Abstract
The assessment of soil quality is essential to track changes in soils as a result of management practices. Although a range of soil physical and chemical properties have been used internationally to track change in soil quality, work to evaluate a range of soil biological indicators as a means of monitoring soil quality has been more limited. In order to identify key biological indicators of soil quality, a long-term field trial at the Indian Agricultural Research Institute, New Delhi, was conducted which included tillage (main plots), water (subplots) and nutrient (sub-subplots) treatments for both rice and wheat. Here the combined influence of tillage, water and nutrient management after eight cropping cycles of rice–wheat, on selected soil microbial properties, was assessed. Results showed that non-puddling significantly enhanced dehydrogenase activity (5%), microbial biomass carbon (3%) and potentially mineralizable nitrogen (5%) over puddling, whereas the latter treatment hugely benefited soil respiration (48%) and metabolic quotient (41%) in rice. No-tillage resulted in higher values of soil biological indicators under wheat cultivation. Partial substitution of fertilizer N by farmyard manure, sewage sludge and a combination of (FYM + biofertilizer + crop residues/green manure) increased indicators at higher magnitudes, like dehydrogenase activity (36%), microbial biomass carbon (33%) and potentially mineralizable nitrogen (57%), but reduced the metabolic quotient which implied an accumulation of stable organic C under organic nutrient management uniformly after both the crops. The drainage of irrigation water in rice also increased dehydrogenase activity and microbial biomass carbon. In contrast more frequent (five times and three times) irrigations in wheat significantly increased these indicators. Principal component analysis revealed that both microbial metabolic quotient and dehydrogenase activity were the most promising indicators of soil biological quality in the present experimental setup.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Rice–wheat cropping systems of Indo-Gangetic plains of India are among the most ancient and important cropping systems of the world, providing employment, income and livelihood for hundreds of millions of rural and urban poor of South Asia. However, the productivity of crops under rice–wheat cropping systems has, at a minimum, reached a plateau and shows a declining trend in many areas of India, highlighting the issue of declining soil quality. It is, therefore, important to identify sensitive soil indicators responsible for changes in soil quality and to identify those suitable as soil quality indicators to inform the appropriate crop management practices to address long-term agricultural sustainability. Till last decade much of the international literature has focused on soil physical and chemical indicators of soil quality and its response to management pressures (Govaerts et al. 2006; Fuentes et al. 2009). However, more recently interest has grown with respect to soil biological properties that can serve as early and more sensitive indicators of agro-ecosystems in response to soil management (Islam and Weil 2000).
Soil biota is considered an important component of soil organic matter involved in energy and nutrient cycling. It is well established that the more dynamic soil characteristics such as microbial biomass, soil enzyme activity and respiration respond more quickly to changing crop management or environmental conditions than total or available soil organic matter (Dick 1992; Doran et al. 1996) or other available soil nutrients. For example, dehydrogenase reflects the total range of oxidative activity of soil microflora and hence provides a good indicator of microbiological activity (Nannipieri et al. 1990). This has been found to be an effective indicator of soil quality in rice–wheat–jute and other cropping systems (Chaudhury et al. 2005). Balanced application of nitrogen, phosphorus and potassium (100% NPK) can lead to significant increases in dehydrogenase activity (DHA), although over use (150% NPK) can cause a reduction in activity, reflecting the sensitivity of this enzyme (Masto et al. 2007). Among various management options, sesbania (a familiar green manure crop of Leguminosae family in Southeast Asia, often known as Dhaincha with common cultivated species as S. aculeata, S. rostrata) offered highest values of both soil DHA and microbial biomass carbon (MBC) during maize–wheat cultivation (Jat et al. 2015). Potential mineralizable N (PMN) is another important soil biological indicator (Hseu et al. 1999), which can be defined as the fraction of organic nitrogen converted to plant available (or mineral) forms under specific conditions of temperature, moisture, aeration and time. PMN is an indirect measure of nitrogen availability during the crop-growing season. The application of green manure has been reported to increase potentially mineralizable N more effectively than other organic manures, like FYM and straw or crop residues (Kang et al. 2005). The combined use of mineral with organic fertilizers (cattle manure and bio-organic fertilizer) showed beneficial effects on soil microbial biomass, enzyme activity and carbon resource utility in a drip-irrigated cotton system (Tao et al. 2015). The metabolic quotient (qCO2), a widely used calculated indicator, has been used to indirectly estimate more C-stable system in a long-term rice–wheat systems (Majumder et al. 2008) and maize (Tamilselvi et al. 2015) showing better sustainability in terms of soil C-storage corresponding to organic or integrated nutrient management.
It has also been found that microbial biomass carbon in the surface (0–20 cm) soil layers is significantly higher under no-tillage than under conventional tillage (Purakayastha et al. 2008). Another recent study involving conservation agriculture practices (reduced tillage, cover crops and fertilization) under long-term cotton cultivation showed that no-till practices brought positive effects in microbial structure and activity in relation to C, N and P cycling (Mbuthia et al. 2015). Seven years of zero-tilled soils attributed C sequestration as evidenced by 9% and 30% higher soil C and microbial biomass C, respectively, with an increased presence of aromatic functional groups (preservation of recalcitrant C) supported by reduced respiration rates and greater enzyme activities, over annually tilled soil (Mangalassery et al. 2015). Moisture content remained another important environmental factor controlling microbial growth and activity (Feng and Simpson 2009; Singh et al. 2011) as well as CO2 production in soils (Murphy et al. 2007). Overall it has been found that these soil microbial properties are more sensitive indicators of soil quality with a special reference to long-term cropping scenario (Balota et al. 2014).
Few of the published literatures to date had concentrated on the long-term impact of nutrient management on changes in soil biological properties (Masto et al. 2007; Tamilselvi et al. 2015; Mbuthia et al. 2015; Zhang et al. 2015). However, a lack of information exists on soil biological and biochemical indicators where major crop management practices (tillage–water–nutrient) are being applied simultaneously and on a long-term basis. This is particularly important in rice–wheat systems, where rice being grown anaerobically and therefore requires a contrasting management to that of wheat. Rice–wheat is the most widely practiced crop rotation across India, and particularly in the Indo-Gangetic plains (covering a long stretch of alluvial zone in northern and eastern India). With this background, the objective was to assess the effect of different but combined input management (water, tillage and nutrient) on selected biological soil quality indicators. The study was aimed to (1) evaluate the selected biological soil quality indicators under long-term cultivation, (2) evaluate the effects of various crop management practices on these biological indicators, (3) identify the most promising sensitive biological indicators of soil quality in these farming systems of the Indo-Gangetic plain. These soil indicators play a crucial role to agricultural sustainability since most of the biological indicators are closely linked to the nutrient dynamics in soil thus maintaining a connection between healthier soil and plant interactions.
Materials and methods
Site description
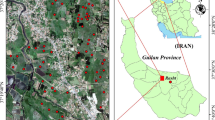
The study site is located at the experimental farm of Indian Agricultural Research Institute (IARI), New Delhi, India (28°37′N latitude, 77°09′E longitude, 228.7 m above MSL). The long-term rice–wheat experiment was continued since 2001. The study area comes under subtropical and semiarid zone of India, specifically the Indo-Gangetic plain. The area recorded mean annual temperatures varied from 18 °C (min) to 35 °C (max) and mean annual precipitation of 650 mm during the experimental seasons of 2008–2009. The rice (Oryza sativa L. cultivar ‘Pusa Sugandh 3’) was transplanted during wet/peak rainy season (June–September) of 2008, and wheat (Triticum aestivum L. cultivar ‘HD 2687’) was sown during dry winter season (October–March) in 2008–2009. The experimental soil was an alluvial, sandy clay loam (sand 48%, silt 29%, clay 23%) of the Holambi series (Typic Haplustepts). Alluvium (Yamuna river transported material) is the parent material of experimental soil, where the topography is flat. Initial soil properties are mentioned in Table 1.
Treatment details
The experiment consisting of three different input managements (tillage, irrigation and nutrient) was designed in a split plot arrangement (Fig. 1). Two tillage treatments, i.e., puddling for transplanted rice and non-puddling for wet seeded rice, were allocated as main plots. The same puddling blocks were allocated to the main plots in succeeding wheat crop. Three water treatments, viz. continuous submergence (W1), irrigation after one day of drainage (W2) and irrigation after three days of drainage (W3) in rice, and five times irrigation, i.e., W1 (crown root initiation, tillering, jointing, flowering and dough stages), three times irrigation, i.e., W2 (crown root initiation, jointing and flowering stages), and two times irrigation, i.e., W3 (crown root initiation and flowering stages), were allocated to the subplots in wheat. Nine nutrient treatments (of three kinds, viz. inorganic, organic and integrated nutrient management) applied to the long-term experiment for both crops as sub-subplots (described in Table 2). For wheat, again splitting was done in each nutrient treatment plot to impose two tillage treatments, i.e., conventional tillage (CT) and no-tillage (NT).
Layout of the field experiment (treatments T1–T9 of the present study were selected from T1, T3, T4, T6, T8, T9, T10, T11, T12 of the original layout of the experiment) (for description, refer to the text and Table 1). Another splitting was done in each nutrient treatment to conventional tillage (CT) and no-tillage (NT) for wheat cultivation
For both the rice and wheat crops, N (120 kg ha−1) was applied as urea following recommended dose of fertilizer. All the treatments including control consisted of the application of recommended P (26.2 kg ha−1) through single super phosphate (SSP) and K (50 kg ha−1) through muriate of potash (MOP). N in three splits and uniform doses of P and K before the transplanting/sowing of the crops were applied. Various organic manures were applied 15 days before transplanting or sowing of the crops at the rate of 2.0 Mg ha−1. The biofertilizer, Azospirillum brasilense CD JA, was applied by coating on the wheat seed surface and by dipping the rice seedling roots in an aqueous suspension culture and maintained a population of about 109 cells g−1 of seed.
Soil sampling and methods of analysis
Composite samples from surface (0–15 cm) soil were collected from treated as well as control plots after eight cropping cycles of rice (number of samples 162) and wheat (number of samples 324) following random sampling technique which completed within first fortnight of October 2008 and March 2009, respectively, after harvesting of rice and wheat. Samples from each plot were stored at 4 °C prior to analysis of the following biological properties. The dehydrogenase activity (DHA) was assessed by colorimetric estimation of triphenylformazan (TPF) formed from triphenyl tetrazolium chloride (TTC) used as an electron acceptor (Klein et al. 1971). Microbial biomass carbon (MBC) was determined by chloroform fumigation and extraction method (Vance et al. 1987), and the carbon content in the extract was determined by alkali trapping of evolved CO2 and titration of the unspent alkali by diluted HCl as per the modified procedure of Jenkinson et al. (1979) and Tate et al. (1988). Potentially mineralizable N (PMN) was determined by anaerobic incubation as described by Keeney (1982). Soil respiration was measured in terms of CO2 evolved during 21 days of incubation at 28 °C using alkali trap in an airtight glass jar by placing 50 g oven dry soil. The alkali trap was replaced at five intervals (1, 3, 7, 10 and 15 days) of the incubation period. The unspent alkali was titrated back with standard HCl using phenolphthalein as an indicator to estimate the CO2–C evolved from soil (Anderson 1982). Metabolic or respiratory quotient (qCO2) was calculated as mg CO2–C evolved per hour per mg of microbial biomass C.
Statistics
The data generated from individual soil biological indicators were analyzed using Windows-based PROC GLM (SAS Institute Inc., 1985), and the least significant difference (LSD) values were tested at 5% level of significance (P = 0.05). Main and interaction effects from main, sub-, and sub-subplot treatments of the split–split plot design were discussed in the results.
Another statistical approach, principal component analysis (PCA), was also used in the present study which provides a non-subjective means to extract and weight information in large and complex data sets (Drinkwater et al. 1996; Reimann et al. 2008). This multivariate statistical technique helped to identify the indicators that best represent variability in a large existing data and to develop a minimum data set of locally relevant function-based soil quality measures (Wander and Bollero 1999). Soil data can be analyzed using PCA to prioritize and reduce the number of indicators or variables that need to be measured in subsequent samplings (Andrews et al. 2001). It may further help to generate a comprehensive understanding of their relative contribution in achieving soil quality in a holistic way. Principal components (PCs) for a data set are defined as linear combinations of the variables that account for maximum variance within the set by describing vectors of closets fit to the n observations in p-dimensional space, subject to being orthogonal to one another (Dunteman 1989). The PCs receiving high eigenvalues followed by variables with high factor loading were assumed as the variables that best represent system attributes (Brejda et al. 2000). Therefore, only the PCs with eigenvalues ≥1 (Kaiser 1960) were examined. At a particular PC, only the variables with high factor loading were retained for minimum data set. In that way the most variable indicators with more factor loadings can be screened and their relative contribution toward soil biological quality can be assumed for the present study, as already established in some earlier researches related to measurement of soil quality (Mandal et al. 2008; Sharma et al. 2008). Data reduction and extraction by PCA and the correlation analysis (both at 5 and 1% probability levels) between indicators were performed by using SPSS version 16.0.
Results
Dehydrogenase activity (DHA)
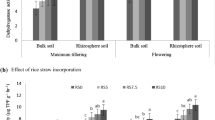
After harvesting of rice, non-puddled soil had significantly higher DHA (0.61 µg TPF g−1 h−1) showing an increase of 5% over the puddled soil (0.58 µg TPF g−1 h−1) (Table 3). Drainage of irrigation water improved the DHA following the trend: W3 > W2 > W1 and increased the DHA value at 14% (W2) and 35% (W3) over W1. T4, FYM-supplemented treatment, also increased DHA (0.81 µg TPF g−1 h−1), followed by T9 where 100% N was substituted by organic sources.
After harvesting of wheat, previously non-puddled soil showed higher DHA in all the water management treatments except for W2 (data not shown). The application of green manure (T5) was most effective (0.52 µg TPF g−1 h−1) in increasing 58% enzymatic activity in soil over T1. No-tillage (NT) showed higher DHA with an increase of 11%, over conventional tillage (CT). Overall irrigation treatments showed the trend W1 > W2 > W3, and with the exception of W3, the water regimes significantly enhanced the DHA under no-tillage. Most of the nutrient management treatments exhibited higher DHA under non-puddling history and no-tillage, whereas the effect of irrigation was not significant after wheat unlike rice cultivation (Table 5).
Microbial biomass carbon (MBC)
Microbial biomass carbon (MBC) was significantly higher in non-puddled (389.1 mg kg−1) compared with puddled soils (379.6 mg kg−1) after rice (Table 3). Like DHA, drainage of irrigation water again increased soil MBC. The most congenial effect of water management was obtained at W3 which had the highest MBC (456.6 mg kg−1) resulting in a 32% increase compared with W1. In puddled soil, water regimes followed the trend for MBC: W3 > W2 > W1, though in non-puddled soil a different trend was observed. Among the various nutrient treatments, T9 (fully organic N source) exhibited the highest MBC (increase of 33%), followed by T7 (27%) over T1, while the use of NPK at higher than the recommended dose (150% NPK, T3) had a negative effect on MBC in a tune of 53% compared with the T9.
In contrast to rice, previously puddled soil showed significantly higher MBC than previously non-puddled soil and also across all the water regimes after harvest of wheat (data not shown). CT plots showed higher MBC than NT plots, and these tillage practices followed a similar trend in W1 and W3 and for most of the nutrient treatments. NT, however, showed higher MBC under FYM, biofertilizer and crop residue (T4, T6, T8) treatments, while overall the control (T1) had higher MBC followed by T7. Frequent irrigation (W1) showed higher MBC than non-frequent irrigation (W3). MBC was found to be highest under control after harvest of wheat. Water regimes had no significant effect on the various nutrient treatments (Table 5).
Potentially mineralizable nitrogen (PMN)
Cultivation of rice without puddling significantly increased potential mineralizable nitrogen (PMN) (18.9 mg kg−1) over puddling treatment (18.0 mg kg−1) (Table 3). Among three water regimes, PMN showed considerably lower variation between continuous submergence (W1) and irrigation after three days of drainage (W3), but both the water regimes showed significantly higher PMN over alternate submergence and drainage (W2). Among the various nutrient treatments, the effect of green manure (T5) seemed to be most prominent followed by addition of FYM (T4) that resulted in higher PMN over T1, T2 and T3. Under puddling, no significant variation existed between W2 and W3, but PMN (18.6 mg kg−1) was significantly higher at W1 over the former water regimes. In both puddled and non-puddled soil T5 invariably showed highest PMN.
After wheat harvest, the previously puddled soil increased the PMN but neither water regimes nor tillage management showed any specific trend with respect to PMN under the two puddling history conditions (data not shown). Generally, NT proved better over CT to improve PMN. However, the fully organic treatment (T9) showed the largest increase in PMN with 73% PMN value compared with the control, followed by 25% N substituted by biofertilizers (T6). Most of the treatments supplemented with organic nutrients showed higher PMN under CT (except T5, T6), whereas the treatments receiving chemical fertilizers showed better response under NT, except T3. Among water management treatments, W2 exhibited higher PMN than others (Table 5).
Soil respiration (RESP)
Soil respiration in terms of CO2–C released (µg g−1 soil h−1) was measured on a cumulative basis up to 21 days of incubation. Results showed a significant positive change (48%) in the puddled treatment (221.2 µg g−1 soil h−1) compared with the non-puddled treatment (149.9 µg g−1 soil h−1) (Table 4). Water regimes showed a decreasing trend on CO2–C evolved as: W1 > W2 > W3; thus, drainage caused a negative impact on RESP (W3 caused −11% change over W1). Across various nutrient treatments, control plots (T1) evolved significantly higher CO2, followed by T2, whereas the other organic treatments were significantly lower than T1. Under the puddled treatment, W2 increased soil respiration, while in non-puddled condition, W1 evolved more CO2–C. Among the various nutrient treatments under puddled condition, T1 followed by T9 evolved more CO2–C, whereas under non-puddled condition, T3 followed by T2 evolved more CO2–C than the other treatments.
In general, the amount of CO2–C evolved under wheat-cropped soil was much less than that of previously rice-cropped soil. No-tillage evolved more CO2–C than conventional tillage irrespective of puddling history (puddled, non-puddled) conditions (data not shown). Overall, NT plots released significantly (5%) more CO2–C than CT. T3 followed by T6 released the maximum CO2–C. Three times irrigations (W2) during the crop growth period increased soil respiration. It is worth mentioning that most of the nutrient treatments were found to release significantly more amount of CO2 under NT compared with CT (Table 5) indicating more actively functioning soil biota present in NT.
Microbial metabolic quotient (qCO2)
The metabolic quotient (qCO2) represents respiration per unit of microbial biomass indirectly measures C-flow through microbial biomass, that is, the energy needed to support a given biomass. A high qCO2 is prevalent in microbial communities in initial stages of development as well as with a large ratio of active to dormant biomass (Anderson 1994). After harvest of rice the qCO2 was significantly higher under puddled soil (0.65) and showed almost 41% increases over non-puddled soil (0.46) (Table 4). Across the water regimes, qCO2 followed the decreasing trend: W1 > W2 > W3, showing the importance of alternate submergence–drainage for achieving a more C-stable system. Continuous submergence during rice cultivation showed a higher qCO2, indicating the role of favorable soil moisture content increasing soil respiration. The quotient was significantly higher under control (T1), 100% NPK (T2) and 150% NPK (T3) as compared to the treatments receiving chemical fertilizers supplemented with organic sources. Among these treatment of sewage sludge (T7) showed minimum qCO2 (0.31) suggesting a stable fraction of carbon under organic nutrient supplementation. Under both the puddling treatments, the effect of water regime on qCO2 followed the same trend (W1 > W2 > W3). T1 and T2 both maintained higher quotient (less stable system) under both the puddling conditions.
After harvesting of wheat, the previously non-puddled soil showed higher qCO2 (data not shown). The water regimes did not show any specific trend across the nine nutrient management options. Overall, higher quotients were found under T6 and T3, implying less C stored in these treatments. However, most of the other treatments showed lower values of qCO2, viz. T4 (0.31), T7 (0.31) and T9 (0.32), indicated a stable C-system. Three times irrigation (W2) lowered the value of qCO2 during wheat. The control and the treatments receiving only chemical fertilizers decreased the quotient values under CT, whereas the treatments receiving partial substitution of N through organic sources showed lower values under NT (except T7 and T9) (Table 5).
Correlations, principal component analysis and minimum data set
After rice crop, indicators were found to be independent, except for qCO2, which was significantly correlated (at 1% probability level) both with soil respiration (r = 0.711) and negatively with MBC (r = −0.618), which could be predicted as qCO2 was derived from these two factors (respiration and MBC) only (Table 6A). Similarly after wheat crop, indicators again did not show much correlation (Table 6B), though qCO2 was correlated with soil respiration (r = 0.426) and MBC (r = −0.858). Interestingly, MBC had a significantly negative correlation (r = −0.254) with soil respiration for wheat-grown soil. Yield of the crops was not well correlated with indicators, except soil respiration and rice yield.
Collecting a minimum data set helps to identify locally relevant soil indicators and to evaluate the link between selected indicators and significant soil and plant properties. A minimum set of indicators is also required to obtain a comprehensive understanding of the soil attributes evaluated. More importantly they serve as a useful tool for screening the condition, quality and health of soil (Doran and Parkin 1994). After harvest of both the crops, analyzed soil biological indicators were screened through PCA. Five PCs summed up of 100% cumulative variation were chosen ranging the eigenvalues from 2.000 to 0.082 (Fig. 2a). Under PC1, qCO2 that exhibited highest factor loading (0.685) was chosen directly for minimum data set (MDS). Under PC2, DHA emerged with 0.726 as highest factor loading. Under PC3, the highly loaded factor PMN (0.961) was chosen for MDS. Under PC4 and PC5, again DHA and qCO2 showing highest factor loadings, respectively, were already selected for MDS. So, for the present study most sensitive and variable biological indicators screened for MDS in rice-cropped soil were qCO2, DHA and PMN.
On the other hand, wheat-cropped aerobic soil created contrasting physical–chemical and biological environment, so separate PCA was carried out to identify most variable biological indicator(s) under this situation. Here also five PCs summed up of 100% cumulative variation were chosen ranging the eigenvalues from 2.114 to 0.103 (Fig. 2b). qCO2 again exhibited highest factor loading (0.655) under PC1 was prioritized to enter into MDS. In PC2, DHA emerged with highest factor loading (0.969). For PC3, the highly loaded factor is PMN (0.965) which was chosen for MDS. Under PC4 and PC5, again RESP and qCO2 showed highest factor loadings, respectively. So, most variable biological indicators for MDS under wheat-cropped soil were qCO2, DHA, PMN and RESP, quite similar to the set of indicators screened for MDS for rice soil. It can also be predicted from PCA that for rice–wheat system the set of indicators that contribute most to soil biological quality are almost similar for both the crops.
Discussion
As non-puddling and repeated submergence–drainage under rice significantly increased DHA, it might be assumed that aerobic condition might have stimulated enzyme activity through enhancing the active microbial population responsible for the enzyme production (Singh et al. 2009). After harvest of rice, similar condition also increased the MBC significantly; this might be due to enhanced storage of organic carbon and creation of aerobic environments which ultimately boost the living microbial biomass pool (Singh et al. 2009). Increased PMN in non-puddled rice-cropped soil probably occurred due to the existence of an aerobic environment promoting mineralization. However, puddling increased soil respiration, perhaps due to proliferation of root biomass and added more C substrates to soil leads to enhanced heterotrophic respiration by soil biota. On the other hand, no-till treatments in wheat soils facilitated better storage of carbonaceous substrates (Bernacchi et al. 2005; Purakayastha et al. 2008) that usually supports higher MBC as well as fuels microbes to enhance dehydrogenase activity. MBC being a labile fraction quickly responds to C availability; thus, NT soils had higher MBC in some organic-treated plots. Similar observations were also reported elsewhere (Masto et al. 2008; Purakayastha et al. 2008). Other than carbon sequestration, no-tillage management is also suggested as a major mechanism for immobilization of N near the soil surface over conventional tillage (Holland and Coleman 1987). This occurs due to the accretion of organic matter and greater microbial activity near the soil surface with no-tillage (Doran 1987) resulting in more PMN in the former treatment in the present study. On the other hand, conventional tillage promoted faster mineralization of organic sources in the standing wheat crop which resulted in higher PMN contributed by residual pool after its harvest. Significantly more CO2 evolution from NT plots across all the nutrient treatments in wheat-cropped soil was probably due to less disturbance in the population and diversity of soil organisms but could be hampered by mechanical destruction under CT, and similar trends have also been reported elsewhere (Purakayastha et al. 2008). In the present study, higher MBC and decreased qCO2 under NT represented a more stable system where soil C is used by microbes more efficiently, similarly reported under long-term cultivation in a Brazilian Oxisol (Balota et al. 2014) and Chinese Inceptisol (Hu et al. 2013).
Alike DHA, intermittent drainage in rice also favored the soil MBC, possibly due to the increment in redox potential which in turn helps to enhance activity of aerobic microflora (Singh et al. 2011). Decreasing trend of soil respiration from continuous submergence (W1) to irrigation after 1 day (W2) and 3 days (W3) of drainage possibly due to reduction in the emission of CO2 from soils when soil moisture content was low (Quemada and Menacho 2001; Singh et al. 2009). Moreover, continuous submergence during rice cultivation led to a higher qCO2, because of favorable soil moisture content which enhanced the rate of soil respiration caused more C-loss from soil, whereas less frequent irrigation under NT improved the microbial efficiency (lowering the qCO2) established as more stable biological system which is in accordance with previous findings (Eleftheriadis and Turrión 2014).
The application of FYM and other organic sources (sewage sludge, biofertilizers, crop residues and combination of these) improved the DHA in both rice- and wheat-cultivated soils, possibly due to the synergistic effect of the stimulation of microbial growth by the organic substrates added and better retention of soil moisture (Kumar et al. 2014). The present results corroborated with findings of other researchers (Kang et al. 2005; Chaudhury et al. 2005; Singh et al. 2011). Likewise, the treatments receiving different organic sources exhibited higher MBC over chemical fertilizers, and this could be due to better proliferation of microbes (Chaudhury et al. 2005; Manjaiah and Singh 2001; Rudrappa et al. 2006). However, quality and content of added organic manures may be a crucial factor for enhancing soil microbial potential or overall biological quality of soil, and crop residues had limited effects on soil microbial functions (Giacometti et al. 2013). Conversely, MBC was found highest under non-treated (control) plots after harvest of wheat, possibly due to presence of more number of oligotrophic microbes which can survive without any organic C for growth and capable of living in nutrient-deficient environments. Among various nutrient treatments, green manure was found most effective in increasing PMN, followed by FYM, indicating the faster degradation of green manures than other organic nutrients (Kang et al. 2005; Beri et al. 1989). Another important observation emanated from the study was that most of the organic treatments exhibited higher PMN under CT, whereas sole chemical fertilizer treatments showed higher PMN under NT. This often happens due to partial immobilization of nutrients in sole fertilizer treatments. Moreover, existence of wide C/N ratio in no-till soils made these phenomena more prevalent. Control plots (T1) followed by 100% NPK (T2)-treated plots evolved significantly higher CO2, while rest of the organic treatments showed significantly lower soil respiration. Moreover, a higher rate of soil respiration in both control and urea-treated plots attributed to microbial communities lived under nutrient starvation (low supply of organic carbon substrates) (Singh et al. 2009). This resulted in a decline in microbial biomass but accelerated the efficiency of utilization of carbon that caused higher rate of respiration under such unfavorable condition (Hu and Cao 2007). The higher metabolic quotient (respiration rate per unit of microbial biomass) values in T1 and T2 supported the previous finding. The organic (sewage sludge)-treated plots showed minimum qCO2, suggesting that stable carbon fractions dominated under this particular organic treatment compared to chemical fertilizer treatments (Lupwayi et al.1998; Rudrappa et al. 2006; Majumder et al. 2008). Most of the organic supplemented (including FYM, green manure, biofertilizers and crop residues) treatments had lower quotient values under the no-till system; thus, it could be suggested that NT offered an effective carbon preservation management option (Purakayastha et al. 2008). Overall organic nutrient management resulted higher soil quality indices in long-term rice–wheat system by enhancing the values of measured soil quality indicators under combined tillage–water–nutrient management condition (Bhaduri et al. 2014a, b; Bhaduri and Purakayastha 2014). Although under long-term fertilization trials, a combination of organic manure and chemical fertilizer better ensured the biological soil health (Tamilselvi et al. 2015; Zhang et al. 2015). MBC that was found to be negatively correlated with soil respiration after wheat harvest may be due to C substrates present in the soil having been sequestered in the long 21-day incubation period, which may otherwise result a contrasting correlation over short-term (7–10 days) incubation, when fast rate of respiration is often observed. Moreover, the relationship of these soil biological indicators with crop yield has previously been mentioned for long-term rice–wheat system (Bhaduri et al. 2014b).
Conclusions
It is concluded from the study that the selected biological indicators of soil quality are found to be very responsive under the long-term rice–wheat experimental condition. Cultivation of rice without puddling could be used as a soil management practice to enhance important biological indicators of soils like MBC, DHA and PMN, though soil respiration enhanced under puddled rice owing to more stored carbon source. No-tillage in wheat could be practiced for benefitting these soil biological indicators. Partial substitutions of chemical fertilizers with different organic sources were found useful for enhancing the aforesaid bioindicators. Drainage of irrigation water in rice soils was favorable for DHA and MBC apart from saving water economy. Respiratory quotient emerged as a key indicator of soil biological processes, indicating the carbon balance in soil across contrasting input management in rice–wheat cropping system, while DHA remained as second most important indicator to influence soil biological quality. The present study infers that bioindicators should be periodically monitored to judge the qualitative status of soil under cereal based agroecosystem for betterment of soil health and agricultural sustainability.
References
Anderson JPE (1982) Soil respiration. In: Page AL et al (eds) Methods of soil analysis. Part 2, 2nd edn. Agron. Monogr. 9. ASA and SSSA, Madison, pp 831–871
Anderson TH (1994) Physiological analysis of microbial communities in soil: applications and limitations. In: Ritz K et al (eds) Beyond the biomass. British Society of Soil Science. Wiley, New York, pp 67–76
Andrews SS, Mitchell JP, Mancinelli R, Karlen DL, Hartz TK, Horwath WR (2001) On-farm assessment of soil quality in California’s central valley. Agron J 94:12–23
Balota EL, Calegari A, Nakatani AS, Coyne MS (2014) Benefits of winter cover crops and no-tillage for microbial parameters in a Brazilian Oxisol: a long-term study. Agric Ecosyst Environ 197:31–40
Beri V, Meelu OP, Khind CS (1989) Biomass production, N accumulation, symbiotic effectiveness and mineralization of green manures in relation to yield of wetland rice. Trop Agric 66:11–16
Bernacchi CJ, Hollinger SE, Meyers T (2005) The conversion of the corn/soybean ecosystem to no-till agriculture may result in a carbon sink. Global Change Biol 11:1867–1872
Bhaduri D, Purakayastha TJ (2014) Long-term tillage, water and nutrient management in rice–wheat cropping system: assessment and response of soil quality. Soil Till Res 144:83–95
Bhaduri D, Purakayastha TJ, Patra AK, Chakraborty D (2014a) Evaluating soil quality under a long-term integrated tillage–water–nutrient experiment with intensive rice–wheat rotation in a semi-arid Inceptisol, India. Environ Monit Assess 186(4):2535–2547
Bhaduri D, Purakayastha TJ, Bhar LM, Patra AK, Sarkar B (2014b) Impact of integrated management on yield sustainability in relation to soil quality under a rice–wheat cropping system. Natl Acad Sci Lett 37(1):25–31
Brejda JJ, Karlen DL, Smith JL, Allan DL (2000) Identification of regional soil quality factors and indicators: II. Northern Mississipi loess hills and Pallouse prairie. Soil Sci Soc Am J 64:2125–2135
Chaudhury J, Mandal UK, Sharma KL, Ghosh H, Mandal B (2005) Assessing soil quality under long-term rice-based cropping system. Commun Soil Sci Plant Anal 36:1141–1161
Dick RP (1992) A review: long-term effects of agricultural systems on soils biochemical and microbial parameters. Agric Ecosyst Environ 40:25–36
Doran ZW (1987) Microbial biomass and mineralizable nitrogen distributions in no-tillage and plowed soils. Biol Fertil Soils 5:68–75
Doran JW, Parkin TB (1994) Defining and assessing soil quality. In: Doran JW et al (eds) Defining soil quality for a sustainable environment, SSSA Special Publication No. 35, SSSA and ASA, Madison, Wisconsin, USA, pp 3–21
Doran JW, Sarrantonio M, Liebig MA (1996) Soil health and sustainability. Adv Agron 56:1–54
Drinkwater LE, Letourneau DK, Worken F, van Bruggen AHC, Shennan C (1996) Fundamental differences between conventional and organic tomato agroecosystems in California. Ecol Appl 5:1098–1112
Dunteman GH (1989) Principal Components Analysis. Sage Publications, London
Eleftheriadis A, Turrión MB (2014) Soil microbiological properties affected by land use, management, and time since deforestations and crop establishment. Eur J Soil Biol 62:138–144
Feng XJ, Simpson MJ (2009) Temperature and substrate controls on microbial phospholipid fatty acid composition during incubation of grassland soils contrasting in organic matter quality. Soil Biol Biochem 41:804–812
Fuentes M, Govaerts B, De León F, Hidalgo C, Dendooven L, Sayre KD, Etchevers J (2009) Fourteen years of applying zero and conventional tillage, crop rotation and residue management systems and its effect on physical and chemical soil quality. Eur J Agron 30(3):228–237
Giacometti C, Demyan MS, Cavani L, Marzadori C, Ciavatta C, Kandeler E (2013) Chemical and microbiological soil quality indicators and their potential to differentiate fertilization regimes in temperate agroecosystems. Appl Soil Ecol 64:32–48
Govaerts B, Sayre KD, Deckers J (2006) A minimum data set for soil quality assessment of wheat and maize cropping in the highlands of Mexico. Soil Till Res 87(2):163–174
Holland EA, Coleman DC (1987) Litter placement effects on microbial and organic matter dynamics in an agroecosystem. Ecology 68:425–433
Hseu ZY, Chen ZS, Tsai CC (1999) Selected indicators and conceptual framework for assessment methods of soil quality in arable soils of Taiwan. Soil Environ 2:77–88
Hu C, Cao Z (2007) Size and activity of soil microbial biomass and soil enzyme activity in long term field experiments. World J Agric Sci 3(1):63–70
Hu J, Zhu A, Wang J, Dai J, Wang T, Chen R, Lin X (2013) Soil microbial metabolism and invertase activity under crop rotation and no-tillage in North China. Plant Soil Environ 59(11):511–516
Islam KR, Weil RR (2000) Soil quality indicator properties in mid-Atlantic soils as influenced by conservation management. J Soil Water Conserv 55(1):69–78
Jat HS, Singh G, Singh R, Choudhary M, Jat ML, Gathala MK, Sharma DK (2015) Management influence on maize–wheat system performance, water productivity and soil biology. Soil Use Manag 31(4):534–543
Jenkinson DS, Davidson SA, Powlson DS (1979) Adenosine triphosphate and microbial biomass in soil. Soil Biol Biochem 11:521–527
Kaiser HF (1960) The application of electronic computers to factor active soil organic matter pools. Soil Sci Soc Am J 58:1130–1139
Kang GS, Beri V, Sidhu BS, Rupela OP (2005) A new index to assess soil quality and sustainability of wheat-based cropping systems. Biol Fertil Soils 41:389–398
Keeney DR (1982) Nitrogen availability indices. In: Page AL, Miller RH, Keeney DR (eds) Methods of soil analysis, part 2, chemical and microbiological properties, 2nd edn. Agron. Monogr. 9, ASA and SSSA, Madison, Wisconsin, USA, pp 711–733
Klein DA, Loh TC, Goulding RL (1971) A rapid procedure to evaluate dehydrogenase activity of soils low in organic matter. Soil Biol Biochem 1:385–387
Kumar S, Patra AK, Singh D, Purakayastha TJ (2014) Long-term chemical fertilization along with farmyard manure enhances resistance and resilience of soil microbial activity against heat stress. J Agron Crop Sci 200:156–162
Lupwayi NZ, Rice WA, Clayton GW (1998) Soil microbial biomass and carbon dioxide flux under wheat as influenced by tillage and crop rotation. Cand J Soil Sci 79:273–280
Majumder B, Mandal B, Bandyopadhyay PK, GangopadhyayA Mani PK, Kundu AL, Mazumdar D (2008) Organic amendments influence soil organic carbon pools and rice–wheat productivity. Soil Sci Soc Am J 72:775–785
Mandal UK, Warrington DN, Bhardwaj AK, Bar-Tal A, Kautsky L, Minz D, Levy GJ (2008) Evaluating impact of irrigation water quality on a calcareous clay soil using principal component analysis. Geoderma 144(1):189–197
Mangalassery S, Mooney SJ, Sparkes DL, Fraser WT, Sjögersten S (2015) Impacts of zero tillage on soil enzyme activities, microbial characteristics and organic matter functional chemistry in temperate soils. Eur J Soil Biol 68:9–17
Manjaiah KM, Singh D (2001) Soil organic matter and biological properties after 26 years of maize–wheat–cowpea cropping as affected by manure and fertilization in a Cambisol in semiarid region of India. Agric Ecosyst Environ 86:155–162
Masto RE, Chhonkar PK, Singh D, Patra AK (2007) Soil quality response to long-term nutrient and crop management on a semi-arid Inceptisol. Agric Ecosyst Environ 118(1–4):130–142
Masto RE, Chhonkar PK, Purakayastha TJ, Patra AK, Singh D (2008) Soil quality indices for evaluation of long term land use and soil management practices in semi-arid sub-tropical India. Land Degrad Dev 19:1–14
Mbuthia LW, Acosta-Martinez V, De Bryun J, Schaeffer S, Tyler D, Odoi E, Mpheshea M, Walker F, Eash N (2015) Long term tillage, cover crop, and fertilization effects on microbial community structure, activity: implications for soil quality. Soil Biol Biochem 89:24–34
Murphy DV, Stockdale EA, Poulton PR, Willison TW, Goulding KWT (2007) Seasonal dynamics of carbon and nitrogen pools and fluxes under continuous arable and leyarable rotations in a temperate environment. Eur J Soil Sci 58:410–424
Nannipieri P, Greco S, Ceccanti B (1990) Ecological significance of the biological activities in soil. In: Bollog JM, Stotzky G (eds) Soil biochemistry, vol 6. Marcel Dekker Inc., New York, pp 293–355
Purakayastha TJ, Huggins DR, Smith JL (2008) Carbon sequestration in native prairie, perennial grass, no-till, and cultivated palouse silt loam. Soil Sci Soc Am J 72:534–540
Quemada M, Menacho E (2001) Soil respiration 1 year after sewage sludge application. Biol Fertil Soils 33:344–346
Reimann C, Filzmoser P, Garrett G, Dutter R (2008) Principal component analysis (PCA) and factor analysis (FA). In: Reimann C et al (eds) Statistical data analysis explained: applied environmental statistics with R, 1st edn. Wiley, Chichester, UK, pp 211–232
Rudrappa L, Purakayastha TJ, Singh D, Bhadraray S (2006) Long-term manuring and fertilization effects on soil organic carbon pools in a Typic Haplustept of semi-arid sub-tropical India. Soil Till Res 88:180–192
Sharma KL, Grace JK, Mandal UK, Gajbhiye PN, Srinivas K, Korwar GR, Bindu VH, Ramesh V, Ramachandran K, Yadav SK (2008) Evaluation of long-term soil management practices using key indicators and soil quality indices in a semi-arid tropical Alfisol. Soil Res 46(4):368–377
Singh G, Kumar D, Marwaha TS, Singh AK (2009) Influence of tillage, water regimes and integrated nitrogen management practices on soil quality indices in rice (Oryza sativa L.) in the Indo-Gangetic plains. Arch Agron Soil Sci 55(4):439–450
Singh G, Kumar D, Marwaha TS, Singh AK, Srinivasmurthy K (2011) Conservation tillage and integrated nitrogen management stimulates soil microbial properties under varying water regimes in maize–wheat cropping system in northern India. Arch Agron Soil Sci 57:507–521
Tamilselvi SM, Chinnadurai C, Ilamurugua K, Arulmozhiselvan K, Balachandar D (2015) Effect of long-term nutrient managements on biological and biochemical properties of semi-arid tropical Alfisol during maize crop development stages. Ecol Indic 48:76–87
Tao R, Liang Y, Wakelin SA, Chu G (2015) Supplementing chemical fertilizer with an organic component increases soil biological function and quality. Appl Soil Ecol 96:42–51
Tate HS, Rost DJ, Feltham CW (1988) A direct extraction method to estimate soil microbial C: effects of experimental variables and some different calibration procedures. Soil Biol Biochem 20:329–335
Vance ED, Brookes PC, Jenkinson DS (1987) An extraction method for measuring soil microbial biomass. Soil Biol Biochem 19:703–707
Wander MW, Bollero GA (1999) Soil quality assessment of tillage impacts in illinois. Soil Sci Soc Am J 63:961–971
Zhang L, Chen W, Burger M, Yang L, Gong P, Wu Z (2015) Changes in soil carbon and enzyme activity as a result of different long-term fertilization regimes in a greenhouse field. PLoS ONE 10(2):e0118371. doi:10.1371/journal.pone.0118371
Acknowledgements
First author is grateful to IARI, New Delhi, for providing Senior Research Fellowship and to Dr. A.K. Singh and all the technical staff of IARI-Mega Project. Authors also acknowledge Dr. R. Dey, ICAR-DGR for giving suitable suggestions in the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bhaduri, D., Purakayastha, T.J., Patra, A.K. et al. Biological indicators of soil quality in a long-term rice–wheat system on the Indo-Gangetic plain: combined effect of tillage–water–nutrient management. Environ Earth Sci 76, 202 (2017). https://doi.org/10.1007/s12665-017-6513-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12665-017-6513-0