Abstract
The assimilation of wastewater effluent into wetlands is used to combat land loss in Southeast Louisiana. This study reports on changes in benthic insect diversity at a wetland assimilation site over the course of multiple years of sampling, during which time an initially healthy marsh degraded to open water and subsequently partially-revegetated. Insects are commonly used to monitor the health of streams and rivers, but have rarely been used to study wetlands. Three year-long sampling bouts resulted in a collection of 3984 individuals, representing 33 families and 86 species. Insect diversity tracked the overall condition of the marsh over time. Simpson’s diversity was highest before degradation occurred, lowest at the height of degradation, and intermediate during the period of partial recovery. Species richness, however, was highest in the partially revegetated marsh community. Although this community included species characteristic of both the intact and degraded communities, it shared greatest affinity with the intact marsh. The dominant taxa present in these communities shifted from various beetles to chironomid flies and then back to beetles. Our finding that insect families readily track wetland condition will be a valuable management tool given the dramatically decreased time and expertise needed to identify specimens to family versus species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Human caused habitat loss, in the form of land transformation or habitat destruction, is widely understood to be the leading cause of biodiversity loss (Vitousek 1994; Pimm and Raven 2000; WWF 2016). The wetlands of the Mississippi Delta are a habitat of particular concern, as multiple factors are contributing to their rapid degradation and disappearance (e.g. logging, oil exploration and extraction, introduction of invasive species, hydrological isolation, and rising sea levels) (Day et al. 2007; Lopez 2009; Shaffer et al. 2009). Between 1932 and 2010, Louisiana lost an estimated 1883 mi2 of wetlands, equaling a quarter of the total wetland area present in 1932. This loss is continuing at an estimated rate of ~16 mi2/year, which is the highest rate globally, and accounts for 90% of coastal land loss in the United States (Britsch and Dunbar 1993; Barras et al. 2003; Barras 2006; Couvillion et al. 2011).
To understand the extent of damages and degradation to any ecological system, and the efficacy of any restoration efforts, reliable indicators are needed that reveal the relative condition, or integrity, of these habitats, from intact to degraded. Ecological integrity is an emergent property of a mosaic of multiple factors, such as community composition and diversity, the functioning and efficiency of certain ecological processes, and the suite of abiotic and biotic environmental conditions that facilitate these communities and processes (Karr 1991; Dale and Beyeler 2001). Because an overwhelming amount of information is present for any environment, collected data must be sifted through in order to identify key surrogates, or ecological indicators, that can reveal the condition of the whole system (Karr 1981; Dale and Beyeler 2001; Niemi and McDonald 2004). In wetland systems, water quality metrics, such as pH, dissolved oxygen, salinity and temperature, have often been used to quantify and track habitat condition. However, as these physical data are not themselves a part of the living system, their value as indicators is limited. Sampling components of the living system, such as plant, microbial, or insect communities, provides a biological snapshot that better captures effects of multiple stressors over time (Barbour et al. 1999).
Insects possess multiple qualities that make them ideal ecological indicators. They are hyperdiverse, accounting for more than 80% of the Earth’s total species (Wilson 1992); fill a wide range of functional roles in ecosystems, including pollination, pest control, prey for vertebrates, decomposition, and nutrient cycling; often have large population sizes; and have relatively quick life cycles and thus respond rapidly to environmental changes (Mattson and Addy 1975; Madden and Fox 1997; Schowalter et al. 1998; Bossart and Carlton 2002). In many cases the relative diversity of insect communities has been directly tied to that of plant and vertebrate communities and to the condition of the overall ecosystem (Cox et al. 1998, Shokri et al. 2009). Insect communities in healthy habitats are often characterized by higher diversity, containing a wide variety of families within many orders, relative to these same habitats when they are degraded (Helgen 2002; Weigel and Dimick 2011). Habitat condition may also be indicated by the presence or absence of specific taxa. For example, in rivers and streams, dominance by chironomid flies is associated with degraded or polluted habitats, whereas a significant presence of the orders Ephemeroptera, Plecoptera and Tricoptera indicates intact habitats (Weigel and Dimick 2011).
Insects are abundant in aquatic habitats and have long been used to assess the relative condition of streams and rivers (Wallace and Webster 1996). While similar studies of wetland habitats are less common, these have been steadily increasing in number (Batzer and Wissinger 1996). Wetlands in temperate regions of North America have received the most attention and these constitute a variety of wetland types, including natural and constructed marsh (Murkin et al. 1992; Chow-Fraser et al. 1998; Galbrand et al. 2007; Drinkard et al. 2011; Culler et al. 2014), seasonal woodland ponds (Batzer et al. 2004; Brooks 2000), and peatlands (Mallory et al. 1994, Spitzer and Danks 2006). Relatively few wetland studies have been conducted in subtropical areas and nearly all have been of forested wetlands (Wehrle et al. 1995; Golladay et al. 1997; Harper et al. 1997; Braccia and Batzer 2001; Batzer et al. 2005; Beganyi and Batzer 2011). Only a handful of studies have investigated wetland insect communities in Louisiana, and except for a single coastal marsh study (Bolduc and Afton 2004), these have focused entirely on various aspects of forested wetlands (Ziser 1978; Day et al. 2006; Parys et al. 2013). Yet, despite the wide variety of wetland habitats and regions represented by published studies, certain broad patterns are apparent. Unlike river and stream communities, intact wetland assemblages tend to be dominated by various families of Odonata, Coleoptera, Hemiptera, and Diptera. But similar to river and stream communities, the dipteran family, Chironomidae, is still an indicator of habitat degradation, occurring at higher abundances with increasing coverage of open water.
Tertiary treatment of wastewater via wetlands assimilation has been advanced as a mechanism for increasing the integrity of natural or degraded wetlands, and is known to be an energy and cost effective alternative to traditional water treatment methods (Breaux et al. 1995; Ko et al. 2004; Kadlec and Wallace 2009). There is a large body of research outlining the positive effects of this type of treatment, including multiple success stories of assimilation projects, both regionally (Day et al. 2004; Hunter et al. 2009) and globally (Kadlec and Wallace 2009; Vymazal 2010), including examples specific to tropical ecosystems (Tanaka et al. 2011), and less developed nations (Kivaisi 2001). Wastewater assimilation has the potential benefit of reintroducing nitrogen and phosphorous, essentially mimicking nutrient inputs from seasonal river flooding that contribute to the natural creation of deltas and still sustain wetlands outside of levee boundaries (Reddy et al. 1993; Rybczyk et al. 2002; Day et al. 2004). Freshwater input, whether by wastewater assimilation or river diversions, also helps buffer wetlands from saltwater intrusion (McKee et al. 2004; Martin and Shaffer 2005; Lane et al. 2007).
Objectives and Predictions
The primary objective of this study is to quantify changes that occurred in the insect assemblage at Four Mile Marsh, a natural marsh in the Lake Pontchartrain Basin that was targeted for tertiary processing of secondarily treated wastewater. Initially, we had hypothesized that insect diversity would increase in concert with the influx of the nutrient rich discharge of water into the marsh as a consequence of increased vegetative quality. Instead, the discharge of wastewater resulted in an unexpected nearly complete shift in vegetative cover, transforming the marsh from an intact, fully vegetated habitat, to a heavily degraded, extensively open water habitat after approximately two and a half years. Here we report on how this dramatic shift in vegetative cover, and subsequent management modifications to water flow in the marsh, influenced the resident insect community. First we quantify insect community diversity associated with three discrete time periods, corresponding to when the marsh was intact, degraded, and partially recovered. Then we compare and contrast community similarity/dissimilarity across these three habitat conditions, and identify the specific taxonomic changes that occurred. We expected our data analysis would reveal both decreased diversity and a change in the taxa present as a consequence of the marsh degradation. But we also expected to find that as a result of the vegetative re-growth associated with the management changes, insect diversity would rebound and the community would shift back towards that initially observed. As predicted, insect community diversity shifted dramatically in concert with the shift in marsh condition, influencing both species numbers and the actual species present. Statistical analysis of this community shift revealed potential biological indicator taxa that could be used to track condition of marsh wetlands of the Mississippi Delta.
Methods
Study Site
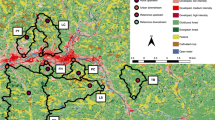
In 2003, Four Mile Marsh, a natural marsh located in the Lake Pontchartrain Basin in Southeast Louisiana, was targeted as a tertiary wastewater treatment site for the nearby municipality of Hammond, Louisiana. Predicted benefits included economic savings, higher water quality, and better fish and wildlife habitat. The project was also expected to improve overall marsh health via reduced salinity and increased nutrient inputs (Day et al. 2005). Four Mile Marsh constitutes a 300 ha plot just south of Ponchatoula, Louisiana (Fig. 1). The entrance is located at 30°24′25.05″N latitude and 90°25′54.91″W longitude. The marsh is bounded to the south and east by the Joyce Wildlife Management Area (JWMA), to the west by Interstate 55, and to the north by an earthen levee separating it from the adjacent South Slough wetlands. The levee and interstate on its north and west sides create a state of hydrologic isolation from potential freshwater inputs such as periodic seasonal flooding of water bodies that lie beyond these barriers. The marsh is susceptible to saltwater intrusion from the south during droughts, as well as from tide and wind driven high water events (Day et al. 2005).
In fall 2006, the continuous release of 11–15,000 m3 of secondarily treated wastewater effluent per day into Four Mile Marsh began. A large pipeline carries wastewater underground from a facility in Hammond, where primary and secondary treatment occurs, to the assimilation site. At the site, the pipeline emerges from underground to an altitude of 2 m and extends along the north bank of the marsh for approximately 1.2 km. The water is distributed into the marsh via 1 m lengths of three inch diameter PVC pipe that extend outward toward the marsh and perpendicular to the larger pipeline (Fig. 2).
Project leaders expected that the site would experience an increase in vegetation, as had been the case at other wastewater assimilation sites in the vicinity (Brantley et al. 2008), and indeed this increase was initially documented (Lundberg et al. 2011). However, the site unexpectedly began to degrade after approximately two years of effluent release. Significant areas of the marsh had converted to open water by late 2008, and these areas increased in extent into 2009, which drew negative scrutiny of the project from local media and residents (David 2009a, b; Eustis 2012). A number of hypotheses for the degradation have been posited, including disease, excess nutrients, and increased nutria (Myocastor coypus) and waterfowl herbivory (Shaffer et al. 2015). Although there is disagreement (David 2009a, b; Eustis 2012), subsequent studies suggest that nutria herbivory and a lack of drainage may be the primary factors impeding recovery (Shaffer et al. 2015). Nutria were hunted aggressively, resulting in over 2000 of them being shot during 2008 and 2009. Additionally, culverts on either side of the site were opened in 2009 to reduce and better control the water level and flow. Although the extent of open water at the marsh was significantly reduced by summer 2011, the site had still not fully revegetated.
Benthic Insect Sampling
Insects were collected at designated distances along transects extending perpendicular from the outfall pipe into the marsh. Samples were collected approximately once a month from June 2007 through July 2008, and from October 2008 through September 2009. A second period of sampling was carried out from January 2012 to January 2013, with samples collected approximately once every three weeks. The two separate sampling periods were coincident with three specific time periods that corresponded with distinct changes in marsh condition based on relative vegetative cover versus open water: fully vegetated (June 2007–July 2008), extensive open water (Oct 2008 – Sep 2009), and intermediate (Jan 2012 – Jan 2013). Hereafter, these periods are referred to as Year 1, Year 2 and Year 3, respectively, for ease of discussion.
Insects were collected using dip net measured sweeps as described in DiFranco (2006) and strained through four stacked metal sieves (with mesh widths of 25, 12.5, 6, and 4 mm from top to bottom) to separate out vegetation and debris. The remaining mixtures of water, organic matter, and insects (including terrestrial species that were dislodged from emergent and floating vegetation) were placed in 500 ml sampling containers and brought back to the lab of Dr. J.L. Bossart at Southeastern Louisiana University for subsequent sorting and identification. Insects were preserved in 95% ethanol. Individuals were identified to the lowest taxonomic level possible, usually species or morphospecies. Multiple taxonomic texts were used to identify specimens (e.g. Wright and Peterson 1944, Young 1979, Stehr 1987, 1991, Daigle 1992, Epler 1996, Merritt and Cummins 1996, Larson et al. 2000, Arnett and Thomas 2001, Epler 2001, Smith 2001, Abbott 2005, Triplehorn and Johnson 2005, Epler 2006, Thorp and Covich 2009). Identifications were confirmed in conjunction with Louisiana State University Arthropod Museum personnel (primarily Dr. C. Carlton and Dr. M. Ferro) by comparison to museum specimens.
Statistical Analysis
Diversity Analysis
Diversity was quantified for each sampling period using univariate approaches including: (a) rarefied and extrapolated species accumulation curves with 95% confidence intervals; (b) estimates of total species richness via multiple non-parametric richness estimators; and (c) Simpson’s Index of Diversity. Rarefied species accumulation curves were generated using EstimateS 9.1.0 (Colwell 2013) to evaluate sampling completeness and compare species richness across sampling periods. Species richness is strongly dependent on sample size. Rarefaction is a robust statistical technique that allows for meaningful standardization and comparison of datasets that differ in size by plotting the average number of species, calculated by repeated random resampling of the complete collection, for a given subsample size (Gotelli and Colwell 2001). EstimateS 9.1.0 allows for rarefaction of individual based data sets, generation of true or unconditional confidence intervals, and statistically sound extrapolation beyond the size of the reference sample (Colwell et al. 2012). This removes the longstanding limitation of only being able to compare different species accumulation curves at the level of the smallest sample and thus having to disregard data from larger samples.
EstimateS was also used to calculate multiple nonparametric estimators of total species richness. These estimators calculate total richness based on a variety of data attributes, and differ with respect to how they handle rare species (Magurran 2004). The estimators used were: ACE and Chao 1 (abundance based); and Chao 2, ICE, Jacknife1 and Jacknife2 (incidence based). Simpson’s index of diversity, which combines both richness and relative abundance, was calculated to compare estimates of overall community diversity. The index is reported here in inverse form, where larger numbers equate to higher diversity. Simpson’s Index represents the probability that two individuals randomly drawn from a sample are different species (Magurran 2004; Lande 1996), and is considered the most robust and biologically meaningful of the diversity indices available (Magurran 2004).
Changes in Insect Assemblages across Sampling Periods
Multivariate statistical techniques were used to evaluate the changes in insect assemblages between sampling years, as these complex community datasets are inherently multivariate. These included a) analysis of similarity (ANOSIM), a multivariate analogue of ANOVA; graphical investigations of natural groupings and significant differences between sampling periods via (b) non-metric multidimensional scaling (NMDS) incorporating (c) similarity profile (SIMPROF) analysis. These permutation based multivariate analyses were carried out in PRIMER 6 (Clarke and Gorely 2006). Sampling data were combined within each of the three main sampling periods to generate a single community data set of species and their abundances for each time period. Raw abundance data were converted to relative abundances and then square root transformed to down weight the contributions of dominant species (Clarke and Gorely 2006). The Bray-Curtis resemblance matrix generated from the standardized, square root transformed data was used for several subsequent analyses.
An analysis of similarity (ANOSIM) tests the null hypothesis that there is no difference between groups of samples belonging to pre-determined factors (Clarke and Gorely 2006), in this instance defined as sampling period (three sampling periods) and distance from outfall (three distances). ANOSIM tests for overall differences and for pairwise differences, reporting statistical difference with an R value ranging from −1 to 1. Values close to negative one indicate strong within group differences (and thus negative values generally should not occur), values close to zero indicate no differences, and values close to positive one indicate strong between group differences. Significance of the R value is reported in percentages.
Spatial clustering of the data was investigated using non-metric multidimensional scaling (NMDS). This ordination positions data points in multi dimensional space (usually 2D or 3D), such that the relative distance between points corresponds as close as possible to their similarity in the initial resemblance matrix (Clarke and Gorely 2006). NMDS gives an easily interpretable graphical presentation of natural groupings and/or differences, visible as points either close together or far apart, respectively. A measure of how faithfully the relationships among samples are able to be represented in dimensional space is given as a value of stress. The lower the stress value, the more faithful the representation (Clarke and Gorely 2006; Clarke and Warwick 2001). A SIMPROF analysis, which also calculates a Bray-Curtis resemblance matrix, was combined with the NMDS to test whether any significant internal structure was present, i.e. clustering of sites/communities based on their similarity/dissimilarity.
Identification of Bioindicators
A similarity percentage (SIMPER) analysis was used to identify individual species that contributed to significant groupings and differences, i.e. potential indicator species. SIMPER uses average dissimilarities between all pairs of samples from the Bray-Curtis resemblance matrix to calculate the percent contribution of each taxa to group formation. These results can then be combined with NMDS to provide visual representations of differences attributed to particular taxa.
Results
Diversity Analysis
On a per sampling effort basis, the number of individuals collected was much higher in Year 2 during the open-water period (Table 1). Yet, despite this much larger overall collection, the number of species collected was lowest (Table 1; Fig. 3). This is illustrated in Fig. 3, wherein the asymptote on the species accumulation curve for Year 2 occurred at a lower number of species than for either of the other years. Additionally, rarefied richness values for Year 2 ranged from 30 to 46% lower than those for Years 1 and 3, regardless of whether smaller samples were extrapolated to the size of the larger samples or whether larger samples were interpolated to the size of the smaller samples. Interestingly, species richness was also always highest for Year 3 (Table 2; Fig. 3), with many new species still being collected when sampling was terminated (Fig. 3).
Simpson’s Index of overall diversity based on Year 1 data was three times higher than the measure of diversity documented for the degraded period, when much of the marsh was open water, and more than 30% higher than that documented for Year 3, when vegetation in the marsh was beginning to rebound (Table 1).
Changes in Insect Assemblages across Sampling Periods
A total of 3984 individuals was collected over the course of the three sampling periods (Table 1), representing 86 species and 33 families. Communities collected during the different sampling periods were statistically significantly different (i.e. Table 3; significant global ANOSIM R value). The greatest difference existed between Years 1 and 2, with smaller but still significant differences occurring between Years 1 and 3, and between Years 2 and 3 (Table 3). Significant differences were also detected between distances from the outfall, but these differences were only weakly supported (R values near 0 and a global p-value only slightly less than the 0.05 threshold) and associated with communities nearest the outfall versus the rest (Table 3). The results of those analyses are addressed in more detail in Weller (2014).
The NMDS ordination indicated spatial separation of data between sampling periods (Fig. 4), which is further evidence of differences among communities. At 52% within-group community similarity, three groups corresponding to the three different time periods were statistically supported (SIMPROF; p < 0.05). However, the main dichotomy occurred at 48% within-group similarity (Fig. 4) and produced a group consisting of only Year 2 communities and a second group that included Year 1 and Year 3 (SIMPROF; p < 0.05), indicating that the community collected in Year 2 was unique from the communities collected in Years 1 and 3.
Identification of Bioindicators
SIMPER analysis identified multiple species, representing three different Orders, contributing to dissimilarities between these communities (Table 4). Changes were also detected at the family level. Taxonomic families represented in the communities changed over time (Fig. 5), tracking changes in marsh condition. The most striking shift in community membership was associated with a drop in abundance of Coleoptera and an associated large increase in abundance of the dipteran family, Chironomidae, during Year 2, a trend that reversed in Year 3 (Fig. 5; Appendix Table 5). Also notable, the relative abundance of odonates dropped by 62% from Year 1 to 2, and then by an additional 50% from Year 2 to 3. The “Other” category also almost quadrupled in Year 3, reflecting an increased number of species collected that were represented by one or only a few individuals.
Discussion
Over the three sampling periods, the insect community at Four Mile Marsh clearly changed in tandem with the visible shift in vegetative cover that occurred as marsh condition declined and then partially recovered. Namely, the insect community of the originally healthy marsh in 2007 (Year 1) transitioned to a community dominated by taxa indicative of degraded habitat by 2008–2009 (Year 2), and by 2012 (Year 3) had shifted back toward one more characteristic of the initial intact community. The taxonomic shifts were apparent at both the level of family and of species. This study also revealed taxa that could potentially be used as ecological indicator species in wetland marshes, a habitat that is relatively understudied in regards to how their associated insect communities change with changes in ecosystem condition.
Community Diversity and Marsh Condition
Multiple lines of evidence indicate the marsh was in a state of recovery from the mostly open water degraded state it had experienced during 2009 by the time the third bout of insect sampling commenced in 2012. First, the species accumulation curve for Year 3 was much higher than the others. Simpson’s Index of overall diversity had also rebounded to nearly twice that calculated for Year 2, indicating species richness and evenness had increased. These patterns make sense since habitats with more heterogeneity, or places where multiple habitats adjoin, are expected to have higher diversity due to increased niche availability (MacArthur and MacArthur 1961; Magurran 2004). Although significant vegetative regrowth had occurred by our third period of sampling, areas of open water persisted. Hence, the marsh comprised two habitat states - that of both pre- and post-restoration efforts, which would have effectively increased the number of ecological niches available for species to occupy.
Partial recovery was also evidenced by the return of species characteristic of the intact (Year 1) marsh community, combined with the continued presence of species characteristic of the degraded (Year 2) marsh community. Notably, the community present in Year 3 also included member species not found in either the intact or degraded communities. The vegetative regrowth that occurred after restoration efforts were implemented comprised a plant community consisting of a higher proportion of annuals vs. perennials, relative to species present in the intact marsh community (Shaffer et al. 2015). The addition of these new insect species is likely at least partially due to this observed shift in the plant community.
Finally, Year 1 and Year 3 samples clustered more closely in multi-dimensional space than either did to those from Year 2 (Fig. 4). Thus, even though the community collected during Year 3 retained some elements of the degraded insect community of Year 2, it was more similar to the intact community collected in Year 1 than it was to the Year 2 community.
Ecological Indicator Taxa
Some taxa were entirely or largely only collected before the marsh degraded and converted to open water, whereas other taxa were entirely or largely only collected once the marsh became degraded. Species whose presence or absence in a community depends on the environmental conditions present are known as indicator species (National Research Council 2000). Ecological indicators measure characteristics of the structure, composition, or function of ecological systems (Niemi and McDonald 2004). The presence or absence of an indicator species is easily interpretable and can be used as a surrogate for the analysis of a complex system that cannot feasibly be measured, such as environmental or habitat condition (National Research Council 2000). Such species can be used to track environmental degradation and can additionally be used in the development of an Index of Ecological or Biological Integrity. Integrity indices integrate multiple measures (e.g. species richness, presence/absence of specific species) that respond in a predictable fashion to environmental change and combine these to generate an easy to understand single number that represents the health or condition of the particular ecosystem (Barbour et al. 1999).
Taxa present indicating a stronger affinity with intact habitat included species of the main coleopteran families (Dytiscidae, Hydrophilidae, and Noteridae), specifically the noterid beetle, Suphisellus bicolor. The re-emergence of coleopteran families in the Year 3 community, in particular, indicates the improvement of habitat from Year 2 to Year 3. Suphisellus bicolor is an herbivorous scavenger known to be found in thick aquatic vegetation (Young 1979) and thus its increased presence fits with the increased vegetative cover present in these years. Coleoptera as a whole appear to be more tolerant of declining habitat quality than the more sensitive, more commonly used benthic macroinvertebrates, such as Ephemeroptera and Plecoptera. Nonetheless, their tolerance level ranks below that of most other insect Orders (Hilsenhoff 1988; Mandaville 2002), and Coleoptera have the added benefit of occurring across a wider range of aquatic habitats. In this context they may prove to be a particularly valuable indicator of wetland condition versus taxa predominately restricted to fast moving waters.
Taxa indicating affinity to the degraded habitat-associated community include waterboatmen (family Corixidae), represented by the species Palmacorixa buenoi, and two morphospecies of chironomid flies. This species of waterboatmen is generally found in ponds and ditches, and is associated with more open water habitats, where it consumes algae and detritus (Epler 2006; Thorp and Covich 2009). Its presence in both the Year 2 and Year 3 communities indicates the open water still remaining in Year 3 was sufficient to support an organism with strong swimming habits. According to the Biological Monitoring Working Party biotic index, a water quality metric based on tolerance of organic pollutants, collection of high numbers of Corixidae is indicative of moderate water quality (Galbrand et al. 2007). Higher numbers of Corixidae have been associated with decreased habitat quality in other indices as well (Bressler et al. 2006; Yimer and Mengistou 2009). The pattern this group showed at Four Mile Marsh is consistent with these earlier findings, as its abundance increased with an observed decrease in vegetation.
High relative abundances of chironomids were collected (they made up approximately 70% of the total sample) when the marsh was degraded. Although their numbers were much decreased by the third bout of sampling, this group still had a major presence (Fig. 5, Appendix Table 5). Chironomidae are commonly associated with degraded habitats in rivers and streams (Mandaville 2002; Weigel and Dimick 2011), as well as marshes (Chow-Fraser et al. 1998), so their abundance during the ‘open-water’ period was not unexpected. The fact they persisted in such high numbers into the third sampling period additionally indicates the marsh had still not fully recovered despite the significant increase in vegetative cover that occurred after management practices were modified.
Interestingly, the order Odonata did not prove useful for tracking marsh condition. This group is typically associated with high quality habitat (Galbrand et al. 2007; Villalobos-Jimenez et al. 2016) and we expected the Odonata would have declined as the marsh converted to open water and then to have rebounded once the marsh was recovering. Although relative abundance of Odonata did indeed drop in Year 2, this decrease was largely due to the dramatic increase in Chironomids collected. Additionally, not only was there no rebound in Year 3, but the most common species present in Year 3 was not among the most common species present in the intact community. It is possible that these unexpected patterns were related to changes in vegetation. Although Year 3 saw a rebound in vegetation coverage, more annual and fewer perennial species were present in the recovering marsh relative to when effluent release began (Shaffer et al. 2015). Regardless, the total number of Odonata collected in Year 3 was very low and any conclusions regarding their indicator potential based on these data are tentative at best.
Management Implications
This project identified multiple species and families that could potentially be used by stakeholders as ecological indicators to gauge marsh condition or measure the success of restoration efforts. Perhaps most notably, differences between communities were readily visible at the family level. As a management tool, the ability to use insect families as indicators, versus relying on species, would be a major benefit as this would greatly reduce the time required to identify insect specimens, which can be extremely time consuming when carried to species level. Species identifications are also generally not feasible for amateurs or individuals who lack access to sophisticated identification equipment and expensive taxonomic reference materials. In contrast, traits that identify families can often be easily learned and observed.
Southeast Louisiana’s unique wetlands are disappearing at an alarming rate and the implementation of strategic management tactics will be critical to their conservation. At the very least, successful management requires regular monitoring and evaluation. Here we show that similar to rivers and streams, survey of benthic insects of the region’s deltaic marshes will readily reveal the biological condition of these economically and inherently valuable ecosystems. While assimilation wetlands often provide a win-win situation, providing both economically efficient water treatment and combating wetlands loss, the changes at Four Mile Marsh occurring after outflow was initiated indicate such outcomes are not a given. Monitoring the insect communities of assimilation wetlands should become a cornerstone of management programs and efforts to mitigate wetland decline in the southeastern United States.
References
Abbott, JC (2005) Dragonflies and damselflies of Texas and the South-Central United States: Texas, Louisiana, Arkansas, Oklahoma, and New Mexico. Princeton University Press
Arnett RH, Thomas MC (2001) American beetles. CRC Press, Boca Raton
Barbour MT, Gerritsen J, Snyder BD, Stribling JB (1999) Rapid bioassessment protocols for use in streams and wadeable rivers: periphyton, benthic macroinvertebrates and fish. 2nd Edition. EPA 841-B-99-002. U.S. Environmental Protection Agency, Washington, DC
Barras JA (2006) Land area change in coastal Louisiana after the 2005 hurricanes—a series of three maps: U.S. Geological Survey Open-File Report 2006–1274, http://pubs.usgs.gov/of/2006/1274/
Barras JA, Beville S, Britsch D, Hartley S, Hawes S, Johnston J, Kemp P, Kinler Q, Martucci A, Porthouse J, Reed D, Roy K, Sapkota S, Suhayda J (2003) Historical and projected coastal Louisiana land changes: 1978–2050: USGS Open File Report 03–334
Batzer DP, Wissinger SA (1996) Ecology of insect communities in nontidal wetlands. Annual Review of Entomology 41:75–100
Batzer DP, Palik BJ, Buech R (2004) Relationships between environmental characteristics and macroinvertebrate communities in seasonal woodland ponds of Minnesota. Journal of the North American Benthological Society 23:50–68
Batzer DP, George BM, Braccia A (2005) Aquatic invertebrate response to timber harvest in a bottomland hardwood wetland of South Carolina. Forest Science 51:284–291
Beganyi SR, Batzer DP (2011) Wildfire induced changes in aquatic invertebrate communities and mercury bioaccumulation in the Okefenokee swamp. Hydrobiologia 669:237–247
Bolduc F, Afton AD (2004) Relationships between wintering waterbirds and invertebrates, sediments and hydrology of coastal marsh ponds. Waterbirds 27:333–341
Bossart JL, Carlton CE (2002) Insect conservation in America. American Entomologist 48:83–91
Braccia A, Batzer DP (2001) Invertebrates associate with woody debris in a southeastern U.S. forested floodplain wetland. Wetlands 21:18–31
Brantley CG, Day JW, Lane RR, Hyfield E, Day JN, Ko JY (2008) Primary production, nutrient dynamics, and accretion of a coastal freshwater forested wetland assimilation system in Louisiana. Ecological Engineering 34:7–22
Breaux A, Farber S, Day JW (1995) Using natural coastal wetlands systems for wastewater treatment: an economic benefit analysis. Journal of Environmental Management 44:285–291
Bressler D, Stribling J, Paul M, Hicks M (2006) Stressor tolerance values for benthic macroinvertebrates in Mississippi. Hydrobiologia 573:155–172
Britsch L, Dunbar J (1993) Land loss rates: Louisiana coastal plain. Journal of Coastal Research 9:324–338
Brooks RT (2000) Annual and seasonal variation and the effects of hydroperiod on benthic macroinvertebrates of seasonal forest (“vernal”) ponds in Central Massachusetts, USA. Wetlands 20:707–715
Chow-Fraser P, Lougheed V, Le Thiec V, Crosbie B, Simser L, Lord J (1998) Long-term response of the biotic community to fluctuating water levels and changes in water quality in Cootes paradise marsh, a degraded coastal wetland of Lake Ontario. Wetlands Ecology and Management 6:19–42
Clarke KR, Gorely RN (2006) PRIMER v6: User Manual/Tutorial. PRIMER-E, Plymouth
Clarke KR, Warwick R (2001) Change in marine communities: an approach to statistical analysis and interpretation, 2nd edn. PRIMER-E, Plymouth
Colwell RK (2013) EstimateS: Statistical estimation of species richness and shared species from samples. Version 9. Persistent URL http://purl.oclc.org/estimates
Colwell RK, Chao A, Gotelli NJ, Lin SY, Mao CX, Chazdon RL, Longino JT (2012) Models and estimators linking individual-based and sample-based rarefaction, extrapolation, and comparison of assemblages. Journal of Plant Ecology 5:3–21
Couvillion BR, Barras JA, Steyer GD, Sleavin W, Fischer M, Beck H, Trahan N, Griffin B, Heckman D (2011) Land area change in coastal Louisiana from 1932 to 2010: U.S. Geological Survey Scientific Investigations Map 3164
Cox R Jr, Hanson M, Roy C (1998) Mallard duckling growth and survival in relation to aquatic invertebrates. Journal of Wildlife Management 62:124–133
Culler LE, Smith RF, Lamp WO (2014) Weak relationships between environmental factors and invertebrate communities in constructed wetlands. Wetlands 34:351–361
Daigle JJ (1992) Florida dragonflies (Anisoptera): a species key to the aquatic larval stages. State of Florida, Department of Environmental Regulation
Dale VH, Beyeler SC (2001) Challenges in the development and use of ecological indicators. Ecological Indicators 1:3–10
David B (2009a) Wetlands project questioned. The Hammond Star May 13
David B (2009b) Profs ponder nutria, nutrients at Joyce The Hammond Star July 17
Day JW Jr, Ko JY, Rybczyk J, Sabins D, Bean R, Berthelot G, Brantley C, Cardoch L, Conner W, Day JN, Englande AJ (2004) The use of wetlands in the Mississippi Delta for wastewater assimilation: a review. Ocean and Coastal Management 47:671–691
Day JW, Lane R, Lindsey J, Day J (2005) Hammond wetland wastewater assimilation use attainability analysis (UAA) www.saveourlake.org (April)
Day JW Jr, Westphal A, Pratt R, Hyfield E, Rybczyk J, Kemp GP, Day JN, Marx B (2006) Effects of long-term municipal effluent discharge on the nutrient dynamics, productivity, and benthic community structure of a tidal freshwater forested wetland in Louisiana. Ecological Engineering 27:242–257
Day JW, Boesch DF, Clairain EJ, Kemp GP, Laska SB, Mitsch WJ, Orth K, Mashriqui H, Reed DJ, Shabman L, Simenstad CA (2007) Restoration of the Mississippi Delta: lessons from hurricanes Katrina and Rita. Science 315:1679–1684
DiFranco J (2006) Protocols for sampling aquatic macroinvertebrates in freshwater wetlands. State of Maine, Department of Environmental Protection
Drinkard MK, Kershner MW, Romito A, Nieset J, Szalay FA (2011) Responses of plants and invertebrate assemblages to water-level fluctuation in headwater wetlands. Journal of the North American Benthological Society 30:981–996
Epler JH (1996) Identification manual for the water beetles of Florida. State of Florida, Department of Environmental Protection
Epler JH (2001) Identification manual for the larval Chironomidae of north and South Carolina. State of North Carolina, Department of Environmental and Natural Resources
Epler JH (2006) Identification manual for the aquatic and semi-aquatic Heteroptera of Florida. State of Florida, Department of Environmental Protection
Eustis S (2012) The collapse of Four Mile Marsh. Gulf Restoration Network. Wave Makers News 7(1). http://healthygulf.org/sites/healthygulf.org/files/march_2012_wave_makers.pdf
Galbrand C, Lemieux IG, Ghaly AE, Côté R, Verma M (2007) Assessment of constructed wetland biological integrity using aquatic macroinvertebrates. OnLine Journal of Biological Sciences 7:52–65
Golladay SW, Taylor BW, Palik BJ (1997) Invertebrate communities of forested lime sink wetlands in Southwest Georgia, USA: habitat use and influence of extended inundation. Wetlands 17:383–393
Gotelli NJ, Colwell RK (2001) Quantifying biodiversity: procedures and pitfalls in the measurement and comparison of species richness. Ecology Letters 4:379–391
Harper D, Mekotova J, Hulme S, White J, Hall J (1997) Habitat heterogeneity and aquatic invertebrate diversity in floodplain forests. Global Ecology and Biogeography Letters 6:275–285
Helgen JC (2002) Methods for evaluating wetland condition: developing an invertebrate index of biological integrity for wetlands. #9 US Environmental Protection Agency
Hilsenhoff WL (1988) Rapid field assessment of organic pollution with a family-level biotic index. Journal of the North American Benthological Society 7:65–68
Hunter RG, Day JW Jr, Lane RR, Lindsey J, Day JN, Hunter MG (2009) Impacts of secondarily treated municipal effluent on a freshwater forested wetland after 60 years of discharge. Wetlands 29:363–371
Kadlec RH, Wallace SD (2009) Treatment wetlands, 2nd edn. CRC Press, Boca Raton
Karr JR (1981) Assessment of biotic integrity using fish communities. Fisheries 6:21–27
Karr JR (1991) Biological integrity: a long-neglected aspect of water resource management. Ecological Applications 1:66–84
Kivaisi AK (2001) The potential for constructed wetlands for wastewater treatment and reuse in developing countries: a review. Ecological Engineering 16:545–560
Ko JY, Day JW, Lane RR, Day JN (2004) A comparative evaluation of money-based and energy-based cost–benefit analyses of tertiary municipal wastewater treatment using forested wetlands vs. sand filtration in Louisiana. Ecological Economics 49:331–347
Lande R (1996) Statistics and partitioning of species diversity, and similarity among multiple communities. Oikos 76:5–13
Lane RR, Day JW, Marx BD, Reyes E, Hyfield E, Day JN (2007) The effects of riverine discharge on temperature, salinity, suspended sediment and chlorophyll a in a Mississippi delta estuary measured using a flow-through system. Estuarine, Coastal and Shelf Science 74:145–154
Larson DJ, Alarie Y, Roughley RE, Conseil national de recherches (Canada) (2000) Predaceous diving beetles (coleoptera: Dytiscidae) of the Nearctic region, with emphasis on the fauna of Canada and Alaska. NRC Research Press, Ottawa
Lopez JA (2009) The environmental history of human-induced impacts to the Lake Pontchartrain Basin in southeastern Louisiana since european settlement – 1718 to 2002. Journal of Coastal Research 54:1–11
Lundberg CJ, Shaffer GP, Wood WB, Day JW Jr (2011) Growth rates of baldcypress (Taxodium distichum) seedlings in a treated effluent assimilation marsh. Ecological Engineering 37:549–553
MacArthur RH, MacArthur JW (1961) On bird species diversity. Ecology 42:594–598
Madden KE, Fox BJ (1997) Arthropods as indicators of the effects of fluoride pollution on the succession following sand mining. Journal of Applied Ecology 34:1239–1256
Magurran A (2004) Measuring biological diversity. Blackwell Science Ltd, Malden
Mallory ML, Blancher PJ, Weatherhead PJ, McNicol DK (1994) Presence or absence of fish as a cue to macroinvertebrate abundance in boreal wetlands. Hydrobiologia 279(280):345–351
Mandaville SM (2002) Benthic macroinvertebrates in freshwaters: taxa tolerance values, metrics, and protocols. Soil & Water Conservation Society of Metro Halifax, Nova Scotia
Martin SB, Shaffer GP (2005) Sagittaria biomass partitioning relative to salinity, hydrologic regime, and substrate type: implications for plant distribution patterns in coastal Louisiana, United States. Journal of Coastal Research 21:167–174
Mattson WJ, Addy ND (1975) Phytophagous insects as regulators of forest primary production. Science 190:515–522
McKee KL, Mendelssohn IA, Materne MD (2004) Acute salt marsh dieback in the Mississippi River deltaic plain: a drought-induced phenomenon? Global Ecology and Biogeography 13:65–73
Merritt RW, Cummins KW (1996) An introduction to the aquatic insects of North America, 3rd edn. Kendall/Hunt Publishing Company, Dubuque, IA
Murkin EJ, Murkin HR, Titman RD (1992) Nektonic invertebrate abundance and distribution at the emergent vegetation-open water interface in the Delta marsh, Manitoba, Canada. Wetlands 12:45–52
National Research Council (2000) Ecological indicators for the nation. National Academy Press, Washington DC
Niemi GJ, McDonald ME (2004) Application of ecological indicators. Annual Review of Ecology, Evolution, and Systematics 35:89–111
Parys KA, Gimmel ML, Johnson SJ (2013) Checklist of insects associated with Salvinia minima baker in Louisiana, USA. Chemicke Listy 9:1488–1495
Pimm SL, Raven P (2000) Extinction by numbers. Nature 403:843–845
Reddy KR, DeBusk WF, DeLaune RD, Koch MS (1993) Long-term nutrient accumulation rates in the Everglades. Soil Science Society of America Journal 57:1147–1155
Rybczyk JM, Day JW, Conner WH (2002) The impact of wastewater effluent on accretion and decomposition in a subsiding forested wetland. Wetlands 22:18–32
Schowalter TD, Zhang YL, Sabin TE (1998) Decomposition and nutrient dynamics of oak Quercus spp. logs after five years of decomposition. Ecography 21:3–10
Shaffer GP, Wood WB, Hoeppner SS, Perkins TE, Zoller J, Kandalepas D (2009) Degradation of baldcypress-water tupelo swamp to marsh and open water in southeastern Louisiana, USA: an irreversible trajectory? Journal of Coastal Research Special Issue 54:152–165
Shaffer GP, Day JW, Hunter RG, Lane RR, Lundberg CJ, Wood WB, Hillmann ER, Day JN, Strickland E, Kandalepas D (2015) System response, nutria herbivory, and vegetation recovery of a wetland receiving secondarily-treated effluent in coastal Louisiana. Ecological Engineering 79:120–131
Shokri MR, Gladstone W, Kepert A (2009) Annelids, arthropods or molluscs are suitable as surrogate taxa for selecting conservation reserves in estuaries. Biodiversity and Conservation 18:1117–1130
Smith DG (2001) Pennak's freshwater invertebrates of the United States: Porifera to Crustacea, 4th edn. John Wiley & Sons, New York
Spitzer K, Danks HV (2006) Insect biodiversity of boreal peat bogs. Annual Review of Entomology 51:137–161
Stehr F (1987) Immature insects volume 1. Kendall Hunt Publishing Co, Dubuque
Stehr F (1991) Immature insects volume 2. Kendall Hunt Publishing Co, Dubuque
Tanaka N, Ng WJ, Jindasa KBSN (2011) Wetlands for tropical applications: wastewater treatment by constructed wetlands. Imperial College Press, London
Thorp JH, Covich AP (2009) Ecology and classification of north American freshwater invertebrates. Academic Press, Cambridge
Triplehorn CA, Johnson NF (2005) Borror and Delong’s introduction to the study of insects, 7th edn. Thomson Brooks/Cole, Belmont
Villalobos-Jimenez G, Dunn AM, Hassall C (2016) Dragonflies and damselflies (Odonata) in urban ecosystems: a review. European Journal of Entomology 113:217–232
Vitousek P (1994) Beyond global warming: ecology and global change. Ecology 75:1861–1876
Vymazal J (2010) Constructed wetlands for wastewater treatment. Water 2:530–549
Wallace JB, Webster JR (1996) The role of macroinvertebrates in stream ecosystem function. Annual Review of Entomology 41:115–139
Wehrle BW, Kaminski RM, Leopold BD, Smith WP (1995) Aquatic invertebrate resources in Mississippi forested wetlands during winter. Wildlife Society Bulletin 23:774–783
Weigel BM, Dimick JJ (2011) Development, validation, and application of a macroinvertebrate-based index of biotic integrity for nonwadeable rivers of Wisconsin. Journal of the North American Benthological Society 30:665–679
Weller MO (2014) Analysis of invertebrate diversity dynamics in a wastewater assimilation marsh in Southeast Louisiana. Master’s Thesis, Southeastern Louisiana University
Wilson EO (1992) The diversity of life. W.W. Norton, New York
Wright M, Peterson A (1944) A key to the genera of Anisopterous dragonfly nymphs of the United States and Canada (Odonata, suborder Anisoptera). The Ohio Journal of Science 44:151–166
WWF (2016) Living Planet Rerport 2016. Risk and resilience in a new era. WWF International, Gland, Switzerland
Yimer HD, Mengistou S (2009) Water quality parameters and macroinvertebrates index of biotic integrity of the Jimma wetlands, southwestern Ethiopia. Journal of Wetlands Ecology 3:79–99
Young FN (1979) Water beetles of the genus Suphisellus crotch in the Americas north of Colombia (coleoptera: Noteridae). The Southwestern Naturalist 24:409–429
Ziser SW (1978) Seasonal variations in water chemistry and diversity of the phytophilic macroinvertebrates of three swamp communities in southeastern Louisiana. The Southwestern Naturalist 23:545–562
Acknowledgements
We are especially indebted to Ryan Clark and multiple undergraduate students at Southeastern Louisiana University; specifically, Joshua Austin, Michelle Bell, Andrew Carson, Ashlee Minor, Leah Poché, Kristen Ramsey, and Chris Stiegler. The identifications of difficult species were verified by Dr. Chris Carlton and personnel at the Louisiana State Arthropod Museum. Funding support was provided by the Environmental Protection Agency and the Lake Pontchartrain Basin Research Program. This material is based in part on work done whilst JLB was serving at the National Science Foundation. The views expressed in this paper do not necessarily reflect those of the National Science Foundation or the United States Government.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
Rights and permissions
About this article
Cite this article
Weller, M.O., Bossart, J.L. Insect Community Diversity Tracks Degradation and Recovery of a Wastewater Assimilation Marsh in Southeast Louisiana. Wetlands 37, 661–673 (2017). https://doi.org/10.1007/s13157-017-0897-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13157-017-0897-1