Abstract
Purpose
Infection and malignancy represent two common complications after solid organ transplantation, which are often characterized by poorly specific clinical symptomatology. Herein, we have evaluated the role of 18F-fluoro-2-deoxy-D-glucose (FDG) positron emission tomography/computed tomography (PET/CT) in this clinical setting.
Methods
Fifty-eight consecutive patients who underwent FDG PET/CT after kidney, lung or heart transplantation were included in this retrospective analysis. Twelve patients underwent FDG PET/CT to strengthen or confirm a diagnostic suspicion of malignancies. The remaining 46 patients presented with unexplained inflammatory syndrome, fever of unknown origin (FUO), CMV or EBV seroconversion during post-transplant follow-up without conclusive conventional imaging. FDG PET/CT results were compared to histology or to the finding obtained during a clinical/imaging follow-up period of at least 6 months after PET/CT study.
Results
Positive FDG PET/CT results were obtained in 18 (31 %) patients. In the remaining 40 (69 %) cases, FDG PET/CT was negative, showing exclusively a physiological radiotracer distribution. On the basis of a patient-based analysis, FDG PET/CT’s sensitivity, specificity, PPV and NPV were respectively 78 %, 90 %, 78 % and 90 %, with a global accuracy of 86 %. FDG PET/CT was true positive in 14 patients with bacterial pneumonias (n = 4), pulmonary fungal infection (n = 1), histoplasmosis (n = 1), cutaneous abscess (n = 1), inflammatory disorder (sacroiliitis) (n = 1), lymphoma (n = 3) and NSCLC (n = 3). On the other hand, FDG PET/CT failed to detect lung bronchoalveolar adenocarcinoma, septicemia, endocarditis and graft-versus-host disease (GVHD), respectively, in four patients. FDG PET/CT contributed to adjusting the patient therapeutic strategy in 40 % of cases.
Conclusions
FDG PET/CT emerges as a valuable technique to manage complications in the post-transplantation period. FDG PET/CT should be considered in patients with severe unexplained inflammatory syndrome or FUO and inconclusive conventional imaging or to discriminate active from silent lesions previously detected by conventional imaging particularly when malignancy is suspected.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Solid organ transplantation represents a valuable therapeutic option for patients with end-stage organ dysfunction related to chronic diseases such as chronic respiratory failure due to cystic fibrosis, chronic obstructive pulmonary disease, chronic kidney diseases or heart failure [1]. Both infectious diseases and malignancies represent two groups of common complications in the post-transplant period. These complications are often characterized by poorly specific clinical symptoms particularly in early stages [2, 3].
The long-term follow-up of patients with solid organ transplantation is challenging including standard clinical and biological examinations, the assessment of organ function, infectious status and monitoring of immunosuppressive drugs. Moreover, when a specific complication is suspected, more investigations are required including further biological analysis and diagnostic imaging examinations. Unfortunately, both biological tests and imaging techniques are often poorly sensitive and not specific in transplant patients. Finally, biopsy represents a diagnostic alternative, but it is not free of further complications [4–8].
18F-fluoro-2-deoxy-D-glucose (FDG) positron emission tomography (PET) is a noninvasive imaging tool with high diagnostic accuracy and therapeutic impact in oncological clinical practice [9, 10]. FDG PET provides the in vivo estimation of pathologically increased glycolytic activity. Thereafter, the latest generation of high-resolution PET scanners is coupled with multidetector CT (PET/CT), allowing a simultaneous co-registration of morphological and functional data [11]. Activated inflammatory cells such as neutrophils, macrophages and lymphocytes present an increased FDG uptake, causing significant FDG accumulation during inflammation [12, 13]. Hence, the slight specificity of FDG PET/CT emerges as a potential advantage in the management of patients with inflammatory or infectious diseases [14]. Moreover, FDG PET/CT seems to be useful in the setting of unexplained inflammatory syndrome and fever of unknown origin (FUO) in both the overall population [15–18] and immunosuppressed patients [19, 20]. Finally, FDG PET/CT plays a role in assessing treatment efficacy in neoplastic [21] and selected non-neoplastic conditions [22, 23], leading to the detection of residual tumoral or inflammatory localizations. Accordingly, FDG PET/CT may represent an interesting technique in the post-transplantation follow-up in patients with inflammatory syndrome of unknown origin and unspecific clinical symptomatology [2, 24–27].
In real-life practice, FDG PET/CT often represents a second-line diagnostic investigation in patients with negative or inconclusive conventional radiologic examinations. This diagnostic condition may be explained by the necessity to obtain a definitive and prompt diagnosis in this particular high-risk patient population as well as by the more important availability of conventional radiography and CT devices. Therefore, patients often undergo FDG PET/CT after other diagnostic procedures, without a structured protocol or diagnostic hypothesis and, at worst, after the beginning of an empiric treatment. Accordingly, the main objective of this retrospective single-center study was to evaluate the diagnostic role and therapeutic impact of FDG PET/CT in daily clinical practice in a cohort of patients with suspicion of malignant or infectious complications after renal, lung or heart transplantation.
Materials and Methods
Patient Population
Fifty-eight consecutive patients with suspicion ofa complication after renal, lung or heart transplantation and presenting to our institution between August 2004 and September 2012 for FDG PET/CT were retrospectively included in this study. Patient mean age at the time of PET/CT was 48 ± 2 years ranging from 21 to 80 years. Patients underwent a standard evaluation including: (1) history and physical examination, (2) standard biological tests comprising the measurement of C-reactive protein (CRP), fibrinogen, hepatic transaminases, lactate dehydrogenase (LDH) and leucocyte count, (3) Epstein-Barr virus (EBV) and cytomegalovirus (CMV) DNA PCR quantification and (4) radiological evaluation including standard radiography and/or high-resolution chest and/or abdominopelvic CT and/or magnetic resonance imaging (MRI) and/or ultrasonography (US). Suspicion of a post-transplant complication was based on a variable combination of clinical symptoms (32 patients), biological test results (27 patients) and radiological abnormalities (14 patients). Follow-up data were obtained until final diagnosis or when the diagnosis of a post-transplant complication was discarded after a spontaneous regression of clinical symptomatology and/or normalization of biological tests without specific medical treatment within 6-month follow-up. Besides neoplastic suspicion, carrying out FDG PET/CT was justified by a cross-disciplinary discussion between an organ transplant specialist and nuclear medicine physician.
Patient Clinical Assessment
The selected patients had previously undergone lung (15 patients), kidney (30 patients) or heart transplantation (13 patients). Table 1 details the patient clinical situation requiring transplantation. A post-transplant complication was suspected about 61 ± 3 months after organ transplantation, ranging from 11 days to 21 years. Eighteen patients (30 %) had a history of pre-transplant malignant tumors including three cases of Hodgkin lymphoma (HL), seven cases of non-Hodgkin lymphoma (NHL), two cases of basal-cell carcinoma, one case of leukemia, one case of rhabdomyosarcoma, one case of Kaposi’s sarcoma, one case of squamous cell carcinoma, one case of prostate carcinoma and one case of in situ uterine cervical adenocarcinoma. CRP, leucocyte count, LDH and gamma glutamyl transferase (GGT) were abnormally increased in 60 %, 3 %, 48 % and 28 %, respectively, and 7 % of patients had fever.
Twelve patients underwent FDG PET/CT to strengthen or confirm a diagnostic suspicion of malignancies (3 lymphomas, 1 adrenal mass of unknown etiology and 8 lung cancers) on conventional imaging. The remaining 46 patients presented with (1) unexplained inflammatory syndrome and FUO, defined as persistent fever up to 38.5 °C without any diagnosis reached from the usual investigations, or (2) CMV or EBV seroconversion during the post-transplant follow-up period. In these 46 patients, conventional imaging was not conclusive.
At the time of FDG PET/CT, all patients were treated in accordance with the international guidelines [28] with a variable combination of tacrolimus (20 patients), mycophenolate mofetil (34 patients) or mycophenolate sodium (3 patients), corticosteroids (40 patients), cyclosporine (16 patients), azathioprine (5 patients) and everolimus (17 patients).
FDG PET-CT Technical Features and Interpretation Criteria
A combined FDG PET/CT scanner was employed for all patients (Discovery ST, GE Medical System, Milwaukee, WI, USA). In order to obtain a serum glucose level less than 6.6 mmol/l, patients fasted for 6 h before intravenous injection of 5.5 MBq/kg FDG (Flucis, CIS Bio International, France). Five milligrams of diazepam and 80 mg of phloroglucinol (musculotropic antispasmodic) had been previously administered to patients. Whole-body (WB) PET/CT acquisitions started about 60 min after tracer injection, including a head to midthigh CT scan (140 kV, 80 mAs, 0.8 s/rotation) during current breathing, followed by a PET scan (7 fields of view, 15 cm/field, 4 min/field, 3.27 mm slice thickness). In selected patients, PET acquisitions (6–7 fields of view, 3 min/field) and low-dose CT were performed on the lower limbs. PET data were reconstructed with and without CT-based attenuation correction by iterative algorithm [ordered subset expectation maximization (OSEM), 2 iterations, 15 subsets, 128 × 128 matrix]. CT, PET (corrected) and combined PET/CT images were displayed on a Xeleris workstation (GE Medical System, Milwaukee, WI, USA) for visual interpretation. The maximum standardized uptake value (mSUV) per focus was employed for semiquantitative analysis of FDG uptake. FDG PET/CT data analysis of all patients was done by an experienced nuclear medicine physician, who was aware of the clinical situation and pathological background and had access to patients’ biological results. Each focus of increased extra-physiologic uptake of FDG was recorded and interpreted according to each patient’s situation. The original FDG PET/CT report was considered for the analysis of each examination result.
FDG was used following marketing authorization. Conforming to local institutional guidelines, all included patients gave free and informed consent for the use of anonymous personal medical data extracted from their file for scientific purposes. The Local Institutional Review Board approved this retrospective study.
Data Analysis
In the present study, FDG PET/CT results were compared to histology after biopsy or more complex surgical procedures (when available) or to clinical follow-up (after FDG PET/CT study) within 6 months including a variable combination of physical examination, biological tests, conventional imaging (CT, MRI, US) and virologic, bacteriologic and mycologic investigations.
The FDG PET/CT was considered contributory to modifying the diagnostic workup and therapeutic strategy when it allowed any of the following: (1) identification of the presence and site of infection or malignancy (other conventional tests performed at the time of FDG PET/CT imaging were negative and/or noncontributory); (2) elimination of the radiological hypothesis of malignancy; (3) determination of the extent of the infection or malignancy in a specific organ or the involvement of other organs; (4) targeting the diagnostic procedure; (5) removing the infection site; (6) changing the duration of antibiotic therapy.
Statistical Analysis
Results are expressed as mean ± SD, range and percentage. Diagnostic performances of FDG PET/CT were evaluated according to a patient-based analysis. FDG PET/CT studies were interpreted as:
-
True positive (TP) when positive FDG PET/CT findings were confirmed by histology or clinical/imaging follow-up;
-
True negative (TN) when FDG PET/CT was interpreted as normal and the diagnosis of post-transplant complication was finally discarded;
-
False positive (FP) when FDG PET/CT findings were not finally confirmed by complementary investigations;
-
False negative (FN) when FDG PET/CT failed to detect the post-transplant complication diagnosed by complementary investigation.
From the TP, FP, TN and FN results, FDG PET/CT’s sensitivity (Se), specificity (Sp), positive predictive value (PPV), negative predictive value (NPV) and overall accuracy were determined. Finally, the Youden Index, which is independent from the prevalence of the disease, was estimated as (Sp + Se)-1. A p value < 0.05 was considered statistically significant. All statistical analyses were performed using SPSS 17.0 software.
Results
A post-transplant complication was finally diagnosed in 18 (31 %) patients, corresponding to 7 deep infections, 1 septicemia of unknown origin, 1 endocarditis, 1 inflammatory disorder, 1 GVHD, 3 aggressive lymphomas and 4 lung cancers.
Overall FDG PET/CT Results
Analysis was performed to evaluate FDG PET/CT as a tool for the detection of both infectious and neoplastic post-transplant complications. Positive FDG PET/CT results were obtained in 18 patients. In the remaining 40 cases (69 %), FDG PET/CT was negative, showing exclusively a physiological radiotracer distribution.
Among the 18 patients (31 %) with positive FDG PET/CT:
-
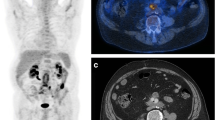
Fourteen patients were TP: seven deep infections [four bacterial pneumonias, one pulmonary fungal infection, one histoplasmosis (Fig. 1), one cutaneous abscess (Fig. 2)], 1 inflammatory disorder (sacroiliitis), 2 large B cell lymphomas (LBCLs) of stage II and and IV, one Hodgkin lymphoma (stage I) and three NSCLCs presented with solitary, small and irregularly shaped nodules sometimes associated with emphysematous parenchymal dystrophy.
Fig. 1 Anterior view of FDG PET whole-body maximum intensity projection (MIP) image (upper panel) and axial FDG PET/CT slice (lower panel) in a 56-year-old male with inflammatory syndrome and a history of kidney transplantation. FDG PET/CT showed pathological uptake in the pyloric region. a Further investigations following FDG PET/CT investigations allowed the diagnosis of gastric histoplasmosis. After 2 years of itraconazole treatment, FDG PET/CT showed the complete regression of FDG uptake abnormalities (b) allowing the withdrawal of the antifungal treatment
Fig. 2 a Coronal, b axial and c sagittal FDG PET/CT images in a 27-year-old lung-transplanted male with inflammatory syndrome showing intense focal uptake in the left thoracic wall. The following ultrasound examination revealed a 35 × 15-mm abscess in the subcutaneous tissue, around the fifth rib, at the periphery of the surgical scar (d). Note the complete regression of tracer uptake abnormalities after surgical treatment e–g
-
Four patients were FP: (1) a kidney-transplanted patient with fever and inflammatory syndrome spontaneously improving during follow-up showed a mild increase of FDG uptake in the hepatic hilum lymph nodes; (2) a kidney-transplanted patient with previous EBV-induced bowel lymphoma and a high serum LDH value presented with increased FDG uptake in the cecum and terminal bowel suggesting tumoral relapse. Histological examination after surgical biopsy showed benign inflammatory granulomas (Fig. 3); (3) a kidney-transplanted patient with unexplained inflammatory syndrome showing a focally increased FDG uptake in the lower esophagus suggesting a primary tumor, with endoscopic biopsy evaluation showing grade C reflux esophagitis; (4) a heart-transplanted patient with radiological suspicion of primary lung cancer because of a mildly hypermetabolic nodule that remained stable in size during a 3-year follow-up.
Fig. 3 Anterior view of FDG PET whole-body maximum intensity projection (MIP) and axial FDG PET/CT image in a kidney-transplanted (*) 27-year-old male with a history of digestive non-Hodgkin lymphoma (white arrow; a) considered in complete metabolic remission at the end of chemotherapy (b). Because an increased serum LDH value and general weakness appeared during follow-up, the patient underwent FDG PET/CT, which showed intense FDG uptake in the right colon and distal ileum (red arrow; c) suggesting disease relapse. The pathological examination after surgical biopsy revealed benign granuloma without tumoral relapse
The intensity of FDG uptake (as measured by mSUV) for the 18 patients with positive FDG PET/CT results was on average 8.2 (ranging from 3.2 to 22.5) (Table 2). There was no statistically significant difference between malignancies and infectious diseases in terms of mSUV.
Among the 40 patients (69 %) with negative FDG PET/CT:
-
Thirty-six patients were TN. A definitive negative diagnosis of post-transplantation complication was retained after a spontaneous regression of clinical symptomatology and/or normalization of biological tests without specific medical treatment within the 6-month follow-up. Moreover, in three patients, FDG PET/CT showed functional thyroid nodules and post-traumatic hypermetabolic bone fractures. In one additional patient, stage II sarcoidosis was suggested and afterwards confirmed by pathological examination.
-
Four patients were FN: one with histologically proven lung bronchoalveolar adenocarcinoma (mSUV: 1.4) and three with septicemia of unknown origin with proved endocarditis and GVHD, respectively, showing no FDG uptake abnormalities.
Based on our patient-based analysis, FDG PET/CT’s sensitivity, specificity, PPV and NPV were respectively 78 %, 90 %, 78 % and 90 %, with a global accuracy of 86 %. The Youden Index was estimated to be 0.68. Clinical presentation and the biological characteristics of 18 patients with true-positive and false-negative FDG PET/CT results are summarized in Table 3.
Impact of FDG PET/CT on the Diagnostic Workup and Therapeutic Strategy
Among the 12 patients with radiological suspicion of malignancies, FDG PET/CT allowed eliminating the hypothesis of malignancy in seven patients and strengthening the diagnosis in three other patients (1 LH, 2 NSCLC) with previous history of lung (n = 2) and kidney (n = 1) transplantation. In the remaining two patients, FDG PET/CT was false negative [i.e., histologically proven lung bronchoalveolar adenocarcinoma (mSUV: 1.4)] and false positive [i.e., a lung hypermetabolic nodule suspected of neoplasia (mSUV: 9.1)], but stable in size during a 3-year follow-up) without inducing a modification of patient management.
Among the remaining 46 patients with clinical and/or biological abnormalities and inconclusive conventional imaging, the FDG PET/CT contributed to patient management in 13 cases (22 %) by:
-
Guiding the diagnostic interventional radiologic procedure in three patients. FDG PET/CT allowed the diagnosis of two LBCLs and one NSCLCs in patients with a previous history of lung (n = 1), heart (n = 1) and kidney (n = 1) transplantation;
-
Suggesting deep infectious disease in seven patients allowing the diagnosis of lung aspergillosis (n = 1), gastro-duodenal histoplasmosis (n = 1), bacterial pneumonia (n = 3), cutaneous abscess (n = 1) and sacroiliitis (n = 1).
The TN FDG PET/CT result enabled stopping antifungal treatment for dubious active aspergillosis and diagnosing immunosuppressive treatment intolerance in two patients with a history of cardiac transplantation.
Finally, in one kidney transplant patient with fever and biological inflammatory syndrome, FDG PET/CT exclusively showed some enlarged and hypermetabolic lymph nodes of the mediastinum and both lung hila, suggesting a stage II sarcoidosis, successively confirmed by pathological examination.
Accordingly, FDG PET/CT contributed to adjusting the patient therapeutic approach in 23 cases (40 %). The clinical presentation and therapeutic impact of 23 patients for whom FDG PET/CT contributed to adjusting the therapeutic approach are summarized in Table 4.
Discussion
The present study evaluates FDG PET/CT’s accuracy in diagnosing post-transplant complications in solid organ transplant recipients in clinical practice. Fifty-eight patients with lung, heart and kidney transplantation were included in this retrospective monocentric study. Furthermore, we investigated the proportion of patients whose management strategy was modified according to FDG PET/CT results.
Post-transplant lymphoproliferative disorders (PTLDs) are the most frequent post-transplant malignancy in the pediatric population and the second in adult patients. PTLD represents a wide spectrum of monomorphic and polymorphic lymphoproliferative disorders, and large B cell lymphoma is the most frequent type [3, 29–32]. PTLDs are often extranodal, with graft, cerebral or digestive involvement [33, 34]. Clinical symptoms depend on tumoral localization and are often poorly specific [2, 34, 35]. Other types of malignancies, especially cutaneous tumors [36] and virus-related diseases, are also more frequent in immunocompromised patients compared to the general population [3]. Moreover, single-lung transplant confers a significantly elevated risk of developing primary lung cancer in native lung [37]. Infectious diseases are also life-threatening complications in an immunosuppressive context, and they can potentially affect several organs [38]. Therefore, a diagnostic tool able to detect the appearance of post-transplant complications during follow-up with high sensibility is mandatory to optimize the therapeutic strategy. FDG PET/CT is recognized to have a great impact on diagnosing, staging and restaging cancer patients [39–41]. Recently, Kubota et al. [42] evaluated the impact of FDG PET findings on decisions regarding patient management strategies (i.e., the strategy modification rate) in a prospective multicenter trial in neoplastic patients. The authors reported a strategy modification rate of 71.6 % and 70.0 %, respectively, for patients with lung cancer and malignant lymphoma. Von Flack et al. [24] compared FDG PET/CT to conventional imaging such as MRI and CT in the primary staging and therapy monitoring in 17 pediatric patients with post-transplant lymphoma. The authors concluded that FDG PET/CT seemed more efficient than conventional imaging to evaluate early treatment efficacy with potential consequences for patient management. In our series, FDG PET/CT contributed to adjusting the patient’s therapeutic strategy in 23 of 58 (40 %) patients. In particular, FDG PET/CT allowed confirming or excluding lymphoma and other malignancies in 10 of 12 patients with inconclusive CT results. FDG PET/CT also suggested infectious complications in seven patients (12 %), guiding the following diagnostic procedures. Hence, independently from the cost and access to this type of medical imaging, FDG PET/CT could have a potential role in optimizing the treatment planning in both neoplastic and selected non-neoplastic conditions during the post-transplant period. FDG PET/CT allows morphofunctional cartography of residual disease after the end or during the course of treatment. Moreover, the evolution of radiotracer uptake intensity reflects the efficacy of treatment, permitting either a better modulation of the drug dosage or a radical modification of the therapeutic strategy.
Graute et al. [26] retrospectively evaluated the diagnostic role of FDG PET or FDG PET/CT in 17 patients with symptoms suspicious of malignancy after heart transplantation. They reported a global accuracy of 71 %, ranging from 43 % to 90 % in patients examined by FDG PET or FDG PET/CT, respectively. Our study population included not only heart transplant patients, but also patients who underwent kidney and lung transplantation. Moreover, the presence of both infectious diseases and malignancies was investigated obtaining an overall diagnostic accuracy of 86 %. Compared to Graute’s results, we obtained a higher specificity value, which is probably due to the systematic use of FDG PET/CT scanners but also to a possible bias related to the studied population. Panagiotidis et al. [43] investigated the role of FDG PET/CT in the diagnosis of PTLD in 40 patients who had previously undergone solid organ or hematopoietic cell transplantation. In this study, FDG PET/CT’s sensitivity and specificity were 88 % and 91 %, respectively. Dierickx et al. [44] analyzed 170 FDG PET scans in 150 patients with suspicion of PTLD after solid organ and hematopoietic cell transplantation, reporting a sensitivity and specificity of 89 %.
Guy et al. [19] assessed the diagnostic performances of FDG PET/CT in 20 patients with febrile neutropenia, reporting a sensitivity of about 93 %. Dong et al. [16] published a meta-analysis on the value of FDG PET/CT in the evaluation of FUO, reporting a pooled sensitivity of 83 %. In our patient cohort, four patients were investigated because of FUO without neutropenia, and FDG PET/CT was false negative in only one patient showing septicemia at the time of examination.
In our series, false-negative FDG PET/CT results were obtained in a case of histologically proven lung adenocarcinoma, in two patients with pyelonephritis and GVH disease and in two patients with septicemia. It is known that FDG PET/CT’s sensitivity is not optimal in certain tumoral types such as MALT lymphoma or carcinoid tumor. Moreover, low uptake intensity has been previously described in pulmonary adenocarcinoma [45]. Urinary FDG excretion and the physiological and heterogeneous intestinal FDG uptake probably reduced the sensitivity of FDG PET/CT for detecting pyelonephritis and GVH reactions. Concerning septicemia, other authors reported false-negative results in this clinical situation [26].
Although interesting results were obtained from an important cohort of kidney, lung and heart-transplanted patients, this study has several limitations such as its retrospective design and the lack of histopathologic confirmation for all lesions. Moreover, the high number of true-negative patients could represent a bias with an impact on the sensitivity and specificity.
Conclusion
Our study supports the potential of FDG PET/CT in the diagnostic workup of complications in the post-transplant period. FDG PET/CT should be considered in transplanted patients with severe unexplained inflammatory syndrome/FUO and normal CT or to discriminate active from silent lesions previously detected by conventional imaging without definitive interpretation when malignancy is suspected.
Considering the recent development of interventional radiology procedures guided by FDG PET/CT [46–48], we can foresee an evolution toward multimodality imaging allowing the definition of a metabolic target for the diagnostic biopsy. However, prospective and larger multicentric studies are needed to completely define the place of FDG PET/CT in the post-transplantation period.
References
Mahillo B, Carmona M, Álvarez M, White S, Noel L, Matesanz R. 2009 global data in organ donation and transplantation: activities, laws, and organization. Transplantation. 2011;92:1069–74.
Parker A, Bowles K, Bradley JA, Emery V, Featherstone C, Gupte G, et al. Diagnosis of post-transplant lymphoproliferative disorder in solid organ transplant recipients—BCSH and BTS Guidelines: Guideline. Br J Haematol. 2010;149:675–92.
Taylor AL, Marcus R, Bradley JA. Post-transplant lymphoproliferative disorders (PTLD) after solid organ transplantation. Crit Rev Oncol Hematol. 2005;56:155–67.
Richardson CM, Pointon KS, Manhire AR, Macfarlane JT. Percutaneous lung biopsies: a survey of UK practice based on 5444 biopsies. Br J Radiol. 2002;75:731–5.
Cohen MB, A-Kader HH, Lambers D, Heubi JE. Complications of percutaneous liver biopsy in children. Gastroenterology. 1992;102:629–32.
Eiro M, Katoh T, Watanabe T. Risk factors for bleeding complications in percutaneous renal biopsy. Clin Exp Nephrol. 2005;9:40–5.
Wiener RS, Schwartz LM, Woloshin S, Welch HG. Population-based risk for complications after transthoracic needle lung biopsy of a pulmonary nodule: an analysis of discharge records. Ann Intern Med. 2011;155:137–44.
Anastasiadis A, Zapała L, Cordeiro E, Antoniewicz A, Dimitriadis G, De Reijke T. Complications of prostate biopsy. Expert Rev Anticancer Ther. 2013;13:829–37.
Poeppel TD, Krause BJ, Heusner TA, Boy C, Bockisch A, Antoch G. PET/CT for the staging and follow-up of patients with malignancies. Eur J Radiol. 2009;70:382–92.
Kwee TC, Kwee RM. Combined FDG-PET/CT for the detection of unknown primary tumors: systematic review and meta-analysis. Eur Radiol. 2009;19:731–44.
Townsend DW, Carney JPJ, Yap JT, Hall NC. PET/CT today and tomorrow. J Nucl Med Off Publ Soc Nucl Med. 2004;45(1):4S–14S.
Braun JJ, Kessler R, Constantinesco A, Imperiale A. 18F-FDG PET/CT in sarcoidosis management: review and report of 20 cases. Eur J Nucl Med Mol Imaging. 2008;35:1537–43.
Treglia G, Taralli S, Calcagni ML, Maggi F, Giordano A, Bonomo L. Is there a role for fluorine 18 fluorodeoxyglucose-positron emission tomography and positron emission tomography/computed tomography in evaluating patients with mycobacteriosis? A systematic review. J Comput Assist Tomogr. 2011;35:387–93.
Jamar F, Buscombe J, Chiti A, Christian PE, Delbeke D, Donohoe KJ, et al. EANM/SNMMI guideline for 18F-FDG use in inflammation and infection. J Nucl Med. 2013;54:647–58.
Federici L, Blondet C, Imperiale A, Sibilia J, Pasquali J-L, Pflumio F, et al. Value of (18)F-FDG-PET/CT in patients with fever of unknown origin and unexplained prolonged inflammatory syndrome: a single centre analysis experience. Int J Clin Pract. 2010;64:55–60.
Dong M, Zhao K, Liu Z, Wang G, Yang S, Zhou G. A meta-analysis of the value of fluorodeoxyglucose-PET/PET-CT in the evaluation of fever of unknown origin. Eur J Radiol. 2011;80:834–44.
Okumura W, Iwasaki T, Toyama T, Iso T, Arai M, Oriuchi N, et al. Usefulness of fasting 18F-FDG PET in identification of cardiac sarcoidosis. J Nucl Med. 2004;45:1989–98.
Balink H, Collins J, Bruyn GA, Bruyn G, Gemmel F. F-18 FDG PET/CT in the diagnosis of fever of unknown origin. Clin Nucl Med. 2009;34:862–8.
Guy SD, Tramontana AR, Worth LJ, Lau E, Hicks RJ, Seymour JF, et al. Use of FDG PET/CT for investigation of febrile neutropenia: evaluation in high-risk cancer patients. Eur J Nucl Med. 2012;39:1348–55.
Sturm E, Rings EHHM, Schölvinck EH, Gouw ASH, Porte RJ, Pruim J. Fluordeoxyglucose positron emission tomography contributes to management of pediatric liver transplantation candidates with fever of unknown origin. Liver Transplant. 2006;12:1698–704.
Kazama T, Faria SC, Varavithya V, Phongkitkarun S, Ito H, Macapinlac HA. FDG PET in the evaluation of treatment for lymphoma: clinical usefulness and pitfalls1. RadioGraphics. 2005;25:191–207.
Imperiale A, Riehm S, Braun JJ. Interest of [18F]FDG PET/CT for treatment efficacy assessment in aggressive phenotype of sarcoidosis with special emphasis on sinonasal involvement. Q J Nucl Med Mol Imaging Off Publ Ital Assoc Nucl Med AIMN Int Assoc Radiopharmacol IAR Sect Soc Radiopharm Chem Biol. 2013;57:177–86.
Sathekge M, Maes A, Kgomo M, Stoltz A, Van de Wiele C. Use of 18F-FDG PET to predict response to first-line tuberculostatics in HIV-associated tuberculosis. J Nucl Med. 2011;52:880–5.
von Falck C, Maecker B, Schirg E, Boerner AR, Knapp WH, Klein C, et al. Post transplant lymphoproliferative disease in pediatric solid organ transplant patients: a possible role for [18F]-FDG-PET(/CT) in initial staging and therapy monitoring. Eur J Radiol. 2007;63:427–35.
McCormack L, Hany TI, Hubner M, Petrowsky H, Mullhaupt B, Knuth A, et al. How useful is PET/CT imaging in the management of post-transplant lymphoproliferative disease after liver transplantation? In four patients with PTLD investigated with PET/CT, this method provided new information that changed medical management in three of the four patients. Am J Transplant. 2006;6:1731–6.
Graute V, Jansen N, Sohn H-Y, Becker A, Klein B, Schmid I, et al. Diagnostic role of whole-body [18F]-FDG positron emission tomography in patients with symptoms suspicious for malignancy after heart transplantation. J Heart Lung Transplant. 2012;31:958–66.
Jigang Yang HZ. The role of 18F-FDG PET/CT in the evaluation of pediatric transplant patients. Hell. J. Nucl. Med. 2015;18
Helderman JH, Bennett WM, Cibrik DM, Kaufman DB, Klein A, Takemoto SK. Immunosuppression: practice and trends. Am J Transplant. 2003;3:41–52.
Vajdic CM, van Leeuwen MT. Cancer incidence and risk factors after solid organ transplantation. Int J Cancer. 2009;125:1747–54.
Penn I, Starzl TE. Malignant tumors arising de novo in immunosuppressed organ transplant recipients. Transplantation. 1972;14:407–17.
Thomas JA, Crawford DH, Burke M. Clinicopathologic implications of Epstein-Barr virus related B cell lymphoma in immunocompromised patients. J Clin Pathol. 1995;48:287.
Opelz G, Döhler B. Lymphomas after solid organ transplantation: a collaborative transplant study report: lymphomas after solid organ transplantation. Am J Transplant. 2004;4:222–30.
Bakker NA, van Imhoff GW, Verschuuren EAM, van Son WJ. Presentation and early detection of post-transplant lymphoproliferative disorder after solid organ transplantation. Transpl Int. 2007;20:207–18.
Tsao L, Hsi ED. The clinicopathologic spectrum of posttransplantation lymphoproliferative disorders. Arch Pathol Lab Med. 2007;131:1209–18.
Caillard S, Porcher R, Provot F, Dantal J, Choquet S, Durrbach A, et al. Post-transplantation lymphoproliferative disorder after kidney transplantation: report of a nationwide French registry and the development of a new prognostic score. J Clin Oncol. 2013;31:1302–9.
Watt KDS, Pedersen RA, Kremers WK, Heimbach JK, Sanchez W, Gores GJ. Long-term probability of and mortality from de novo malignancy after liver transplantation. Gastroenterology. 2009;137:2010–7.
Dickson RP, Davis RD, Rea JB, Palmer SM. High frequency of bronchogenic carcinoma after single-lung transplantation. J Heart Lung Transplant. 2006;25:1297–301.
Humar A, Michaels M. AST ID working group on infectious disease monitoring. American Society of Transplantation recommendations for screening, monitoring and reporting of infectious complications in immunosuppression trials in recipients of organ transplantation. Am J Transplant. 2006;6:262–74.
Yap CS, Seltzer MA, Schiepers C, Gambhir SS, Rao J, Phelps ME, et al. Impact of whole-body 18F-FDG PET on staging and managing patients with breast cancer: the referring physician’s perspective. J Nucl Med. 2001;42:1334–7.
Seltzer MA, Yap CS, Silverman DH, Meta J, Schiepers C, Phelps ME, et al. The impact of PET on the management of lung cancer: the referring physician’s perspective. J Nucl Med. 2002;43:752–6.
Meta J, Seltzer M, Schiepers C, Silverman DH, Ariannejad M, Gambhir SS, et al. Impact of 18F-FDG PET on managing patients with colorectal cancer: the referring physician’s perspective. J Nucl Med. 2001;42:586–90.
Kubota K, Matsuno S, Morioka N, Adachi S, Koizumi M, Seto H, et al. Impact of FDG-PET findings on decisions regarding patient management strategies: a multicenter trial in patients with lung cancer and other types of cancer. Ann Nucl Med. 2015;29:431–41.
Panagiotidis E, Quigley A-M, Pencharz D, Ardeshna K, Syed R, Sajjan R, et al. 18F-fluorodeoxyglucose positron emission tomography/computed tomography in diagnosis of post-transplant lymphoproliferative disorder. Leuk Lymphoma. 2013;55:515–9.
Dierickx D, Tousseyn T, Requilé A, Verscuren R, Sagaert X, Morscio J, et al. The accuracy of positron emission tomography in the detection of posttransplant lymphoproliferative disorder. Haematologica. 2013;98:771–5.
Mamede M, Higashi T, Kitaichi M, Ishizu K, Ishimori T, Nakamoto Y, et al. [18F]FDG uptake and PCNA, Glut-1, and hexokinase-II expressions in cancers and inflammatory lesions of the lung. Neoplasia. 2005;7:369–79.
Shyn PB. Interventional positron emission tomography/computed tomography: state-of-the-art. Tech Vasc Interv Radiol. 2013;16:182–90.
Imperiale A, Garnon J, Bachellier P, Gangi A, Namer IJ. Simultaneous 18F-FDOPA PET/CT-guided biopsy and radiofrequency ablation of recurrent neuroendocrine hepatic metastasis: further step toward a theranostic approach. Clin. Nucl. Med. 2015
Cornelis F, Silk M, Schoder H, Takaki H, Durack JC, Erinjeri JP, et al. Performance of intra-procedural 18-fluorodeoxyglucose PET/CT-guided biopsies for lesions suspected of malignancy but poorly visualized with other modalities. Eur J Nucl Med Mol Imaging. 2014;41:2265–72.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Nastassja Muller, Romain Kessler, Sophie Caillard, Eric Epailly, Fabrice Hubelé, Céline Heimburger, Izzie-Jacques Namer, Raoul Herbrecht, Cyrille Blondet and Alessio Imperiale declare that they have no conflict of interest.
Ethical Statement
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. For this type of study formal consent is not required.
Rights and permissions
About this article
Cite this article
Muller, N., Kessler, R., Caillard, S. et al. 18F-FDG PET/CT for the Diagnosis of Malignant and Infectious Complications After Solid Organ Transplantation. Nucl Med Mol Imaging 51, 58–68 (2017). https://doi.org/10.1007/s13139-016-0461-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13139-016-0461-6