Abstract
Plant biomass and its allocation patterns are instrumental in understanding global carbon sinks; however, knowledge is still limited, especially in high-altitude peatlands. We investigated aboveground and belowground biomass allocation in the Zoige peatland of the Tibetan plateau, China, and its relationship with environmental factors using data collected from 32 sites across the peatland during 2011 and 2012. Standardized major axis, multiple factor analysis and linear regression functions were used to perform data analysis. The average aboveground biomass, belowground biomass, total biomass and root:shoot ratio for the Zoige alpine peatland were 341.01, 3262.93, 3620.36 g m−2 and 10.32, respectively. On average, approximately 86% of the root biomass was located in the top 30 cm of soil. There was positive allometric relationship (p < 0.01) between belowground biomass and aboveground biomass. The water conditions, soil organic carbon and soil nitrogen were the main factors that influenced plant biomass and biomass allocation in the Zoige peatland.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Peatlands are an important component of terrestrial carbon storage and represent a major carbon sink. Peatlands cover only approximately 3% of the world”s land area, but they potentially store approximately 30% of the global terrestrial ecosystem carbon, equivalent to 455 Pg C (Gorham 1991; Blodau 2002). Both the belowground and aboveground components of plants are primary sources of labile C to peatland soil. The decomposition of plant material is a key component of nutrient cycling and a major contributor to soil CO2 flux (Murphy and Moore 2010). Understanding plant aboveground biomass (AGB) and belowground biomass (BGB) allocation patterns across natural gradients is necessary for an in-depth understanding of how plants may respond to future climate change.
Compared with the considerable number of studies on AGB in peatlands, little is known about the belowground component (Weltzin et al. 2000; O’Driscoll et al. 2011; Byrd et al. 2011). Currently, using default values of root:shoot (R/S) ratios and AGB of different vegetation types to estimate large-scale root carbon storage is a practical interim method (IPCC 2003). This method is also permitted and applied by nations to estimate BGB and carbon stocks for national greenhouse gas inventory purposes (Snowdon et al. 2000; Australian Greenhouse Office 2002; Eamus et al. 2002; Mokany et al. 2006). Many peatland C models also rely on R/S ratios to estimate belowground biomass (Murphy et al. 2009a). However, some vegetation types do not have the IPCC default R/S values; such as tundra, cool temperate arid shrublands and alpine peatlands (Mokany et al. 2006). According to Murphy (2009), the BGB may be equal to or greater than the AGB in peatland ecosystems. A wide variation in R/S ratios (3:1–30:1) and a mean value of 11:1 have been suggested for arctic tundra ecosystems (Dennis and Johnson 1970). Relatively few researchers have considered the variations in R/S ratios across gradients of plant types and environments within and between peatland ecosystems (Finér and Laine 2000; Murphy et al. 2009a). Miller (2011) examined the effects of long-term drainage on plant community composition and biomass in boreal continental peatlands (bogs and fens) but only considered the AGB. In summary, we still do not know the R/S ratios in alpine peatland environments or understand their relationships with natural gradients within alpine peatlands.
The optimal partitioning theory and the allometric biomass partitioning theory are two important biomass allocation hypotheses. Under the optimal partitioning theory, plants allocate biomass to the organ that acquires the most limiting resource. This means that plants should allocate more biomass to roots when water or nutrients are limiting and shift more biomass aboveground in higher nutrient or moisture conditions (McConnaughay and Coleman 1999; McCarthy and Enquist 2007; Kobe et al. 2010;). Allometric partitioning theory states that plant allocation between components is mainly regulated by total plant size and follows a scale relationship between compartments (West et al. 1999; Genet et al. 2011). These two hypotheses have been studied extensively in woody plants and grasslands, mainly at the individual level (Enquist and Niklas 2002; Murphy et al. 2009b; ). Recently, Murphy and Moore (2010) reported that the relationships between AGB and BGB of shrubs in an ombrotrophic peatland complied with the allometric relationships, whereas herbs did not. Moreover, variations in water conditions may change the allometric relationship between AGB and BGB (Murphy et al. 2009). In comparison with information at the individual level, little evidence is available at the community level. The results of Yang et al. (2009) support the isometric allocation hypothesis for the AGB and BGB relationship in Tibetan grasslands. However, whether this relationship holds true across alpine peatlands types is unknown.
The Zoige wetland of the Tibetan plateau is an alpine peatland at permanent low temperatures. The existing studies are limited and mainly focused on methane fluxes (Chen et al. 2009; Chen et al. 2011) and landscape pattern changes (Bai et al. 2008), and little work has been conducted to directly compare the biomass of vegetation communities, vertical distribution of roots and effects of environmental factors on biomass allocation. The major objectives of this study were to 1) characterize differences in different plant communities with respect to both AGB and BGB, 2) quantify belowground root distribution with depth, and 3) determine the relationship between biomass and environmental factors.
Material and Methods
Site Description
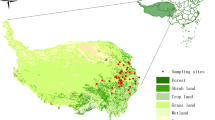
The high-altitude Zoige peatland (32° 100 N–34° 100 N, 101° 450 E–103° 250 E) is located on the eastern margin of the Qinghai–Tibet Plateau (Fig. 1). It is the highest and largest peat marsh in China and was, formed during the Early Holocene (9355 ± 115 BP) (Chai and Jin 1963). The Zoige peatland contains 26% of the total peatland area of China and 45% of the total peat reserves (Joosten 2004). Mean annual temperature in the area is 0.6–1.0 °C because of the high altitude (3400–3600 m), and the majority of the 580–860 mm of annual precipitation falls in summer (Chen and Bloemendal 1999, Zhang and Jiang 2008). The dominant vegetation in the majority of this area is perennial herb (Carex muliensis, Heleocharis uniglumis, Blysmus sinocompressus). The distribution of species is related to water table position, with Carex muliensis and Heleocharis uniglumis found mostly in wetter areas of the peatland while Kobresia pusilla and Potentilla fulgens prefer drier areas (Han et al. 2011, 2012). In this study, we explore biomass allocation and relationships between biomass allocation and environmental factors using data surveyed from 32 sites across the Zoige peatland during 2011–2012. The 32 sites essentially represent the major plant communities and surface water status variation of the Zoige alpine peatland.
Field Biomass Survey and Soil Features
The AGB and BGB samples were collected at 32 sites on the Zoige peatland in 2011 and 2012 during July and August, approximately the time of peak biomass production. At each 10 × 10 m site, the site surface bare spot area, water table and plant species composition were investigated. Within each of the 32 sample plots, five replicates of vegetation were harvested at the ground surface from 50 × 50 cm squares.
The BGB was estimated from three replicate 5 cm diameter soil cores collected at depth intervals of 10 cm down to the maximum root depth using a soil auger. The maximum root depth was determined by soil carbon storage estimation (0–2 m) and as described in another paper (Ma 2013). Dates from the upper 80 cm of soil were used here because almost all plant roots were concentrated within this zone in all samples from the Zoige peatland. Cores were washed through a 0.5 mm sieve to remove soil. Both AGB and BGB samples were oven-dried at 65 °C until a constant mass was reached. Total biomass was the sum of AGB and BGB.
Soil samples were collected at depth intervals of 10 cm to the maximum root depth using a soil auger. Six replicate 5 cm diameter soil cores were collected from each site. Three of these were oven-dried at 105 °C until a constant mass was reached to measure gravimetric soil water content and soil bulk density (BD). The remaining soil cores were air-dried for analysis of soil physicochemical properties. Soil organic carbon (SOC) was determined with the K2Cr2O7 titration method after digestion (Nelson and Sommers 1975). Total nitrogen (TN) was determined using the semi-micro-Kjeldahl method (Lu 1999). Total phosphorus (TP) was determined colorimetrically after wet digestion with H2SO4 plus HClO4 (Parkinson and Allen 1975).
Data Analysis
We calculated the mean values of AGB, BGB, total biomass and R/S ratio for all sampling sites. In addition, differences in AGB, BGB and R/S ratio between the seven types of plant communities were evaluated using ANOVA. To avoid pseudoreplication (Hurlbert 1984; Weishampel 2009), data from subplots of the same cover type within a given plot were pooled to form a single unit rather than treated as independent replicates.
Vertical root distributions were modeled using the asymptotic equation described by Gale and Grigal (1987) and Jackson (1996) (Eq. 1):
where Y is the cumulative percentage of root biomass from the soil surface to depth d (cm) and β is the estimated parameter. Values of β can range from 0.1 to 1, where 1 indicates that all production is at depth and 0.1 that all root production is at the surface (Murphy et al. 2009). We then calculated the percentage of root biomass found in the upper 30 cm of soil for each biome, based on their respective β values.
To study the relationships between AGB and BGB, the data were analyzed with a linear regression function. The analysis was conducted using SPSS software (SPSS version 18.0; SPSS Inc., Chicago, IL).
We then investigated the relationships between each environmental variables and AGB, BGB, total biomass, R/S ratio and β value. Multiple factor analysis (MFA) (BARALOTO 2011; Le et al 2008) was used to obtain an overview of the plots and the variables describing them. The advantage of MFA is that variables are separated into groups, each of which is given equal weight in the analysis. Environmental variables are separated into three groups: soil physical and chemical properties (soil water content, BD, SOC, soil TP, soil TN, C:N ratio and N:P ratio), site surface bare spot area and water table. The MFA analyses were conducted using the package FactoMineR (Le et al. 2008) in the R language and environment for statistical computing version 2.11.1 (R Core Development Team 2009). Finally, the relationships between AGB, BGB, total biomass, R/S ratio, β and environmental variables (soil water content, water table, SOC, TN, TP, C:N, N:P) were tested individually by regression analyses, and the correlations among environmental variables in the Zoige peatland were studied using correlation analyses. Regression analyses and correlation analyses were performed using SPSS software (SPSS version 18.0; SPSS Inc., Chicago, IL).
Results and Discussion
AGB, BGB and R/S Ratio
The dominant vegetation community types in the majority of this area are Carex muliensis, Heleocharis uniglumis, Blysmus sinocompressus, Carex lasiocarpa, Kobresia pusilla and others. The distribution of species is related to water table position, with Carex muliensis and Heleocharis uniglumis found mostly in permanent water areas of the peatland while Kobresia pusilla, Elymus nutans and Potentilla fulgens prefer drier areas. The environmental characteristics of the seven vegetation communities are given in Table 1.
The mean AGB, BGB, total biomass and R/S ratio values by total and major plant communities in the Zoige peatland are given in Fig. 2. The average AGB, BGB and total biomass in the Zoige alpine peatland were 341.01, 3262.93 and 3620.36 g m−2, respectively, and the average R/S ratio was 10.32. The CM and EPC communities had the largest BGB and total biomass followed by the CK and BCP communities, and finally the KP, EPA and PA communities. Total biomass and BGB were smallest in the PA community and largest in the CM community. Our survey also indicates that the CM, EPC, BCP and CK communities were usually located in areas with a permanent or seasonal surface water regime, and KP, EPA and PA communities were usually located at the humid soil surface.
We found that roots represent a quantitatively important biomass in the Zoige peatland. The R/S (BGB/AGB) ratio (10.32) for the Zoige peatland (Fig. 2) was higher than values of 0.28–1.38 observed in temperate peatland by Murphy (2009), 0.72–1.27 in an ombrotrophic bog by Moore et al. (2002) and 7.5 (1.5–21.9) in an average wetland by Cˇížková (1999). Mokany et al. (2006) reported that shrublands and grasslands possessed a much greater range in R/S ratios (0.34–26.03) than forests and woodlands. According to a global analysis of root distributions in terrestrial biomes conducted by Jackson (1996), R/S ratios were highest for tundra, grasslands and cold deserts (ranging from 4 to 7) out of all terrestrial ecosystems. Our estimated R/S ratio is comparable to estimates made for semi-wet arctic tundra ecosystems (R/S ratio of 11) by Dennis and Johnson (1970). This result is in accordance with the significant increase in the R/S ratio with declining temperature (Hui and Jackson 2005; Mokany et al. 2006) and increasing altitude (Leuschner et al. 2007; McCarthy and Enquist 2007). The higher R/S ratio in alpine peatland could be due to the relatively slow depletion of root carbohydrates in response to low respiration rates in cold environments (Yang et al. 2009) and might be associated with slower root turnover in colder regions (Gill and Jackson 2000; Yang et al. 2009).
The communities located in areas with occasional surface saturation have been shown to allocate more biomass to roots than the communities in areas where the surface is permanently wet and in areas where the surface is never saturated. Similar results have been reported by Olsrud and Christensen (2011) for a subarctic mire ecosystem, where the semi-wet ecosystem had a higher R/S ratio compared with the wet minerotrophic ecosystem. This shift in biomass allocation may arise from waterlogging (McFarlane et al. 2003) or adverse soil chemical conditions (Leuschner et al. 2007; Lambers et al., 1998).
Vertical Distribution of Roots
Asymptotic modeling of the vertical root distribution in the Zoige peatland produced an average biomass depth distribution parameter (β) of 0.921 (Fig. 3). The mean for the whole Zoige peatland was approximately 86% of roots were in the upper 30 cm of soil. Higher β values denote a greater proportion of roots at depth. Plant communities varied in their rooting depth distribution (Table 2). This difference in the seven communities could be attributed to their different species composition. Although absolute differences between the average β values of the seven communities appear to be small, these differences lead to large differences in vertical root distributions. For example, mean β values from the E community (β = 0.8383) and F community (β = 0.9442) indicate that approximately 99% of the roots of the E community are located above 30 cm depth but only 82% of roots of the F community are located above 30 cm depth. Differences in the parameter β do not reflect differences in root biomass but only in the vertical root distributions relative to depth.
The mean β in our alpine peatlands showed shallower rooting profiles (β = 0.921, r2 = 0.96) than those observed in global assessments of grasses (β = 0.952, r2 = 0.88) and values were closer to those observed in tundra ecosystems (β = 0.914, r2 = 0.91) (Jackson 1996). Plants are likely to concentrate their root production in the surface layers of the soil profile in response to high concentrations of N, P, and K (Jobbágy and Jackson 2001; Murphy and Moore 2010). In addition, oxygen deficiencies are least likely to occur in shallow soil layers of peatlands (Schenk and Jackson 2002). Our results indicate that there are some similarities in vertical root distribution profiles between high-altitude peatland ecosystems and ecosystems at high latitudes. The shallower rooting profiles in these ecosystems are partly caused by the physical barriers inhibiting root growth in cold regions, such as permafrost (Jackson 1996; Kane et al. 1992). In addition, waterlogging usually limits root growth (Kane et al. 1992) by reducing water absorption and transpiration (Ladiges et al. 1981). These, among other factors, contribute to the fact that tundra ecosystems are the most shallowly rooted of all biomes.
Allometric Relationship between AGB and BGB
There was a positive allometric relationship (P < 0.01) between AGB and BGB. The relationship between BGB and AGB in the Zoige peatland could be characterized by a linear regression function of BGB =4.81 AGB + 1749.36 (r2 = 0.19, P < 0.01) (Fig. 4). There have been numerous studies on the allometric relationships between aboveground biomass and belowground biomass (for example, Feliciano et al. 2014; Njana et al. 2015; Ward 2015). Our results suggested that belowground biomass could be estimated from the aboveground biomass in the Zoige peatland. This information is valuable given the difficulty in measuring belowground biomass.
Biomass Allocation and Environmental Factors
MFA, regression analyses and correlation analyses were used to examine the relationship between environmental variations in addition to their relationships with biomass. Two major gradients in environmental variables were observed and they explained 61.2% of the variance in the dataset (Fig. 5). The first MFA dimension has strong contributions of the principal components of the soil physical and chemical properties, water table and surface conditions group. In particular, this dimension represents a gradient of increasing N:P ratios, C:N ratios and soil water content. The second dimension has weak contributions of soil chemical properties. Mean total biomass and BGB were positively correlated with soil water content, water table, SOC, and soil C:N ratios, and tended to increase along the first dimension of soil water content and soil chemical properties and to decrease with increasing surface bare spot area (Dimension 2) (Fig. 5).
Ordination of variables and sample plots using multiple factor analysis (MFA), in which biomass variables are illustrated along the resulting dimensions. The panel shows the correlation circle with groups of soil, water, and surface variables. The meanings of the acronyms are as follows: Biomass depth distribution parameter (β); Total phosphorus (TP); Aboveground biomass (AGB); Total nitrogen (TN); Soil organic carbon (SOC); Water table (WT); Soil water content (SWC); Soil N:P ratio (N:P); Soil C:N ratio (C:N); Total biomass (TB); Belowground biomass (BGB); Belowground biomass: Aboveground biomass (R:S); Bare spot area (BSA); Soil bulk density (BD)
Both AGB and BGB were positively correlated with the water table (r2 = 0.167, P < 0.05 for AGB, r2 = 0.320, P < 0.001 for BGB) and soil C:N ratio (r2 = 0.138, P < 0.05 for AGB, r2 = 0.418, P < 0.001 for BGB) (Table 3). However, the R/S ratio did not show any significant change along the gradient of the water table but did show a significant change with soil C:N ratio. This indicates that the rising water table did not initiate the reduction in the aboveground biomass.
Water conditions were positively correlated with total nitrogen (r2 = 0.45, P < 0.05 for water table, r2 = 0.59, P < 0.01 for soil water content) and soil organic carbon (r2 = 0.65, P < 0.01 for water table, r2 = 0.78, P < 0.01 for soil water content) (Table 4). Similar results from previous studies have shown that the increase in soil water availability may accelerate soil N cycling (Lü et al. 2014; Van Groenigen et al. 2014). Moreover, water availability might increase fine root production, allowing plants to explore more of the soil volume for available N (Li et al. 2011; Lü et al., 2014).
Moreover, both SOC (r2 = 0.15, P < 0.05) and soil water content (r2 = 0.485, P < 0.001) were positively correlated with BGB (Table 3). In addition, soil water content showed a significant positive correlation with soil organic carbon content (r2 = 0.78, P < 0.01) (Table 4). This result indicated that soil water content, soil organic carbon and their interaction could influence belowground biomass. Correlation analysis further confirmed that the water conditions, soil organic carbon and soil nitrogen were the main factors that influenced the plant biomass and biomass allocation in the Zoige peatland. These results suggested that allocation patterns were different in response to differences in water and nutrient availability.
Conclusion
This study is the first to document information on biomass allocation and its relationship with environmental factors in alpine peatlands on the Qinghai–Tibetan Plateau. In the Zoige alpine peatland, the average AGB, BGB and total biomass were 341.01, 3262.93 and 3620.36 g m−2, respectively, and the R/S ratio was 10.32. The CM and EPC communities had the largest BGB and total biomass, followed by the CK and BCP communities, and finally the KP, EPA and PA communities. Total biomass and BGB were smallest in the PA community and largest in the CM community. We found that the overall R/S ratio (10.32) in alpine peatlands was higher than that observed in temperate peatland (0.28–1.38) and is comparable to estimates made in a wetland arctic tundra ecosystem (R/S ratio of 11). Zoige peatlands have a much shallower root distribution than temperate grasslands, with 86% of roots in the top 30 cm of soil. This unique distribution of roots in alpine peatlands should, therefore, be incorporated into biogeochemical models that examine the feedbacks of alpine peatland vegetation to climatic change, because current global biogeochemical models never consider such a pattern of root distribution (Jackson 1996; Schenk and Jackson 2002). There was a positive allometric relationship (P < 0.01) between AGB and BGB. The water conditions, soil organic carbon and soil nitrogen were the main factors that influenced plant biomass and their biomass allocation in the Zoige peatland.
References
Australian Greenhouse Office (2002) Greenhouse gas emissions from land use change in Australia: an integrated application of the National Carbon Accounting System. Australian Greenhouse Office, Canberra
Bai J, Hua YH, Cui BS, Wang QG, Chen H (2008) Changes in landscape pattern of alpine wetlands on the Zoige plateau in the past four decades. Acta Ecologica Sinica 28:2245–2252
Baraloto C, Rabaud S, Molto Q (2011) Disentangling stand and environmental correlates of aboveground biomass in Amazonian forests. Global Change Biology 17(8): 2677-2688
Blodau C(2002) Carbon cycling in peatlands A review of processes and controls. Environmental Reviews 10(2):111-134
Byrd K, Blanchard S, Schile L, Kolding S, Kelly M, Windham-Myers L, Miller R (2011) Advanced remote sensing to quantify temperate peatland capacity for belowground carbon capture. AGU Fall Meeting 0611
Chai X,Jin SR(1963)Types and and Development of the Ruoergai Plateau. Acta Geographica Sinica(3): 219-240
Chen F, Bloemendal J (1999) An 800 ky proxy record of climate from lake sediments of the Zoige Basin, eastern Tibetan plateau. Palaeogeography Palaeoclimatology Palaeoecology 151(4):307–320
Chen H, Wu N, Gao Y, Wang Y, Luo P, Tian J (2009) Spatial variations on methane emissions from Zoige alpine wetlands of Southwest China. Science of the Total Environment 407:1097–1104
Chen H, Wu N, Wang Y, Gao Y, Peng C (2011) Methane fluxes from alpine wetlands of Zoige plateau in relation to water regime and vegetation under two scales. Water, Air, and Soil Pollution 217:173–183
Čížková H (1999) Growth dynamics and ecophysiology of Phragmites in relation to the climatic conditions in boreal-Mediterranean and oceanic-continental gradients. In:Final Project for contracts ENV4-CT95-0147 and IC20-CT-960020 report. Denmark, University of Aarhus
Dennis JG, Johnson PL (1970) Shoot and rhizome-root standing crops of tundra vegetation at barrow, Alaska. Arctic and Alpine Research 2:253–266
Eamus D, Chen X, Kelley G (2002) Root biomass and root fractal analyses of an open eucalyptus forest in a savanna of North Australia. Australian Journal of Botany 50:31–41
Enquist BJ, Niklas KJ (2002) Global allocation rules for patterns of biomass partitioning in seed plants. Science 295:1517–1520
Feliciano EA, Wdowinski S, Potts MD (2014) Assessing mangrove above-ground biomass and structure using terrestrial laser scanning: a case study in the Everglades National Park. Wetlands 34(5):955–968
Finér L, Laine J (2000) The ingrowth bag method in measuring root production on peatland sites. Scandinavian Journal of Forest Research 15:75–80
Gale MR, Grigal DF (1987) Vertical root distributions of northern tree species in relation to successional status. Canadian Journal of Forest Research 17(8):829-834
Genet A, Wernsdörfer H, Jonard M, Pretzsch H, Rauch M, Ponette Q, Nys C, Legout A, Ranger J, Vallet P (2011) Ontogeny partly explains the apparent heterogeneity of published biomass equations for Fagus sylvatica in Central Europe. Forest Ecology and Management 261:1188–1202
Gill RA, Jackson RB (2000) Global patterns of root turnover for terrestrial ecosystems. The New Phytologist 147:13–31
Gorham E (1991) Northern peatlands: role in the global carbon cycle and probable response to climate warming. Ecological Applications 1:182–193
Han DY, Yang YX, Yang Y (2012) Changes of plant species diversity and interspecific correlation in a degraded swamp community along drainage gradients on the Zoigê plateau of China. Chinese Journal of Plant Ecology 36(5):411–419 (in Chinese)
Han DY, Yang YX, Yang Y, Ke L (2011) Species composition and succession of swamp vegetation along grazing gradients in the Zoige Plateau, China. Actaecologica Sinica 31(20) (in Chinese)
Hui DF, Jackson RB (2005) Geographical and interannual variability in biomass partitioning in grassland ecosystems: a synthesis of field data. The New Phytologist 169:85–93
Hurlbert SH (1984) Pseudoreplication and the design of ecological field experiments. Ecological Monographs 54:187–211
IPCC (2003) Good practice guidance for land use, land-use change and forestry. Intergovernmental Panel on Climate Change Geneva, Switzerland
Jackson (1996) A global analysis of root distributions for terrestrial biomes. Oecologia 108(3):389–411
Jobbágy EG, Jackson RB (2001) The distribution of soil nutrients with depth: global patterns and the imprint of plants. Biogeochemistry 53:51–77
Joosten H (2004) The IMCG global peatland database www.imcg.net/gpd/gpd.htm.
Kane DL, Hinzman LD, Woo M, Everett KR (1992) Arctic ecosystems in a changing climate. Academic Press, San Diego, pp 35–51
Kobe K, Iyer M, Walters MB (2010) Optimal partitioning theory revisited: nonstructural carbohydrates dominate root mass responses to nitrogen. Ecology 91:166–179
Ladiges PY, Foord PC, Willis R (1981) Salinity and waterlogging tolerance of some populations of Melaleuca ericifolia Smith. Australian Journal of Ecology 6:203–215
Lambers H, Chapin III FS, Pons TL(1998) Photosynthesis, respiration, and long-distance transport. Plant physiological ecology 10-153
Lê S, Josse J, Husson F (2008) FactoMineR: an R package for multivariate analysis. Journal of statistical software 25(1): 1-18
Leuschner C, Moser G, Bertsch C, Röderstein M, Hertel D (2007) Large altitudinal increase in tree root/shoot ratio in tropical mountain forests of Ecuador. Basic and Applied Ecology 8:219–230
Li JZ, Lin S, Taube F, Pan QM, Dittert K (2011) Above and belowground net primary productivity of grassland influenced by supplemental water and nitrogen in Inner Mongolia. Plant and Soil 340:253–264
Lu R (1999) Analytical methods of soil agrochemistry. China Agricultural Science and Technology Press, Beijing, pp 85–96
Lü X T, Dijkstra F A, Kong D L (2014) Plant nitrogen uptake drives responses of productivity to nitrogen and water addition in a grassland. Scientific reports, 2014, 4
Ma QF (2013) Ecosystem carbon storage in Zoige alpine marsh (Doctoral dissertation)
McCarthy M, Enquist B (2007) Consistency between an allometric approach and optimal partitioning theory in global patterns of plant biomass allocation. Functional Ecology 21:713–720
McConnaughay KDM, Coleman JS (1999) Biomass allocation in plants: ontogeny or optimality? A test along three resource gradients. Ecology 80:2581–2593
McFarlane N, Ciavarella T, Smith KF (2003) The effects of waterlogging on growth, photosynthesis and biomass allocation in perennial ryegrass (Lolium perenne L.) genotypes with contrasting root development. The Journal of Agricultural Science 141:241–248
Miller CA (2011) The Effect of Long-Term Drainage on Plant Community Composition, Biomass, and Productivity in Boreal Continental Peatlands (Doctoral dissertation)
Mokany K, Raison R, Prokushkin AS (2006) Critical analysis of root: shoot ratios in terrestrial biomes. Global Change Biology 12:84–96
Moore TR, Bubier JL, Frolking SE, Lafleur PM, Roulet NT (2002) Plant biomass and CO2 exchange in an ombrotrophic bog. Journal of Ecology 90:25–36
Murphy MT, Moore TR (2010) Linking root production to aboveground plant characteristics and water table in a temperate bog. Plant and Soil 336:219–231
Murphy (2009a) Variations in above- and below-ground vascular plant biomass and water table on a temperate ombrotrophic peatland. Botany 87:845–853
Murphy M, Laiho R, Moore TR (2009b) Effects of water table drawdown on root production and aboveground biomass in a boreal bog. Ecosystems 8:1268–1282
Nelson D, Sommers L (1975) A rapid and accurate procedure for estimation of organic carbon in soils. Proceedings of the Indiana Academy of Sciences 84:456–462
Njana MA, Eid T, Zahabu E (2015) Procedures for quantification of belowground biomass of three mangrove tree species. Wetlands Ecology and Management 23(4):749–764
O’Driscoll C, Rodgers M, O’Connor M, Asam ZZ, de Eyto E, Poole R, Xiao L (2011) A potential solution to mitigate phosphorus release following clearfelling in peatland forest catchments. Water, Air, and Soil Pollution 221:1–11
Olsrud M, Christensen TR (2011) Carbon partitioning in a wet and a semiwet subarctic mire ecosystem based on in situ 14C pulse-labelling. Soil Biology and Biochemistry 43(2):231–239
Parkinson J, Allen S (1975) A wet oxidation procedure suitable for the determination of nitrogen and mineral nutrients in biological material. Communications in Soil Science and Plant Analysis 6(1):1–11
R Development Core Team (2009) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Schenk HJ, Jackson RB (2002) The global biogeography of roots. Ecological Monographs 72:311–328
Snowdon P, Eamus D, Gibbons P (2000) Synthesis of Allometrics, review of root biomass and Design of Future Woody Biomass Sampling Strategies. In: NCAS technical report 17. Canberra, Australian Greenhouse Office
Van Groenigen KJ, Qi X, Osenberg CW et al (2014) Faster decomposition under increased atmospheric CO2 limits soil carbon storage. Science 344(6183):508–509
Weishampel P, Kolka R, King JY (2009) Carbon pools and productivity in a 1-km2 heterogeneous forest and peatland mosaic in Minnesota, USA. Forest Ecology and Management 257(2):747-754
Ward PJ (2015) Prediction intervals: placing real bounds on regression-based allometric estimates of biomass. Biometrical Journal 57(4):695–711
Weltzin JF, Pastor J, Harth C, Bridgham SD, Updegraff K, Chapin CT (2000) Response of bog and fen plant communities to warming and water-table manipulations. Ecology 81:3464–3478
West GB, Brown JH, Enquist BJ (1999) A general model for the structure and allometry of plant vascular systems. Nature 400:664–667
Yang Y, Fang J, Ji C, Han W (2009) Above-and belowground biomass allocation in Tibetan grasslands. Journal of Vegetation Science 20:177–184
Zhang G, Jiang N (2008) Methanogenesis from methanol at low temperatures by a novel psychrophilic methanogen,“Methanolobus psychrophilus” sp. nov., prevalent in Zoige wetland of the Tibetan plateau. Applied and Environmental Microbiology 74(19):6114–6120
Acknowledgements
The authors would like to thank Luan Junwei for useful suggestions on improving the manuscript. This research was funded by Forestry industry research special funds for public welfare project “Typical lake and marsh ecosystem services evaluation research” (No. 201204201) and Jilin province science and technology office project “Jilin wetland ecosystem services valuation” (No. 2013020).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ma, Q., Cui, L., Song, H. et al. Aboveground and Belowground Biomass Relationships in the Zoige Peatland, Eastern Qinghai–Tibetan Plateau. Wetlands 37, 461–469 (2017). https://doi.org/10.1007/s13157-017-0882-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13157-017-0882-8