haemodialysis: strengthening the Achilles’ heel. Nat. Rev. Nephrol. 9(6):348–357, 2013.
Rothuizen, T. C., C. Wong, P. H. A. Quax, A. J. Van Zonneveld, T. J. Rabelink, and J. I. Rotmans. Arteriovenous access failure: More than just intimal hyperplasia? Nephrol. Dial. Transplant. 28(5):1085–1092, 2013.
Roy-Chaudhury, P., L. Arend, J. Zhang, M. Krishnamoorthy, Y. Wang, R. Banerjee, A. Samaha, and R. Munda. Neointimal hyperplasia in early arteriovenous fistula failure. Am. J. Kidney Dis 50(5):782–790, 2007.
Roy-Chaudhury, P., B. S. Kelly, M. A. Miller, A. Reaves, J. Armstrong, N. Nanayakkara, and S. C. Heffelfinger. Venous neointimal hyperplasia in polytetrafluoroethylene dialysis grafts. Kidney Int. 59(6):2325–2334, 2001.
Roy-Chaudhury, P., B. S. Kelly, J. Zhang, A. Narayana, P. Desai, M. Melhem, H. Duncan, and S. C. Heffelfinger. Hemodialysis vascular access dysfunction: From pathophysiology to novel therapies. Blood Purification 21(1):99–110, 2003.
Roy-Chaudhury, P., and T. Lee. Vascular stenosis: biology and interventions. Curr. Opin. Nephrol. Hypertens. 16(6):516–522, 2007.
Roy-Chaudhury, P., V. P. Sukhatme, and A. K. Cheung. Hemodialysis vascular access dysfunction: A cellular and molecular viewpoint. J. Am. Soc. Nephrol. 17(4):1112–1127, 2006.
Roy-Chaudhury, P., Y. Wang, M. Krishnamoorthy, J. Zhang, R. Banerjee, R. Munda, S. Heffelfinger, and L. Arend. Cellular phenotypes in human stenotic lesions from haemodialysis vascular access. Nephrol. Dial. Transplant 24(9):2786–2791, 2009.
Sarmast, M., H. Niroomand-Oscuii, F. Ghalichi, and E. Samiei. Evaluation of the hemodynamics in straight 6-mm and tapered 6- to 8-mm grafts as upper arm hemodialysis vascular access. Med. Biol. Eng. Comput. 52(9):797–811, 2014.
Shtilman, L., E. Levich, S. A. Orszag, R. B. Pelz, and A. Tsinober. On the role of helicity in complex fluid flows. Phys. Lett. 113A:32–37, 1985.
Sigovan, M., V. Rayz, W. Gasper, H. F. Alley, C. D. Owens, and D. Saloner. Vascular remodeling in autogenous arterio-venous fistulas by MRI and CFD. Ann. Biomed. Eng. 41(4):657–668, 2012.
Silva, J. D. A., J. Karam-filho, and C. C. H. Borges. Computational analysis of anastomotic angles by blood flow conditions in side-to-end radio-cephalic fistulae used in hemodialysis. Biomed. Sci. Eng. 8(3):131–141, 2015.
Sivanesan, S., T. V. How, and A. Bakran. Sites of stenosis in AV fistulae for haemodialysis access. Nephrol. Dial. Transplant. 14(1):118–120, 1999.
Steinman, D. A., J. B. Thomas, H. M. Ladak, J. S. Milner, B. K. Rutt, and J. David Spence. Reconstruction of carotid bifurcation hemodynamics and wall thickness using computational fluid dynamics and MRI. Magn. Reson. Med. 47(1):149–159, 2002.
Stracke, S., K. Konner, I. Köstlin, R. Friedl, P. M. Jehle, V. Hombach, F. Keller, and J. Waltenberger. Increased expression of TGF-β1 and IGF-I in inflammatory stenotic lesions of hemodialysis fistulas. Kidney Int. 61(3):1011–1019, 2002.
Sung, K. H., H. Kim, and H. S. Ryou. Numerical study on the effect of separate cannulation method on hemodynamics in an arteriovenous graft. J. Mech. Sci. Technol. 30(2):963–970, 2016.
Van Canneyt, K., U. Morbiducci, S. Eloot, G. De Santis, P. Segers, and P. Verdonck. A computational exploration of helical arterio-venous graft designs. J. Biomech. 46(2):345–353, 2013.
Van Canneyt, K., U. Morbiducci, S. Eloot, G. De Santis, P. Segers, and P. Verdonck. A computational exploration of helical arterio-venous graft designs. J. Biomech. 46(2):345–353, 2013.
Van Canneyt, K., T. Pourchez, S. Eloot, C. Guillame, A. Bonnet, P. Segers, and P. Verdonck. Hemodynamic impact of anastomosis size and angle in side-to-end arteriovenous fistulae: A computer analysis. J. Vasc. Access 11(1):52–58, 2010.
Van Tricht, I., D. De Wachter, J. Tordoir, and P. Verdonck. Hemodynamics and complications encountered with arteriovenous fistulas and grafts as vascular access for hemodialysis: A review. Ann. Biomed. Eng. 33(9):1142–1157, 2005.
Van Tricht, I., D. De Wachter, J. Tordoir, and P. Verdonck. Comparison of the hemodynamics in 6 mm and 4-7 mm hemodialysis grafts by means of CFD. J. Biomech. 39(2):226–236, 2006.
Wentzel, J. J., F. J. H. Gijsen, J. C. H. Schuurbiers, R. Krams, P. W. Serruys, P. J. De Feyter, and C. J. Slager. Geometry guided data averaging enables the interpretation of shear stress related plaque development in human coronary arteries. J. Biomech. 38(7):1551–1555, 2005.
Acknowledgments
The Irish Research Council for Science Engineering and Technology, GOIPG/2014/1450. The European Union’s Seventh Framework Programme for Research, Technological Development and Demonstration under Grant Agreement No. 324487.
Funding
This study was funded by The Irish Research Council for Science Engineering and Technology, under Grant Number GOIPG/2014/1450 as well as by The European Union’s Seventh Framework Programme for research, technological development and demonstration, under Grant Number 324487.
Conflict of Interest
All authors declare that there is no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editors: James E Moore Jr. and Ajit P. Yoganathan oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Cunnane, C.V., Cunnane, E.M. & Walsh, M.T. A Review of the Hemodynamic Factors Believed to Contribute to Vascular Access Dysfunction. Cardiovasc Eng Tech 8, 280–294 (2017). https://doi.org/10.1007/s13239-017-0307-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13239-017-0307-0