Abstract
The use of natural polymers, such as gelatin and other proteins, has increased in an attempt to replace part of the consumption of petroleum-based packaging. This study evaluated the influence of green tea extract and lemon nanoemulsion on mechanical, thermal and permeability properties of gelatin matrix. The results showed that green tea increased the gelatin tensile strength (TS) from 86 ± 7 MPa to 101 ± 5 MPa, on the other hand, the nanoemulsion decreases to 78 ± 8 MPa. The incorporation of green tea and nanoemulsion enhanced the water vapor permeability of gelatin film; this could be due to the interacting with the hydrophobic domains of gelatin. This was indicated by melting point (Tm) in differential scanning calorimetry (DSC) and degradation temperatures in the thermogravimetric analysis (TG), respectively. Changes in FTIR spectra of gelatin film were observed when the green tea and nanoemulsion were incorporated. Therefore, this study showed a new characterization and formulation of gelatin films incorporated by green tea extract and lemon nanoemulsion and their potential for edible film.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Fossil derivatives are the main source of polymers to produce packaging that are known, however they take a long time to degrade, leading serious waste accumulation problems (Liu et al. 2017). In this context, the interest for biodegradable natural polymers, such as some proteins, have been increased to replace part of petroleum-based packaging (Hong et al. 2009; Otoni et al. 2014).
Gelatin (G) is a biodegradable water-soluble polymer and mainly based on its gel/film-forming properties is used in the pharmaceutical industry, food and cosmetics (Vanin et al. 2014; Cozmuta et al. 2015; Steyaert et al. 2016). The gelatin extraction is obtained through the controlled chemical hydrolysis of the insoluble fibrous collagen from animal skin and bones during the slaughter (Ahmad et al. 2012; Chen et al. 2009). Gelatin has a triple helix structure formed by seven different amino acids and a denaturation temperature around 40 °C (Alparslan et al. 2016). The resistance, solubility and viscosity of gelatin are favorable film-forming properties, which has been instigating several studies (Nur Hanani et al. 2014).
The latest research in packaging includes the incorporation of natural active substances in polymer matrices (Alparslan et al. 2016; Kumari et al. 2017). In this context, components that have antimicrobial and antioxidant properties, such as essential oils and green tea extract, have been used in certain packages in order to improve stability to prolong the shelf life of certain foods (Muriel-Galet et al. 2015).
Green tea (Camellia sinensis)(GT) is considered to be a product with antioxidative and antimicrobial properties (Nunes et al. 2018). In addition, green tea has a complex chemical composition and the most important bioactive constituents include polyphenols and flavonoids, such as catechins and alkaloids (Wu et al. 2013). The European Union (EU) classifies the green tea extracts as a food additive and due to the advantageous properties, as improvement of mechanical properties, it has been incorporated in food packages (Giménez et al. 2013; Wrona et al. 2017).
In this study it lemon essential oil nanoemulsion (LN) was used due to its attractive organoleptic properties and for improvement in antimicrobial and antioxidant properties of the films. The Citrus lemon is used to extract this oil and its major components are the valencene, limonene and ocimene (Muriel-Galet et al. 2015). Therefore, the purpose of this research was to apply green tea extract in gelatin polymer matrix films incorporated with lemon nanoemulsion to study and characterize the effects of these compounds on the film forming capacity.
Experimental
Materials
Colorless gelatin powder (Royal®), Sorbitan monooleate, commercially known as Tween 80 was procured from Synth (Diadema, SP, Brazil). Green tea extract and lemon essential oil was purchased from Ferquima (Vargem Grande Paulista—SP).
Emulsion preparation
The emulsions were obtained by adding 1% (w/v) lemon essential oil and 0.75% (m/v) Tween 80 to distilled water, followed by mixing in a homogenizer (T25 ULTRA-TURRAX IKA® Werke GmbH & Co., Staufen, Germany) at 15,000 rpm/min for 5 min. Suspensions were visually evaluated for opalescence, homogeneity, presence or absence of sediments and phase separation after resting of the solution.
Film preparation
Films were prepared by hydrating 5 g of gelatin in 100 mL of water at 20 °C to 25 °C and produced by casting technique. Subsequently, the mixture was heated on a magnetic stirrer until it reached 60 °C. To the second sample of film, the gelatin was hydrated directly in 100 mL of water with green tea extract (5% m/v). Then, 50 mL of each film-forming solution were spread on a rectangular support in the dimensions of 15 cm × 20 cm and dried at room temperature (25 °C to 30 °C) for 24 h.
Characterization
Zeta potential and droplet size
The zeta potential (ζ) and average particle size of emulsion droplets were determined in a Zetasizer Nano Series (Malvern Instruments Inc., Worcestershire, U.K.) by dynamic light scattering.
Film thickness determination
A digital micrometer (No. 7326, Mitutoyo Corp., Kanogawa, Japan) was used to measure the film thickness at five random positions along the samples. The mean values were used to calculate mechanical properties of the films.
Water vapor permeability (WVP)
The permeability to water vapor was measured by a gravimetric method to determine the relative humidity (RH) at the film underside, using the modified version of ASTM E96-80 (ASTM 1980), described by McHugh et al. (McHugh et al. 1993). Samples exempt of physical defects, such as bubbles, cracks or pinholes, were shaped into circles and fixed in poly (methyl methacrylate) flat-bottom cups (50.8 mm—dia.), filled with 6 mL of distillated water to increase the RH environment inside the cups. Another plate was placed over the film and fixed with four symmetrical screws. Then, the plates were maintained at 25 °C in an oven containing silica gel to control the inner relative humidity. The amount of water permeated through the films was determined by periodic weighing of the cells using an analytical balance (± 0.0001 g) for one day. Five replications were used to calculate WVP in g mm/kPa h m2 according to McHugh et al. 1993 (McHugh et al. 1993).
Water solubility of the films
Solubility in water was determinate according to the method modified by Gontard et al. (1994). Firstly, three disks (2 cm in diameter) of each sample were cut and dried in an oven at 100 °C for 24 h and weighed in analytical balance to determine the initial percentage of dry matter. This was considered as the initial weight (Wi). The samples were immersed in 50 mL of water and agitated slowly and periodically at 25 ± 2 °C for 24 h. Then, the samples were placed in an oven at 100° C for 24 h for the gravimetric determination of water content of the films. This was taken as the final weight (Wf). The solubility percentage (S%) was determined by using the formula: S (%) = [(Wi−Wf)/Wi] × 100.
Mechanical properties
The elongation at break (EB), tensile strength (TS) and elastic modulus (EM) were determined according to ASTM method D882-97 (ASTM, 1997) using an Instron Universal Testing (Model 3369, Instron Corp., Canton, Mass., USA). The films were equilibrated at 33 ± 2% relative humidity (RH) for 48 h before test. At least ten samples of each film in the dimensions of 100 mm length × 13 mm width were cut and analyzed.
Thermogravimetric analysis (TG)
Samples between 6 and 10 mg were submitted to a thermal analysis TA Instruments TGA Q-500 with temperature range of 0 to 700.00 °C and heating speed of 10 °C/min under a nitrogen flow rate of 40 mL/min.
Differential scanning calorimetry (DSC)
The thermal properties of films were investigated through a TA Instruments DSC Q100 device, Standard Cell FC. Weighed film samples (6 to 8 mg) were placed in a closed aluminum steel pan and DSC was performed over the temperature range (−50 °C to 300 °C) at heating rate of 10 °C/min, and nitrogen flow rate of 50 mL/min to detect glass transition temperature (Tg) and the maximum of the melting temperature peak (Tm).
Fourier transformation infrared spectroscopy (FTIR)
Films were macerated with potassium bromide (Br) powder and then pressed into pastilles. A spectrophotometer (Nicolet—NEXUS 670 FT-IR) was used to obtain the infrared spectra, with 128 scans in the frequency range of 400 to 4000 cm−1 with 2 cm−1 resolution.
Results and discussion
Thickness of films
The thickness of gelatin films incorporated with GTE is shown in Table 1. The films did not differ significantly from each other.
Hoque et al. (2011a) and Wu et al. (2013) also observed that thickness of gelatin film incorporated with different herb extract was similar to the samples. The Fig. 1 shows all the types of films prepared.
Zeta potential and droplet size
A nanoemulsion (oil-in-water, O /W) consists in oil droplets of mean size ranging from 20 to 200 nm dispersed in aqueous medium by a surfactant (Donsí and Ferrari 2016). The obtained nanoemulsion had a mean particle size of 193.4 ± 0.5 nm and it was visually opalescent, without phase separation or precipitation after preparation. Acevedo-Fani et al. (2015) presented similar results with essential-oil-loaded nanoemulsion in sodium alginate film. The zeta potential of the nanoemulsion was approximately −10 mV. Generally, the electric charge of the oil droplets is directed by the charge of adsorbed surfactants around them, which may be cationic, anionic or non-ionic. In this study, Tween 80 (a non-ionic surfactant) was used, which lead to an expected electric charge near zero. When the value is high enough the droplets are prevented from aggregating due to electrostatic repulsion between them.
Water vapor permeability (WVP)
Water vapor permeability ensues due to the difference of relative humidity (RH) inside flat-bottom cups. This analysis measures the capacity of water vapor to diffuse from a larger region of higher water vapor pressure and penetrate into a region with lower water vapor pressure environment. As shown in the Table 2 the WVP increased from 0.15 to 0.25 g mm/kPa h m2 to the incorporation of green tea extract and lemon nanoemulsion in gelatin matrix.
Atarés et al. (2010) reported that the WVP value is dependent of the microstructural characteristics of film related with the formation of a heterogeneous surface and increasing porosity with the addition of essential oil in the gelatin matrix. Gelatin is a hydrophilic material and the addition of these components changed the water vapor transmission through films. LN and GT can be interacting with the hydrophobic domains of gelatin and this phenomenon hinders interactions between the peptide chains and water molecules, which increases the volume of the system, favoring the mobility of the matrix chains and the passage of water vapor molecules. This is a result that can be favorable depending of its application, once it is in accordance with the results reported by Altiok et al. (2010) and Kavoosi et al. (2014).
Solubility determination of films
The Table 2 summarized the water solubility percentages (weight loss) of the gelatin films. The gelatin films presented a solubility percentage of 39.31 ± 0.04%, but with the incorporation of the green tea extract and the lemon nanoemulsion, there was an increase in the solubilities of the films to 63.57 ± 0.01%. This result corroborates with what was observed in the water vapor permeability test, in which the addition of these components increased the water vapor passage, since the hydrophobic characteristics of the oil and certain components of the tea extract favored its interactions with the hydrophobic domains of the gelatin.
As a similar result, Gómez-Estaca et al. (2010) reported that the solubility of bovine gelatin films increased from 26.2 ± 1.5% to 41.1 ± 1.5% when clove essential oil was inserted. Kavoosi et al. (2014) incorporated essential oil of Zataria multiflora into gelatin film also observed increased solubility and permeability to water vapor of the material. The increased solubility of the gelatin film with the addition of the nanoemulsion may be occurring due to the interactions between the hydrophobic domains of the gelatin and the essential oil.
Mechanical Properties
The elongation at break (EB) and tensile strength (TS) are associated with certain mechanical properties and chemical structures of materials. The EB is related to film stretching capacity and the TS indicates the resistance to tensile forces (Otoni et al. 2014; Rojas-Graü et al. 2007). The lemon nanoemulsion incorporated into the gelatin film caused an increase in EB and decreased TS, the values are shown in the Table 1.
This behavior can be attributed to the lower interaction between the gelatin chains caused by lemon oil, resulting in a discontinuity and decreased cohesion. Tongnuanchan et al. (2015) and Kavoosi et al. (2014) also reported that gelatin films incorporated with essential oil decreased tensile strength, however increased elongation at break in comparison to the gelatin film without oil. Mechanical properties of gelatin films incorporated with green tea extract are also depicted in the Table 1. When the green tea extract was added into gelatin film, the TS increased from 86 ± 7 to 101 ± 5 MPa and the EB reduced from 4 ± 1 to 2.82 ± 0.3%. Likewise, GTE added on GLN increased TS to 98 ± 8 and the EB reduced to 3.5 ± 0.4. It is known that the green tea has many groups of polyphenolic compounds and these hydroxyl groups could be able to interact with the hydrogen acceptors of gelatin molecule by the hydrogen bonds and hydrophobic interaction (Gómez-Guillén et al. 2009). Similar results were found by Wu et al. (2013). with the incorporation of green tea extract in silver carp skin gelatin.
Thermogravimetric analysis (TG)
The thermal degradation behavior of the films was analyzed using the TG tests. As shown in the Table 3, the first degradation temperature (Td1) of all the films occurred between 23 °C and 41.79 °C.
This weight loss at this range temperature is associated with the loss of bond and free water absorbed in the films. Similar results were reported by Tongnuanchan et al. (2015) when fish skin gelatin was added in palm oil and Hoque et al. (2011b) in cuttlefish skin gelatin films. The second stage of degradation temperature (Td2) appeared approximately between 172.62 and 192.57 °C. Gelatin film showed higher temperature and the incorporation of green tea extract and lemon essential oil decreased this initial stage. According to Hoque et al. (2011a), this may be due to decomposition of protein fractions of lower molecular weight. In the third stage (Td3), 377.87–453.16 °C, films added by lemon nanoemulsion presented minor degradation temperature, while the green tea extract addition increased this initial temperature. These results revealed that films incorporated with LN presented lower Td2 and Td3 than gelatin films without LN. The addition of LN decreased the heat resistance, lowering the intra/intermolecular protein interaction of the gelatin matrix. Similar results were found by Tongnuanchan et al. (2015) with a lower heat resistance of fish skin gelatin film added with palm oil. On the other hand, gelatin films with GTE showed higher heat resistance than the gelatin film pure. These results suggest, there may be a greater bonding between gelatin and phenolic compounds of green tea extract, resulting in an increase of thermal stability in these type of film (Hoque et al. 2011a).
Differential scanning calorimetry (DSC)
Differential scanning calorimetry can be used to characterize thermal properties of food proteins (Guerrero et al. 2011). In this study DSC, it was used to investigate thermal properties of gelatin films with and without lemon nanoemulsion and green tea extract (Table 4).
DSC thermograms of the gelatin pure films showed two sharp endothermic peaks at 104.50 °C and 225.03 °C. After extract addition, the values decreased to 99.26 °C and 217.45 °C. A peak around 100 °C meant the evaporation of free water and the unstable tea extracts components. The minor melting temperature (Tm) after the green tea incorporation was suggested by Peng et al. (2013), were in decrement of intermolecular force and it could be associated between the polymeric chains and depolymerization of tea extracts in the gelatin/tea extracts composite films. The change in enthalpy values obtained in the second peak were 7.22 J/g (pure gelatin) and 12.78 J/g (gelatin and green tea). The heat absorbed (ΔH) by green tea pure gelatin was larger than pure gelatin films and these higher ΔH values may be attributed to the increase of gelatin-green tea hydrogen bonds, which could reduce polymer chain movement and induce crystalline growth (Peng et al. 2013; Sahraee et al. 2017).
The green tea powder is a complex mixture of polyphenols, lipids, cellulose, minerals and vitamins, and all these components exert influence to the thermal properties (Wrona et al. 2017). When the lemon nanoemulsion were added into gelatin films, it was two sharp endothermic peaks were also observed at 98.35 °C and 224.0 °C, similar results with gelatin film. However, the heat absorbed (ΔH) in these events reduced to 176.4 J/g and 1.7 J/g, respectively. Sahraee et al. (2017) reported that it can be associated with the heterogeneous dispersion of oil in the polymer matrix. The gelatin films with LN and GTE exhibited endothermic peaks at 104.60 and 216.34 °C and ΔH values of 195.3 and 12.2 J/g. Therefore, the decrease of the first value of ΔH of GGTLN may be due to a reduction in the number of hydrogen bonds and increase in the extent of cross-covalent bonds (Bigi et al. 2001).
Fourier transformation infrared spectroscopy (FTIR)
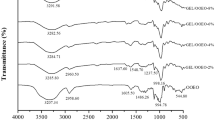
FTIR spectroscopy method is used to investigate the chemical components and intermolecular interactions by streaming infrared waves across the material, measuring the transmittance at 4000–400 cm−1 with 2 cm−1 resolution (Siripatrawan and Harte 2010).
As can be seen in Fig. 2, there is a band between 3250–3650 cm−1 that can be related to O–H stretch. In the gelatin films this region is wider than films with green tea extract. In this band, it can also be observed stretching vibrations of N–H bond which indicates the presence of intra-molecular hydrogen bond between gelatin chains (Duhoranimana et al. 2017).
A peak near 2960 cm−1 was observed in all the films spectra and, it is assigned to the C-H stretching and bending vibrations and amide II bond (Gómez-Guillén et al. 2007). The weak peak in 1455 cm−1 also refers to C–H bending vibration (Sahraee et al. 2017). The peak at 1643 cm−1 in all the spectra is attributed to the C=O stretching vibration of amide I, this is the most useful peak among the absorption of secondary structures of gelatin.
The peak at 1514 cm−1 in the green tea extract indicates the presence of aromatic groups (–C=C) and is also observed in GGTLN with lower intensity and overlapping amide II (Jayaramudu et al. 2017). The peaks 760 cm−1 and 825 cm−1 related to OH deformation of catechin and epicatechin, respectively. Green tea extract powder has this same peak of gelatin and they end up overlapping in GGT film (Arrondo et al. 1993). The peaks at 1244 cm−1 and 1546 cm−1 presented the bending of C-N bond (amide III) and N–H bond (amide II), respectively (Wrona et al. 2017; Duhoranimana et al. 2017). The characteristic peaks of lemon essential oil at 797 cm−1, 886 cm−1 and 948 cm−1 indicate the presence of limonene and α-pinene; β-pinene and cafene and γ-terpinenes, respectively. These peaks are also present in the gelatin film with nanoemulsion but shifted to a lower wavenumber (Silvertein et al. 2006; Barros et al. 1986).
Conclusion
In this study, gelatin nanocomposite films containing lemon nanoemulsion and green tea extract have been synthesized. To the mechanical test, GTE increased the tensile strength of gelatin film and its combination with LN raised elongation at break, compared with GLN. Moreover, the barrier property (WVP) had decreased with LN and GTE in gelatin film. According to DSC and TG results, nanocomposite films GGTLN had discretely lower thermal stability than pure gelatin films. These changes were verified in FTIR and could be attributed to the interactions and the good compatibility of gelatin and the composites. These results indicate that gelatin nanocomposite films can be appropriate for the specific applications, although additional studies are required to use this film as an edible film.
References
Acevedo-Fani A, Salvia-Trujillo L, Rojas-Graü MA, Martín-Belloso (2015) Edible films from essential-oil-loaded nanoemulsions: physicochemical characterization and antimicrobial properties. Food Hydrocoll 47:168–177
Ahmad M, Benjakul S, Prodpran T, Agustini TW (2012) Physico-mechanical and antimicrobial properties of gelatin film from the skin of unicorn leatherjacket incorporated with essential oils. Food Hydrocoll 28:189–199
Alparslan Y, Yapici HH, Metin C, Baygar T, Baygar AGT (2016) Quality assessment of shrimps preserved with orange leaf essential oil incorporated gelatin. LWT Food Sci Technol 72:457–466
Altiok D, Altiok E, Tihminlioglu F (2010) Physical, antibacterial and antioxidant properties of chitosan films incorporated with thyme oil for potential wound healing applications. J Mater Sci Mater Med 21:2227–2236
Arrondo JLR, Muga A, Castresana J, Goñi FM (1993) Quantitative studies of the structure of proteins in solution by Fourier-Transform Infrared Spectroscopy. Prog Biophys Mol Biol 59:23–56
ASTM—American Society for Testing and Materials (1980) Standard test method for water vapor transmission of materials. In: Annual Book of American Standard Testing Methods. Philadelphia, American Society for Testing and Materials, p E96-80
ASTM—American Society for Testing and Materials (1997) Standard test method for tensile properties of thin plastic sheeting. In: Annual Book of American Standard Testing Methods. Philadelphia, American Society for Testing and Materials, pp D882–D897
Atarés L, Bonilla J, Chiralt A (2010) Characterization of sodium caseinate-based edible films incorporated with cinnamon or ginger essential oils. J Food Eng 100:678–687
Barros CB, Yabiku HY, Pinto AJD (1986) Óleos essenciais cítricos especificações. Fundação Cargill, Campinas
Bigi A, Cojazzi G, Panzavolta S, Rubini K, Roveri N (2001) Mechanical and thermal properties of gelatin at different degrees of glutaraldehyde crosslinking. Biomater 22:763–768
Chen X, Jia Y, Feng L, Sun S, An L (2009) Numerical simulation of coil–helix transition processes of gelatin. Polymer 50:2181–2189
Cozmuta AM, Turila A, Apjok R, Ciocian A, Cozmuta LM, Peter A, Nicula C, Galic N, Benkovic T (2015) Preparation and characterization of improved gelatin films incorporating hemp and sage oils. Food Hydrocoll 49:144–155
Donsí F, Ferrari G (2016) Essential oil nanoemulsions as antimicrobial agents in food. J Biotechnol 233:106–120
Duhoranimana E, Karangwa E, Lai L, Xu X, Yu J, Xia S, Zhang X, Muhoza B, Habinshuti I (2017) Effect of sodium carboxymethyl cellulose on complex coacervates formation with gelatin: Coacervates characterization, stabilization and formation mechanism. Food Hydrocoll 69:111–120
Giménez B, Moreno S, López-Caballero ME, Montero P, Gómez-Guillén MC (2013) Antioxidant properties of green tea extract incorporated to fish gelatin films after simulated gastrointestinal enzymatic digestion. LWT - Food Sci Technol 53:445–451
Gómez-Estaca J, López de Lacey A, López-Caballero M, Gómez-Guillén MC, Montero P (2010) Biodegradable gelatin chitosan films incorporated with essential oils as antimicrobial agents for fish preservation. Food Microbiol 27:889–896
Gómez-Guillén MC, Ihl M, Bifani V, Silva A, Montero P (2007) Edible films made from tuna-fish gelatin with antioxidant extracts of two different murta ecotypes leaves (Ugni molinae Turcz). Food Hydrocoll 21:1133–1143
Gómez-Guillén MC, Pérez-Mateos M, Gómez-Estaca J, López-Caballero E, Giménez B, Montero P (2009) Fish gelatin: a renewable material for developing active biodegradable films. Trends in Food Sci Technol 20:3–16
Gontard N, Duchez C, Cuq JL, Guilbert S (1994) Edible composite films of wheat gluten and lipids: water vapour permeability and other physical properties. Int J Food Sci Technol 29:39–50
Guerrero P, Nur Hanani ZA, Kerry JP, de la Caba K (2011) Characterization of soy protein-based films prepared with acids and oils by compression. J Food Eng 107:41–49
Hong YH, Lim GO, Song KB (2009) Physical properties of Gelidium corneum–gelatin blend films containing grapefruit seed extract or green tea extract and its application in the packaging of Pork Loins. J Food Sci 74:6–10
Hoque MS, Benjakul S, Prodpran T (2011a) Properties of film from cuttlefish (Sepia pharaonis) skin gelatin incorporated with cinnamon, clove and star anise extracts. Food Hydrocoll 25:1085–1097
Hoque MS, Benjakul S, Prodpran T, Songtipya P (2011b) Properties of blend film based on cuttlefish (Sepia pharaonis) skin gelatin and mungbean protein isolate. Int J Biol Macromol 49:663–673
Jayaramudu T, Varaprasad K, Kim HC, Kafy A, Kim JW, Kim J (2017) Calcinated tea and cellulose composite films and its dielectric and lead adsorption properties. Carbohyd Polym 171:183–192
Kavoosi G, Rahmatollahi A, Dadfar SMM, Purfard A (2014) Effects of essential oil on the water binding capacity, physico-mechanical properties, antioxidant and antibacterial activity of gelatin films. LWT Food Sci Technol 57:556–561
Kumari M, Mahajan H, Joshi R, Gupta M (2017) Development and structural characterization of edible films for improving fruit quality. Food Packag Shelf Life 12:42–50
Liu F, Chiou BS, Avena-Bustillos RJ, Zhang Y, Li Y, McHugh TH, Zhong F (2017) Study of combined effects of glycerol and transglutaminase on properties of gelatin films. Food Hydrocoll 65:1–9
McHugh TH, Avena-Bustillos FL, Krochta JM (1993) Hydrophilic edible films: modified procedure for water vapor permeability and explanation of thickness effects. J Food Sci 58:899–903
Muriel-Galet V, Cran MJ, Bigger SW, Hernández-Muñoz P, Gavara R (2015) Antioxidant and antimicrobial properties of ethylene vinyl alcohol copolymer films based on the release of oregano essential oil and green tea extract components. J Food Eng 149:9–16
Nunes JC, Melo PTS, Aouada FA, de Moura MR (2018) Influência da nanoemulsão de óleo essencial de limão em filmes à base de gelatina. Quim Nova 41:1006–1010
Nur Hanani ZA, Roos YH, Kerry JP (2014) Use and application of gelatin as potential biodegradable packaging materials for food products. Int J Biol Macromol 71:94–102
Otoni CG, de Moura MR, Aouada FA, Camilloto GP, Cruz RS, Lorevice MV, Soares NF, Mattoso LHC (2014) Antimicrobial and physical-mechanical properties of pectin/papaya puree/cinnamaldehyde nanoemulsion edible composite films. Food Hydrocoll 41:188–194
Peng Y, Wu Y, Li Y (2013) Development of tea extracts and chitosan composite films for active packaging materials. Int J Biol Macromol 59:282–289
Rojas-Graü MA, Avena-Bustillos RJ, Olsen C, Friedman M, Henika PR, Martín-Belloso O, Pan Z, McHugh TH (2007) Effects of plant essential oils and oil compounds on mechanical, barrier and antimicrobial properties of alginate–apple puree edible films. J Food Eng 81:634–641
Sahraee S, Milani JM, Ghanbarzadeh B, Hamishehkar H (2017) Physicochemical and antifungal properties of bio-nanocomposite film based on gelatin-chitin nanoparticles. Int J Biol Macromol 97:373–381
Silvertein RM, Webster FX, Kiemle DJ (2006) Identificação Espectrométrica de Compostos Orgânicos. LTC, Rio de Janeiro
Siripatrawan U, Harte BR (2010) Physical properties and antioxidant activity of an active film from chitosan incorporated with green tea extract. Food Hydrocoll 24:770–775
Steyaert I, Rahier H, Vlierberghe SV, Olijve J, de Clerck K (2016) Gelatin nanofibers: analysis of triple helix dissociation temperature and cold-water. Food Hydrocoll 57:200–208
Tongnuanchan P, Benjakul S, Prodpran T, Nilsuwan K (2015) Emulsion film based on fish skin gelatin and palm oil: physical, structural and thermal properties. Food Hydrocoll 48:248–259
Vanin FM, Hirano MH, Carvalho RA, Moraes ICF, Bittante AMQB, Sobral PJA (2014) Development of active gelatin-based nanocomposite films produced in an automatic spreader. Food Res Int 63:16–24
Wrona M, Cran MJ, Nerín C, Bigger SW (2017) Development and characterisation of HPMC films containing PLA nanoparticles loaded with green tea extract for food packaging applications. Carbohydr Polym 156:108–117
Wu J, Chen S, Ge S, Miao J, Li J, Zhang Q (2013) Preparation, properties and antioxidant activity of an active film from silver carp (Hypophthalmichthys molitrix) skin gelatin incorporated with green tea extract. Food Hydrocoll 32:42–51
Acknowledgements
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001. 2019. Grant 2019/06170-1 and 2013/07296-2 CEPID, São Paulo Research Foundation (FAPESP). The authors are thankful to Embrapa Instrumentation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nunes, J.C., Melo, P.T.S., Lorevice, M.V. et al. Effect of green tea extract on gelatin-based films incorporated with lemon essential oil. J Food Sci Technol 58, 1–8 (2021). https://doi.org/10.1007/s13197-020-04469-4
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-020-04469-4