Abstract
The characteristics of mercury in the aquatic environment have been intensively studied in mining areas with heavy mercury pollution but little work has been conducted in urban areas, with no significant Hg source. This paper presents a study of the Haihe River, which flows through an urban area in North China. The concentrations of total mercury (THg) and methylmercury (MeHg) in the river water were 3.6–31.2 and 0.12–3.21 ng/l, and the corresponding values in river sediment were 22.9–374.8 and 0.03–0.46 μg/kg. These values are lower than the reported values from mining areas. The THg concentration in sediment samples collected from the urban areas was higher than that from the rural areas and the global background levels, indicating the influence of urbanization on mercury contamination. Samples of typical riparian and floating plants, reed and hornwort, were collected. Correlation analysis showed that sediment is the major source of THg and MeHg in reed and water is the major source of MeHg in hornwort. The higher bioaccumulation factor of reed indicates its higher potential to accumulate MeHg from the environment.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Mercury is one of the most toxic metals and a global environmental pollutant (Lindqvist et al. 1991; Li et al. 2009). In China, the developing industrial technologies and the lack of waste treatment facilities mean a considerable amount of Hg is transported to aquatic systems through sludge, fertilizers, lime and manures as well as atmospheric deposition. Inorganic Hg is converted to toxic methylmercury (MeHg) under certain conditions, which bioaccumulates in the aquatic food chain. Pollution incidents such as those that occurred in Japan and Iraq (Bakir et al. 1973; Hylander 2001) highlight the toxicity of MeHg to humans. Mercury in the environment has recently received increased attention in China (Streets et al. 2005; Zhang and Wong 2007; Fu et al. 2011). Coal-fired power plants, smelters and other industries are responsible for approximately a quarter of the total global Hg emission per year (Fthenakis et al. 1995; Pacyna and Pacyna 2002). Wu et al. (2006) calculated that total Hg emissions from all anthropogenic sources in China and found that it increased at an average annual rate of 2.9 % between 1995 and 2003, reaching 696 (±307) metric tons in 2003. This could become an important source of Hg in the river system by direct emission or by dry/wet atmospheric deposition. The methylation of inorganic Hg renders these emissions a threat to aquatic systems.
In aquatic systems, sediment is an important Hg sink. The Hg in surface sediments can be transferred to the water column through diffusion and re-suspension, where it enters the aquatic food web (Mason and Lawrence 1999). Many studies have been conducted to gain understanding of the mechanism of MeHg bioaccumulation from soil and sediment to plants. A high level of Hg bioaccumulation in aquatic organisms can occur even with extremely low sediment Hg concentrations (Hammerschmidt and Fitzgerald 2004). Zhang et al. (2010a) reported that rice is an intensive bioaccumulator of MeHg in a mining area of Guizhou, China. García-Sánchez et al. (2009) found that plants growing in soils with higher elemental Hg concentrations had greater Hg contents. Ullrich et al. (2007) found that Hg concentrations were much higher in narrow-leaf cattail than in sediments. Mishra et al. (2009) found that aquatic macrophytes, such as Pistia stratiotes and Azolla pinnata, were efficient in the accumulation and removal of Hg from a coal mine effluent. Studies on Hg accumulation by plants from water have indicated that Hg in the water could also be an important source for aquatic plants (Skinner et al. 2007; Lafabrie et al. 2011). Most research to date on Hg accumulation in plants has focused on areas with Hg levels far above normal or with significant point sources, such as mining areas or coal fired power plants, or on specific crop plants, such as rice and vegetables. Little attention has been paid to rivers in urban areas, with no significant Hg sources. Urbanization may generate some point and non-point sources that may contribute to Hg pollution in aquatic systems. The urban aquatic plants have received little research interest due to their low economic value.
Urbanization is increasing rapidly in China so the study of Hg in urban river systems is extremely valuable. The aim of this work was to investigate the pollution status and distribution of Hg in an urban section of the Haihe River, and to investigate the accumulation of Hg by riparian and floating plants. The relative concentrations of Hg in water, sediment and plants were investigated to show the role of plants in Hg accumulation and transfer between water and sediment.
Materials and methods
Study area
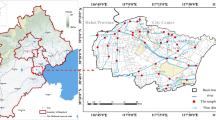
Tianjin is approximately 100 km southeast of Beijing and the second largest industrial and commercial city in North China (Fig. 1). It has an area of approximately 11, 200 km2 and a population of 10 million. As one of the important water sources to the city of Tianjin, the Haihe River has played an important role in maintaining the city’s rapid industrialization and urbanization. It is the largest river system in North China, with many tributaries upstream of Tianjin and flows into the Bohai Sea at Dagukou. The rainy season is concentrated in a very short period (summer) in the Haihe River Basin, and the average annual precipitation is 548 mm in this area. The average width and depth of the river are 180 and 11 m, respectively. The main water source for the Haihe River is Luanhe River, and the water input was 20,000,000 m3 in 2009 (data from Tianjin Water Conservancy Bureau).
In the past decade, the Haihe River has been seriously polluted by the input of industrial, domestic and agricultural effluents. This has had a detrimental effect on the aquatic ecosystem (Qin et al. 1998; Zhen et al. 1999). The continuous input of pollutants associated with rapid population growth and incomplete wastewater treatment has decreased water quality dramatically. The reported concentrations of biochemical oxygen demand (BOD) and chemical oxygen demand (COD) were very high. In 2005–2006, the COD in Haihe River ranged from 6.80 to 11.60 mg/l (Sun et al. 2008), which was much higher than the natural unpolluted rivers. Tianjin is located in a semi-arid area and the low river flow in dry seasons increased the pollution. To reduce seawater intrusion and improve the water quality, the Erdaozha Dike was built approximately 30 km west of the Haihe estuary. The dike is only opened during the flood season but no floods have occurred over the past 10 years, meaning the dike was seldom opened. The main water sources upstream of the dike are tributaries, artificial channels and rainfall. From the dike to the estuary, the river water becomes salty gradually due to sea water intrusion caused by surface tidal waves from the Bohai Sea.
Sample collection and preparation
The sampling area was divided into saline water section (from the Erdaozha Dike to the estuary) and fresh water section (from the upstream to the Erdaozha Dike). The sampling sites are depicted in Fig. 1. The three saline water sites were labeled S1, S2 and S3, and the six fresh water sites were labeled F1, F2, F3, F4 (urban areas), F5 and F6 (suburban areas). Control sites, C1 and C2, were selected at the saline water section and fresh water section respectively. C1 is on the upstream of the Haihe River, and C2 is approximately 15 km east of the Erdaozha Dike. Both of the two sites were located at the agricultural area with no significant human activity.
The sampling was conducted during summer 2010. Sediment samples (0–20 cm) were collected using a cylindrical sediment sampler (Beeker, Holland), and immediately bagged. Water samples (30 cm under the surface) were collected using a Teflon bailer and immediately bottled. The sampling equipment was cleaned thoroughly with deionized water between each site to avoid cross contamination. Water quality parameters [temperature, pH, electric conductivity (EC), dissolved oxygen (DO)] were determined at the time of sampling with a portable water analyzer. Duplicate samples of reed (Phragmites australis) were collected from eight sites (C2, F1, F2, F3, F5, F6, S1 and S2), and hornwort (Myriophyllum spicatum L.) were collected from four sites (C1, F1, F5 and F6).
All plant and sediment samples were individually sealed and cooled during the same-day transportation to the laboratory. Sediment and plant samples were frozen at −20 °C and water samples were kept at 4 °C until analysis.
Laboratory analysis
The water samples were acidified with 20 % HNO3 (v/v) immediately on laboratory arrival. The Hg levels and TOC were determined within 3 days of sampling. The sediment samples were homogenized with a blender after removing large particles (stones and plant residues), freeze-dried for 4 days, and sieved to 100-mesh before analysis.
Plant samples were washed with ultrapure water at least three times. Reed samples were separated into leaf, stem and root but this proved too difficult with hornwort and so leaves and stems were combined in these samples. Following preparation, the samples were covered with aluminum foil, and freeze-dried for 3 days and then grinded into a powder with a grinder (MM400, Retsch, Germany). The grinder was thoroughly cleaned with ultrapure water and dried between samples to avoid cross contamination. The samples were subsequently sealed in acid-cleaned glass bottles and stored in a dryer for analysis.
The THg concentrations of the freeze-dried solid samples and water samples were determined using a Mercury analyzer (DMA-80, Milestone, Italy). The working process of DMA-80 includes thermal composition, catalytic reduction, amalgamation, desorption, and atomic absorption spectroscopy (CVAAS). The instrument calibration and measurement were conducted according to EPA 7473. The absolute detection limit of THg for DMA-80 was 0.005 ng.
The sequential MeHg extraction procedure was based on Maggi’s three-step sequential extraction method (Maggi et al. 2009; Tong et al. 2012). First, solid samples were hydrolyzed using 48 % hydrobromic acid (HBr), then mixed with 20 ml toluene to extract the MeHg from the hydrolyzed liquid phase. At this stage, double extraction was applied to the samples with low MeHg concentrations. After shaking for 30 min and centrifugation at 4,500 rpm for 10 min, 1 ml l-cysteine (0.5 % solution) was added to bind the MeHg in the toluene phase. The MeHg in the cysteine aqueous solution was determined with the DMA-80 Mercury analyzer.
Extraction of MeHg from water samples was conducted following a similar procedure. 200 ml water was added to a 500 ml centrifugal pot with 20 ml HBr (48 %) and mixed with 40 ml toluene. Shaking and centrifugation was the same as that for plant and sediment samples. Double toluene extraction was applied, and 3 ml of 0.5 % (v/w) l-cysteine aqueous solution was used to strip MeHg from the toluene phase and the MeHg in the cysteine aqueous solution was determined with a mercury analyzer.
The limit of quantification (LOQ) for MeHg in 4.0000 g of sediment was 0.017 ng/g, while that for 1.0000 g of dry plant sample and 200 ml of unfiltered water was 0.068 ng/g and 0.24 ng/l, respectively.
The THg determination method was verified by comparison with two certified reference materials (GBW-10029, 850 ± 30 ng/g and SRM-2976, 61 ± 3.6 ng/g). Our results for THg were 834 ± 6 ng/g (n = 7) in GBW10029 and 62.4 ± 1.5 ng/g (n = 7) in SRM-2976. Solid sample MeHg detection was validated with certified reference materials GBW-10029 (840 ± 30 ng/g), SRM-2976 (28.1 ± 0.31 ng/g) and ERM-CC580 (75 ± 4 ng/g). Our corresponding detection values were 814 ± 15 (n = 7), 27.5 ± 0.4 (n = 7) and 68.2 ± 1.4 (n = 7) ng/g. Water sample MeHg detection was validated with diluted MeHg stock solution (0.5 ng/l). Our detection value was 0.519 ± 0.052 ng/l (n = 6). TOC of water was measured using TOC-5000A (Shimadzu, Japan).
Results and discussion
Hg levels in water and sediment
The concentrations of THg and MeHg in the water samples are shown in Table 1. The non-detectable values from C2, F2 and F5 were replaced with 1/2 of the LOQ (0.12 ng/l). The means (and ranges) of THg in water samples were 10.5 (3.6–24.7) ng/l for fresh water, 17.1 (7.4–31.2) ng/l for saline water and 11.9 (11.0–12.9) ng/l for control sites. The corresponding MeHg concentrations were 0.27 (0.12–0.40), 1.40 (0.37–3.21), and 0.25 (0.12–0.37) ng/l. The average aqueous concentration of Hg in natural systems is reported to be less than 30 ng/l (Babiarz et al. 2000, 2003) and all our sampling sites (except S2) were <30 ng/l. The high THg and MeHg values observed at S2 are possibly due to a nearby industrial area with a wastewater outlet into the river.
The concentrations of THg and MeHg in sediments are shown in Table 2. Means (and ranges) of THg in surface sediment were 202.26 (23.77–374.82) μg/kg for the fresh water, 68.40 (59.95–79.28) μg/kg for saline water and 26.96 (22.89–31.03) μg/kg for control. The corresponding MeHg concentrations were 0.18 (0.03–0.46), 0.17 (0.10–0.21), and 0.11 (0.07–0.15) μg/kg. The THg and MeHg concentrations in sediment from the control sties were significantly lower than those from the fresh water and saline water sites and the THg concentration of the saline water sites was much lower than that of the fresh water sites. Across all the sampling sites, the percentage of THg that occurred as MeHg was between 0.03 and 0.48 % with an average of 0.21 %. This is comparable to the reported values in Hudson River sediment (0.07–0.3 %) (Heyes et al. 2004) and in the contaminated sites such as Lavaca Bay (0.6 %) (Bloom et al. 1998).
Table 3 compares the concentrations of THg and MeHg in the Haihe sediments with other estuarine–coastal systems. The concentrations of THg in our study are similar to the reported values from South Florida Estuaries, Lot-Garonne and Lake Shihwa. MeHg values in the Haihe River sediment are similar to those from South Florida Estuaries, Lake Shihwa and the Anadyr Estuary.
Global background levels of THg are reported to be 50 μg/kg in river sediments, 100–300 μg/kg in lake sediments and 50–80 μg/kg in sea sediments (Fujii 1976). The concentrations of THg from control sites (22.89 and 31.03 μg/kg) and saline sites (59.95–79.28 μg/kg) were within the range for uncontaminated sediments, indicating that human activity had no significant impact at these sites. The THg contents (23.77–374.82 μg/kg) in sediments collected from fresh water sites were higher. This section flows through urban and suburban areas, indicating the impact of urbanization on THg concentration in sediment. Both THg and MeHg concentrations from this study were much lower than those reported in soil or sediment from contaminated areas (Hintelmann and Wilken 1995; Ding et al. 2009; Zhang et al. 2010a).
Hg levels in the plants
Tables 4 and 5 show the measured THg and MeHg concentrations of the reed and hornwort in different sampling sites. Elevated THg concentrations were found in the all the samples of leaf of reed, with a mean (and range) of 197.2 (109.9–290.3) μg/kg. The mean THg concentration (and range) found in reed stem was 171.4 (74.2–327.7) μg/kg and the mean (and range) found in reed root was 132.4 (48.6–388.3) μg/kg. The THg concentration in hornwort was low, with its mean (and range) of 68.8 (57.9–91.0) μg/kg. The high THg content in reed leaf can be explained by plant Hg uptake directly from the air through the leaf or stem surface in addition to the uptake from sediment by roots. Previous studies have shown that atmospheric mercury pollution is serious in North China (Fang et al. 2001a, b; Wang et al. 2006, 2009). Ericksen et al. (2003) found that the mean deposition rate of atmospheric Hg on forest foliage was 0.25 ± 0.15 ng/(m2 h) when atmospheric Hg concentrations ranged from 10 to 30 ng/m3. Rutter et al. (2011) measured the uptake of mercury by the leaf of White Ash and found uptake values of 1.3 ± 0.1 × 10−4 ng/(m2 s) in atmospheric Hg concentrations of 4–6 ng/m3.
Mean (and range of) MeHg concentrations in all the samples of the leaf, stem and root of reed were 0.35 (0.18–0.60), 0.65 (0.12–1.98), and 3.73 (0.69–19.30) μg/kg, respectively. The distribution of MeHg in reed shows similar trends to that measured in rice in China (Zhang et al. 2010a). The mean (and range of) MeHg concentration in hornwort was 3.80 (2.11–5.98) μg/kg. The THg and MeHg concentrations measured in the plants of this study are low compared with those reported in rice from mining areas in China but are close to previously reported values from barnyard grass (Echinochloa crusgalli) in the city of Huludao, approximately 400 km east of the sampling area for this study (Zhang et al. 2010b).
A significant correlation in MeHg contents between the leaf and stem of reed (Fig. 2a, R 2 = 0.55, p < 0.05, n = 18) was observed. This indicates that the concentrations of MeHg in the leaf and stem were affected by the same translocation effects. For the plants, the MeHg in the sediment was the main source, and the contribution from MeHg in air was considered to be negligible. No significant correlation (Fig. 2b, R 2 = 0.003, p > 0.05, n = 18) in THg contents was observed between the leaf and stem of reed. This is possibly because THg in the reed leaves originated partly from ambient air through leaf surface absorption, in addition to the THg coming from sediments through root uptake (Zhang et al. 2010a).
Hg accumulation mechanisms for reed and hornwort
Previous studies (Zhang et al. 2010a, b) have reported that in heavily contaminated areas, THg in plants is much lower than that of the soil, while in our study, the mean THg concentrations in plants were close to those in the corresponding sediments. On the other hand, the MeHg concentrations in reed and hornwort were significantly higher than the concentrations in corresponding sediments. The mean site-specific bioaccumulation factor (BAFs, defined as the root/sediment concentration ratios) for THg in reed was approximately 1.19. The corresponding BAF for MeHg was 20.29. A higher accumulation of MeHg than THg has previously been reported in other plants such as vascular plants and bilberry (Gnamus et al. 2000; Schwesig and Krebs 2003). Plants have a natural resistance against some toxic pollutants such as mercury, and have detoxification mechanisms. The detoxification process has been observed in the reduction of Hg2+ in plants, but has not been found effective for MeHg in plants (Hussein et al. 2007).
Previous studies (Gnamus et al. 2000; Zhang et al. 2010a, b) have reported BAFs of rice or barnyard grass usually lower than 0.5; the BAF of THg in reed root in this study was much higher than this. The average BAF of MeHg in reed root in the present study was also higher than the reported values for other plants, which were usually planted in contaminated soil. Plants with a BAF value higher than one are usually classified as intensive bioaccumulators. Some authors have reported that aquatic plants are bioaccumulators of mercury (Ullrich et al. 2007; Mishra et al. 2009) meaning reed has the potential to accumulate mercury from sediment. Many studies investigating Hg accumulation have focused on areas with high soil mercury concentrations (usually over 1,000 μg/kg). The sampling area in our study was much less contaminated. Plant to soil ratios have also been reported high for low Hg control areas in other studies (Sierra et al. 2009).
A positive correlation between THg in the root of reed and sediment was observed (Fig. 3a, R 2 = 0.69, p < 0.05, n = 18), indicating the importance of sediment as a THg source for roots. A significant correlation between MeHg in reed root and sediment was also observed (Fig. 3b, R 2 = 0.70, p < 0.05, n = 18), suggesting that MeHg in sediment may be a major contributor to MeHg in reed roots.
Hornwort is a submerged plant with no roots. It is suspended in the water column and can only absorb nutrients from water. Toxic metal ions are thought to enter cells through the same uptake processes that move essential micronutrient metal ions. Bioaccumulation of MeHg from water has been shown to be significant, and that a higher concentration of Hg in plants can indicate a proportional increase in the Hg level in the water (Mauro et al. 2001). In our study, a positive relationship (p < 0.05, R 2 = 0.554, n = 12) was found between the MeHg in hornwort and water, suggesting that MeHg in water is an important source for the hornwort. No such positive relationship (p > 0.1, R 2 = 0.084, n = 12) was found for THg. It is possible that the THg in hornwort could also originate from suspended particles beside the water. The bioavailability of MeHg and THg to aquatic organisms is influenced by the chemical forms of MeHg and Hg(II) in water. Physical and chemical properties, such as pH, DOC, DO, activity of sulfate-reducing bacteria could also affect the adsorption of Hg by plants by influencing the chemical forms of Hg (Gorski et al. 2008; Merritt and Amirbahman 2009).
Conclusions
This paper presents a study on mercury in the Haihe River in North China. The concentration of THg and MeHg in the water was in the range of natural levels (30 ng/l), except for a higher concentration observed close to an industrial area. The THg and MeHg concentrations in sediments were much lower than the reported soil values from mining areas in China. The THg concentrations at the fresh water sites were slightly higher than the global background values. The results illustrate that THg and MeHg in river water and sediment from non-mining areas are far below that of mining areas, but that urbanization and industrialization influence mercury content in the aquatic environment. For most of the sampled sites, MeHg concentration was positively correlated to THg concentrations, suggesting that MeHg contents were partly controlled by the THg in sediment.
Reed and hornwort are typical species of riparian and floating plants, in the Haihe River. The concentration of THg in reed was higher than that in hornwort, while the MeHg concentrations in the hornwort were much higher than that in the leaves and stems of reed. For reed, the concentrations of THg and MeHg in roots were positively correlated with those in sediment, indicating that sediment is an important source of mercury for reed root. A positive correlation between plant and water Hg concentrations was observed for hornwort, suggesting water is an important source for hornwort. The BAF value of MeHg was 20.29 for reed root which means that reed has a stronger potential to accumulate MeHg from the environment.
References
Babiarz CL, Hoffman SR, Shafer MM, Hurley JP, Andren AW, Armstrong DE (2000) A critical evaluation of tangential-flow ultrafiltration for trace metal studies in freshwater systems: total mercury and methylmercury. Environ Sci Technol 34(16):3428–3434
Babiarz CL, Hurley JP, Krabbenhoft DP, Gilmour C, Branfireun BA (2003) Application of ultrafiltration and stable isotope amendments to field studies of mercury partitioning to filterable carbon in lake and overland runoff. Sci Total Environ 304(1–3):295–303
Bakir F, Damluji SF, Amin-Zaki L, Murtadha M, Khalidi A, al-Rawi NY, Tikriti S, Dahahir HI, Clarkson TW, Smith JC, Doherty RA (1973) Methylmercury poisoning in Iraq. Science 181(4096):230–241
Bloom NS, Gill GA, Cappellino S, Dobbs C, McShea L, Driscoll C, Mason R, Rudd J (1998) Speciation and cycling of mercury in Lavaca Bay, Texas, sediments. Environ Sci Technol 33(1):7–13
Canário J, Vale C, Caetano M (2005) Distribution of monomethylmercury and mercury in surface sediments of the Tagus estuary (Portugal). Mar Pollut Bull 50:1142–1145
Canário J, Branco V, Vale C (2007) Seasonal variation of monomethylmercury concentrations in surface sediments of the Tagus estuary (Portugal). Environ Pollut 148(1):380–383
Ding ZH, Liu JL, Li LQ, Lin HN, Wu H, Hu ZZ (2009) Distribution and speciation of mercury in surficial sediments from main mangrove wetlands in China. Mar Pollut Bull 58(9):1319–1325
Ericksen JA, Gustina MS, Schorranb DE, Johnson DW, Lindberg SE, Coleman JS (2003) Accumulation of atmospheric mercury in forest foliage. Atmos Environ 37(12):1613–1622
Fang FM, Wang QC, Liu RH, Ma ZW, Hao QJ (2001a) Atmospheric particulate mercury in Changchun city, China. Atmos Environ 35(25):4265–4272
Fang FM, Wang QC, Li JF (2001b) Atmospheric particulate mercury concentration and its dry deposition flux in Changchun city, China. Sci Total Environ 281(1–3):229–236
Fthenakis VM, Lipferta FW, Moskowitz PD, Saroff L (1995) An assessment of mercury emissions and health risks from a coal-fired power plant. J Hazard Mater 44(2–3):267–283
Fu XW, Feng XB, Sommar J, Wang SF (2011) A review of studies on atmospheric mercury in China. Sci Total Environ 421–422:73–81
Fujii M (1976) Mercury distribution in lithosphere and atmosphere. In: Kitamura S, Kondo M, Takizawa Y, Fujii M, Fujiki M (eds) Mercury. Kodansha Scientific, Tokyo
García-Sánchez A, Murciego A, Álvarez-Ayuso E, Santa Regina I, Rodríguez-González MA (2009) Mercury in soils and plants in an abandoned cinnabar mining area (SW Spain). J Hazard Mater 168(2–3):1319–1324
Gnamus A, Byrne AR, Horvat M (2000) Mercury in the soil-plant-deer- predator food chain of a temperate forest in Slovenia. Environ Sci Technol 34(16):3337–3345
Gorski PR, Armstrong DE, Hurley JP, Krabbenhoft DP (2008) Influence of natural dissolved organic carbon on the bioavailability of mercury to a freshwater alga. Environ Pollut 154(1):116–123
Hammerschmidt CR, Fitzgerald WF (2004) Geochemical controls on the production and distribution of methylmercury in near-shore marine sediments. Environ Sci Technol 38(5):1487–1495
Heyes A, Millera C, Mason RP (2004) Mercury and methylmercury in Hudson River sediment: impact of tidal resuspension on partitioning and methylation. Mar Chem 90(1–4):75–89
Hintelmann H, Wilken RD (1995) Levels of total mercury and methylmercury compounds in sediments of the polluted Elbe River: influence of seasonally and spatially varying environmental factors. Sci Total Environ 166(1–3):1–10
Hussein HS, Ruiz ON, Terry N, Daniell H (2007) Phytoremediation of mercury and organomercurials in chloroplast transgenic plants: enhanced root uptake, translocation to shoots, and volatilization. Environ Sci Technol 41(24):8439–8446
Hylander LD (2001) Global mercury pollution and its expected decrease after a mercury trade ban. Water Air Soil Poll 125(1):331–344
Kannan K, Falandysz J (1998) Speciation and concentrations of mercury in certain coastal marine sediments. Water Air Soil Poll 103(1–4):129–136
Kannan K, Smith RG, Lee RF, Windom HL, Heitmuller PL, Macauley JM, Summers JK (1998) Distribution of total mercury and methyl mercury in water, sediment, and fish from south Florida estuaries. Arch Environ Contam Toxicol 34(2):109–118
Lafabrie C, Major KM, Major CS, Cebrián J (2011) Arsenic and mercury bioaccumulation in the aquatic plant, Vallisneria neotropicalis. Chemosphere 82(10):1393–1400
Li P, Feng XB, Qiu GL, Shang LH, Li ZG (2009) Mercury pollution in Asia: a review of the contaminated sites. J Hazard Mater 168(2–3):591–601
Lindqvist O, Johansson K, Bringmark L, Timm B, Aastrup M, Andersson A, Hovsenius G, Håkanson L, Iverfeldt Å, Meili M (1991) Mercury in the Swedish environment-recent research on causes, consequences and corrective methods. Water Air Soil Poll 55(1–2):1–261
Maggi C, Berducci MT, Bianchi J, Giani M, Campanella L (2009) Methylmercury determination in marine sediment and organisms by direct mercury analyser. Anal Chim Acta 641(1–2):32–36
Mason RP, Lawrence AL (1999) Concentration, distribution, and bioavailability of mercury and methylmercury in sediments of Baltimore Harbor and Chesapeake Bay, Maryland, USA. Environ Toxicol Chem 18(11):2438–2447
Mauro JBN, Meili M, Karlsson AL, Sundbom M, Coelho-Souza SA, Guimarães JRD, Machado MC, Hylander (2001) Simultaneous dynamics of microbial activity and Hg methylation in the periphyton of macrophyte dominated wetlands, 6th International Conference on mercury as a global pollutant. Minamata, Japan
Merritt KA, Amirbahman A (2009) Mercury methylation dynamics in estuarine and coastal marine environments: a critical review. Earth Sci Rev 96(1–2):54–66
Mikac N, Niessen S, Ouddane B, Wartel M (1999) Speciation of mercury in sediments of the seine estuary (France). Appl Organomet Chem 13(10):715–725
Mishra VK, Tripathi BD, Kim KH (2009) Removal and accumulation of mercury by aquatic macrophytes from an open cast coal mine effluent. J Hazard Mater 172(2–3):749–754
Oh S, Kim MK, Yi SM, Zoh KD (2010) Distributions of total mercury and methylmercury in surface sediments and fishes in Lake Shihwa, Korea. Sci Total Environ 408(5):1059–1068
Ouddane B, Mikac N, Cundy AB, Quillet L, Fischer JC (2008) A comparative study of mercury distribution and methylation in mudflats from two macrotidal estuaries: the Seine (France) and the Medway (United Kingdom). Appl Geochem 23(4):618–631
Pacyna EG, Pacyna JM (2002) Global emission of mercury form anthropogenic sources in 1995. Water Air Soil Poll 137(1–4):149–165
Qin BP, Zhai DH, Yuan Q, Zhang Y (1998) Study on the aquatic ecosystem of Haihe River. J Urban Environ Urban Ecol (in Chinese) 11:48–51
Rutter AP, Schauer JJ, Shafer MM, Creswell JE, Olson MR, Robinson M, Collins RM et al (2011) Dry deposition of gaseous elemental mercury to plants and soils using mercury stable isotopes in a controlled environment. Atmos Environ 45(4):848–855
Schafer J, Blanc G, Cossa D, Audry S, Bossy C (2006) Mercury in the Lot-Garonne river system (France): sources, fluxes and anthropogenic component. Appl Geochem 21(3):515–527
Schwesig D, Krebs O (2003) The role of ground vegetation in the uptake of mercury and methylmercury in a forest ecosystem. Plant Soil 253(2):445–455
Sierra MJ, Millán R, Esteban E (2009) Mercury uptake and distribution in Lavandula stoechas plants grown in soil from Almadén mining district (Spain). Food Chem Toxicol 47(11):2761–2767
Skinner K, Wright N, Porter-Goff E (2007) Mercury uptake and accumulation by four species of aquatic plants. Environ Pollut 145(1):234–237
Streets DG, Hao JM, Wu Y, Jiang JK, Chan M, Tian HZ, Feng XB (2005) Anthropogenic mercury emissions in China. Atmos Environ 39(40):7789–7806
Sun XM, Wen W, Sun SJ, Huang SL (2008) Contamination status of nutrient salt of nitrogen and phosphor, and COD in Haihe River, Tianjin. Modern Agril Sci Technol (in Chinese) 11:362–366
Tong YD, Zhang W, Hu XD, Ou LB, Hu D, Yang TJ, Wei W, Wang XJ (2012) Model description of trophodynamic behavior of methylmercury in a marine aquatic system. Environ Pollut 166:89–97
Ullrich SM, Ilyushchenko MA, Uskov GA, Tanton TW (2007) Mercury distribution and transport in a contaminated river system in Kazakhstan and associated impacts on aquatic biota. Appl Geochem 22(12):2706–2734
Wan Q, Feng XB, Lu J, Zheng W, Song XJ, Han SJ, Xu H (2009) Atmospheric mercury in Changbai Mountain area, northeastern China I. The seasonal distribution pattern of total gaseous mercury and its potential sources. Environ Res 109(3):201–206
Wang ZW, Zhang XS, Chen ZS, Zhang Y (2006) Mercury concentrations in size-fractionated airborne particles at urban and suburban sites in Beijing, China. Atmos Environ 40(12):2194–2201
Wu Y, Wang SX, Streets DG, Hao J, Chan M, Jiang JK (2006) Trends in anthropogenic mercury emissions in China from 1995 to 2003. Environ Sci Technol 40(17):5312–5318
Zhang L, Wong MH (2007) Environmental mercury contamination in China: sources and impacts. Environ Int 33(1):108–121
Zhang H, Feng XB, Larssen T, Shang LH, Li P (2010a) Bioaccumulation of methylmercury versus inorganic mercury in rice (Oryza sativa L.) grain. Environ Sci Technol 44(12):4499–4504
Zhang ZS, Wang QC, Zheng DM, Zheng N, Lu XG (2010b) Mercury distribution and bioaccumulation up the soil-plant-grasshopper-spider food chain in Huludao city, China. J Environ Sci 22(8):1179–1183
Zhen MZ, Qian XC, Cheng G, Lu DF (1999) The environmental function and quality standard of surface water in Tianjin. J Urban Environ Urban Ecol (in Chinese) 12:10–12
Acknowledgments
This study was funded by the National Natural Science Foundation of China (41001350, 40971247, 41130535) and the Ministry of Education of China (306019), and was supported by the China Postdoctoral Science Foundation (201003003).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tong, Y., Zhang, W., Hu, D. et al. Behavior of mercury in an urban river and its accumulation in aquatic plants. Environ Earth Sci 68, 1089–1097 (2013). https://doi.org/10.1007/s12665-012-1810-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12665-012-1810-0