Abstract
Purpose
Our aim was to identify couples at risk of having a homozygous or compound heterozygous child with a severe hemoglobinopathy by antenatal screening and prenatal diagnosis in Surat, South Gujarat.
Method
Pregnant women were screened for hemoglobinopathies by means of red cell indices, the solubility test, cellulose acetate electrophoresis tests, and confirmation by HPLC. Husbands of the pregnant women having hemoglobinopathies were counseled and screened for hemoglobinopathies. The couples at risk were again counseled and referred to the National Institute of Immunohematology, where mutations in parents and fetuses were identified by molecular analysis. After prenatal diagnosis, the continuing pregnancies were followed up and infants were tested at birth.
Results
Out of 3,009 women, 37.04, 52.6, and 10.3 % were in the first, second, and third trimester of pregnancy, respectively. Among those having hemoglobinopathies, 102 (3.38 %) had the β-thalassemia trait, 46 (1.5 %) the Sickle cell trait, and 26 (0.86) had hemoglobin variants like Hb DPunjab, Hb E, Hb DIran, Hb QIndia, Hb JParis-I, and Hb OIndonesia. Out of the 14 couples at risk of having an affected child, 11 (78.5 %) couples opted for prenatal diagnosis. Three fetuses had homozygous β-thalassemia and hence the pregnancies were terminated. Follow up of normal or heterozygous fetuses confirmed the diagnosis.
Conclusion
During antenatal screening, we found many Hb variants of β and α globin chains. Late antenatal registration, non-cooperation of the husband for investigation, and refusal for prenatal diagnosis are the main hurdles in the hemoglobinopathy prevention program and awareness is necessary.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hemoglobinopathies, the inherited disorders of hemoglobin (Hb) are divided into three groups: structural hemoglobin variants, the thalassemias characterized by the reduced rate of synthesis of one or more globin chains, and the condition in which fetal hemoglobin synthesis persists beyond the neonatal period, collectively known as hereditary persistence of fetal hemoglobin [1].
Because of social, cultural, and religious taboos, as well as ignorance about the disease and family influences, control of thalassemia is a major problem in India. The approaches for prevention include mass screening for carriers, premarital counseling, antenatal screening, and prenatal diagnosis. In India, many studies have shown that antenatal screening followed by prenatal diagnosis is the most feasible for the prevention of birth of homozygous children [2, 3].
In this study, we aimed to detect couples at risk and offer them genetic counseling and the option of prenatal diagnosis by antenatal screening for hemoglobinopathies in Surat city. This has also enabled us to know the prevalence of hemoglobinopathies in the population in Surat.
Materials and Methods
The study was approved by the Institutional ethics committee. We obtained permission from leading gynecologists and collected pregnant women’s samples from different maternity hospitals of Surat city for antenatal screening of hemoglobinopathies. After taking informed consent in the local language, two ml blood was collected in EDTA. All the samples were tested for red blood cell indices [4] on an automated cell counter MEK-5216K (NIHON KOHDEN, Japan), solubility test [5], and cellulose acetate electrophoresis at pH 8.9 [4]. The samples showing Hb A2 >3.5 % were diagnosed as β-thalassemia trait (BTT). The doubtful cases (Hb A2 3.5–3.9 %), all heterozygous and structural Hb variants, were confirmed by high performance liquid chromatography (HPLC) on a Hb variant testing system (BioRad Laboratories, Hercules, USA) [6]. Husbands of pregnant women having hemoglobinopathies were called for counseling and screened for hemoglobinopathies. The couples at risk for hemoglobinopathies were again counseled for prenatal diagnosis and referred to the National Institute of Immunohaematology (NIIH), Mumbai. Chorionic villus sampling (CVS) was performed by the gynecologist transabdominally under the ultrasound guidance between 9.5 and 13 weeks gestation and fetal blood collection between 18 and 21 weeks gestation. The villus tissue was cleaned under a dissecting microscope (Nikon, Tokyo, Japan) to remove any maternal residual contamination.
The fetal DNA from CVS or the fetal blood sample was extracted using the Qiagen Mini Blood Kit and was collected in 50 μL of the AE buffer provided. Maternal contamination in the fetal cells was checked by the nested PCR method for a variable number of tandem repeats (VNTR).
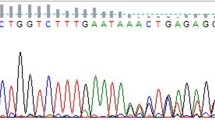
The mutation was detected by a stepwise approach. In the first step, six common Indian mutations [IVS 1 nt 5 (G → C), IVS 1 nt 1 (G → T), Cd 8/9 (+G), Cd 41/42 (−CTTT), Cd 15 (G → A), and Cd 30 (G → C)] causing β-thalassemia along with Hb S and Hb E were screened by covalent reverse dot blot hybridization using China 1 and PCO 6 (Sigma Genosys, Bangalore, India) and a hybridization step [7]. If no mutations were detected in these samples, then they were analyzed for the 7th common mutation (619 bp deletion) and 28 other known, but less common, mutations reported among Indians by means of the amplification refractory mutation system (ARMS) [8]. The uncharacterized samples were directly sequenced on the ABI PRISM™ BIO automated DNA sequencer using the Big Dye Terminator Kit (Applied Biosystems, USA).
After prenatal diagnosis, the continuing pregnancies were followed up and the infants rechecked at birth or at 6 months of age for confirmation of the diagnosis.
Results
Out of 3,009 women, 37.04, 52.6, and 10.3 % were in the first, second, and third trimester of pregnancy, respectively.
The pregnant women were mainly from the Hindu, Jain, and Muslim communities. The prevalence of various hemoglobinopathies in them is presented in Table 1. Among those having hemoglobinopathies, 102 (3.38 %) had the β-thalassemia trait (BTT), 46 (1.5 %) the sickle cell trait (SCT), and 26 (0.86) had other hemoglobin variants including 11 (0.36 %) cases of Hb DPunjab trait, Hb E trait 7 (0.23 %), Hb DIran trait 2, Hb QIndia triat 2, Hb JParis-I 3, and one case of Hb OIndonesia trait.
Hb D Family Variants
A total of 11 cases (0.36 %) had Hb DPunjab trait with normal or reduced MCV (Mean ± SD = 72.8 ± 10.1) and MCH (Mean ± SD = 22.5 ± 5.0) among the antenatal women. Further analysis suggested that among them, two women were Rajput from Surat and Madhya Pradesh, two were Shaikh, and one each of the Koli, Leva, Vasava, Marwadi, Jain, and Suthar communities. The cellulose acetate electrophoresis pattern at alkaline pH showed a band at the Hb S position, and the HPLC histogram showed a peak in the D-window (Mean ± SD = 32.1 ± 4.3 %) (R.T. 4.12–4.14 min) which confirms Hb DPunjab heterozygosity.
Hb D-Iran heterozygous was found in two cases belonging to the Muslim community from Rajasthan. The MCV (84.2, 73.7 fL) and MCH (24.6, 21.2 pg) were reduced or normal. On electrophoresis, they showed a band in the Hb S position and on HPLC, high Hb A2 (41.8, 42 %) (R.T. 3.64 min) which confirmed Hb DIran.
Hb E
Seven cases (0.36 %) of Hb E trait were found with normal or reduced MCV (Mean ± SD = 77.9 ± 7.0), MCH (Mean ± SD = 23.5 ± 3.0). They were Maratha (2), Bengali (2), Kshtriya from Orissa (1), Kadva Patel (1), and Chaudhary (1) from Surat. The cellulose acetate electrophoresis pattern at alkaline pH showed a band at the Hb A2 position and the HPLC histogram showed a peak in Hb A2 window (Mean ± SD = 28.2 ± 1.8) (R.T. 3.65 min).
Hb QIndia
Two cases showing normal red cell indices were identified as Hb Q-India during screening. On cellulose acetate electrophoresis, they showed a band in Hb S position and on HPLC, an unknown peak (17.7 and 16.5 %) at R.T. 4.77 min. One woman was Punjabi and another Sindhi.
Hb OIndonesia
This woman was of the Marwadi community from Rajasthan with reduced MCV (68.6 fL) and MCH (18.2 pg). Cellulose acetate electrophoresis revealed an abnormal band between Hb A2 and Hb S, and HPLC analysis showed an abnormal peak in the C window (11.2 % R.T. 4.92). α-gene sequencing (done at NIIH, Mumbai) showed a heterozygous G → A substitution at codon 116 (glutamine to lysine) of the alpha 1 globin gene corresponding to Hb O Indonesia.
Hb J-Paris I
Three cases showing a fast-moving band on electrophoresis were further run on HPLC and showed an abnormal peak (P3 = 27.8 % at R.T. 1.73 min). They were of the Muslim community (Shaikh, Soni) from Surat. The red cell indices were normal with reduced MCH. α-gene sequencing showed heterozygous C → A substitution at codon 12 (alanine to aspartate) of the alpha 2 globin gene corresponding to Hb J Paris—I.
The husbands of 148 pregnant women having BTT or SCT were requested to visit our laboratory for investigation and 125 (84.4 %) were investigated for hemoglobinopathies. Among them, 14 were found to have BTT. Table 2 gives the data of mutations identified in them.
These couples were again counseled for prenatal diagnosis, but three couples were not convinced. Out of 11 couples who agreed for prenatal diagnosis, three had thal major (Homozygous) fetuses, and hence they opted for medical termination of pregnancy (MTP). Pregnancies with normal or BTT fetuses were continued. We investigated the infants 6 months after the birth. All were found to be thalassemia minor or normal, and hence the findings correlated with the prenatal diagnosis results.
Discussion
The approaches to prevent the birth of a thalassemia major child include carrier screening, premarital counseling, and prenatal diagnosis. Different counties have had varied experiences and no single strategy can meet the needs of every population. Premarital screening for β-thalassemia is not widely acceptable in India because of social and cultural taboos. Antenatal screening is the important step to identify women having the risk of producing a child affected with hemoglobinopathy. In India, many studies [2, 3] have reported the success of antenatal screening followed by prenatal diagnosis. Gujarat has many high-risk communities [9] which need antenatal screening and prenatal diagnosis for hemoglobinopathies.
The carrier rate of β-thalassemia varies from 1 to 17 % in India with an average of 3–4 % [10]. The overall prevalence of BTT was 3.38 % among antenatal women. Bhatia and Rao [11] have reported the prevalence of SCT in tribal populations in South Gujarat, which ranges from 0 to 31.4 %. As a majority of these tribes are in the Valsad and Dang regions, antenatal women from Surat showed only a 1.5 % SCT prevalence.
Hb D-Punjab also known as Hb D-Los Angeles is seen mainly in North-western India, but sporadic cases have been reported from Gujarat [9]. This study reports 11 cases of Hb D-Punjab trait mainly from Gujarat, except two cases which were from Rajasthan and Madhya Pradesh. This study also reports two unrelated Muslim cases, both from Rajasthan having Hb DIran trait.
Hb E is widely distributed in the North-eastern states of India [9]. This study reports Hb E trait in seven individuals. They belonged to the Maratha community from Maharashtra, Bengalis, Odia from Orissa, Kadva Patel, and Chaudhary from Surat.
Hb O-Indonesia is also known as Hb Buginese-X. It was reported in India in a Muslim from Maharashtra by Nair et al. [12]. We detected it in a Marwadi woman from Rajasthan.
Hb J Paris-I also known as Hb J-Alijezur was first described in a woman of Spanish origin living in Paris. Subsequently, it was observed in Portugal, Iran, Yugoslavia, and Italy. In India, it was first observed in a North Indian of Punjabi Hindu origin [13]. In this study, we found three Muslim cases (Shaikh, Soni) from Surat having Hb JParis I trait.
The first case of Hb Q-India was from India, reported by Sukumaran et al. in 1972 [14] in a Sindhi family with associated β-thalassemia. Phanasgaonkar et al. [15] reported 64 cases, a majority of them were originally from Sindh in Pakistan and the remaining were North Indians. We found two cases from the Sindhi and Punjabi communities.
In our study, the majority of the women had registered in the antenatal clinic after completion of the first trimester, and 10.3 % of them had registered after completion of the second trimester when MTP is not possible. Thus, the main hurdle for prenatal diagnosis is the late visit to the antenatal clinic. Out of 148 husbands of BTT/SCT cases, 23 (15.6 %) refused to be investigated, which shows a lack of awareness to understand the importance of screening for hemoglobinopathy. Three out of 14 couples requiring prenatal diagnosis refused to give consent for prenatal diagnosis. The other obstacles were late registration and non-cooperation of the family for further investigation.
Conclusion
During antenatal screening, we found many Hb variants of β and α globin chains in Surat city. Late antenatal registration, non-cooperation of the husband for investigation, and refusal for prenatal diagnosis are the main hurdles in the hemoglobinopathy prevention program, and awareness is necessary.
References
Weatherall DJ, Clegg JB. The thalassemia syndromes. 4th ed. Oxford: Blackwell Scientific Publication; 2001.
Colah RB, Gorakshakar A, Surve R, et al. Feasibility of antenatal screening of β-thalassemia in Mumbai, India. Acta Heamatol. 2001;105:252.
Colah R, Surve R, Wadia M, et al. Carrier screening for β-thalassemia during pregnancy in India: a 7 year evaluation. Genet Test. 2008;12:181–6.
Wild BJ, Bain BJ. Investigation of abnormal haemoglobins and thalassemia. In: Lewis SM, Bain BJ, Bates I, editors. Dacie and Lewis practical hematology. 9th ed. Edinburgh: Churchill Livingstone; 2001. p. 231–68.
Huntsman RG, Barclay GPT, Canning DM, et al. A rapid whole blood solubility test to differentiate the sickle cell trait from sickle cell anemia. J Clin Path. 1970;23:781–3.
Joutovsky A, Hazdi-Nesic J, Nardi MA. Retention time as a diagnostic tool for hemoglobin variants and hemoglobinopathies: a study of 60,000 samples in a clinical diagnostic laboratory. Clin Chem. 2004;50:1736.
Colah RB, Gorakshakar AC, Lu CY, et al. Application of covalent reverse dot blot hybridization for rapid prenatal diagnosis of the common Indian thalassemia syndromes. Ind J Hematol Blood Transf. 1997;15:10–3.
Varawalla NY, Old JM, Sarkar R, et al. The spectrum of β-thalassemia mutations on the Indian subcontinent: the basis for prenatal diagnosis. Br J Hematol. 1991;78:242–7.
Sharma A. Haemoglobinopathies in India. In: Peoples of India: some genetical aspects. New Delhi: ICMR; 1983. p. 31–49.
Modell B, Petrou M. The problem of the hemoglobinopathies in India. Ind J Hematol. 1983;1:5–16.
Bhatia HM, Rao VR. Genetic atlas of Indian tribes. Mumbai: Institute of Immunohaematology (ICMR); 1987.
Nair S, Nadkarni A, Warang P, et al. Five α-globin chain variants identified during screening for hemoglobinopathies. European J Clin Invest. 2010;40:226–32.
Dash S, Huisman TH. First observation of hemoglobin J-Paris I (alpha2-12 (A10 alanine-aspartic acid beta -2) in the Indian subcontinent. Acta Haematol. 1988;79:117.
Sukumaran PK, Merchant SM, Desai MP. Hemoglobin Q India (alpha 64(E13) aspartic acid → histidine) associated with beta-thalassemia observed in three Sindhi families. J Med Genet. 1990;9:436–9.
Phanasgaonkar S, Colah R, Ghosh K, et al. Hb Q-India and its interaction with β-thalassemia: a study of 64 cases from India. Br J Biomed Sci. 2007;64:160–3.
Acknowledgments
We thank the Indian Council of Medical Research for financial support and the National Institute of Immunohaematology, Mumbai for providing training to set up the facility in Surat city. We thank the doctors of the different maternity hospitals of Surat for sending antenatal samples to our centre and Dr. Sona Nair from NIIH, Mumbai for alpha gene sequencing.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bhukhanvala, D.S., Sorathiya, S.M., Sawant, P. et al. Antenatal Screening for Identification of Couples for Prenatal Diagnosis of Severe Hemoglobinopathies in Surat, South Gujarat. J Obstet Gynecol India 63, 123–127 (2013). https://doi.org/10.1007/s13224-012-0271-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13224-012-0271-4



