Abstract
Salinity and inundation influence both wetland plant species diversity and productivity and are likely to change with predicted climate change. We investigated the degree to which these factors affect aboveground annual net primary production (ANPP) of pickleweed, Sarcocornia pacifica, at three tidal marshes spanning a salinity gradient in the San Francisco Bay Estuary, California, USA, including two natural marshes and one restored marsh. We collected S. pacifica biomass four times throughout the growing season, with roughly half of the collections in well-drained areas near channels and half away from channels, in areas of poor drainage. We examined the effects of site, channel drainage, and pore-water salinity on ANPP. Plots in well-drained areas had significantly greater biomass than those in poorly-drained areas. Most striking was the interaction between soil salinity and marsh drainage on aboveground productivity; salinity had a strong negative impact on ANPP in poorly-drained plots but had no effect in well-drained plots. In a recently-restored brackish marsh, productivity decreased with increasing distance to the nearest channel. These results suggest that within site hydrological dynamics have a strong influence over S. pacifica ANPP, with potential implications for the likely effects of shifts in salinity and inundation associated with climate change.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Salinity and inundation regimes are two critical factors influencing vegetation distribution and productivity in salt and brackish marshes (Sanchez et al. 1996; Mendelssohn and Morris 2000). Tidal fluctuations, local precipitation, and freshwater inputs drive most variations in salinity and inundation across tidal marshes and can influence vegetation through both primary tidal movement and secondary water-logging of marsh soils, creating a large range of hydrologic microhabitats for vegetation growth (Ungar 1991). Infrequent tidal flushing combined with evapotranspiration results in more saline soil conditions and, thus, more stressful growing conditions, with likely interactions among these factors. These interactions are amplified in estuarine Mediterranean-type salt and brackish marshes where little to no rainfall occurs during the growing season, when water salinity can range between 5‰ in February and 25‰ in August (L. Schile, unpublished data).
Both salinity and inundation regimes within tidal marshes are likely to be affected by future climate change through increases in rates of global sea-level rise and changes in local precipitation and watershed runoff (Adam 2002; Hopkinson et al. 2008; Vermeer and Rahmstorf 2009). Locally accelerated sea-level rise in a New England salt marsh caused a landward shift in distribution for low marsh plants, resulting in a decrease in area of high marsh plant communities (Donnelly and Bertness 2001), and increases in sea level have been found to reduce high salt marsh metabolism (Miller et al. 2001). Large-scale changes in tidal marshes have occurred in coastal areas with high rates of relative sea-level rise due to local subsidence, such as Louisiana and Chesapeake Bay (Baumann et al. 1984; Ward et al. 1998). Restoration of tidal marshes also will be affected by climate change, as sediment accretion and the relative elevation of the marsh are influenced not only by available sediment supply, but also by the rate of sea-level rise and successful plant recruitment (Callaway et al. 2007).
Tidal marsh plants have specific morphological and physiological characters that enable them to persist in saline and regularly inundated environments; however, there are limitations associated with growing in such environments that can result in reduced performance and even mortality. Salt is directly toxic to most plant species, but it also alters a plant’s osmotic potential, modifying water uptake in the short term. In more stressful saline conditions, this can cause dehydration and wilting, as well as reduced CO2 uptake (Rozema 1991; Ewing et al. 1995). Some species, like pickleweed, store salts or employ other molecules to change osmotic potential. Under water-logged conditions, hydrogen sulfide or other toxins can be produced as a result of the reduced soil environment and adversely affect rhizomes and roots (Cronk and Fennessy 2001) and limit nutrient uptake, growth, and biomass (Mendelssohn and Seneca 1980; Koch and Mendelssohn 1989), among other deleterious effects. The dominant effects of flooding are excessive inundation and the reduction in soil aeration (Chapman 1974; Ungar 1991). Marsh plants tolerate low oxygen conditions by morphological adaptations like aerenchyma and physiological modifications, and their roots oxygenate the rhizosphere, all of which facilitate growth in environments that most plants cannot tolerate. Increased inundation combined with increased salinity compounds stressful conditions for plant growth, which in turn could have cascading effects on the wetland ecosystem.
We focused our investigation on the combined effects of increased inundation and salinity on the above-ground productivity of Sarcocornia pacifica (Standl.) A. J. Scott (perennial pickleweed; formerly Salicornia virginica). This cosmopolitan species dominates the majority of marsh area in San Francisco Bay salt marshes as well as in other Pacific Coast marshes (Macdonald 1977) and is found throughout many of North American salt and brackish marshes (Cronk and Fennessy 2001). Congeners are also important in Mediterranean-type marshes throughout the world (Ibanez et al. 1999; Alvarez-Rogel et al. 2007; Saintilan 2009). Sarcocornia pacifica has a high salt tolerance (Barbour and Davis 1970) and a broad elevation range (L. Schile, unpublished data). Despite the fact that S. pacifica is the dominant plant in Pacific coast salt marshes and other similar perennial shrubs dominate many Mediterranean-type marshes, only a small number of studies have addressed productivity of perennial halophytic shrubs in tidal marshes (e.g., Mahall and Park 1976a; Balling and Resh 1983; Curco et al. 2002; Scarton et al. 2002). Considering that primary productivity is an important metric of ecosystem services, and that few papers have examined the combined effects of salinity and inundation on plant productivity in Mediterranean-type salt marshes, we investigated primary production both within a site and at a broader landscape context to add to our understanding of this basic ecosystem function.
For our investigation, we analyzed trends in S. pacifica productivity across three sites characterized by differences in drainage and salinity and monitored changes in pore-water salinity at different depths. Based on research with this and other salt marsh species, we hypothesized that increases in either salinity or inundation rates would lead to decreases in productivity of S. pacifica. Specifically, we asked 1) does productivity decrease at different marsh sites along an increasing estuarine salinity gradient; 2) does productivity decrease with increasing inundation; and 3) do salinity and drainage interact on their influence on productivity within and across multiple sites?
Methods
Site Description
The San Francisco Bay Estuary (hereafter referred to as “Estuary”) covers approximately 4,096 km2 of the central California coastal region and includes a broad mix of salt, brackish, and tidal freshwater marsh ecosystems (Atwater et al. 1979; Josselyn 1983). The marsh landscape is a complex mosaic of remaining historic marshes, recently developed marshes, restored marshes, and potentially restorable diked bayland sites, located within one of the country’s largest urban areas (Baye et al. 2000). Despite impacts from development and exotic species, these ecosystems are of critical regional importance for biodiversity, harboring a number of rare plant and animal species, including almost 50 species of special conservation concern (Olofson 2000).
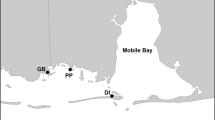
Total standing biomass of S. pacifica was monitored for one growing season at three tidal marshes located along a salinity gradient in northern San Francisco Bay: China Camp State Park, Coon Island, and Pond 2a (Fig. 1). China Camp (122°29′31″W, 38°0′47″N) is a natural salt marsh located along the fringes of San Pablo Bay and is one of two sites in the San Francisco Bay National Estuarine Research Reserve (NERR). The bulk of the marsh plain is dominated by S. pacifica, bordered along the Bay edge by Spartina foliosa Trin. Coon Island (122°19′30″W, 38°11′43″N) is a natural brackish marsh located on the Napa River that is dominated by S. pacifica, Bolboschoenus maritimus Palla (alkali bulrush), Typha angustifolia L. (cattail), and Schoenoplectus acutus (Muhlenberg ex Bigelow) Á. Löve & D. Löve, (tule), with S. foliosa around the perimeter of the site. Pond 2a (122°19′15″W, 38°9′10″N), also located off of the Napa River, is a former salt pond that was restored in 1997. Presently, Pond 2a is almost entirely vegetated by B. maritimus, with small patches of S. pacifica, S. foliosa, Schoenoplectus americanus (Persoon) Volkart ex Schinz & R. Keller (common three square or chairmaker’s bulrush), and T. angustifolia. All sites exhibit a mixed semi-diurnal tide (MHW 1.8 m). The climate is characterized as a Mediterranean-type climate with almost the entire annual rainfall occurring between October and April (Josselyn 1983).
Experimental Design
In February and early March of 2005, prior to the initiation of spring growth, we established 20 1.5 × 1.5-m plots at each site in visually monospecific S. pacifica stands of roughly uniform height and density; at this time plants had little to no green succulent tissue. Plots were haphazardly selected within the monospecific stands. The spatial coordinates were recorded for each plot using a WAAS-enabled (wide area augmentation system) Garmin GPSmap 76S handheld unit, the corners were staked, and each plot was divided into nine 50 × 50-cm subplots.
At Coon Island and China Camp, half of the plots were randomly located in areas of frequent tidal flushing, adjacent or close to channels (hereafter well-drained plots), and the other half were located on the inner marsh plain, in sites where we observed several cm of standing water that persisted between tides (poorly-drained plots). However, after plots were established at Coon Island, two of the ‘poorly-drained’ plots were discovered to be next to channels and were thus reclassified as well-drained. Due to the patchy distribution of S. pacifica at Pond 2a and in order to reduce trampling of newly-colonizing vegetation, plots were established near a boardwalk that spanned the 2.5 km-wide site rather than in areas that were specifically well or poorly drained, and we used the distance to the nearest creek as a metric for plot drainage at this site.
Data Collection
We measured pore-water salinity, S. pacifica biomass, and average height four times throughout the growing season in 2005. Biomass was collected at China Camp on February 25, May 16, July 12, and August 22; at Coon Island on February 22, June 9, July 15, and August 23; and at Pond 2a on March 2, May 17, July 13, and September 3. The subplot for each data collection event was randomly selected from unharvested subplots; only the center and corner subplots were designated for destructive sampling to minimize the effects of biomass removal on subsequent subplot collections.
After each biomass collection, a soil core was removed from within the subplot to measure soil salinity at average depths of 5, 10, and 20 cm. Salinity was determined by extracting water from a 2.8-cm diameter soil core using a 20-ml syringe with filter paper at the tip and measuring the pore-water salt content with a handheld refractometer calibrated for salinity (Reichert Analytical Instruments, Depew, NY, USA). We recorded the average plant height within a subplot before all aboveground biomass was removed and placed the harvested plant material in plastic bags for sorting in the lab. Plant material was rinsed with fresh water to remove salt, dirt, and any other debris; sorted by species; placed into paper bags; dried to a constant weight at 60°C for 2 days; and weighed.
Many studies report biomass as standing crop based on a single collection of plant material. This method does not account for variability in biomass over time, which is particularly important for perennial species, and increases the chances of failing to sample at peak biomass. We defined biomass as the mass resulting from a single material collection and annual net primary productivity (ANPP) as the peak biomass minus the initial biomass (Cronk and Fennessy 2001).
Data Analysis
All data were analyzed using SAS version 9.1 (SAS Institute 2006). With the exception of the single nonparametric test that was run, all data fit the assumptions for conducting parametric analyses (homogeneity of variance and normality). Because we could not decisively distinguish well-drained and poorly-drained areas at Pond 2a, we analyzed its salinity, biomass, height, and ANPP data separately from Coon Island and China Camp.
Changes in pore water salinity over time at depths of 5, 10, and 20 cm were analyzed using a repeated measures analysis of variance (rmANOVA). All repeated measures analyses were conducted using MANOVA test criteria in PROC GLM. Soil salinity data from Pond 2a were averaged across all plots for each depth and analyzed with a rmANOVA. For all subsequent analyses incorporating salinity data, we chose the salinity values taken at the 10-cm depth during the last sampling period because the majority of the S. pacifica root system occurs within the top 15 cm of soil (Mahall and Park 1976a) and to capture the maximum salinity values measured during the experiment.
Changes in standing biomass over time were analyzed with a rmANOVA using data averaged 1) within well-drained and poorly-drained areas at Coon Island and China Camp and, in a separate analysis, 2) across all plots at Pond 2a. We conducted a Kruskal-Wallis test to determine if the time of peak biomass differed among sites and tested two methods of calculating ANPP for S. pacifica: highest biomass minus lowest biomass and biomass from the first sampling event minus the last. For all sites, aboveground ANPP calculated by subtracting the initial biomass from the largest biomass per plot was greater than subtracting the first sampling period from the last (China Camp, t 19 = 7.14, p < 0.001; Pond 2a, t 19 = 4.62, p < 0.001; Coon Island, t 19 = 2.76, p = 0.010). In light of the observation that the harvest method can result in an underestimate of productivity (Onuf et al. 1978; Day et al. 1989) and to incorporate the differences in timing of peak biomass, we chose the method of using the highest and lowest biomass values to estimate ANPP. We tested for the effects of site and drainage on ANPP at Coon Island and China Camp using an Analysis of Covariance test with salinity as the covariate.
Because the conditions at Pond 2a were not comparable to the other sites due to its recent restoration, we investigated the effect of distance to the nearest channel on ANPP, salinity, and average plant height. Using ArcMap version 9.1 (ESRI 2007), a channel network layer was created based on an ortho-rectified color infra-red image flown in 2004, and the distance in meters between each productivity plot and the nearest channel in the layer was calculated using the ‘near’ function under ‘Proximity’ tools. Using a simple linear regression, we individually tested for effects of distance from channel on salinity, ANPP, and the maximum average plant height, in addition to any effects of salinity on ANPP.
At China Camp and Coon Island, we chose the maximum value of the average plant height per plot from across the four sampling periods and conducted a multiple linear regression to examine the effects of salinity and plot drainage on average plant height.
Results
Soil Salinity
Soil salinity increased over time at China Camp (Fig. 2a) and Coon Island (Fig. 2b), and the rate differed depending on soil depth, drainage, and site (Table 1). At China Camp, the average soil salinity ranged from 33‰ in February to 43‰ in August; at Coon Island, the salinity values ranged from 12‰ in February to 18‰ in August; and at Pond 2a, salinity values ranged from 12‰ in early March to 22‰ in early September. No significant interaction among time, drainage, and depth, or time, drainage, and site was found. Salinity changes over time varied by depth and across sites (Table 1). Salinity at each depth was greater at China Camp than Coon Island (Table 1). In addition, poorly-drained areas had greater salinity than well-drained areas for each sampling period (p < 0.001 for all comparisons). At Pond 2a, salinity significantly increased over time at each soil depth (F 6,108 = 3.94, p = 0.001; Fig. 2c); however, salinity levels at depths of 5 and 10 cm were only significantly different from those at 20 cm during the first sampling period (p < 0.002 for both comparisons), after which no significant differences among depths were found (p > 0.15). Salinity at each plot within Pond 2a averaged over time significantly increased as the distance to the nearest channel increased (Fig. 3; F 1,18 = 30.96, p < 0.001).
Changes in Biomass over Time
At China Camp and Coon Island, biomass of S. pacifica increased significantly over time, and the increase varied with site and drainage (Table 1; Fig. 4). No significant interaction occurred among time, drainage, and site (Table 1). At China Camp, biomass was significantly greater in well-drained plots versus poorly-drained plots at all sampling events except for February (p < 0.030 for all significant comparisons); however, biomass in well-drained plots at Coon Island was significantly greater than in poorly-drained plots only in July (p = 0.008). Biomass was greater at Coon Island than China Camp in June and September but not in February or July (p < 0.001; p < 0.001; p = 0.21; p = 0.45, respectively). Additionally, average S. pacifica biomass significantly increased over time at Pond 2a (F 3,17 = 12.56, p < 0.001; Fig. 4). The timing of peak biomass differed among sites (Kruskal-Wallis test; χ2 2 = 11.63, p = 0.003): plots in China Camp had peak biomass in July (Fig. 4), and plots in Pond 2a peaked in late August/early September (Fig. 4); whereas, Coon Island’s peak biomass varied depending on inundation (Fig. 4).
Aboveground Annual Net Primary Productivity
ANPP was significantly affected by salinity, drainage, and site at China Camp and Coon Island (F 7,32 = 5.56, p < 0.001; Fig 5a). The model was reduced to include only significant terms. As a result, the three-way interaction and the interaction between site and salinity were removed. The reduced model included site, salinity, drainage, and the individual interactions of drainage with site and salinity. ANPP decreased with increasing salinity in poorly-drained plots but not in well-drained plots (Table 1; Fig. 5a), and the slopes of the regression lines were significantly different (poorly-drained slope = −19.8; well-drained slope = 0.4; t 34 = 2.11, p = 0.025). Additionally, the effect of plot drainage on ANPP varied between sites (data not shown; Table 1). At Pond 2a, ANPP was not significantly affected by salinity or distance to nearest channel (data not shown; F 1,18 = 0.13, p = 0.73; F 1,18 = 0.32, p = 0.58, respectively).
Plant Height
At China Camp and Coon Island, the maximum average height decreased with increasing salinity in both well-drained and poorly-drained areas, but the relationship was stronger in poorly-drained plots (Table 1; Fig 5b; R 2 = 0.23 and R 2 = 0.68, respectively). Additionally, the maximum average height increased significantly with ANPP in poorly-drained areas but not in well-drained areas (data not shown; Table 1; poorly-drained: R 2 = 0.41; well-drained: R 2 = 0.11). At Pond 2a, the maximum average plant height decreased slightly with increasing distance from the nearest channel, but the effect was not significant (data not shown; F 1,20 = 2.48, p = 0.13).
Discussion
The most striking influence on S. pacifica productivity was the combined effect of soil salinity and marsh drainage. Previous studies have evaluated effects of salinity and drainage simultaneously from oligohaline to salt marshes on the Gulf and Atlantic Coasts (see below) or in greenhouse studies (Kuhn and Zedler 1997; Callaway and Zedler 1998), but no field studies have evaluated combined effects on Mediterranean salt marsh shrubs, such as S. pacifica. Spalding and Hester (2007) evaluated effects of inundation and salinity on three oligohaline species (Spartina patens (Aiton) Muhl., Panicum hemitomon Schult., and Sagittaria lancifolia L.) from Louisiana; they found differential effects on each species and a number of interactions among salinity and inundation. Similar to our findings, the effects of increased salinity were exacerbated by increased inundation for P. hemitomon and S. lancifolia. Similar interactions were found by Willis and Hester (2004) where inundation increases caused no reduction in P. hemitomon biomass, unless accompanied by increased salinity.
As was found for differences in growth forms for Spartina (Valiela et al. 1978; Mendelssohn and Morris 2000), tidal channels are important in modifying the marsh plain environment, and subsequently Sarcocornia productivity. Bertness et al. (1992) also demonstrated that Iva frutescens L., a high marsh shrub in New England salt marshes, was able to grow to lower elevations adjacent to creeks. Tidal channels are the primary conduits for water, sediments, and nutrients and have local effects on drainage and soil chemistry. The influence of channels in Pacific Coast tidal marsh systems has been well-identified for a number of processes (Zedler et al. 1999; Sanderson et al. 2000). In San Francisco Bay Estuary marshes, for example, poorly-drained areas away from tidal channels had greater salinity levels than those located in areas with frequent tidal flushing (Figs. 2 and 3; Ustin et al. 1982; Balling and Resh 1983). Regionally, salinity decreases with distance from the Pacific Ocean in the Estuary, and, within a site, decreases with frequency of tidal flushing.
Proximity to tidal channels influences a number of biological responses, such as species composition (Hopkins and Parker 1984; Sanderson et al. 2000), and, specifically for S. pacifica, biomass (Fig. 4), ANPP (Fig. 5a), and height (Fig. 5b). Productivity was particularly influenced by the degree of drainage (Fig. 5a). Two other analogous patterns correspond with these results. Artificial mosquito-control ditches might be expected to influence drainage and salinity in a manner similar to natural tidal channels, and biomass was found to be greater next to these ditches (Balling and Resh 1983). However, Balling and Resh (1983) found lower productivity next to a natural channel compared to productivity on the marsh plain and along a mosquito ditch. This effect could be due to lack of replication, since only one site within one marsh was sampled. While S. pacifica is most abundant around mean higher high water (MHHW), inundation impacts Sarcocornia growth and survival at low marsh elevations where dominance shifts to S. foliosa in San Francisco Bay marshes (Mahall and Park 1976a, c). Overall, in marshes along a salinity gradient in the Estuary, we found S. pacifica to be insensitive to changes in salinity as long as areas were well-drained, but productivity decreased with increasing salinity in areas with poorly-drained soils.
Effects of Individual Factors: Inundation and Salinity
As with other tidal marsh species, our data suggest that S. pacifica can tolerate a broad range of salinity and inundation but performs better at well-drained, tidally flushed sites. The relatively high elevation sites on the marsh plain in San Francisco Bay marshes are only infrequently covered by spring tides to shallow depths of 5–10 cm, and plants in these locations often were growing in 1–10 cm of standing water that was rarely flushed with tidal activity (L. Schile, personal observation). Water-logging of the soil and the creation of anaerobic conditions for longer periods of time would depress growth compared to well-flushed locations (Ungar 1991; Mendelssohn and Morris 2000). Sarcocornia pacifica responds to increased inundation by increasing the amount of aerenchymatous tissue (Seliskar 1985) but can lose competitive ability through delayed and occasionally reduced growth (Mall 1969). Increases in productivity at higher marsh elevations undoubtedly relates to tidal flushing that replenishes oxygenated water and removes anoxic water and sulfides, thereby converting waterlogged soils into conditions more optimal for plant growth (Pennings and Callaway 1992). Productivity also decreases with increased depth of tidal submergence adjacent to the low marsh in San Francisco Bay (Mahall and Park 1976c; Josselyn 1983). However, previous submergence studies in the Estuary have focused on the lower elevation limits of S. pacifica and its competition with low marsh species, with little attention given to the variable responses of S. pacifica where it dominates the marsh plain.
Sarcocornia pacifica typifies a plant that tolerates saline conditions; however, saline conditions have been observed to reduce its growth and stature (Josselyn 1983; Pearcy and Ustin 1984; Brenchley-Jackson 1992; Callaway and Zedler 1998). Mahall and Park (1976b) observed that S. pacifica mortality did not increase with higher salinity, but lower shoot growth tended to occur under more saline conditions. Zedler (1983) observed a 160% increase in S. pacifica productivity after a freshwater flood in a hypersaline southern California marsh, which could have been a result of the freshwater pulse or the presence of tidal flow. In our study, S. pacifica height increased with decreasing salinity, similar to the other studies that have measured S. pacifica height, biomass, or productivity (Zedler 1983; Seliskar 1985; Brenchley-Jackson 1992; Callaway and Zedler 1998, but see Zhang et al. 1997); however, salinity alone had little effect on aboveground productivity (Table 1).
Soil salinity is of particular interest in Mediterranean-type marshes due to high rates of summer evapotranspiration and extremely infrequent summer rains. At our sites, soil salinity varied throughout the growing season, increasing at all depths over time during the growing season. Soil salinity levels across all sites initially were greatest at the lowest depth, 20 cm, and gradually increased throughout the summer until the upper soil depths were equal or greater than the samples from 20-cm depth (Fig. 2). These trends are consistent with results from Mahall and Park (1976b) who also worked in a San Pablo Bay marsh. In this Mediterranean-type climate, the majority, if not all, of the rainfall occurs between October and April; during the rainy season (winter and early spring) pore water salinity decreases, and surface soil salinities can be reduced to lower levels than in deeper soil layers. Soil salinity increases over the spring and summer as the rains cease, freshwater inflow greatly decreases, temperatures rise, and evapotranspiration increases, creating the most stressful salinity conditions during the growing season.
Similar effects of individual factors of salinity and inundation have been observed with other Mediterranean perennial salt marsh shrubs. While positive effects on biomass have been observed for an annual (Cooper 1982) and a perennial (Barbour and Davis 1970) pickleweed species, the effect of salinity is negative at higher salinities (2.2% salt). Sarcocornia fruticosa (L.) A. J. Scott (formerly Salicornia fruticosa) in the Rhône Delta of France had higher ANPP in a site with regular sea water exchange compared to a nearby impounded site that was often hypersaline (Ibanez et al. 1999). Additionally, S. fruticosa growth in the Po Delta, Italy was influenced by increased inundation relative to other European sites, resulting in an increase in below-ground biomass, and height increased with increasing ANPP (Scarton et al. 2002). Modified hydrology in a Spanish salt marsh, lead to increases in S. fruticosa relative to most other species (Alvarez-Rogel et al. 2007). Arthrocnemum subterminale (Parish) Standl. (formerly Salicornia subterminalis) had decreased growth under prolonged periods of saturation, which affected both above and below ground biomass (Kuhn and Zedler 1997). In South Africa, Sarcocornia perennis (Mill.) A.J. Scott (formerly Salicornia perennis) growth was affected more by prolonged inundation than by high salinity levels (>35‰; Adams and Bate 1995).
Management Implications: Climate Change and Restoration
In light of increasing rates of sea-level rise (Church and White 2006; Church et al. 2008; Vermeer and Rahmstorf 2009), estuaries throughout the world will experience longer durations of inundation. Specific climate change scenarios for the Estuary watershed (Sierra Nevada Mountains) predict reduced snowpack and earlier snowmelt, which in turn will reduce freshwater flow in the late spring and summer (Hayhoe et al. 2004; Cayan et al. 2008b). Salinity regimes will be significantly altered in this Mediterranean-type climate because of reduced freshwater flow and increased evapotranspiration from warmer temperatures. Combined with increasing rates of sea-level rise, the Estuary’s tidal marshes are predicted to experience longer periods of inundation, increased summer salinities, and a larger reach of saline water into historically fresh portions of the estuary (Cayan et al. 2008a, b). The implications for the tidal marsh plant communities are profound, particularly for restored sites where newly recruiting vegetation is likely to be highly sensitive to increasing salinity and inundation (Callaway et al. 2007). Understanding the complex effects of inundation regimes and salinity gradients on current plant communities will aid in predicting changes in plant productivity and survival. Our results suggest a significant decrease in productivity and height with the combined stresses of prolonged inundation and salinity in a ubiquitous and tolerant plant species, changes that are likely to have cascading effects across all marsh and pelagic species.
References
Adam P (2002) Saltmarshes in a time of change. Environmental Conservation 29:39–61
Adams JB, Bate GC (1995) Ecological implications of tolerance of salinity and inundation by Spartina maritima. Aquatic Botany 52:183–191
Alvarez-Rogel J, Jimenez-Carceles FJ, Roca MJ, Ortiz R (2007) Changes in soils and vegetation in a Mediterranean coastal salt marsh impacted by human activities. Estuarine Coastal and Shelf Science 73:510–526
Atwater BF, Conard SG, Dowden JN, Hedel CW, MacDonald RL, Savage W (1979) History, landforms, and vegetation of the estuary’s tidal marshes. In: Conomos TJ (ed) San Francisco Bay: the urbanized estuary. Pacific Division, American Association for the Advancement of Science, San Francisco, pp 347–385
Balling SS, Resh VH (1983) The influence of mosquito control recirculation ditches on plant biomass, production and composition in two San Francisco Bay salt marshes. Estuarine Coastal and Shelf Science 16:151–161
Barbour MG, Davis CB (1970) Salt tolerance of five California salt marsh plants. The American Midland Naturalist 84:262–265
Baumann RH, Day JW Jr, Miller CA (1984) Mississippi deltaic wetland survival: sedimentation versus coastal submergence. Science 224:1093–1095
Baye PR, Faber PM, Grewell B (2000) Tidal marsh plants of the San Francisco Estuary. In: Olofson PR (ed) Bayland ecosystem species and community profiles: life histories and environmental requirements of key plants, fish, and wildlife. San Francisco Bay Regional Water Quality Control Board, Oakland, pp 9–33
Bertness MD, Wikler K, Chatkupt T (1992) Flood tolerance and the distribution of Iva frutescens across New England salt marshes. Oecologia 91:171–178
Brenchley-Jackson JE (1992) Factors controlling the vegetation dynamics of Spartina foliosa and Salicornia virginica in California’s coastal wetlands. Ph.D. Dissertation. University of California, Davis, CA
Callaway JC, Zedler JB (1998) Interactions between a salt marsh native perennial (Salicornia virginica) and an exotic annual (Polypogon monspeliensis) under varied salinity and hydroperiod. Wetlands Ecology and Management 5:179–194
Callaway JC, Parker VT, Vasey MC, Schile LM (2007) Emerging issues for the restoration of tidal marsh ecosystems in the context of predicted climate change. Madrono 54:234–248
Cayan DR, Bromirski PD, Hayhoe K, Tyree M, Dettinger MD, Flick RE (2008a) Climate change projections of sea level extremes along the California coast. Climatic Change 87:S57–S73
Cayan DR, Maurer EP, Dettinger MD, Tyree M, Hayhoe K (2008b) Climate change scenarios for the California region. Climatic Change 87:S21–S42
Chapman VJ (1974) Salt marshes and salt deserts of the world, 2nd edn. J. Cramer, Berlin
Church JA, White NJ (2006) A 20th century acceleration in global sea-level rise. Geophysical Research Letters 33:L01602. doi:10.1029/2005GL024826
Church JA, White NJ, Aarup T, Wilson WS, Woodworth PL, Domingues CM, Hunter JR, Lambeck K (2008) Understanding global sea levels: past, present and future. Sustainability Science 3:9–22
Cooper A (1982) The effects of salinity and waterlogging on the growth and cation uptake of salt marsh plants. The New Phytologist 90:263–275
Cronk JK, Fennessy MS (2001) Wetland plants: biology and ecology. Lewis Publishers, Boca Raton
Curco A, Ibanez C, Day JW, Prat N (2002) Net primary production and decomposition of salt marshes of the Ebre delta (Catalonia, Spain). Estuaries 25:309–324
Day JW Jr, Hall CAS, Kemp WM, Yánez-Arancibia A (1989) Estuarine ecology. Wiley, New York
Donnelly JP, Bertness MD (2001) Rapid shoreward encroachment of salt marsh cordgrass in response to accelerated sea-level rise. Proceedings of the National Academy of Sciences of the United States of America 98:14218–14223
ESRI (Environmental Science Research Institute) (2007) ArcMap version 9.1 Available from http://esri.com/. ESRI, Redlands, CA
Ewing K, McKee KL, Mendelssohn IA, Hester MW (1995) A comparison of indicators of sub-lethal nutrient stress in the salt marsh grass, Spartina patens. Environmental and Experimental Botany 35:331–343
Hayhoe K, Cayan D, Field CB, Frumhoff PC, Maurer EP, Miller NL, Moser SC, Schneider SH, Cahill KN, Cleland EE, Dale L, Drapek R, Hanemann RM, Kalkstein LS, Lenihan J, Lunch CK, Neilson RP, Sheridan SC, Verville JH (2004) Emissions pathways, climate change, and impacts on California. Proceedings of the National Academy of Sciences of the United States of America 101:12422–12427
Hopkins DR, Parker VT (1984) A study of the seed bank of a salt marsh in northern San Francisco Bay. American Journal of Botany 71:348–355
Hopkinson CS, Lugo AE, Alber M, Covich AP, Van Bloem SJ (2008) Forecasting effects of sea-level rise and windstorms on coastal and inland ecosystems. Frontiers in Ecology and the Environment 6:255–263
Ibanez C, Day JW, Pont D (1999) Primary production and decomposition of wetlands of the Rhone delta, France: Interactive impacts of human modifications and relative sea level rise. Journal of Coastal Research 15:717–731
Josselyn M (1983) The ecology of San Francisco Bay tidal marshes: a community profile. U.S. Fish and Wildlife Service, Division of Biological Services, Washington
Koch MS, Mendelssohn IA (1989) Sulfide as a soil phytotoxin: differential responses in two marsh species. Journal of Ecology 77:565–578
Kuhn NL, Zedler JB (1997) Differential effects of salinity and soil saturation on native and exotic plants of a coastal salt marsh. Estuaries 20:391–403
MacDonald KB (1977) Coastal salt marsh. In: Barbour M, Major J (eds) Terrestrial vegetation of California. Wiley, New York, pp 263–294
Mahall BE, Park RB (1976a) The ecotone between Spartina foliosa Trin. and Salicornia virginica L. in salt marshes of northern San Francisco Bay: I. Biomass and production. Journal of Ecology 64:421–433
Mahall BE, Park RB (1976b) The ecotone between Spartina foliosa Trin. and Salicornia virginica L. in salt marshes of northern San Francisco Bay: II. Soil water and salinity. Journal of Ecology 64:793–809
Mahall BE, Park RB (1976c) The ecotone between Spartina foliosa Trin. and Salicornia virginica L. in salt marshes of northern San Francisco Bay: III. Soil aeration and tidal immersion. Journal of Ecology 64:811–819
Mall RE (1969) Soil-water-salt relationships of waterfowl food plants in the Suisun Marsh of California. Wildlife Bulletin 1. California Department of Fish and Game, Sacramento
Mendelssohn IA, Morris JT (2000) Eco-physiological controls on the productivity of Spartina alterniflora Loisel. In: Weinstein MP, Kreeger DA (eds) Concepts and controversies in tidal marsh ecology. Kluwer Academic Publishers, Boston, pp 59–80
Mendelssohn IA, Seneca ED (1980) The influence of soil drainage on the growth of the salt marsh cordgrass Spartina alterniflora in North Carolina. Estuarine Coastal and Shelf Science 11:27–40
Miller WD, Neubauer SC, Anderson IC (2001) Effects of sea level induced disturbances on high salt marsh metabolism. Estuaries 24:357–367
Olofson PR (ed) (2000) Bayland ecosystem species and community profiles: life histories and environmental requirements of key plants, fish, and wildlife. San Francisco Bay Regional Water Quality Control Board, Oakland
Onuf CP, Quammen ML, Schaffer GP, Peterson CH, Chapman JW, Cernak J, Holmes RW (1978) An analysis of the values of central and Southern California coastal wetlands. In: Greeson PE, Clark JR, Clark JE (eds) Wetland functions and values: the state of our understanding. American Water Resources Association, Minneapolis, pp 186–199
Pearcy RW, Ustin SL (1984) Effects of salinity on growth and photosynthesis of three California tidal marsh species. Oecologia 62:68–73
Pennings SC, Callaway RM (1992) Salt marsh plant zonation: the relative importance of competition and physical factors. Ecology 73:681–690
Rozema J (1991) Growth, water and ion relationships of halophytic monocotyledonae and dicotyledonae: a unified concept. Aquatic Botany 39:17–33
Saintilan N (2009) Distribution of Australian saltmarsh plants. In: Saintilan N (ed) Australian saltmarsh ecology. CSIRO Publishing, Collingwood, pp 23–74
Sanchez JM, Izco J, Medrano M (1996) Relationships between vegetation zonation and altitude in a salt-marsh system in northwest Spain. Journal of Vegetation Science 7:695–702
Sanderson EW, Ustin SL, Foin TC (2000) The influence of tidal channels on the distribution of salt marsh plant species in Petaluma Marsh, CA, USA. Plant Ecology 146:29–41
SAS Institute (2006) SAS, version 8.2. SAS Institute, Inc., Cary
Scarton F, Day JW, Rismondo A (2002) Primary production and decomposition of Sarcocornia fruticosa (L.) Scott and Phragmites australis Trin. ex Steudel in the Po Delta, Italy. Estuaries 25:325–336
Seliskar DM (1985) Morphometric variations of five tidal marsh halophytes along environmental gradients. American Journal of Botany 72:1340–1352
Spalding EA, Hester MW (2007) Interactive effects of hydrology and salinity on oligohaline plant species productivity: implications of relative sea-level rise. Estuaries and Coasts 30:214–225
Ungar IA (1991) Ecophysiology of vascular halophytes. CRC Press, Boca Raton
Ustin SL, Pearcy RW, Bayer DE (1982) Plant water relations in a San-Francisco Bay salt marsh. Botanical Gazette 143:368–373
Valiela I, Teal JM, Deuser WG (1978) Nature of growth forms in salt marsh grass Spartina alerniflora. The American Naturalist 112:461–470
Vermeer M, Rahmstorf S (2009) Global sea level linked to global temperature. Proceedings of the National Academy of Sciences of the United States of America 106:21527–21532
Ward LG, Kearney MS, Stevenson JC (1998) Variations in sedimentary environments and accretionary patterns in estuarine marshes undergoing rapid submergence, Chesapeake Bay. Marine Geology 151:111–134
Willis JM, Hester MW (2004) Interactive effects of salinity, flooding, and soil type on Panicum hemitomon. Wetlands 24:43–50
Zedler JB (1983) Freshwater impacts in normally hypersaline marshes. Estuaries 6:346–355
Zedler JB, Callaway JC, Desmond JS, Vivian-Smith G, Williams GD, Sullivan G, Brewster AE, Bradshaw BK (1999) Californian salt-marsh vegetation: an improved model of spatial pattern. Ecosystems 2:19–35
Zhang M, Ustin SL, Rejmankova E, Sanderson EW (1997) Monitoring Pacific coast salt marshes using remote sensing. Ecological Applications 7:1039–1053
Acknowledgments
This research was supported by the CALFED Science Program Grant No. 4600002970, as part of the Integrated Regional Wetland Monitoring (IRWM) pilot project, Science Grant #1037 from the California Bay-Delta Authority, and the National Institute of Climate Change Research (Department of Energy, Coastal Center). We thank the California Department of Fish and Game and the San Francisco Bay National Estuarine Research Reserve (NERR) for granting permission to access and collect at the study sites. We greatly appreciate the many hours of field and laboratory work put in by D. Benner, T. Cornelisse, Z. Diggory, T. Engle, S. Haines, and S. Santos. Additionally, we thank E. Herbert and two anonymous reviewers for editorial input.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schile, L.M., Callaway, J.C., Parker, V.T. et al. Salinity and Inundation Influence Productivity of the Halophytic Plant Sarcocornia pacifica . Wetlands 31, 1165–1174 (2011). https://doi.org/10.1007/s13157-011-0227-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13157-011-0227-y