Abstract
In this study, seed and seed shell extracts of Cola nitida were investigated for the biogenic synthesis of silver nanoparticles (AgNPs) under ambient condition. The biosynthesized AgNPs were characterized through visual development of colour, UV–vis spectroscopy, Fourier transform infrared spectroscopy (FTIR) and transmission electron microscopy (TEM). The antibacterial activities of the AgNPs were determined using some multidrug-resistant clinical isolates. The biosynthesized AgNPs depicted brown and yellowish orange colour using seed and seed extract, respectively, with maximum absorbance readings at 457.5 and 454.5 nm. The FTIR spectrum showed strong peaks at 3292.49, 2086.98 and 1631.78 cm−1 for seed extract-mediated AgNPs, while peaks of 3302.13, 2086.05 and 1633.71 cm−1 were obtained for seed shell extract-mediated AgNPs, all indicating that proteins were the capping and stabilization molecules in the biogenic synthesis of AgNPs. The AgNPs were spherical in shape with sizes ranging from 8 to 50 and 5 to 40 nm for seed and seed shell-mediated AgNPs, respectively. The energy-dispersive X-ray (EDX) analysis showed the presence of silver as a prominent metal, while the selected area electron diffraction (SAED) pattern conformed to the face-centred cubic phase and crystalline nature of AgNPs. At various concentrations ranging from 50–150 μg/ml, the AgNPs inhibited growth of multidrug-resistant strains of Klebsiella granulomatis, Pseudomonas aeruginosa and Escherichia coli to the tune of 10–32 mm. Comparatively, seed shell extract-mediated AgNPs had better activities with minimum inhibitory concentration (MIC) of 50 μg/ml against all the tested isolates, while the MICs of seed extract-mediated AgNPs were obtained as 50, 80 and 120 μg/ml against E. coli, P. aeruginosa (wound) and P. aeruginosa (burn), respectively. This study has demonstrated the feasibility of eco-friendly biogenic synthesis of AgNPs using seed and seed shell extracts of C. nitida, and the report to the best of our knowledge is the first reference to extracts of C. nitida for the green synthesis of AgNPs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
The numerous potential applications of nanoparticles have played a prominent role in the search for eco-friendly processes of producing nanoparticles using different biological materials, since the conventional chemical synthesis involves the use of toxic solvents, high pressure and energy which may be harmful to the environment. Such synthesis is capable of yielding nanoparticles of unique attributes and properties, which may influence their applications. Owing to the rich biodiversity of microbes and plants, the potential of biological materials in the synthesis of nanoparticles is yet to be fully explored. In this regard, several biological resources of the tropics, particularly those of Nigeria, have not been adequately evaluated for their potentials at synthesizing nanoparticles. For instance, in our laboratory, we have recently reported the isolation of a local strain of Bacillus safensis LAU 13 (GenBank accession no KJ461434) with potent ability to degrade keratins [1, 2] and whose keratinase was used for the green synthesis of silver nanoparticles [3]. This was the first report of its isolation in Nigeria and demonstration of the capacity of the bacterium to produce keratinase. More recently, the novel utilization of spider cobweb for the green synthesis of silver nanoparticles (AgNPs) was first reported in our laboratory [4].
The green synthesis of nanoparticles has continued to receive unprecedented attention due to the simplicity of the process, less handling of chemicals and eco-friendliness [5]. In addition, the availability of several biological macromolecules/substances that can serve as capping and stabilization molecules for the green synthesis of nanoparticles has also contributed to the steady rise of this process. Several authors have used bacteria, fungi, algae and plant extracts for the green synthesis of metal nanoparticles [3, 5–13]. We have also documented the first review on the green reduction of graphene oxides [14].
Kola is a caffeine-containing nut, and the two common species are Cola nitida and Cola acuminata [15]. The nut is chewed in many West African countries, as it is known to ease hunger pangs. It also has ethnomedicinal values with the report of its use to treat whooping cough and asthma, and it contains caffeine, essential oils, phenolic compounds and alkaloids [16–18]. It is also a flavouring ingredient used in the production of flavoured beverages and chocolates, as well as in the production of dyes [19]. Cola sp., an evergreen plant originally endemic to West Africa, mainly Cote d’Ivoire, Ghana, Liberia, Nigeria and Sierra Leone, has been introduced to many tropical countries [20]. The exotic species can be found in countries such as Jamaica, India and USA. The world production of kola nuts from C. nitida and allied species was estimated as ca. 180,000 t of which about 120,000 t is produced in Nigeria and used either internally or in neighbouring countries [21]. Despite the huge potentials that kola nut offers, it has not been fully exploited for applications in food and pharmaceutical industries. This work therefore seeks to extend the frontiers of the potential applications of the extracts of the seed and seed shell of kola with particular interest in nanobiotechnology. The work is the pioneering effort at evaluating metabolites of kola for the green synthesis of nanoparticles.
Metal nanoparticles and their alloys such as silver, iron, cadmium, zinc, platinum, gold and graphene have diverse applications in different aspects of human endeavours, such as catalytic activity [9, 22, 23], optical [24], electronic [25], magnetic, antimicrobial [3, 7, 10, 13, 26] and biomedical usefulness [27, 28]. AgNPs, for instance, are of interest because of their unique properties (size- and shape-dependent optical, electrical and magnetic properties) which can be incorporated into coating materials, antimicrobial applications, biosensor materials, composite fibres and electronic components [29]. Amongst metal nanoparticles, AgNPs have shown excellent inhibitory and bactericidal effects among several metallic nanoparticles [30]. The antibacterial activities of AgNPs have been attributed to the interaction of AgNPs with sulphur- and phosphorus-containing biomolecules in the bacterial cell. As the particles enter into the cell, cell killing is initiated through the attack of the respiratory chain and cell division [31].
In this work, we report the green synthesis of AgNPs using the seed and seed shell of C. nitida and evaluation of its antibacterial activities using some drug-resistant strains of bacteria. To the best of our knowledge, this is the first report of the green synthesis of nanoparticles using C. nitida.
2 Materials and Methods
2.1 Sample Collection
C. nitida plant with the voucher number LH0431 was obtained from Aroje village, Ogbomoso, and deposited in the herbarium of the Department of Pure and Applied Biology, Ladoke Akintola University of Technology, Ogbomoso. The fruit parts were separated to obtain the seed and seed shell, which were cut into pieces, air-dried for 5 days under ambient condition and then milled into powdery form using an electric blender.
2.2 Preparation of Extracts
One gram of the milled seed (CSE) and seed shell (CSS) were weighed and suspended in 10 ml of distilled water. The extracts were obtained by heating in water bath at 60 °C for 1 h. The extracts were then filtered using Whatman No. 1 filter paper and then centrifuged at 4000 rpm for 20 min. The supernatants were collected and used for further processes.
2.3 Biogenic Synthesis of AgNPs
The respective supernatant obtained was used to synthesize AgNPs as previously described [3, 4]. About 1 ml of the extract was added to the reaction vessel containing 40 ml of 1-mM silver nitrate (AgNO3) solution for the reduction of silver ion. The reaction was carried out in static condition at room temperature (30 ± 2 °C) for 2 h. The formation of AgNPs was monitored through visual observation of the change of colour and measurement of the absorbance spectrum of the reaction mixture using UV–visible spectrophotometer (Cecil, USA).
Fourier transform infrared (FTIR) spectroscopy analysis was carried out using IRAffinity-1S Spectrometer (Shimadzu, UK) on the powder sample of AgNPs according to Bhat et al. [32]. The AgNP solution was centrifuged at 10,000 rpm for 20 min. The solid residue obtained was then dried at room temperature, and the powder obtained was used for FTIR measurement using KBr pellets.
The transmission electron microscopy (TEM) micrograph was obtained as follows. A drop of nanoparticles in suspension was placed on a 200-mesh hexagonal copper grid (3.05 mm) (Agar Scientific, Essex, UK) coated with 0.3 % formvar dissolved in chloroform. The particles were allowed to settle for 3–5 min on the grid, the excess liquid flicked off with a wick of filter paper, and the grids were then air-dried before TEM viewing. Micrograph was obtained using a JEM-1400 (JEOL, USA) operating at 200 kV.
2.4 Antimicrobial Activities of Synthesized AgNPs
The antibacterial property of the synthesized AgNPs was investigated using the agar diffusion method as previously described [4]. Clinical isolates of Escherichia coli, Klebsiella granulomatis and Pseudomonas aeruginosa obtained from LAUTECH Teaching Hospital, Ogbomoso, were used as test organisms. Each bacterium was grown overnight in peptone water, and 18-h culture was used to seed the plates of Mueller–Hinton Agar (Lab M Ltd., UK) with the aid of sterile cotton swab stick. The plates were then bored using cork borer (7 mm) to create wells. The wells were irrigated with 100 μl of graded concentrations of AgNPs prepared by dispersion in sterile distilled water. The plates were thereafter incubated at 37 °C for 24 h. At the end of incubation, the plates were examined for the zones of inhibition, which were measured.
2.5 Antibacterial Susceptibility Test
The test bacterial isolates were screened for susceptibility using a panel of antibiotics on Mueller–Hinton Agar plates by disc diffusion as previously demonstrated [33, 34]. The bacterial isolates were tested on the discs (Abtek Biologicals Ltd., UK) impregnated with antibiotics containing (μg) the following: ceftazidime (Caz), 30; cefuroxime (Crx), 30; gentamicin (Gen), 10; cefixime (Cxm), 5; ofloxacin (Ofl), 5; augmentin (Aug), 30; nitrofurantoin (Nit), 300; and ciprofloxacin (Cpr), 5. The plates were incubated at 37 °C for 48 h, and afterwards, the zones of inhibition were examined and interpreted accordingly [35] considering the appropriate breakpoints [36].
3 Results and Discussion
3.1 Biogenic Formation of AgNPs
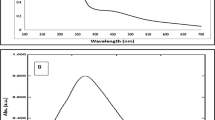
The formation of AgNPs was facilitated by the seed and seed shell extracts within a period 10 min, with the colour stabilizing within 20 min. This led to the development of yellowish orange and brown colour for the seed and seed shell-mediated AgNPs, respectively (Fig. 1). Colloidal AgNP solutions of varying colours including yellowish, brown and dark brown have been reported by several authors [3, 4, 37, 38] due to composition of different macromolecules responsible for the catalytic formation and stabilization of the particles. The biosynthesized AgNPs showed maximum absorbance readings at wavelengths of 457.5 and 454.5 nm (Figs. 2a and 3a) for seed and seed extract-mediated AgNPs, respectively, indicating the formation of AgNPs. These values are within the range reported for AgNPs [3, 4, 39–42]. On storage, the AgNPs showed stability without any form of aggregation or deterioration in UV–vis absorption spectra after 3 months (Figs. 2b and 3b).
The FTIR results showed strong peaks at 3292.49, 2086.98 and 1631.78 cm−1 for seed extract-mediated AgNPs (Fig. 4), while peaks of 3302.13, 2086.05 and 1633.71 cm−1 (Fig. 5) were obtained for seed shell extract-mediated AgNPs, all indicating that proteins were the capping and stabilization molecules in the biogenic synthesis of AgNPs. The bands 3292 and 3302 are typical of N–H bond of amines, while 1631 and 1633 are indicative of C=C stretch of alkenes or C=O stretch of amides, respectively [43]. The active principles of kola nut which include the presence of ~9.5 % crude protein and richness in alkaloids, caffeine (2.8 %), theobromine (0.05 %) and kolatine [21] are biomolecules rich in these bonds.
The AgNPs were spherical in shape with sizes ranging from 8 to 50 and 5 to 40 nm for seed and seed shell-mediated AgNPs, respectively (Figs. 6a and 7a). These shape and sizes are in agreement with those earlier reported [3, 4, 40, 41]. The particles were well dispersed, an indication of good stability to avoid aggregation. The energy-dispersive X-ray (EDX) patterns (Figs. 6c and 7c) showed the presence of silver in the AgNP solution [4, 5, 44] to the tune of 8.6 and 45.1 % in the seed and seed shell-mediated AgNPs, respectively, with the characteristic ring-like selected area electron diffraction (SAED) patterns (Figs. 6b and 7b) typical of the face-centred cubic crystalline structure of silver [43]. It can therefore be concluded from these results that seed and seed shell extracts of C. nitida can find applications in the biogenic and eco-friendly synthesis of AgNPs, thereby establishing the nanobiotechnological relevance of the plant.
3.2 Antibacterial Activities of Biosynthesized AgNPs
The biosynthesized AgNPs displayed considerable inhibitory activities against some clinical isolates of bacteria (Figs. 8 and 9). While the bacterial isolates were multidrug-resistant strains showing resistance to two to eight antibiotics (Table 1), the AgNPs at concentrations of 50–150 μg/ml effectively inhibited strains of E. coli, P. aeruginosa and K. granulomatis to the tune of 10–32 mm. Comparatively, seed shell extract-mediated AgNPs had better activities with minimum inhibitory concentration (MIC) of 50 μg/ml against all the tested isolates, while the MICs of seed extract-mediated AgNPs were obtained as 50, 80 and 120 μg/ml against E. coli, P. aeruginosa (wound) and P. aeruginosa (burn), respectively (Figs. 8 and 9). The overall greater potency of seed shell extract-mediated AgNPs may be attributed to the high concentration of Ag (45.1 %) in the colloidal AgNPs as obtained in the EDX data compared to 8.6 % of seed extract-mediated AgNPs. Nevertheless, the antibacterial activity of the AgNPs against multidrug-resistant bacteria is noteworthy, in view of widespread of resistant bacteria in the environment. We have previously reported high level of antibiotic resistance among bacterial isolates obtained from diverse sources in Nigeria [33, 34, 45–51]. The antibacterial activities of AgNPs as obtained in this study are similar to those reported in previous studies [3–5, 7, 10, 42, 43, 52]. For instance, Salem et al. [5] reported zones of inhibition of 7–19 mm for AgNPs biosynthesized using leaf and latex of Ficus sycomorus against some bacterial strains, while Augustine et al. [7] obtained zones of 8–12 mm for strains of E. coli and Staphylococcus aureus using AgNPs synthesized from leaf extract of Piper nigrum. However, the present study has shown more potent activities of AgNPs against multidrug-resistant bacteria, thereby demonstrating the efficacy of the particles, which can be applied for biomedical applications to combat drug-resistant bacteria.
4 Conclusion
This study has demonstrated the feasibility of using seed and seed shell extracts of C. nitida for the biogenic, green and eco-friendly synthesis of AgNPs. This led to the formation of spherical-shaped AgNPs with sizes ranging from 5 to 50 nm. The particles displayed high potency against multidrug-resistant clinical bacterial isolates, thereby establishing the relevance of biosynthesized AgNPs in biomedical applications. The seed shell extract-mediated AgNPs with MIC of 50 μg/ml were found to have better antibacterial activities compared to seed extract mediated-AgNPs with MIC in the range of 50–120 μg/ml. It can therefore be concluded that extracts of C. nitida can find useful applications in the biosynthesis of novel nanoparticles, thereby expanding the biotechnological potential of the plant. To the best of our knowledge, this report represents the first reference to C. nitida in the green synthesis of AgNPs.
References
Lateef, A., Adelere, I. A., Gueguim-Kana, E. B. (2015). Bacillus safensis LAU 13: a new source of keratinase and its multi-functional biocatalytic applications. Biotechnology and Biotechnological Equipment, 29, 54–63.
Lateef, A., Adelere, I. A., Gueguim-Kana, E. B. (2015). The biology and potential biotechnological applications of Bacillus safensis. Biologia, 70, 411–419.
Lateef, A., Adelere, I. A., Gueguim-Kana, E. B., Asafa, T. B., Beukes, L. S. (2015). Green synthesis of silver nanoparticles using keratinase obtained from a strain of Bacillus safensis LAU 13. International Nano Letters, 5, 29–35.
Lateef, A., Ojo, S.A., Azeez, M.A., Asafa, T.B., Yekeen, T.A., Akinboro, A., Oladipo, I.C., Gueguim-Kana, E.B., Beukes, L.S. (2015). Cobweb as novel biomaterial for the green and eco-friendly synthesis of silver nanoparticles. Applied Nanoscience, doi: 10.1007/s13204-015-0492-9.
Salem, W. M., Haridy, M., Sayed, W. F., Hassan, N. H. (2014). Antibacterial activity of silver nanoparticles synthesized from latex and leaf extract of Ficus sycomorus. Industrial Crops and Products, 62, 228–234.
Shivaji, S., Madhu, S., Singh, S. (2011). Extracellular synthesis of antibacterial silver nanoparticles using psychrophilic bacteria. Process Biochemistry, 46, 1800–1807.
Augustine, R., Kalarikkal, N., Thomas, S. (2014). A facile and rapid method for the black pepper leaf mediated green synthesis of silver nanoparticles and the antimicrobial study. Applied Nanoscience, 4, 809–818.
Kumar, B., Smita, K., Cumbal, L., Debut, A. (2014). Sacha inchi (Plukenetia volubilis L.) oil for one pot synthesis of silver nanocatalyst: an ecofriendly approach. Industrial Crops and Products, 58, 238–243.
Mishra, A., Kumari, M., Pandey, S., Chaudhry, V., Gupta, K. C., Nautiyal, C. S. (2014). Biocatalytic and antimicrobial activities of gold nanoparticles synthesized by Trichoderma sp. Bioresource Technology, 166, 235–242.
Nazeruddin, G. M., Prasad, N. R., Prasad, S. R., Shaikh, Y. I., Waghmare, S. R., Adhyapak, P. (2014). Coriandrum sativum seed extract assisted in situ green synthesis of silver nanoparticle and its anti-microbial activity. Industrial Crops and Products, 60, 212–216.
Shanmugam, N., Rajkamal, P., Cholan, S., Kannadasan, N., Sathishkumar, K., Viruthagiri, G., et al. (2014). Biosynthesis of silver nanoparticles from the marine seaweed Sargassum wightii and their antibacterial activity against some human pathogens. Applied Nanoscience, 4, 881–888.
El-Batal, A. I., ElKenawya, N. M., Yassin, A. S., Amin, M. A. (2015). Laccase production by Pleurotus ostreatus and its application in synthesis of gold nanoparticles. Biotechnology Reports, 5, 31–39.
Yallappa, S., Manjanna, J., Dhananjaya, B. L. (2015). Phytosynthesis of stable Au, Ag and Au–Ag alloy nanoparticles using J. sambac leaves extract, and their enhanced antimicrobial activity in presence of organic antimicrobials. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 137, 236–243.
Agharkar, M., Kochrekar, S., Hidouri, S., Azeez, M. A. (2014). Trends in green reduction of graphene oxides, issues and challenges: a review. Material Research Bulletin, 59, 323–328.
Akinoso, R., Aremu, A. K., Balogun, I. S. (2014). Some physical properties of kola nuts—a response surface approach. International Agrophysics, 28, 251–255.
Odebode, A. C. (1996). Phenolic compounds in the kola nut (Cola nitida and Cola acuminata) (Sterculiaceae) in Africa. Revista de Biología Tropical, 44, 513–515.
Asogwa, E. U., Anikwe, J. C., Ihokwunye, F. C. (2006). Kola production and utilization for economic development. African Scientist, 7, 4–5.
Dewole, E. A., Dewumi, D. F. A., Alabi, J. Y. T., Adegoke, A. (2013). Proximate and phytochemical of Cola nitida and Cola acuminata. Pakistan Journal of Biological Science, 2013, 1–4.
Jarvis, G. (2002). The rise and fall of cocaine cola. https://www.lewrockwell.com/2002/05/gail-jarvis/the-rise-and-fall-of-cocaine-cola/ (2002). Accessed 25 April, 2015.
Lowe, H. I. C., Watson, C. T., Badal, S., Peart, P., Toyang, N. J., Bryant, J. (2014). Promising efficacy of the Cola acuminata plant: a mini review. Advances in Biological Chemistry, 4, 240–245.
Orwa, C., Mutua, A., Kindt, R., Jamnadass, R., Anthony, S. (2009). Agroforestry tree database: a tree reference and selection guide version 4.0 (http://www.worldagroforestry.org/sites/treedbs/treedatabases.asp). Accessed on 19 June, 2015.
Edison, T. J. I., & Sethuraman, M. G. (2012). Instant green synthesis of silver nanoparticles using Terminalia chebula fruit extract and evaluation of their catalytic activity on reduction of methylene blue. Process Biochemistry, 47, 1351–1357.
Muniyappan, N., & Nagarajan, N. S. (2014). Green synthesis of silver nanoparticles with Dalbergia spinosa leaves and their applications in biological and catalytic activities. Process Biochemistry, 49, 1054–1061.
Kumar, S. S., Venkateswarlu, P., Rao, V. R., Rao, G. N. (2013). Synthesis, characterization and optical properties of zinc oxide nanoparticles. International Nano Letters, 3, 30–36.
Rajeshkumar, S., Ponnanikajamideen, M., Malarkodi, C., Malini, M., Annadurai, G. (2014). Microbe-mediated synthesis of antimicrobial semiconductor nanoparticles by marine bacteria. Journal of Nanostructure and Chemistry, 4, 96–102.
Gopinath, K., Venkatesh, K. S., Ilangovan, R., Sankaranarayanan, K., Arumugam, A. (2013). Green synthesis of gold nanoparticles from leaf extract of Terminalia arjuna, for the enhanced mitotic cell division and pollen germination activity. Industrial Crops and Products, 50, 737–742.
Fayaz, A. M., Balaji, K., Girilal, M., Yadav, R., Kalaichelvam, P. T., Venketesan, R. (2010). Biogenic synthesis of silver nanoparticles and their synergistic effect with antibiotics: a study against gram-positive and gram-negative bacteria. Nanomedicine: Nanotechnology, Biology and Medicine, 6, 103–109.
Niraimathi, K. L., Sudha, V., Lavanya, R., Brindha, P. (2013). Biosynthesis of silver nanoparticles using Alternanthera sessilis (Linn.) extract and their antimicrobial, antioxidant activities. Colloids and Surfaces B: Biointerfaces, 102, 288–291.
Khanna, A. S. (2008). Nanotechnology in high performance paint coatings. Asian Journal of Experimental Science, 21(2), 25–32.
Cho, K. H., Park, J. E., Osaka, T., Park, S. G. (2005). The study of antimicrobial activity and preservative effects of nanosilver ingredient. Electrochimica Acta, 51, 956–960.
Mahendra, R., Alka, Y., Aniket, G. (2009). Silver nanoparticles as a new generation of antimicrobials. Biotechnology Advances, 27(1), 76–83.
Bhat, R., Deshpande, R., Ganachari, S. V., Huh, D. O., Venkataraman, A. (2011). Photo-irradiated bio-synthesis of silver nanoparticles using edible mushroom Pleurotus florida and their antibacterial activity studies. Bioinorganic Chemistry and Applications, 2011, 1–7.
Lateef, A., Davies, T. E., Adelekan, A., Adelere, I. A., Adedeji, A. A., Fadahunsi, A. H. (2010). Akara Ogbomoso: microbiological examination and identification of hazards and critical control points. Food Science and Technology International, 16, 389–400.
Lateef, A., Ojo, M.O. (2015). Public health issues in the processing of cassava (Manihot esculenta) for the production of ‘lafun’ and the application of hazard analysis control measures. Quality Assurance and Safety of Crops and Foods, doi: 10.3920/QAS2014.0476.
Chortyk, T. O., Severson, R. F., Cutler, H. C., Siesson, V. A. (1993). Antibiotic activities of sugar esters isolated from selected Nicotiana species. Bioscience Biotechnology and Biochemistry, 57, 1355–1356.
Andrews, J. M. (2005). BSAC standardized disc susceptibility testing method (version 4). Journal of Antimicrobial Chemotheraphy, 56, 60–76.
Kalishwaralal, K., Deepak, V., Ramkumarpandian, S., Nellaiah, H., Sangiliyandi, G. (2008). Extracellular biosynthesis of silver nanoparticles by the culture supernatant of Bacillus licheniformis. Material Letters, 62, 4411–4413.
Shaligram, N. S., Bule, M., Bhambure, R., Singhal, R. S., Singh, S. K., Szakacs, G., et al. (2009). Biosynthesis of silver nanoparticles using aqueous extract from the compactin producing fungal strain. Process Biochemistry, 44, 939–943.
Thirumurugan, A., Tomy, N. A., Kumar, H. P., Prakash, P. (2011). Biological synthesis of silver nanoparticles by Lantana camara leaf extracts. International Journal of Nanomaterials and Biostructure, 1(2), 22–24.
Zaki, S., El-Kady, M. F., Abd-El-Haleem, D. (2011). Biosynthesis and structural characterization of silver nanoparticles from bacterial isolates. Material Research Bulletin, 46, 1571–1576.
Kannan, R. R. R., Arumugam, R., Ramya, D., Manivannan, K., Anantharaman, P. (2013). Green synthesis of silver nanoparticles using marine macroalga Chaetomorpha linum. Applied Nanoscience, 3, 229–233.
Priyadarshini, S., Gopinath, V., Priyadharsshini, N. M., Ali, D. M., Velusamy, P. (2013). Synthesis of anisotropic silver nanoparticles using novel strain, Bacillus flexus and its application. Colloids and Surfaces B: Biointerfaces, 102, 232–237.
Shankar, S., Jaiswal, L., Aparna, R. S. L., Prasad, V. (2014). Synthesis, characterization, in vitro biocompatibility, and antimicrobial activity of gold, silver and gold silver alloy nanoparticles prepared from Lansium domesticum fruit peel extract. Material Letters, 137, 75–78.
Shameli, K., Ahmad, M. B., Zargar, M., Wan Yunus, W. M. Z., Ibrahim, N. A., Sha-banzadeh, P., et al. (2011). Synthesis and characterization of silver/montmorillonite/chitosan bionanocomposites by chemical reduction method and their antibacterial activity. International Journal of Nanomedicine, 6, 271–284.
Adewoye, S. O., & Lateef, A. (2004). Assessment of the microbiological quality of Clarias gariepinus exposed to an industrial effluent in Nigeria. The Environmentalist, 24, 249–254.
Lateef, A. (2004). The microbiology of a pharmaceutical effluent and its public health implications. World Journal of Microbiology and Biotechnology, 20, 167–171.
Lateef, A., Oloke, J. K., Gueguim-Kana, E. B. (2004). Antimicrobial resistance of bacterial strains isolated from orange juice products. African Journal of Biotechnology, 3, 334–338.
Lateef, A., Oloke, J. K., Gueguim-Kana, E. B. (2005). The prevalence of bacterial resistance in clinical, food, water and some environmental samples in Southwest Nigeria. Environmental Monitoring and Assessment, 100, 59–69.
Lateef, A., Oloke, J. K., Gueguim-Kana, E. B., Pacheco, E. (2006). The microbiological quality of ice used to cool drinks and foods in Ogbomoso Metropolis, Southwest, Nigeria. Internet Journal of Food Safety, 8, 39–43.
Lateef, A., Yekeen, T. A., Ufuoma, P. E. (2007). Bacteriology and genotoxicity of some pharmaceutical wastewaters in Nigeria. International Journal of Environmental Health, 1(4), 551–562.
Lateef, A., & Yekeen, T. A. (2006). Microbial attributes of a pharmaceutical effluent and its genotoxicity on Allium cepa. International Journal of Environmental Studies, 63(5), 534–536.
Kanmani, P., & Lim, S. T. (2013). Synthesis and structural characterization of silver nanoparticles using bacterial exopolysaccharide and its antimicrobial activity against food and multidrug resistant pathogens. Process Biochemistry, 48, 1099–1106.
Acknowledgments
AL thanked authority of LAUTECH, Ogbomoso, Nigeria, for the provision of some of the facilities used in this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lateef, A., Azeez, M.A., Asafa, T.B. et al. Cola nitida-Mediated Biogenic Synthesis of Silver Nanoparticles Using Seed and Seed Shell Extracts and Evaluation of Antibacterial Activities. BioNanoSci. 5, 196–205 (2015). https://doi.org/10.1007/s12668-015-0181-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12668-015-0181-x