Abstract
Objectives
To determine the efficacy of tranexamic acid in decreasing blood loss in elective/emergency LSCS.
Materials and Methods
A prospective randomised case control study was done in 200 pregnant women undergoing elective/emergency LSCS in the Department of Obstetrics and Gynaecology, at a tertiary care teaching hospital in Mysuru, from December 2018 to September 2019. Women in the age group of 18–35 years were included in the study. Those with anaemia (Hb < 10 gm%), hypertension in pregnancy, bleeding diathesis, GDM on insulin, polyhydramnios, oligohydramnios, cardiac and chronic liver disorders were excluded from the study. Two hundred women undergoing emergency/elective LSCS were divided into case (group 1) or control (group 2) groups using a computer-generated random number table. Tranexamic acid (10 mg/Kg) was given in 100 ml Normal Saline 10 mins prior to skin incision to women in the first group, along with routine care (10 Units of Oxytocin IM soon after extraction of the baby). Routine care, as per institutional protocol, was followed in the second group. The primary outcome was to estimate the intraoperative blood loss. Blood loss was measured by weighing pads, mops, drapes before and after surgery and blood in the suction container after surgery. Two separate suction catheters and containers were used, in order to minimise mixing of blood and amniotic fluid. Total blood loss was calculated as the difference in the weight of the pads, mops and drapes before and after surgery and the sum of the amount of blood in suction container. The difference between the pre-operative and post-operative haemoglobin and haematocrit was compared. The pre-operative, intra-operative and post-operative hemodynamics were also compared.
Results
Statistical analysis was done using MS Excel and R-3.5.1 software. Unpaired and paired t test were used. In our study, there was a significant decrease in intraoperative bleeding in women receiving tranexamic acid. Women in the control group had a significant fall in the postoperative hemoglobin when compared to women who received tranexamic acid. Also, women who received tranexamic acid did not develop any significant hemodynamic changes during or immediately after the surgery.
Conclusion
Tranexamic acid can be safely used as a prophylactic agent to reduce bleeding during elective and emergency LSCS.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obstetric haemorrhage, especially post-partum haemorrhage, is, perhaps one of the most common causes of maternal mortality and severe morbidity like anaemia, need for blood transfusion, prolonged hospital stay and puerperal sepsis [1]. The average blood loss during LSCS is around 800–1000 ml and in normal delivery, it is around 500 ml. Lower segment Caesarean section is the most common surgery performed in obstetrics. Intraoperative blood loss during LSCS causes significant reduction in haemoglobin postoperatively and the need for blood transfusions or intravenous iron therapy was found to be more during postoperative care. Various uterotonic drugs like oxytocin, methyl ergometrine, prostagladin E1 and Prostaglandin F2 alpha have been successfully used for prophylaxis and management of post-partum haemorrhage during and after caesarean section [2]. Tranexamic acid is a potent inhibitor of fibrinolysis, and was first reported by OKAMOTO. It is an analogue of lysine [1]. It inhibits conversion of plasminogen to plasmin by plasminogen activators and thus prevents lysis of formed clot, by blocking the lysine binding locus of plasminogen. It starts acting rapidly, within 5–15 min and lasts for a maximum of 3 h. Tranexamic acid is widely used in the first line management of heavy menstrual bleeding. Tranexamic acid, administered intravenously, has been used for many years to reduce blood loss during and after surgery, for various procedures such coronary artery bypass, spine surgery, surgeries of the oral cavity, liver transplantation, total hip or knee arthroplasty and procedures of the genito-urinary tract [3]. It has shown to be very useful to reduce the requirement of blood transfusion in these surgeries. Therefore, this study was done, in order to determine whether tranexamic acid administered intravenously, as a prophylactic measure, can decrease blood loss during lower segment caesarean section.
Materials and Methods
A randomised case control study was undertaken in 200 pregnant women undergoing elective or emergency LSCS in the Department of Obstetrics and Gynaecology, at a tertiary teaching hospital in Mysore, from December 2018–September 2019. Sample size was calculated using the formula as {2 × SD2 × (Zα2 + Zβ2)2}/{M2 − M1}2. Reference for sample size calculation was based on a prospective randomised controlled trial, conducted by Dhivya et al., in Coimbatore, India. Power of study was set at 80% and two sided confidence interval at 95%. Taking these into account, the minimum number of women needed to produce statistically significant figure was 58 in each group; hence a total of 120 subjects were chosen for the study. As we were able to operate more number of cases during the time period available, we extended the study to 100 patients in each group. Institutional Ethical Committee approval was taken. The patients were randomised using a computer-generated random number table, as cases (group 1) and controls (group 2), respectively. Women in the age group of 18–35 years were included in the study. Women with hypertension in pregnancy, antepartum haemorrhage, cardiovascular diseases, diabetes on insulin, moderate to severe anaemia, bleeding and clotting disorders, chronic liver disorders, detection of postpartum haemorrhage on table, polyhydramnios and oligohydramnios were excluded from the study. A detailed history was taken from the subjects and routine physical examination was done. Informed written consent was taken from eligible patients before enrolling them in the study. Tranexamic acid 10 mg/kg was given in 100 ml Normal saline 10 mins prior to skin incision, in the first group, along with the usual uterotonics, that is 10 units of Oxytocin intramuscular, immediately after extraction of the fetus. The women in the second group were administered 10 units of Oxytocin intramuscular, soon after delivery of the baby, as per institutional protocol. We used two separate suction catheters and two suction containers, in order to minimize mixing of blood and amniotic fluid during surgery. The first suction was disconnected immediately after extraction of the baby. The amount of blood collected in the second suction container was measured. Although some amount of mixing of amniotic fluid did occur, an attempt was made to reduce it as much as possible. The amount of blood loss was measured using the Gravimetric method. All the mops and drapes used during the surgery, as well as the perineal sheet used on the operating table were weighed using an electronic weighing scale, prior to, and after the surgery. The sum of blood absorbed by the soaked mops and drapes (difference in the weight of the mops and drapes) before and after surgery; blood absorbed by the perineal sheet intra operatively, and during toileting of the vagina (wet weight–dry weight) and the volume of blood in the second suction bottle was used to calculate the blood loss. One milligram weight was considered equivalent to one milliliter of blood. The gravimetric method of determining blood loss remains the most practical method, although it gives an approximate estimate of blood loss. Haemoglobin and hematocrit estimation were done 24 h after the surgery for all the patients to compare with the preoperative value. Hemodynamic parameters, such as the pulse rate and blood pressure during pre-operative period, intraoperative and immediate post-operative period were also compared in both groups.
Statistical Analysis
-
1.
The data were analysed using MS Excel and R-3.5.1 software.
-
2.
All the tests of significance were carried out at 5% level of significance.
-
3.
The statistical methods used were:
-
a.
Frequency distribution and percentage
-
b.
Descriptive Statistics (minimum, maximum, mean, standard deviation)
-
c.
Independent sample t-test
-
d.
Paired t-test for comparative variables
-
a.
Results
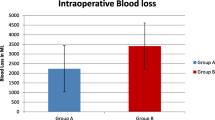
Our patients were in the age group of 18–35 years, with the youngest being 19 years in the case group as well as in the control group, and the oldest was 32 years in the case and 35 years in the control group. Women beyond 34 weeks of gestation were included in the study, and the mean gestational age was 268 days in the case group and 269 days in the control group, respectively. A total of 47% women were Primigravidas, in both the case and control groups, with no significant difference between the two groups. The highest order pregnancy seen was Gravida 4, with previous two lower segment caesarean sections. 10.5% of cases and 22% of controls were operated as elective caesarean, the rest were emergencies. 41.5% of women underwent repeat caesarean in view of previous one caesarean and 2% had undergone previous two caesarean sections. 32% were operated in view of cephalo pelvic disproportion, 11% in view of persistent fetal bradycardia, 5.5% for malpresentations, and others for failed induction, secondary cervical dystocia, contracted pelvis and maternal request. There was no significant difference in the indications for caesarean section between the two groups. The average amount of blood loss was 590 ml in the case group, ranging from 300 to 1280 ml. The control group had an average blood loss of 696.6 ml, and it ranged from 400 to 1160 ml (Fig. 1). The difference in blood was found to be statistically significant, with the case group having significantly less intra operative bleeding as compared to women in the control group. Before applying the parametric tests we have checked for the assumptions-normality, independence and randomness. As sample size is large, we have proceeded with parametric test, as the interpretation of the hypothesis does not vary, whether we apply parametric or nonparametric test. The average difference between haemoglobin in the pre-operative and post-operative period was 0.85 gm% in the cases and 1.16 gm% among the controls, respectively. Average haemoglobin before surgery was 11.5 gm% in case group and 11.98 gm% among the controls. Women in the case group had a mean post-operative haemoglobin of 10.8%, whereas, among the controls it was found to be 11.01gm%. The mean difference in haemoglobin between the cases and controls was found to be statistically significant (Fig. 2; Table 1). We found a similar significant difference in the hematocrit also, between the two groups. There was no significant difference in the pulse rate and blood pressure between the cases and control during the intraoperative and immediate post-operative period, suggesting that tranexamic acid is not associated with significant hemodynamic changes when administered as an infusion.
Discussion
Tranexamic acid acts by competitively blocking the lysine binding site of plasminogen. It thus prevents the lysis of the formed clot. The action of tranexamic acid starts within 5–15 min and lasts for up to 3 h. Tranexamic acid was discovered by Utako Okomoto in 1950s as an outcome of her research during the Sino Japanese war [1]. However, oral TXA was listed as an anti-fibrinolytic agent in the early nineties. Intravenous use of tranexamic acid to reduce blood loss during surgery was approved by the US Food and Drug Administration in 2010. There is sufficient evidence which shows that intraoperative administration of tranexamic acid decreases the need for blood transfusion during various surgeries, especially in trauma and transplant surgery.
Despite the strong evidence favouring intraoperative use of tranexamic acid, a slightly higher risk of thrombo-embolic events has been reported, which may outweigh the benefits of reduced blood use. Recent evidence from the CRASH-2 (Clinical Randomization of an Antifibrinolytic in Significant Haemorrhage) trial of tranexamic acid in bleeding trauma patients showed a statistically significant reduction in mortality with no increase in thromboembolic effects [4]. The CRASH-2 trial was a multicentric, randomized controlled trial, done in predominately low-to moderate income countries proposed that a single dose of 1 g of tranexamic acid in trauma patients with heavy bleeding, should be followed by a 1 g infusion over eight hours. The main outcome of the study was to study the overall mortality rates, which were lower (14.5%) in patients who received tranexamic acid vs the control group (16%). The mortality due to bleeding was also found to be significantly lower (T 4.9% versus P 5.7%) [5].
Shantakumari et al. conducted a randomized controlled trial on a group of 100 women who underwent elective LSCS. One gram of tranexamic acid was administered via the intravenous route to one group, and intraoperative and post-operative blood loss measured [6]. Their findings concur with our results, showing that tranexamic acid significantly decreases blood loss. Hemoglobin and hematocrit measured 24 h after surgery were also significantly higher in the study population, which again is reflected in our study.
Similarly, Abdel-Aleem M. et al. studied 740 women undergoing elective caesarean section and concluded that tranexamic acid given before incision decreases intraoperative and post-operative blood loss [7]. Xu and colleagues conducted a randomized, double-blind, case-controlled study on 174 primigravida undergoing LSCS. One group was given tranexamic acid in the dose of 10 mg/kg prior to incision, and the other was a control group. They determined that tranexamic acid significantly reduces blood loss in the intraoperative and immediate post-operative period [8].
The Military Application of Tranexamic Acid for Trauma Emergency Resuscitation (MATTERs) trial has addressed various other aspects by evaluating patients with combat related injuries, all of whom required surgery, had a specific requirement of an anti-fibrinolytic and at least one unit of packed cell transfusion. The results of this trial established with undoubted certainty, the role of tranexamic acid in decreasing mortality and reducing the need for blood transfusion in patients with penetrating trauma [9].
Based on this evidence, that tranexamic acid significantly improves outcome in trauma patients, the WHO in 2012 recommended the use of tranexamic acid for the prophylaxis and management of post-partum haemorrhage. It was to be used only after three hours of delivery, when uterotonics fail to control bleeding or there is trauma to the genital tract [10].
Tranexamic acid has been used both prophylactically and for management of both atonic as well as traumatic postpartum haemorrhage. Gungorduk et al. prophylactically administered tranexamic acid in 660 women who underwent elective Lower Segment Caesarean Section. According to them, the mean estimated blood loss and the need for additional uterotonics were significantly less as compared to women who did not receive tranexamic acid. They also observed a reduction in transfusion requirements [11]. They used an initial dose of 1 g slow IV bolus followed by another 1 gm four hours later.
Sentilhes et al. have reviewed various randomized controlled trials, which look into the prevention and treatment of PPH with tranexamic acid use. They came to a conclusion that the benefits of tranexamic acid, used prophylactically, in both vaginal and caesarean delivery definitely overscores the side effects [12].
The World Maternal Antifibrinolytic Trial (WOMAN trial) is an International, randomized placebo-controlled trial which studied the effects of intravenous administration of tranexamic acid for the management of post-partum haemorrhage. It showed that early use of IV TXA (within 3 h of delivery of baby and as soon as possible after onset of bleeding) significantly decreases mortality in women with post-partum haemorrhage, irrespective of its cause, and with no adverse maternal effects [13]. The WHO updated its recommendations in 2017, after the results of the WOMAN trial were published. As per the new recommendations, intravenous tranexamic acid is to be administered within three hours after birth and as soon as possible after the onset of post-partum haemorrhage, irrespective of the cause of bleeding [14].
Massimo Franchini et al. conducted a meta-analysis of 18 randomized controlled trials, and they strongly recommend the use of intravenous tranexamic acid for decreasing blood loss and need for blood transfusion in pregnant women undergoing caesarean section [15]. They have not reported any significant adverse effects with the use of tranexamic acid. In our study we found no thrombotic events, nor were there any major incidences of intraoperative hypotension associated with tranexamic acid.
Coming to the methods used to estimate intraoperative blood loss, Khadilkar et al. compared various methods such as gravimetric method, soaked mops and visual estimation. They used plastic drapes, measuring jars, gravimetric method and a training module along with novel clot conversion factor which was designed to validate the visual assessment of blood loss by healthcare professionals. They concluded that visual estimation was unreliable, but periodic training of labour room staff minimised the errors and they recommended that clot conversion factor should be considered useful in quantitative assessment of blood loss [16]. Schorn [17] reviewed the various methods of estimating intrapartum blood loss, and concluded that gravimetric method, along with direct measurement is the most practical method, which is also used in our study.
Conclusion
Tranexamic acid can be used safely and effectively as a prophylactic measure to reduce intraoperative blood loss during elective and emergency caesarean section, with no significant adverse effects.
References
Dhivya SJ, Reena A, et al. Role of tranexamic acid in reducing blood loss during elective LSCS: A randomized controlled study in 2016. J Clin Diagn Res. 2016;10(12):QC17–QC21.
Babita R, Nayak L, et al. Intravenous 1gm tranexamicacid for prevention of blood loss and blood transfusion during caesarean section:a randomized case control study. Int J Reprod Contracept Obstet Gynaecol. 2014;3(2):366–9.
Ker K, Edwards P, et al. Effect of tranexamic acid on surgical bleeding: systematic review and cumulative meta-analysis. BMJ. 2012;344:e3054.
The CRASH-2 Collaborators. Effects of tranexamic acid on death, vascular occlusive events, and blood transfusion in trauma patients with significant hemorrhage (CRASH-2): a randomized, placebo-controlled trial. Lancet. 2010;376:23–322.
Binz S, McCollester J, Thomas S, et al. CRASH-2 study of tranexamic acid to treat bleeding in trauma patients: a controversy fueled by science and social media. J Blood Transfus. 2015;2015:874920. https://doi.org/10.1155/2015/874920.
Sampathkumari S, et al. A study on efficacy of prophylactic tranexamic acid in reducing blood loss during elective Lscs. IOSR J Dent Med Sci. 2018;17(2):59–63.
Abdel-Aleem H, Alhusaini TK, AbdelAleem MA, Menoufy M, Gülmezoglu AM. Effectiveness of tranexamic acid on blood loss in patients undergoing elective cesarean section: Randomized clinical trial. J Matern Fetal Neonatal Med. 2013;26(1):1705–9.
Xu J, Gao W, Ju Y. Tranexamic acid for the prevention of postpartum hemorrhage after cesarean section: a double-blind randomization trial. Arch Gynecol Obstet. 2013;287(3):463–8.
Morrison JJ, Dubose JJ, Rasmussen TE, Midwinter MJ. “Military application of tranexamic acid in trauma emer-gency resuscitation (MATTERs) study. Arch Surg. 2012;147(2):113–9.
World Health Organization (WHO). WHO Recommendations for the Prevention and Treatment of Postpartum Hemorrhage. Geneva: WHO; 2012.
Gungorduk K, Yildirim G, Asicioglu O, Ark C. Efficacy of intravenous Tranexamic acid in reducing blood loss after elective cesarean section: a prospective randomized double blind placebo controlled study. Am J Prenatal. 2011;28(3):233–40.
Sentilhes L, Daniel V, Darsonval A, Deruelle P, Vardon D, Perrotin F, et al. Study protocol: TRAAP-TRAnexamic Acid for Preventing postpartum haemorrhage after vaginal delivery: a multicenter randomized, double-blind, placebo-controlled trial. BMC Preg Childbirth. 2015;15(1):1.
WOMAN Trial Collaborators. Effect of early tranexamic acid administration on mortality, hysterectomy, and other morbidities in women with post-partum hemorrhage(WOMAN): an international, randomized, double-blind, placebo-controlled trial. Lancet. 2017;389(10084):2105–16.
Updated WHO recommendation on Tranexamic acid for the treatment of Postpartum Hemorrhage. Highlights and Key Messages from the World Health Organization’s 2017 Global Recommendations. 2017.
Massimo F, Carlo M, et al. Safety and efficacy of tranexamic acid for prevention of obstetric hemorrhage: An updated systemic review and meta analysis. Blood Trans. 2018;16(4):329–37.
Khadilkar SS, Sood A, Ahire P. Quantification of Peri-partum Blood Loss: Training Module and Clot Conversion Factor. J Obstet Gynecol India. 2016;66:307–14.
Schorn MN. Measurement of blood loss: review of the literature. J Midwifery Womens Health. 2010;55(1):20–7.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study was approved by the Institutional ethical committee, vide letter no. JS/MC/PG/6227/2018-19, dated 02/02/2019. Our study involves human participants. It has been approved by the institutional ethical committee and has been performed in accordance with the ethical standards as laid down in the 1964 declaration of Helsinki and its later amendments or comparable ethical standards.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
L. Hemapriya is a Associate Professor, Department of Obstetrics and Gynaecology, JSS Medical College and Hospital, JSSAHER, Mysore, India, Greeshma More is a Junior resident, Department of Obstetrics and Gynaecology, JSS Medical College and Hospital, JSSAHER, Mysore, India. Anil Kumar is a Associate Professor, Department of Anaesthesiology, JSS Medical College and Hospital, JSSAHER, Mysore, India.
Rights and permissions
About this article
Cite this article
Hemapriya, L., More, G. & Kumar, A. Efficacy of Tranexamic Acid in Reducing Blood Loss in Lower Segment Cesearean Section: A Randomised Controlled Study. J Obstet Gynecol India 70, 479–484 (2020). https://doi.org/10.1007/s13224-020-01351-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13224-020-01351-3