Abstract
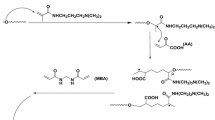
Sodium alginate and poly(acrylamide-co-N-vinylcaprolactam-co-acrylamidoglycolic acid) based dual responsive semi-IPN hydrogels (SA-PAVA) were successfully synthesized by free radical redox polymerization. N, N′-Methylene-bis-acrylamide was used as a crosslinker and 5-fluorouracil, an anti-cancer drug, was loaded onto these semi-IPN hydrogels via equilibrium swelling method. The hydrogels were also used as templates for the production of silver nanoparticles by using NaBH4 as reducing agent. In order to understand the polymer-drug interactions, pristine, as well as drug loaded, SA-PAVA hydrogels were characterized by Fourier transform infrared spectroscopy and differential scanning calorimetry. The formation of silver nanoparticles was confirmed by UV-visible spectroscopy, thermogravimetric analysis (TGA), scanning electron microscopy (SEM) and transmission electron microscopy (TEM). The swelling behavior of the hydrogel was investigated in distilled water under various pH and temperature conditions. In vitro release of 5-fluorouracil from these SA-PAVA hydrogels was carried out in gastro-intestinal fluids different temperatures. The SA-PAVA hydrogel/silver nanocomposites showed excellent anti-bacterial activity towards Escherichia coli and Bacilli.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
D. S. W. Benoit, S. D. Collins, and K. S. Anseth, Adv. Funct. Mater., 17, 2085 (2007).
A. Shikanov, R. M. Smith, M. Xu, T. K. Woodruff, and L. D. Shea, Biomaterials, 32, 2524 (2011).
G. Chen and A. S. Hoffman, Nature, 373, 49 (1995).
T. Gao, W. Wang, and A. Wang, Macromol. Res., 19, 739 (2011).
J. T. Zhang, S. X. Cheng, S. W. Huang, and R. X. Zhuo, Macromol. Rapid Commun., 24, 447 (2003).
D. P. Huynh, C. T. Huynh, and D. S. Lee, Macromol. Res., 18, 589 (2010).
H. Savas and O. Guven, Int. J. Pharm., 224, 151(2001).
K. Joseph and L. Robert, Adv. Drug Deliv. Rev., 46, 125 (2001).
N. Orakdogen, Macromol. Res., 22, 32 (2014).
X. Z. Zhang, D. Q. Wu, and C. C. Chu, Biomaterials, 25, 3793 (2004).
E. Jabbari and S. Nozari, Eur. Polym. J. 36, 2685 (2000).
I. Kaetsu, K. Uchida, H. Shindo, S. Gomi, K. Sutani, Radiat. Phys. Chem., 55, 193 (1999).
K. S. V. Krishna Rao, B. V. K. Naidu, M. C. S. Subha, and T. M. Aminabhavi, Carbohydr. Polym., 66, 333 (2006).
X. Li, W. Wu, J. Wang, and Y. Duan, Carbohydr. Polym., 66, 473 (2006).
B. L. Guo and Q. Y. Gao, Carbohydr. Res., 342, 2416 (2007).
K. M. Reddy, V. R. Babu, K. S. V. K. Rao, M. C. S. Subha, K. C. Rao, M. S. Ram, and T. M. Aminabhavi, J. Appl. Polym. Sci., 107, 2820 (2008).
A. L. Carmen, A. Concheiro, S. D. Alexander, N. V. Grinberg, T. V. Burova, and V. Y. Grinberg, J. Control. Release, 102, 629 (2005).
T. S. Cu, V. D. Cao, C. K. Nguyen, and N. Q. Tran, Macromol. Res., 22, 418 (2014).
K. S. V. K. Rao, P. R. Reddy, Y. I. Lee, and C. Kim, Carbohydr. Polym., 87, 920 (2012).
Y. Kim, V. R. Babu, D. T. Thangadurai, K. S. V. K. Rao, H. Cha, C. Kim, W. Joo, and Y. I. Lee, Bull. Korean Chem. Soc., 32, 553 (2011).
W. R. Gombotz and S. F. Wee, Adv. Drug Deliv. Rev., 31, 267 (1998).
A. Kikuchi, M. Kawabuchi, A. Watanabe, M. Sugihara, Y. Sakurai, and T. Okano, J. Control. Release, 58, 21 (1999).
I. Y. U. Galaev and B. Mattiasson, Enzyme Microb. Technol., 15, 354 (1993).
Y. Liu, L. J. Duan, M. J. Kim, J. H. Kim, and D. J. Chung, Macromol. Res., 22, 240 (2014).
K. S. V. K. Rao, A. B. V. K. Kumar, K. M. Rao, M. C. S. Subha, and Y. I. Lee, Polym. Bull., 61, 81 (2008).
A. C. W. Lau and C. Wu, Macromolecules, 32, 581 (1999).
H. Vihola, A. Laukkanen, L. Valtola, H. Tenhu, and J. Hirvonen, Biomaterials, 26, 3055 (2005).
K. S. V. K. Rao and C. S. Ha, Polym. Bull., 62, 167 (2009).
X. F. Li, X. Q. Feng, S. Yang, T. P. Wang, and Z. X. Su, Iran. Polym. J., 17, 843 (2008).
S. Waxman and K. J. Scanlon, in Clinical Interpretation and Practice of Cancer Chemotherapy, E. M. Greenspan, Ed., Raven press, New York, 1982, p 38.
J. P. Sommadossi, D. A. Gewirtz, R. B. Diasio, C. Aubert, J. P. Cano, and I. D. Goldman, J. Biol. Chem., 257, 8171 (1982).
S. Einmahl, M. Zignani, E. Varesio, J. Heller, J. L. Veuthey, C. Tabatabay, and R. Gurny, Int. J. Pharm., 185, 189 (1999).
N. Aggarwal, H. Hogen Esch, P. Guo, A. North, M. Suckow, and S. K. Mittal, Can. J. Vet. Res., 63, 148 (1999).
O. Garcia, R. M. Trigo, M. D. Blanco, and J. M. Teijon, Biomaterials, 15, 689 (1994).
H. Inomato, S. Goto, and S. Saito, Macromolecules, 23, 4887 (1990).
T. Tokuhiro, T. Amiya, A. Mamada, and T. Tanaka, Macromolecules, 24, 2936 (1991).
R. Yoshida, Y. Okuyama, K. Sakai, T. Okano, and Y. Sakurai, J. Membr. Sci., 89, 267 (1994).
H. E. Sherif, M. E. Masry, and A. Kansoh, Macromol. Res., 19, 1157 (2011).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Reddy, P.R.S., Rao, K.M., Rao, K.S.V.K. et al. Synthesis of alginate based silver nanocomposite hydrogels for biomedical applications. Macromol. Res. 22, 832–842 (2014). https://doi.org/10.1007/s13233-014-2117-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13233-014-2117-7