Abstract
The natural mineral waters of the springs Dona Beja (DBS) and Andrade Júnior (AJS) from Araxá city, in the Brazilian State of Minas Gerais (MG), are well known since the nineteenth century when started the studies of their healing properties for the tuberculosis treatment. In Brazil, the period 1930–1950 corresponded to the construction peak of thermal and non-thermal spas for therapeutic and leisure purposes. In 1944, the President Getúlio Vargas inaugurated a large spa at Barreiro area in Araxá city. The DBS waters have low salinity, high radioactivity, discharge sustained by the recharging rainwater, and expected low residence time in the aquifer due to the maximum recharge–discharge distance of 2–3 km. The AJS waters are hypothermal/thermal, alkaline, sulfured, highly saline, exhibiting deep circulation and discharging in a site dominated by slightly weathered rocks. Barreiro area is also characterized by the exploration of phosphate fertilizer (begun in 1947–1948) and niobium (identified in pyrochlore in 1953). Since the 60s and 70s, the demand for these resources increased, favoring the release of contaminants into the environment. As a consequence, several professionals and institutions belonging to different sectors of the society have pointed out potential environmental problems due to possible contamination of the DBS and AJS waters, with implications for reducing the tourism activities in the region that take an important role on the municipality economy. This study compared the major hydrochemical characteristics of the DBS and AJS waters with those of other surface waters and rainwater occurring in that area for identifying possible degradation of their quality due to the anthropogenic inputs taking place there.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In Brazil, the thermal and mineral waters use is not recent due to the arrival of European immigrants, mainly from Portugal. The discovery of the thermal waters in Caldas Velhas city (Goiás State) occurred in 1722. In 1816, Baron Eschwege did the first official communication to the government of the existence of mineral waters at Araxá city (Minas Gerais State), which were of great therapeutic value (Magalhães 1945).
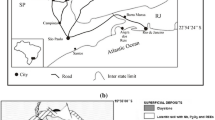
Araxá city is located in southeastern Brazil about 375 km distant from Belo Horizonte (W), 649 km from Brasília (SE), 830 km from Rio de Janeiro (NW), and 581 km from São Paulo (NW) (Fig. 1). The city became very known after a love affair between Dona Beja (Beija) (Anna Jacintha de São José) and a magistrate of the Paracatu town nearby (Dannemann 2013). In the beginning of the nineteenth century, Dona Beja became a leading figure in the local high society (Dannemann 2013) and nowadays Dona Beja Museum hosts some of her belongings in a building located at Araxá city center that served as her past home.

(Modified from DNPM (1987))
Location of the municipality of Araxá in the Brazilian state of Minas Gerais (MG). SP São Paulo State, RJ Rio de Janeiro State
Magalhães (1945) reported the following relevant facts related to the mineral waters of Araxá: the first chemical analyses of the sulfured waters (1886); the study of the healing properties of the waters for the tuberculosis treatment (1890); the discovery by Dr. Andrade Júnior of high radioactive emanations in some waters (1927); and the inauguration by the Brazilian President Getúlio Vargas of a large spa at Barreiro (“Grand Hotel Hydrothermal Complex”, today Grand Hotel Tauá) (1944). This last event was within the period 1930–1950, which corresponded to the construction peak of thermal and non-thermal spas for therapeutic and leisure purposes in the Brazilian states of São Paulo (SP) and Minas Gerais (MG) (Mourão 1992). The Brazilian Code of Mineral Waters (BCMW) was established in that time, under French influence, by Register 7841 published on 8 August 1945 (DFPM 1966).
The distance of Barreiro area from Araxá city center is about 8 km (Fig. 1), occurring there two major springs with different hydrochemical composition: Dona Beja and Andrade Júnior. Such area is also characterized by the presence of phosphate fertilizer and niobium mining activities that have increased since the 60s and 70s following the development of the companies CBMM (Brazilian Company of Mining and Metallurgy), and Bunge/Arafértil (today Vale Fertilizantes) (Viana et al. 1999, Lemos Jr 2012). About 936 × 109 metric tons of Nb2O5 and 20 × 103 tons per year of ground apatite (for use in agriculture) have been produced there (USGS 2018).
Anthropogenic Ba inputs occurred at Barreiro area by the early 1980s due to a leakage occurring at one tailings dam, while other contaminants have been also released there as a consequence of the mining activities, causing intense debate among several professionals and institutions belonging to different sectors of the society (Viana et al. 1999; Santos et al. 2011; Fernandes et al. 2011). This was partially due to possible contamination of the waters of the springs Dona Beja and Andrade Júnior, with implications for reducing the tourism activities in the region that take an important role on the municipality economy.
Under such scenario, this paper reports a comparative hydrochemical study of the natural mineral spring waters Dona Beja and Andrade Júnior of Barreiro area, Araxá city, with surface waters and rainwater occurring there and in other sites nearby, aiming to identify possible contaminants and eventual sources and pathways related to their presence, like surface–groundwater interactions.
Study area and sampling
The regional weather pattern at Araxá city is typically tropical, i.e., dominated by a wet season in the summer (about 50% of the annual precipitation generally falls between December and February) and a dry period in the winter (less than 10% of the annual rainfall often happens between May and September) (Viana et al. 1999). In the period 1972–1996, the lowest, average, and highest annual precipitation in the region corresponded to 102 cm (1990), 156.2 cm, and 203.1 cm (1983), respectively (Viana et al. 1999).
In this study, the natural mineral water samples were taken from the springs Dona Beja (DBS) and Andrade Júnior (AJS), whose discharge is at Barreiro area (Fig. 2). Both springs are linked to different aquifer systems. The water table interception at the terrain surface due to the topography causes the DBS seepage (Beato et al. 2000). FUNTEC (1984) and Beato et al. (2000) classified the aquifer system as granular, free, and semi-confined by clayey levels of the weathered layer, mainly in the intrusive body domain (2 km diameter; variable thickness, reaching up to 200 m at southern portion, close to the niobium mine). The recharge occurs due to direct infiltration in areas of higher altitude (2–3 km is the maximum estimated recharge–discharge distance) and the flow lines at subsurface converge to the Grand Hotel lake and Sal stream. One main recharge area is the 70 ha “green ring” site located upwards of DBS, which allows a significant aquifer replenishment, also contributing the waters of some local dams and of the phosphate mine dyke (Beato et al. 2000).
The AJS seepage is situated in front of the Grand Hotel, possessing a distinct genesis relatively to DBS. The water circulation is deep, discharging in a site dominated by slightly weathered rocks. Faults, fractures, and cracks dominate this free to semi-confined aquifer that mainly occurs in rocks surrounding the carbonatite complex (Beato et al. 2000). The recharge is due to direct rainwater infiltration, in small areas of the outcropping rocks, as well by waters infiltrating in the quartzite residual soil. The groundwater flow is toward several directions as the ore body is circular and exhibits different altitudes (FUNTEC 1984; Beato et al. 2000).
The Barreiro area landscape is dominated by the presence of Grand Hotel lake, basically formed by DBJ waters, and the lake formed by AJS waters (Fig. 2). Two major portions define the surface hydrology (Fig. 2): (1) an eastern one, consisting of Baritina and Mata streams and respective tributaries; (2) a western one, consisting of Cascatinha and Borges streams. The phosphate fertilizer mining area is drained by the surface waters of Cascatinha, Borges, and Baritina streams. The niobium mining area as well discharges waters into Baritina stream and also into Mata stream. All streams flow into the Grand Hotel direction, originating from the Sal stream that corresponds to the unique Barreiro area exit.
COPASA (Sanitation Company of Minas Gerais State) is responsible for the captation of surface waters, its treatment, and distribution in water-supply systems of Araxá city from three sites located outside of Barreiro area: Areia stream (ARS), Fundo stream (FUS), and Feio stream (FES) (Fig. 2). The monitoring points of surface waters included in this study for comparison with the DBS and AJS waters corresponded to Grand Hotel lake (GHL), Sal stream (SS1 and SS2), Areia stream (ARS), Fundo stream (FUS), and Feio stream (FES) (Fig. 2). Additionally, rainwater samples were also collected at two stations (Table 1): (1) RW1—about 1 km distant from the phosphate fertilizer processing unit of Vale Fertilizantes; (2) RW2—COPASA water treatment and distribution unit. The fieldwork involved sampling of waters and sediments in the dry period/beginning of the wet season in the region (from June to October).
Analytical methods
The groundwater and surface water samples (between 10 and 40 L) were stored in polyethylene flasks previously cleaned by an appropriate treatment (acid washing with 10% nitric acid and rinsing with Milli Q water) finished by washing with the same sampled water (APHA 1989). One composite sample (~ 15 L) of rainwater from each site (RW1 and RW2) was recovered by bulk collectors (dry and wet deposition; Cresswell and Bonotto 2008) installed from August to October and coupled to 20 L-polyethylene flasks.
Temperature, pH, conductivity, and potential redox (Eh) readings of the water samples were done in situ adopting the analytical protocols described by Bonotto (2006). Each flask containing the water sample was kept in iceboxes, and transported up to LABIDRO-Isotopes and Hydrochemistry Laboratory of IGCE/UNESP/Rio Claro-SP, Brazil, for the chemical analysis.
The samples were divided into different aliquots for the determination of the main dissolved cations and anions, heavy metals, and radionuclides (210Po and 238U). The filtrate evaporation to dryness of an aliquot in a previously weighed flask yielded the Total Dissolved Solids (TDS) data (Rice et al. 2012). Titration with 0.02 N H2SO4 (Hach 1992) allowed to determine alkalinity in one 100 mL aliquot of each water sample. Additional aliquots provided the data acquisition by AAS—atomic absorption spectrometry (Na), colorimetry (K, Ca, SiO2, Mg, Cl−, NO3−, SO42−, and PO43−), fluoride-selective electrode, and ICP-AES (inductively coupled plasma atomic emission spectrometry) (Fe, Mn, Al, Cd, Cr, Sr, Ba, Co, Cu, Zn, Pb, and Ni). More details of these analyses are given elsewhere (e.g., Hach 1992; van de Wiel 2003; Bonotto and Silveira 2003).
An aliquot of ~ 10 L of each water sample was used for measuring the dissolved 210Po activity concentration (209Po spike = 8 dpm), whereas ~ 20 L for acquiring the 238U activity concentration and 234U/238U activity ratio (AR) data (232U spike = 4.4 dpm). Bonotto et al. (2009) and Bonotto (2010) described the procedures adopted for such readings as done by alpha spectrometry, whose analytical uncertainties were often ± 10–15% (1σ standard deviation, 5% significance level; Young 1962).
Also relevant for this study are the results obtained for six sediment cores (GHL, SS1, SS2, ARS, FUS, and FES) sampled at Araxá city, adopting the protocols described by Bonotto and Garcia-Tenorio (2019). They exhibited depths of 25–40 cm and were cut into slices from 3 to 5 cm of thickness. Then the sediments were analyzed according to their grain size, chemical, and radiometric composition by several methods (spectrophotometry, X-ray fluorescence, α-spectrometry, etc.) (Bonotto and Garcia-Tenorio 2019).
Results and discussion
Hydrochemical highlights
Table 1 reports the results obtained in the analysis of the physicochemical parameters and dissolved constituents in the water samples. According to temperature, the BCMW establishes the following waters classification (DFPM 1966): cold (< 25 °C), hypothermal (25–33 °C), mesothermal (33–36 °C), isothermal (36–38 °C), and hyperthermal (> 38 °C). Using this guideline, the DBS waters are cold, whereas the AJS waters are hypothermal.
WHO (2011) proposed the following guideline values for constituents of health significance in drinking water: Ba—0.7 mg/L, nitrate (as NO3−)—50 mg/L, fluoride—1.5 mg/L, Cr—50 µg/L, Pb—10 µg/L, Cd—3 µg/L, and U—30 µg/L. Depending on the associated cation, taste (not health) thresholds for Cl− were in the range of 200–300 mg/L (WHO 2011). The dataset reported in Table 1 indicates that F− and Ba are the unique chemicals analyzed that exceed such guideline reference values. In this study, F− concentration above 1.5 mg/L was only found at AJS, but Bonotto (2016) reported F− > 1.5 m/L in several other natural mineral waters. However, the AJS water source is not used for daily human consumption as it is only occasionally ingested from taps available for tourists circulating in the area. Ba concentration levels above 0.7 mg/L were found at Barreiro area, reaching the highest value in GHL (1.3 mg/L) and possibly associated to the pyrochlore generations occurring there. Pyrochlore is a complex mineral group of general formula M2T2X7 (M=Na, Ca, Ba, K, U; T=Nb, Ta, Ti) that justifies the accentuated barium dissemination there. Similar to the AJS waters, the water sources DBS and GHL are also not used in the water-supply system of Araxá city for human consumption.
Krauskopf and Bird (1995) pointed out that the proton and electron activities in the environment are generally represented by the parameters pH and Eh, respectively. The data reported in Table 1 and plotted in Fig. 3 show that all waters would occur in environments changing from transitional to reducing conditions; also, RW1 and RW2 exhibit acidity, whereas the water sources DBS, AJS, GHL, SS1, SS2, ARS, FUS, and FES are alkaline. The spring water AJS exhibited the highest value of electrical conductivity (7140 µS/cm) and TDS (2898 mg/L) (Table 1), as a consequence of its deeper circulation in fractured rocks surrounding the carbonatite complex.
Analytical data of this study plotted in an Eh vs. pH diagram as described by Krauskopf and Bird (1995)
The analytical data reported in Table 1 for the surface waters have been plotted in a Gibbs (1970) diagram as shown in Fig. 4. It shows the major features of the hydrochemistry, i.e., dominant influence of the processes of weathering of rocks and soils compared to the dilution effects by rainwater. The points for GHL, SS1, and SS2 coincide as they are in the same hydrographic basin.
Data of the surface waters analyzed in this study plotted in a Gibbs (1970) diagram
The Piper (1944) diagram allows an useful comparison of the hydrochemical dataset reported in Table 1. It shows that the calcium dominance of RW1 and RW2 possibly reflects on enhanced Ca2+ levels of DBS and FUS (Fig. 5). For dissolved anions, it shows the mixed character of FUS and FES and that the bicarbonate facies dominates RW1, DBS, GHL, SS1, and SS2 (Fig. 5). Chloride dominates RW2, indicating unequal chemical composition of the rainwater in the region, which possibly implies on the enhanced concentration of this anion in ARS. Chloride also dominates AJS, greatly differing from DBS that is dominated by HCO3−, thus, indicating the diverse hydrochemical composition of these spring waters related to their different aquifer systems.
Data for major cations and anions of the waters analyzed in this study plotted in a Piper (1944) diagram
Mobility index (M) for the spring waters and surface waters
Different indices have been reported in the literature for characterizing the heavy metals and radionuclides transfer from rocks, soils, and sediments into the waters. Their typical names are distribution coefficient, geochemical enrichment factor, enrichment factor, adsorption coefficient, among other, and they are dimensionless or expressed in cm3/g (or m3/kg) (Szalay 1964; Langmuir 1978; Borovec 1981; AAEC 1983). The reciprocal of these indices (i.e., given in g/cm3 or kg/m3) has been adopted by Bonotto (2015, 2017) for investigating the radionuclides solubility in different situations. It corresponds to the ratio of the weight of the dissolved constituent per unit volume of solution to its weight per unit weight of the solid phase; the numerator corresponds to the liquid phase, whereas the denominator to the solid phase. In this study, the mobility index (M) is represented by M = Cl/Cs where C is the (activity) concentration of the element in the liquid (l) and solid (s) phases.
The chemical analyses of the sediment core sampled at Araxá city are reported in Table 2. These data are needed for estimating M values of the corresponding constituents analyzed in the surface waters. Table 3 shows the chemical data of carbonatites and mica-rich rocks from the Araxá complex (mean value of 41 samples) as reported by Traversa et al. (2001). Such dataset represents the composition of the rock matrices occurring in the aquifers strata of the groundwater systems associated to the mineral waters DBS and AJS, which allows to estimate their M values. The 210Po activity concentration in Table 3 was determined by the [Po] vs. [U] relationship established from the sediment core data (Table 2). The parameters [U] (uranium concentration, in ppm) and [Po] (210Po activity concentration, in dpm/g) defined the straight line [Po] = 1.6795 [U]−0.9559 as plotted in Fig. 6.
The available data shown in Tables 1, 2, 3 allowed to estimate M values for Si, Al, Fe, Na, K, Ca, Mg, P, Ba, Sr, Cr, Zn, Cu, Ni, Pb, U, and Po as reported in Table 4. Their range was 9.5 × 10−4–1958 kg/m3 for groundwaters DBS and AJS and was 9.7 × 10−7–23.04 kg/m3 for surface waters GHL, SS1, SS2, ARS, FUS, and FES. The most notorious aspect of this index is that it highlights the tendency of Si, Al, Fe, P, Cr, Zn, Cu, Ni, Pb, and Po to remain absorbed in the solid phase of all samples analyzed (M < 1 in Table 4), as well the opposite tendency of Na that would remain dissolved in the liquid phase (M > 1 in Table 4). The elements K, Ca, Mg, Ba, Sr, and U exhibited M < 1 and M > 1 values, according to the water type (spring water, lake water or stream water) and sampling point.
The indices reported in Table 4 for the lake water GHL were plotted in log scale against the indices estimated for the stream waters SS1, SS2, ARS, FUS, and FES, yielding significant Pearson correlation coefficients (Fig. 7). The same happened when plotting the indices obtained for the spring waters DBS and AJS (Fig. 7). However, plots of the groundwater indices against the surface waters’ ones did not result in significant relationships. This would suggest the usefulness of the indices for discriminating processes affecting diverse hydrological resources. Congruent dissolution processes occurring in the surface waters would imply on related mobility coefficients, but they would happen in different ways of those involving the subsurface resources. Unlike in the surface waters, several factors take an important role on the transfer of the various elements into the liquid phase such as the aquifers lithologies and groundwater residence time during the water–rock interactions.
The relationships among the mobility index (M) for the elements analyzed in the spring waters and surface waters sampled at the municipality of Araxá in the Brazilian state of Minas Gerais. The codes are reported in Table 1
The comparison of the M index with simple ratios calculated from the concentration of each dissolved constituent in the spring waters and surface waters also confirms its suitability for investigating the water/rock–soil interactions. For instance, MCa corresponds to 0.0023 kg/m3 and 1.27 kg/m3 for spring water AJS and surface water GHL, respectively, implying that MCa in GHL is circa 550 times higher than MCa in AJS. However, the data in Table 1 show that the Ca concentration in GHL is about six times higher than that of AJS. The high Ca concentration (~ 25%) in carbonatites and mica-rich rocks from the Araxá complex suggests that the highest value (550) is more reliable than the lowest (6) for representing the extent of the processes related to the Ca release into both liquid phases.
Figure 7 shows the relationships among the mobility index (M) for the elements analyzed in the spring waters and surface waters sampled at the municipality of Araxá in the Brazilian state of Minas Gerais. The codes are reported in Table 1.
Possible scenarios derived from dissolved radionuclides
210Po is an intermediary member of the natural mass number (4n + 2) 238U decay series that finishes at the stable 206Pb, according to the sequence: 238U (4.49 Ga, α) → 234Th (24.1 d, β−) → 234Pa (1.18 min, β−) → 234U (0.248 Ma, α) → 230Th (75.2 ka, α) → 226Ra (1622 a, α) → 222Rn (3.83 d, α) → 218Po (3.05 min, α) → 214Pb (26.8 min, β−) → 214Bi (19.7 min, β−) → 214Po (0.16 ms, α) → 210Pb (22.26 a, β−) → 210Bi (5 d, β−) → 210Po (138 d, α) → 206Pb. The 238U descendants are produced continuously in rocks and minerals, since uranium is among the main elements contributing to natural terrestrial radioactivity. Because some of the 222Rn escape from the rocks and minerals to the surrounding fluid phase, such as air, 222Rn emanating from land surfaces is responsible for 210Po present in the atmosphere. 210Po produced by 222Rn is removed from the atmosphere by precipitation, yielding values of about 10 mBq/L in the rainwaters of Araxá city (Table 1). Some 210Po atoms escape the rocks and soils to the surrounding fluid, such as groundwater during interactions between the liquid and solid phases. The values found in groundwaters from Barreiro area corresponded to 8–10 mBq/L (Table 1).
210Po in water is a well-documented radionuclide for health risk, as it has been proposed a guidance level of 0.1 Bq/L for the activity concentration in drinking water to not exceed the reference dose level of the committed effective dose equal to 0.1 mSv from 1 year’s consumption (WHO 2011). In terms of the decay sequence 210Pb (22.26 a, β−) → 210Bi (5 d, β−) → 210Po (138 d, α) →··· both radionuclides 210Pb and 210Po are much more restrictive than 210Bi for ingestion in drinking water, since no guideline level has been proposed for 210Bi (WHO 2011). Therefore, the highest 210Po activity concentration of about 10 mBq/L in the waters analyzed in this study is ten times lower than the maximum value allowed by WHO for the ingestion of this radionuclide in drinking water.
Figure 8 shows the data of the dissolved 210Po activity concentration plotted according to the sampling site. The spring waters DBJ and AJS and rainwaters RW1 and RW2 exhibited the highest values, followed by the surface waters of GHL, whose tendency is to have an intermediary composition among two major end members. The first is the group comprised by the surface waters ARS, FES, FUS, SS1, and SS2, whose mean 210Po activity concentration is 1.5 mB/L. The second is the group including DBJ, AJS, RW1, and RW2, whose average 210Po activity concentration is 9.6 mB/L. The global mean value of the 210Po activity concentration is 5.2 mBq/L, which, within experimental errors, practically coincides with the result found for the GHL sample (5.4 mBq/L).
Previous studies held by Bonotto and de Lima (2010) indicated that 210Po is a conservative radionuclide as its activity concentration (0.21 dpm/L) was the same in Corumbataí River upstream and downstream from Rio Claro city, whereas all major chemical data pointed out poorer conditions of the water quality downstream of that municipality. Such conservative behavior apparently also happens in this study, depending on the water types, as shown in Fig. 8. Two major end members can be recognized, the first comprising the surface waters ARS, FES, FUS, SS1, and SS2 (mean value = 1.5 mB/L) and the second embracing the spring and rainwaters (DBJ, AJS, RW1, and RW2) (mean value = 9.6 mBq/L). The lake water GHL exhibits an intermediate 210Po activity concentration (5.4 mBq/L) of these extreme values. However, it is not possible to use such dataset for performing mixing calculations due to the non-existence of direct hydraulic connection among some streams (ARS, FES, and FUS) and the lake (GHL).
On the other hand, the U-isotopes (238U and 234U) modeling may be adopted for this purpose. The highest dissolved 238U concentration in the waters analyzed was 1.7 µg/L (Table 1) that is ~ 18 times lower than the maximum value of 30 µg/L allowed by WHO (2011) for the ingestion of this radionuclide in drinking water. Worldwide soluble U concentrations generally range from 0.1 to 10 μg/L in rivers, lakes, and groundwaters (Ivanovich and Harmon, 1992). 234U is radiogenic, reaching secular equilibrium with 238U circa 1 Ma in all minerals and rocks remaining closed systems for U. Therefore, the 234U/238U activity ratio (AR) is unity in the bulk of such systems, but water–rock/soil interactions frequently result in AR > 1 for dissolved U (Osmond and Cowart 1976; Ivanovich and Harmon 1992; Baskaran 2011).
Cowart and Osmond (1980) plotted the dissolved U concentration and AR data in groundwaters on a two-dimensional diagram containing the following areas of associative significance for the dissolved U concentration (Fig. 9): oxidized aquifer with “normal” values (1–10 μg/L), oxidized aquifer with “enhanced” levels (> 10 μg/L), and reduced aquifer or strata with low levels (< 1 μg/L). AR values between 1 and 2 define a “normal” worldwide situation, AR > 2 suggests the formation of a U-deposit, and AR < 1 indicates the remobilization of a U-accumulation (Cowart and Osmond 1980). The U concentration and AR data of the spring waters DBS and AJS are plotted in Fig. 9 together with the values obtained for the surface waters. Practically, all of them fitted field IV that characterizes reduced environments with low U levels, agreeing with their classification based on the Eh–pH diagram (Fig. 3). The exception was sample SS2 that tended to fit the oxidized field (Fig. 9), contrarily to its highly reducing character (Fig. 3).
Data of the waters analyzed in this study plotted in the log dissolved U concentration vs. 234U/238U activity ratio (AR) diagram as proposed by Cowart and Osmond (1980)
The reciprocal of the dissolved U concentration (S, in L/µg) and the AR of dissolved U data allow to determine the mixing volumes of different groundwater masses (Osmond et al. 1974). For two water sources, the resulting mixture (T) will have S and AR values falling on a straight line joining the corresponding plots of the two components, whose proportions can be calculated (Osmond et al. 1974). Figure 10 shows that the available data in the study area permit to adjust a straight line considering the spring waters DBS and AJS as end members and the lake water GHL as the resultant water. This is compatible with the hydrological and geomorphological features at Barreiro area, with the U-isotopes modeling suggesting that the relative volumes of the mixing waters DBS and AJS for the lake waters (GHL) are 39% and 61%, respectively. Therefore, the uranium isotopes 234U and 238U are useful tools to quantitatively evaluate the mixing proportions of the spring waters DBS and AJS for the lake waters GHL at Barreiro area in Araxá city.
The 234U/238U activity ratio (AR) plotted against the reciprocal of the U concentration (in L/µg) of the spring waters DBS and AJS and lake water GHL (data reported in Table 1)
Conclusions
In this study, (radio)chemical analysis was performed for samples of the natural mineral waters of the springs Dona Beja (DBS) and Andrade Júnior (AJS) from the municipality of Araxá in the Brazilian state of Minas Gerais, which was compared with the results obtained for samples of rainwater and surface water occurring nearby and in other drainage systems of the region and a 35-cm depth sediments core of Grand Hotel lake, and groundwater of Dona Beja and Andrade Júnior springs. Among the constituents of health significance in drinking water as proposed by WHO in 2011, fluoride concentration above 1.5 mg/L was found at AJS waters that is similar to the results obtained for other natural Brazilian mineral waters and elsewhere. However, this water source is not used for daily human consumption as it is only occasionally ingested from taps available for tourists circulating in the area. Barium concentration levels above 0.7 mg/L were found at DBS and AJS waters, reaching the highest value (1.3 mg/L) in the lake waters GHL that are possibly associated to the wide pyrochlore dissemination occurring there. The waters of DBS and GHL are also not used in the water-supply system of Araxá city for human consumption, like the AJS waters. The chemical analysis of sediments provided by previous studies and of carbonatites and mica-rich rocks from the Araxá complex permitted estimating the mobility index M (in kg.m−3) for several constituents analyzed in the water samples. This index highlighted the tendency of Si, Al, Fe, P, Cr, Zn, Cu, Ni, Pb, and Po to remain absorbed in the solid phase of all samples analyzed (M < 1), as well the opposite tendency of Na that would remain dissolved in the liquid phase (M > 1). The spring waters DBJ and AJS and rainwaters (RW1 and RW2) exhibited the highest values of the 210Po activity concentration, whereas the Sal, Areia, Fundo, and Feio stream waters the lowest ones. The lake water GHL exhibited an intermediate 210Po activity concentration (5.4 mBq/L) among these two major end members of water groups. The 234U/238U activity ratio (AR) and reciprocal of the dissolved U concentration in the DBJ and AJS waters allowed to determine their mixing volumes in the lake water (GHL) composition. The DBS waters contribution was 39%, whereas the AJS waters contribution was 61%, showing the usefulness of the U-isotope modeling for such purpose.
References
AAEC (Australian Atomic Energy Commission Research Establishment) (1983) Radionuclide migration around uranium ore bodies: analogue of radioactive waste repositories. Ann Rep, AAEC, Sydney, p 36
APHA (American Public Health Association) (1989) Standard methods for the examination of water and wastewater. APHA, Washington, DC
Baskaran M (2011) Handbook of environmental isotope geochemistry. Springer, New York
Beato DAC, Viana HS, Davis EG (2000) Evaluation and hydrogeological diagnosis of mineral waters aquifers from Barreiro, Araxá, MG, Brazil. In: ABAS (Brazilian Association of Groundwater) (ed) Proc. I Joint World Congress on Groundwater. ABAS, Fortaleza, pp 1–20 (in Portuguese)
Bonotto DM (2006) Hydro(radio)chemical relationships in the giant Guarani aquifer, Brazil. J Hydrol 323:353–386
Bonotto DM (2010) The Poços de Caldas hot spot: a big blast for nuclear energy in Brazil. Nova Science, New York, p 228
Bonotto DM (2015) 226Ra and 228Ra in mineral waters of southeast Brazil. Environ Earth Sci 74:839–853
Bonotto DM (2016) Hydrogeochemical study of spas ground waters from southeast Brazil. J Geochem Explor 169:60–72
Bonotto DM (2017) The dissolved uranium concentration and 234U/238U activity ratio in groundwaters from spas of southeastern Brazil. J Environ Radioact 166:142–151
Bonotto DM, de Lima JLN (2010) Hydrochemistry and weathering rates on Corumbataí River basin, São Paulo State, Brazil. J Hydrol 383:291–306
Bonotto DM, Garcia-Tenorio R (2019) Investigating the migration of pollutants at Barreiro area, Minas Gerais State, Brazil, by the 210Pb chronological method. J Geochem Explor 196:219–234
Bonotto DM, Silveira EG (2003) Preference ratios for mercury and other chemical elements in the Madeira river, Brazil. J S Am Earth Sci 15:911–923
Bonotto DM, Caprioglio L, Bueno TO, Lazarindo JR (2009) Dissolved 210Po and 210Pb in Guarani aquifer groundwater, Brazil. Radiat Meas 44:311–324
Borovec Z (1981) The adsorption of uranyl species by fine clays. Chem Geol 32:45–58
Cowart JB, Osmond JK (1980) Uranium isotopes in groundwater: their use in prospecting for sandstone-type uranium deposits. US DOE, Rep. GJBX-119, vol 80, p 112
Cresswell RG, Bonotto DM (2008) Some possible evolutionary scenarios suggested by 36Cl measurements in Guarani aquifer groundwaters. Appl Radiat Isot 66:1160–1174
Dannemann FK (2013) Dona Beja–Araxá-Minas Gerais. http://www.recantodasletras.com.br/resenhas/1677087. Accessed 15 Dec 2018 (in Portuguese)
DFPM (Division for Supporting the Mineral Production) (1966) The mining code, the mineral waters code and how applying research in a mineral deposit, 8th ed. DFPM, Rio de Janeiro (in Portuguese)
DNPM (National Department of Mineral Production) (1987) Major mineral deposits of Brazil. DNPM, Brasília (in Portuguese)
Fernandes FRC, Enriquez MA, Alamino RCJ (2011) Mineral resources & territorial sustainability, v. 1. CETEM/MCTI, Rio de Janeiro, pp 283–305
FUNTEC (Minas Gerais Technological Center Foundation) (1984) Ecological conflict diagnosis report including recommended works and measures for mitigation of the ecological impact due to mining, v. 1 and 2. Technical Report. ECOS–Geology, Consulting and Services Ltd., Araxá (in Portuguese)
Gibbs RJ (1970) Mechanisms controlling world water chemistry. Science 170:1088–1090
Hach (1992) Water analysis handbook. Hach Co., Loveland, p 831
Ivanovich M, Harmon RS (1992) Uranium series disequilibrium: applications to environmental problems, 2nd edn. Clarendon Press, Oxford
Krauskopf KB, Bird DK (1995) Introduction to geochemistry. McGraw-Hill Inc., New York, p 647
Langmuir D (1978) Uranium solution-mineral equilibria at low temperatures with applications to sedimentary ore deposits. Geochim Cosmochim Acta 42:547–569
Lemos Jr MA (2012) Studies for evaluating the capacity of the niobium wastes reservoir. M.Sc. Dissertation, School of Mines, Federal University of Ouro Preto, Ouro Preto (in Portuguese)
Magalhães MC (1945) The spa of Araxá. Medical Association of Argentina, Buenos Aires, p 124 (in Portuguese)
Mourão BM (1992) Hydrological medicine—modern therapy of mineral waters and healing spas. Municipal Secretary of Education, Poços de Caldas (in Portuguese)
Osmond JK, Cowart JB (1976) The theory and uses of natural uranium isotopic variations in hydrology. Energy Rev 14:621–679
Osmond JK, Kaufman MI, Cowart JB (1974) Mixing volume calculations, sources and aging trends of Floridan aquifer water by uranium isotopic methods. Geochim Cosmochim Acta 38:1083–1100
Piper AMA (1944) A graphic procedure in the geochemical interpretation of water-analyses. Trans Am Geophys Union 25:914–928
Rice EW, Baird RB, Eaton AD, Clesceri LS (2012) Standard methods for the examination of water and wastewater, 22nd ed. American Public Health Association/American Water Works Association/Water Environment Federation, Washington, DC, p 1496
Santos MS, Carneiro LG, Medeiros G, Sampaio C, Martorell ABT, Gouvea S, Cunha KMD (2011) PIXE analyses applied to characterize water samples. Proc. International Nuclear Atlantic Conference—INAC, ABEN (Brazilian Association of Nuclear Energy), Belo Horizonte, pp 1–6
Szalay A (1964) Cation exchange properties of humic acids and their importance in the geochemical enrichment of UO2 2+ and other cations. Geochim Cosmochim Acta 28:1605–1614
Traversa G, Gomes CB, Brotzu P, Buraglini N, Morbidelli L, Principato MS, Ronca S, Ruberti E (2001) Petrography and mineral chemistry of carbonatites and mica-rich rocks from the Araxá complex (Alto Paranaíba Province, Brazil). An Acad Bras Cienc 73:71–98
USGS (United States Geological Survey) (2018) Mineral resources on-line spatial data: Araxá. https://mrdata.usgs.gov/mrds/show-mrds.php?dep_id=10068132. Accessed 29 Jan 2018
van de Wiel HJ (2003) Determination of elements by ICP-AES and ICP-MS. National Institute of Public Health and the Environment (RIVM), Bilthoven, The Netherlands, p 37
Viana HS, Davis EG, Beato DAC, Cabral JAL (1999) Araxá Project: geoenvironmental study of mineral springs. CPRM (Brazilian Geological Survey), Belo Horizonte, pp 1–125 (in Portuguese)
WHO (World Health Organization) (2011) Guidelines for drinking water quality, 4th edn. WHO Press, Geneva, p 541
Young HD (1962) Statistical treatment of experimental data. McGraw Hill, New York, p 172
Acknowledgements
DMB thanks CNPq (Grant Nos. 301992/2016-9, 400700/2016-6) (National Council for Scientific and Technologic Development) and FAPESP (Grant No. 2018/25332-0) (Foundation for Supporting Research in São Paulo State) in Brazil for financial support of this study. Two anonymous referees are greatly thankful for helpful comments that improved the readability of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is a part of the Topical Collection in Environmental Earth Sciences on “Mineral and Thermal Waters” guest edited by Drs. Adam Porowski, Nina Rman, and Istvan Forizs, with James LaMoreaux as the Editor-in-Chief.
Rights and permissions
About this article
Cite this article
Bonotto, D.M., de Oliveira Thomazini, F. Comparative study of mineral and surface waters of Araxá spa, Minas Gerais State, Brazil. Environ Earth Sci 78, 542 (2019). https://doi.org/10.1007/s12665-019-8539-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12665-019-8539-y