Abstract
Following the appearance of symptoms of arsenic toxicity in the inhabitants of villages in the Muteh gold mining region, central Iran, the concentration of this element in various parts of biogeochemical cycle is investigated. For this purpose, rock, groundwater, soil, plant, livestock hair and wool, and human hair samples are collected and analysed. Total arsenic content ranges from 23 to 2,500 mg/kg in rock samples, 7–1,061 μg/l in water, 12–232 mg/kg in soil, 0.5–16 mg/kg in plant samples, 4.10–5.69 mg/kg in livestock hair and wool, and 0.64–5.82 mg/kg in human hair. Arsenic concentration in various parts of biogeochemical cycle near the gold deposit in a metamorphic complex, and also close to the gold-processing plant, is very high and decreases exponentially with increasing distance from them. Arsenic concentration in water from a well close to the Muteh gold mine is above 1 mg/L. Arsenic in hair samples taken from local inhabitants is above the recommended levels, and the control samples in Shahre-Kord city. Arsenic concentration is higher in male population and correlates positively with age. It is suggested that arsenic resulting from the decomposition of ore mineral such as orpiment (As2S3), realgar (As2S2) and arsenopyrite (FeAsS) is responsible for polluting natural resources and the human intake via drinking water and the food chain. Gold mining and processing has undoubtedly enhanced the release of arsenic and intensified the observed adverse effects in Muteh area.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Although at first glance geology may appear far from human health, but as Komatina (2004) has pointed out, human health is in fact directly linked to geology. Human Exposure to high levels of toxic metals usually causes acute toxicity symptoms, while long-term exposure to lower levels can trigger allergies and even cancers (Berkowitz et al. 2008).
Arsenic is a ubiquitous toxicant and a carcinogenic element associated with a wide range of adverse human health effects (WHO 1999; Tseng et al. 2002; Nvas-Acien et al. 2006). Arsenic forms both organic and inorganic compounds in the nature, and inorganic compounds are generally more toxic and mobile than organoarsenic species (WHO 2001; Hughes 2002; Wang and Mulligan 2006a, b). As is the twentieth abundant element in the Earth’s crust (Bhumbla and Keefler 1994; Duker et al. 2005), and occurs in two distinct mineral associations, i.e., sulphides and oxides. Arsenic is also reported in some silicate minerals, such as biotite. Oxide minerals do not take arsenic into their structure, but have a great capacity to adsorb arsenic on their surface. Arsenic can form sulphide mineral such as orpiment (As2S3), realgar (As2S2) and arsenopyrite (FeAsS), and also substitutes for sulphur in more common minerals such as pyrite, chalcopyrite (copper sulphide), galena (lead sulphide) and sphalerite (zinc sulphide). These minerals commonly form in areas of hydrothermal activity, and are often associated with ores (Ravenscroft et al. 2009).
Mining and extraction of As-bearing ore deposits will greatly enrich arsenic in all compartments of the biological cycle, i.e., soil, water, plant, and living organisms including human in the immediate vicinity of the ore deposits (Merian et al. 2004; Robles-Arenas et al. 2006). Schwedt (2001) believes that arsenic is one of the most dangerous elements derived from mining activity. Human exposure to arsenic from mining activity has been recorded in many part of the world including Thailand, Ghana, Greece, Austria, Australia, China, Brazil, Mexico, Canada and part of United States of America (Hinwood et al. 2004; Lu and Zhang 2005; Patel et al. 2005; Figueiredo et al. 2007; Armienta and Segovia 2008; Liu et al. 2011). Gold mining has always been accompanied by hazardous arsenic toxicity both in miners and in the close ecosystems (Patel et al. 2005).
More people are affected by As poisoning in Asia than in the rest of the world combined (Ravenscroft et al. 2009). The magnitude of this problem is severe in Bangladesh, West Bengal in India, and China (e.g., Acharyya et al. 2000; Sun et al. 2001; Rahman and Ravenscroft 2003; Van Geen et al. 2003; Xia and Liu 2004; Ravenscroft et al. 2009; Singh et al. 2010). The so-called Qinghai–Tibet As-rich province of China extends westwards into Afghanistan, Iran and Turkey, and eastward into Thailand and Myanmar (Mianpiang 1997). Groundwater contamination by arsenic and the associated health effects in Kurdistan province of Iran was already investigated by Mosaferi et al. (2007) who reported a mean As concentration of 290 μg/l and a maximum of 1,480 μg/l in groundwater.
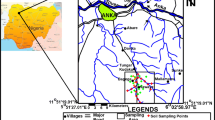
Muteh area is located north of the Isfahan province in central Iran, between longitudes 50°32′30″ E and 33°45′00″ N, and latitudes 55°49′00″ E and 33°28′30″ N (Fig. 1). The climate is arid to semi-arid, with an average annual temperature of −5°C in winter (December–February) and 32°C in summer (June–September). The mean annual rainfall varies between 180 and 300 mm. Muteh is the only active gold-producing mine in Iran. There are 10 gold ore bodies and occurrences in the Muteh area (Fig. 1). Currently, only two mines (Senjedeh and Cah Khaton) are being mined.
The main purpose of this paper is to (a) establishing arsenic content and distribution in compartments of the biogeochemical cycle including rock, soil, water, plant, livestock, and human hair in the Muteh mining district, (b) Identifying the sources of arsenic, (c) determining arsenic content in different soil fractions (d) evaluating localised health problems arising from exposure to arsenic in the Muteh area.
Geological setting
Arsenic rich provinces extend from Tibet area of China westward into Afghanistan, Iran and Turkey, and eastward into Thailand and Myanmar (Ravenscroft et al. 2009). The Muteh metamorphic complex hosts the spatially related gold deposits in central Iran (Fig. 2). This complex is located within the so-called Sanandaj-Sirjan zone of the Zagros orogenic belt, which consists of three parallel zones: (1) the Urumieh Dokhtar magmatic assemblage, (2) the Zagros imbricate zone and (3) the Zagros fold-thrust belt. The Zagros imbricate zone (or “Sanandaj-Sirjan metamorphic Zone”, as redefined by Alavi (1994) after Stocklin (1968, 1977), is a zone of thrust faults that have transported numerous bodies of metamorphosed and non-metamorphosed Phanerozoic stratigraphic units of the Afro-Arabian passive continental margin, as well as its “obducted” ophiolites (Alavi 2004).
The pre-Permian deformed subzone of the Sanandaj-Sirjan which hosts the Muteh gold deposit, is characterised by having a high-grade metamorphic core (mainly gneiss and amphibolite) covered by low-grade metamorphic rocks (mainly greenschist, metarhyolite and marble). Chlorite schist is the predominant host rock in Muteh gold district. Mesozoic and Tertiary sedimentary rocks overlaying Paleozoic sedimentary rocks are abundant in the study area, and are largely covered by quaternary alluvial sediments. The metamorphic complex is intruded by middle to late cretaceous and paleocene-eocene granitic plutons (Moritz et al. 2006). Gold mineralization is related to a protracted geologic evolution of the Sanandaj-Sirjan tectonic zone and magmatic-metamorphic hydrothermal activity (Moritz and Ghazban 1996). Mineralization is similar to mesothermal quartz vein type deposits in orogenic belts (Robb 2005) and is controlled by brittle, ductile to ductile–brittle shear zones. Hydrothermal alteration associated with gold occurrences is characterised by intense, pervasive bleaching of the host rocks (Moritz et al. 2006). Alteration assemblages consist of silicified rock with microcrystalline to crystalline quartz, fine-grained muscovite, pyrite, dolomite-ankerite, and albite overprinting the metamorphic minerals in the host rock (Moritz et al. 2006). Pyritization is the dominant alteration and is rather extensive and related to gold mineralization (Abdollahi et al. 2009). Gold paragenetic minerals include principal sulphide phases as pyrite, arsenopyrite and chalcopyrite. There are no visible gold grains and gold commonly occurs as very fine-grained solid inclusions (1–5 μm) in pyrite and arsenopyrite.
Materials and methods
Sampling, sample preparation and analysis
In this study, sampling was limited to the Muteh mining region (Muteh, Yekke chah, Golcheshmeh and Laibid villages). In all, a total of 198 samples were collected, including 16 rock samples, 17 water samples, 34 soil samples [(17 topsoil (0–5 cm) and 17 subsoil (15–20 cm) samples], 32 plant samples (21 samples from Sieberi sp. and 11 samples from Harmala sp.), 16 samples from sheep’s wool and goat’s hair and 83 samples from human hair (43 males and 40 females).
Water samples were collected from springs, drainages, water wells, aqueducts and mine tunnels in acid washed (0.5 M HCl) 1,000 mL polyethylene bottles. Field parameters pH, EC, Eh and temperature of water were determined on-site using portable devices. Each sample was split into two fractions. The first fraction was filtered through a 0.45 μm filter paper and then acidified by concentrated HNO3 to pH of 2 for arsenic and principal cations analysis by (ICP-MS) (ACME Laboratory, Canada). The second fraction was left unacidified and unfiltered for anion analysis.
Soil sampling was carried out along three parallel linear sections extending from Muteh gold mining complex, towards Muteh village. At least 1 kg of soil was collected from a 1 × 1 m2 at 0–5 and 15–50 cm depths in polyethylene bags. In the laboratory the samples were air-dried for a week, and then were sieved through a <2.0 mm mesh size polyethylene sieve, to remove stones, coarse materials and other extraneous debris. The sieved samples were ground to a fine powder using an agate Tema grinder. To examine the fractionation of As and other heavy metals, the procedure of Tessier et al. (1979) consisting of five steps, i.e., exchangeable fraction (F1), bound to carbonate (F2), bound to Fe/Mn oxides (F3), bound to organic matter (F4) and the residual fraction (F5), was carried out on six soil samples. The sequential extraction was performed progressively on an initial weight of 1.0 g of dry soil samples. The arsenic content of the extracted solutions was determined using ICP-MS (ACME Laboratory, Canada). The accuracy of results was checked by calculating recovery percents using the following formula:
The results are presented in Table 1 and according to Burt et al. (2003) indicate a good agreement between sum of extracted As and total As determined by total analysis.
500 mg plant samples were collected from roots and leaves of two abundant indigenous species, i.e., Artemisia aieberi and Peganum harmala. The collected plant samples were washed by tap water, rinsed with deionized water, and air-dried at room temperature. 0.5 g of the dry sample was mixed with concentrated nitric acid (HNO3) and hydrogen peroxide (H2O2) and sealed in a digestion vessel. For soil and rocks samples, nominally 0.2 g of dry sample was mixed with concentrated hydrochloric acid (HCl) and nitric acid (HNO3) and sealed within a digestion vessel. The vessel is then placed into a microwave digestion machine and heated to around 160–170°C using microwave energy. After digestion, distilled water is added to dilute the acid solution and the sample poured off into a test tube. The elements required are then measured using both ICP-MS and ICP-OES instrument techniques (AMDEL Laboratory, Australia).
The mineralogy of 10 rock samples representing host rocks, ore minerals and waste dumps were examined using X-ray diffractometry (XRD). For this purpose, the samples were ground using a Tema Grinder and a Siemens D 500 diffractometer.
About 30 g of wool and hair samples were collected from sheeps and goats in Muteh village. Also, 5–7 g of human hair samples were collected from male and female inhabitants in Muteh, Yekke chah, Golcheshmeh and Laibid villages. 10 samples were also collected from Shahre-Kord town as control samples. The hair samples cover a wide range of age. Wool and hair samples were washed following the method proposed by IAEA (1978) and involved sequential washing with acetone, water and acetone. The samples were dried in an oven at 35°C. 0.25 g of each sample was weighed and digested in extra pure nitric acid (HNO3) in a digestion bomb at about 150°C. The resulting solution was made to volume by deionized water, and as content was measured using Graphite Furnace Atomic Absorption Spectrometry (GF-AAS).
Results and discussion
Because of the different behaviour of arsenic in rock, water, soil, plant and animal environments, each environment will be discussed separately.
As in Muteh rocks
It is well known that gold is strongly associated with arsenic in most of the world’s gold deposits (Dickson et al. 1975). The host rocks for gold and arsenic in Muteh area are predominantly schists and gneisses related to collisional tectonics. Gold mineralization is mostly associated with late hydrothermal activity and commonly forms fissure-filling sulphide-silica veins of various sizes (Moritz et al. 2006). Minor Cu–As sulphide mineralization also occurs as veins and veinlets in the metamorphic host, especially in Senjedeh mine. The results of X-ray diffractometry are presented in Table 2 and indicate that As-bearing minerals such as realgar (As2S2), orpiment (As2S3), pyrite (FeS2) and chalcopyrite (CuFeS2) along with silicates such as tremolite, actinilite, biotite, muscovite, feldspars and chlorite are the most abundant minerals in both host rocks and the mineralized veins. Furthermore, polished thin sections were prepared and studied in reflected and transmitted light. The results are the same as those obtained from XRD, the only exception is the presence of arsenopyrite in the polished thin sections. Geochemical analysis of 16 host rock samples indicates that arsenic content varies between 23 and 2,500 mg/kg with a mean of 743 mg/kg. Analyses carried out by Afzal et al. (2007) indicate that As content in drilling cores obtained from Muteh host rocks increases in unweathered rock at depth. Arsenic released to the aqueous phase by weathering of As-bearing minerals can remain in solution or be trapped by adsorption on secondary phases and colloids or by coprecipitation reactions (Materaa et al. 2003).
According to Afzal et al. (2007), there is a positive correlation between As and Au content in the gold-bearing host. Apparently, zones with more than 1 mg/kg Au, contain at least 500 mg/kg of arsenic. Seemingly, Gold mining and processing seem to play an important role in releasing As from gold-bearing rocks and veins into the surrounding environment.
As in water
Weathering and erosion of As-bearing minerals releases As into surface water and ground water resources (Wang and Mulligan 2006a, b; Armienta and Segovia 2008; Naidu and Bhattacharya 2009). Water samples were collected from tube wells (n = 4), Senjedeh mine (n = 3), Chah-Khatun mine (n = 2) and drinking water (n = 8) in villages where human hair specimens were also collected (Table 3).
A useful approach in evaluating the degree of pollution and potential hazard posed by water is measuring the permissible quality factor (PQF), calculated by dividing the maximum concentration of a polluting element by the concentration given by the drinking water guidelines by (USEPA 2003) for drinking water (10 μg/l).
The results of water analysis are presented in Table 3. It can be seen that the calculated PQF for As in water samples from Chah-Khatun (W-4) and Senjedeh mine (W-7) (38 and 24, respectively) is higher than the recommended values. Furthermore, As concentration in the water of a well close to the Muteh gold mine (W-6) is 106 times greater than the maximum permissible level given by USEPA (2003) for drinking water. Arsenic content in the water used for drinking in Muteh (W-17), Golcheshmeh (W-13) and Yekkeh Chah (W-14) villages is also above the recommended value, especially in one of the two wells that supply drinking water to Muteh village and is located near the metamorphic complex (W-12).
The quality of water considerably improves with increasing distance from the gold mine and the metamorphic complex (Keshavarzi et al. 2009). For example, arsenic content of a qanat water 10 km south of the metamorphic complex (W-8) is 7 μg/l. The decreasing trend of As concentration is probably related to the decreasing amounts of As-bearing minerals. High sulphate content in water samples north of Muteh area suggests that oxidation of sulphide minerals is responsible for observed high arsenic content. There is very little doubt that anthropogenic contribution mostly comes from gold mining and the mineral dressing plant. In addition, infiltration through the tailing dam undoubtedly plays an important role in polluting the groundwater. The highest As content in groundwater (1,061 μg/l) is encountered in a well south of Muteh gold complex (W-6). It is known that the effective surface of crushed As-bearing sulphide minerals in mine tailings increases considerably, causing a higher rate of oxidation. This may ultimately lead to the higher bioavailability of arsenic and other potentially toxic elements. It is noteworthy that very little acid mine drainage is produced in Muteh gold district. The reason is most probably the arid climate of Muteh area and the presence of a large volume of acid neutralising marble rocks. High Ca and alkali earth elements content in groundwater also indicate high buffering capacity in Muteh region.
As in soil and plants
Food crops generally adsorb arsenic from soil. Two kinds of not clearly differentiated soil safety standard are commonly used. One relates to the health hazard for people (especially children) eating soil directly, and the other, relates arsenic concentrations that interfere with healthy plant growth and satisfactory crop yield (Ravenscroft et al. 2009). The concentrations of some major, minor and trace elements in the Muteh region soil are presented in Table 4. Samples were collected from two horizons, i.e., 0–5 cm (topsoil) and 15–20 cm (subsoil) to assess the role played by clay minerals and Fe–Mn hydroxides in adsorbing arsenic and other trace metals. It can be seen that As and trace metals contents in the two sampled horizons are very similar, reflecting a common source for these elements.
Copper is slightly enriched in some topsoil samples, especially in the vicinity of Senjedeh mine, probably due to higher copper mineralization and surface disturbance caused by gold prospecting activity.
The measured background values for As, Cu, Fe, Mn, Mo, and Pb in Muteh soil are 26.5, 28.2, 38800, 702, 1.9, and 18.5 mg/kg, respectively. Natural, background value for As in Muteh area is higher than both average arsenic concentration in Earth’s crust (1.8 mg/kg) (Mason and Moore 1982) and the mean global concentration given for soils (4.7 mg/kg) (Kabata-Pendias and Mukherjee 2007). Cu, and Fe, background values are also slightly higher than the average concentrations given for the soils (14 mg/kg for Cu, and 21,000 mg/kg for Fe) (Kabata-Pendias and Mukherjee 2007).
Table 4 presents the enrichment factors (Brady 1984), and geoaccumulation index (Eby 2004) for elements in topsoil and subsoil samples. Arsenic displays the highest enrichment factor among the measured elements, followed by S, Mo and Pb. Contents of As in topsoil and subsoil samples are very similar. Arsenic content north and northwest of Muteh mining district (Senjedeh and Chahkhatoon mines) is high. However, the highest As content occurs in the playa sediments east of the Muteh village, probably resulting from groundwater seepage and subsequent evaporation at the ground surface.
The high concentration of arsenic and other trace elements in the vicinity of the Muteh mining zone rapidly decreases towards Muteh village, reflecting decreasing anthropogenic activities (gold mining and processing).
The possible mobilisation of As in the soils, and subsequent leaching into ground or surface water or entry into the human food chain, should always be considered as a serious hazard (Bhattacharya et al. 2007). Hence, it is necessary to show As in different fractions of soils. For this purpose a sequential extraction analysis using Tessier et al. (1979) method was performed. The results are presented in Table 1. The sum of fractions is higher than the total value north of Senjedeh and South of Chahkhatoon mines resulting in inferior recovery rates. The worse recovery rate relates to subsample heterogeneity, which is a typical and unavoidable characteristic when extracting highly contaminated samples (Herreweghe et al. 2003). Significant amounts of As were found in the residual fraction (55.04–72.41% of the total As). These results probably reflect the dominant role of lithology in arsenic speciation. Ion exchangeable and carbonate fractions, which are generally considered as “easily releasable” make up less than 6.29%. Fe–Mn oxide ranges from 14.09 to 34.11%. The organic bound arsenic is less than 13.32%. The low content of extractable As, suggest that the role played by soil colloids such as clay minerals, Fe–Mn hydroxides, and organic matter in adsorbing arsenic is limited and As mostly occurs in the residual fraction. However, considering the high concentration of total arsenic, even small percent of mobile arsenic is considerable.
Although, the main source of arsenic in Muteh area is geogenic, there is very little doubt that soil arsenic content is greatly enhanced by anthropogenic activity, i.e., exploitation of Muteh gold deposit.
A. sieberi and P. harmala species are the most abundant indigenous plant species widely spread in the Muteh arid region. For this reason, these two types of plants were sampled and analysed. The results (Table 5) indicate As, Al, Cu, Fe, and Pb content in A. sieberi is higher than in P. harmala, while concentration of Na, K, Mg, Ca, P and S is higher in the latter. The results suggest that Artemisia sp. tends to bioaccumulate toxic trace elements including arsenic, while Peganum sp. display high tendency to concentrate some major elements. Furthermore, the concentrations of some selected elements (Table 5) in the leaves of Artemisia sp. is higher than in roots.
In Artemisia sp. As is highly correlated with As in both topsoil and subsoil samples, reflecting the bioaccumulating nature of this plant for As. The lack of such correlation in Peganum sp. and the low correlation between trace elements content and soil suggests the low tendency of this species for uptake of trace elements. Since A. sieberi is used as forage in the study area, hence, arsenic maybe transferred via food chain to humans.
As in human and livestock
An important part in medical geology research is the evaluation of the influence of natural and anthropogenic pollutants on biota and humans. Arsenic may extensively bound to intracellular components and use this pathway to accumulate in tissues of the lung, kidney, hair and nails (Vahter and Marafante 1983; Georis et al. 1990). In humans, As pollution is best indicated in toenails (e.g. Karagas et al. 2000; Mandal et al. 2003; Schmitt et al. 2005) and hair (Kurttio et al. 1998; Agusa et al. 2006). For this reason, human hair (male and female) and livestock hair and wool (sheep and goat) were sampled and analysed (Table 6). Ten hair samples from the inhabitant of a safely distant town (Shahre-Kord) were also taken as control samples (Table 6). The results indicate a higher mean arsenic concentration in male hair (2.66 mg/kg) compared with female hair (1.60 mg/kg). Hinwood et al. (2003) also suggest that arsenic is generally more concentrated in male hair. The reason is probably: (1) As commonly concentrates in the lower parts of hair, and men usually have shorter hair than women; (2): Men are more exposed to As-bearing suspended air particulates, and therefore absorb more arsenic than women.
According to Chowdhury et al. (2000) normal arsenic levels in human hair is 0.08–0.25 mg/kg (toxicity level >1 mg/kg). As shown in Table 6, arsenic concentration in all collected hair samples (male and female) in the Muteh area is considerably higher than the concentrations given by Chowdhury et al. (2000).
Armienta et al. (1997) have shown that arsenic levels in human organs vary with age. Figure 3 also indicates that there is a positive correlation between As level in human hair and age. Apparently, arsenic has bioaccumulated in time in Muteh area inhabitants. This conclusion was also reached by other authors (e.g. Yang et al. 2002). Jones et al. (2005) have already noted that the most likely source for arsenic poisoning is polluted drinking water.
This is well supported by the high arsenic levels in the groundwater used for drinking in the Muteh area (Table 3), as well as high As content in the inhabitants hair, especially in Muteh village (Fig. 4). Symptoms of As chronic poisoning (mild keratosis) already can be seen on the palms and sols of some inhabitants. Collected samples from wool and hair of livestock (sheep and goat) also show high arsenic content in the Muteh mining district. Mean As concentration in goats’ hair (5.16 mg/kg) is higher than in sheep’s wool (4.24 mg/kg) probably indicating that goat is more susceptible to arsenic toxicity than sheep.
Conclusions
The close association between As-bearing minerals and gold mineralization is a well-known fact. Economic and environmental geologists. Economic geologists use this association to track down hidden gold deposit, while environmental geologist generally look at this association from arsenic pollution point of view in gold-mining areas, such as Muteh gold district. Thus, the presence of arsenic minerals such as arsenopyrite and orpiment identified in Muteh deposit is thought to be the geogenic source of the observed chronic symptoms seen in the inhabitants of Muteh area.
The result of analyses carried out on different environments ranging from rock to human hair indicate that arsenic have moved through all compartments of the biogeochemical cycle in Muteh region using either direct or indirect pathways. However, the well above standard arsenic content in the drinking water of Muteh, Golcheshmeh and Yekke-Chah villages, indicate that drinking water is the major cause of arsenic poisoning seen in the local inhabitants. The lack of symptoms in control samples (Shahre-Kord inhabitants) confirms this conclusion. Among indigenous plant species, Artemisia sp seems to bioaccumulate arsenic among other heavy metals in its leaves. Since the plant is consumed grazes by sheep and goats in the area, it provides another pathway for arsenic to enter the food chain and be ingested by local inhabitants, as proved by high As content in sheep wool and goats hair.
The observed correlation between arsenic concentration and age in both male and female inhabitant in Muteh region suggests that arsenic has bioaccumulated in time, showing the chronic nature of the observed As-toxicity symptoms. The higher As content in male hair is probably the result of higher outdoor exposure. Continuous monitoring of arsenic content in drinking water, and sustainable exploitation of Muteh gold deposit are essential to reduce arsenic toxicity in the Muteh gold district.
References
Abdollahi MJ, Karimpour MH, Kheradmand A (2009) Petrography and sulphur isotope studies of pyrites in the Muteh gold deposit. Am J App Sci 6:1086–1092
Acharyya SK, Lahiri S, Raymahashay BC, Bhowmik A (2000) Arsenic toxicity of groundwater in parts of the Bengal basin in India and Bangladesh: the role of quaternary stratigraphy and holocene sea-level fluctuation. Environ Geol 39:1127–1137
Afzal P, Aamdari S, Zakariai SJ, Lotfi M (2007) Arsenic distribution in Senjedeh ore deposit, Muteh mining complex. In: Proceeding of 7th Congress on Safety, Health and Environment in Mines and Related Industries, 30 October–1 November. Sarcheshmeh Copper Complex, Iran, pp 1–10 (in Farsi)
Agusa T, Kunito T, Fujihara J, Kubota R, Minh TB, Trang PTK et al (2006) Contamination by arsenic and other trace elements in tube-well water and its risk assessment to humans in Hanoi. Vietnam. Environ Pollut 139:95–106
Alavi M (1994) Tectonics of the Zagros orogenic belt of Iran: new data and interpretations. Tectonophys 229:211–238
Alavi M (2004) Regional stratigraphy of the zagros fold-thrust belt of Iran and its proforeland evolution. Am J Sci 304:1–20
Armienta MA, Segovia N (2008) Arsenic and fluoride in the groundwater of Mexico. Environ Geochem Health 30(4):345–353
Armienta MA, Rodriguez R, Cruz O (1997) Arsenic content in hair of people exposed to natural arsenic polluted groundwater at Zimp, Mexico. J Contam Toxicol 59:583–589
Berkowitz B, Dror I, Yaron B (2008) Contaminant geochemistry: interactions and transport in the subsurface environment. Springer, Heidelberg, p 412
Bhattacharya P, Welch AH, Stollewerk KG, McLaughlin MJ, Bundschuh J, Panaullah G (2007) Arsenic in the environment: biology and chemistry. Sci Total Environ 379:109–120
Bhumbla DK, Keefler RF (1994) Arsenic mobilization and bioavailability in soils. In: Niragu JO (ed) Arsenic in the environment, Part I, cycling and characterization. Wiley, New York, pp 51–82
Brady NC (1984) The Nature and properties of soils. MacMillan, New York
Burt R, Wilson MA, Keck TJ, Dougherty BD, Storm DE, Lindhal JA (2003) Trace element speciation in selected smelter-contaminated soils in Anaconda and Deer Lodge Valley, Montana, USA. Adv environ res 8:51–67
Chowdhury UK, Biswas BK, Chowdhury TR, Samanta G, Mandal BK, Basu GK, Chanda CR, Lodh D, Saha KC, Mukherjee SK, Roy S, Kabir S, Quamruzzaman Q, Chakraborti D (2000) Groundwater arsenic contamination in Bangladesh and West Bengal, India. Environ. Health Perspect 108(5):393–397
Dickson FW, Radtke AS, Weissberg BG, Heropoulos C (1975) Solid solutions of antimony, arsenic and gold in stibnite (Sb2S3), orpiment (As2S3) and realgar (As2S2). Econ Geol 70:591–594
Duker AA, Carranza EJ, Hale M (2005) Arsenic geochemistry and health. Environ Int 31:631–641
Eby GN (2004) Principles of environmental chemistry. In: Edmunds WM. Thomson, UK
Figueiredo BR, Borba RP, Angelica RS (2007) Arsenic occurrence in Brazile and human exposure. Environ Geochem Health 29:109–118
Georis B, Cardenas A, Buchet JP, Lauwerys RR (1990) Inorganic arsenic methylation by rat tissue slices. Toxicology 63:73–84
Herreweghe SV, Swennen R, Vandecasteele C, Cappuyns V (2003) Solid phase speciation of arsenic by sequential extraction in standard reference materials and industrially contaminated soil samples. Environ Pollut 122:323–342
Hinwood AL, Sim MR, Jolley D, Klerk ND, Bastone EB, Gerostamoulos J, Drummer OH (2003) Hair and toenail arsenic concentrations of residents living in areas with high environmental arsenic concentrations. J Environ Med 111:187–193
Hinwood AL, Sim MR, Jolley D, Klerk ND, Bastone EB, Gerostamoulos J, Drummer OH (2004) Exposure to inorganic arsenic in soil increases urinary inorganic arsenic concentration of residents living in old mining areas. Environ Geochem Health 26:27–36
Hughes MF (2002) Arsenic toxicity and potential mechanisms of action. Toxicol Lett 133(1):1–16
International Atomic Energy Agency (IAEA) (1978) IAEA/RL/50, Veina
Jones DC, Duvauchelle C, Ikegami A, Olsen CM, Lau SS (2005) Serotonergic neurotoxic metabolites of ecostasy identified in rat brain. J Pharmacol Exp 313:422–431
Kabata-Pendias A, Mukherjee AB (2007) Trace elements from soils to human. Springer-Verlag, Berlin
Karagas MR, Tosteson TD, Blum J, Klaue B, Weiss JE, Stannard V et al (2000) Measurement of low levels of arsenic exposure: a comparison of water and toenail concentrations. Am J Epidemiol 152:84–90
Keshavarzi B, Moore F, Esmaeieli A, Rastmanesh F (2009) The source of fluoride toxicity in Muteh area, Isfahan, Iran. Environ Earth Sci 61:777–786
Komatina MM (2004) Medical geology: effects of geological environments on human health. Elsevier Science and Technology, London
Kurttio P, Komulainen H, Hakala E, Kahelin H, Pekkanen J (1998) Urinary excretion of arsenic species after exposure to arsenic present in drinking water. Arch Environ Contam Toxicol 34:297–305
Liu B, Wu F, Li X, Fu Z, Deng Q, Mo C, Zhu J, Zhu Y, Liao H (2011) Arsenic, antimony and bismuth in human hair from potentially exposed individuals in the vicinity of antimony mines in Southwest China. Microchem J 97:20–24
Lu X, Zhang X (2005) Environmental geochemistry study of arsenic in Western Hunan mining area, P.R. China. Environ Geochem Health 27(4):313–320
Mandal BK, Ogra Y, Suzuki KT (2003) Speciation of arsenic in human nail and hair from arsenic-affected area by HPLC-inductively coupled argon plasma mass spectrometry. Toxicol Appl Pharmacol 189:73–83
Mason B, Moore CB (1982) Principles of Geochemistry, 4th edn. Wiley, New York
Materaa V, He’chob IL, Laboudiguea A, Thomasa P, Tellierb S, Astrucb M (2003) A methodological approach for the identification of arsenic bearing phases in polluted soils. Environ Pollut 126:51–64
Merian E, Anke M, Ihnat M, Stoeppler M (2004) Elements and their compounds in the environment. Willey, New York, p 350
Mianpiang Z (1997) An Introduction to Saline-alkaline Lakes on the Qinghai-Tibet Plateau. Kluwer, Dordrecht
Moritz R, Ghazban F (1996) Geological and fluid inclusion studies in the Muteh gold district, Sanandaj-Sirjan zone, Esfahan Province, Iran. Schweiz Miner Petrogr Mitt 76:85–89
Moritz R, Ghazban F, Singer BS (2006) Eocene gold ore formation at Muteh, Sanandaj-Sirjan tectonic zone, Western Iran: a result of late-stage extension and exhumation of metamorphic basement rocks within the Zagros Orogen. Econ Geol 101:1497–1524
Mosaferi M, Yunesian M, Dastgiri S, Mesdaghinia A, Esmailnasab N (2007) Prevalence of skin lesions and exposure to arsenic in drinking water in Iran. Sci Total Environ 390(1):69–76
Naidu R, Bhattacharya P (2009) Arsenic in the environment—risks and management strategies. Environ Geochem Health 1:1–8
Nvas-Acien A, Silbergergeld EK, Streeter RA, Clark JM, Burke TA, Guallar E (2006) Arsenic exposure and type 2 diabetes: a systematic review of the experimental and epidemiological evidence. Am J Epidemiol 162:1037–1049
Patel KS, Shrivas K, Brandt R, Jakubowski N, Corns W, Hoffmann P (2005) Arsenic contamination in water, soil, sediment and rice of central India. Environ Geochem Health 27(2):131–145
Rahman AA, Ravenscroft P (eds) (2003) Groundwater resources and development in Bangladesh – background to the arsenic crisis, agricultural potential and the environment. Bangladesh Centre for Advanced Studies. University Press Ltd., Dhaka
Ravenscroft P, Brammer H, Richards KS (2009) Arsenic pollution: a global synthesis. Wiley, Blackwell
Robb L (2005) Introduction to Ore-Forming Processes. Blackwell Publishing, Malden
Robles-Arenas VM, Rodrı’guez R, Garcı’a C, Manteca JI, Candela L (2006) Sulphide-mining impacts in the physical environment: Sierra de Cartagena-La Union (SE Spain) case study. Environ Geol 51:47–64
Ronov AB, Yaroshevski AA (1969) Earths Crust Geochemistry, Encyclopedia of Geochemistry and Environmental Sciences, Fairbridge RW (ed) Van Nostrand, New York, p 850
Schmitt MT, Schreinemachers D, Ning Z, Zhao B, Le XC et al (2005) Human nails as a biomarker of arsenic exposure from well water in inner Mongolia: comparing atomic fluorescence spectrometry and neutron activation analysis. Biomarkers 10:95–104
Schwedt G (2001) Essential guide to environmental chemistry. Willey, New York
Singh M, Singh AK, Swati, Srivastava N, Singh S, Chowdhary AK (2010) Arsenic mobility in fluvial environment of the Ganga Plain, northern India. Environ Earth Sci 59:1703–1715
Stocklin J (1968) Structural history and tectonics of Iran. a review. Am Assoc Petrol Geolog Bull 52:1229–1258
Stocklin J (1977) Structural correlation of the Alpine ranges between Iran and central Asia. Mem Hors-serie Soc Geol Fr 8:333–353
Sun G, Pi J, Li B, Guo X, Yamauchi H, Yoshida T (2001) Progress on researches of endemic arsenism in China: population at risk, intervention actions and related scientific issues. In: Chappell WR, Abernathy CO, Calderon RL (eds) Arsenic exposure and health effects IV. Elsevier, Oxford, pp 79–85
Tessier A, Campbell GC, Bisson M (1979) Sequential extraction procedure for the speciation of particulate trace metals. Anal Chem 51:844–851
Tseng CH, Tseng CP, Chiou HY, Hsueh YM, Chong CK, Chen CJ (2002) Epidemiologic evidence of diabetogenic effect of arsenic. Toxicol Lett 133:69–76
US EPA (2003) Arsenic treatment technology evaluation handbook for small systems. EPA 816-R-03-014, p 126
Vahter M, Marafante E (1983) Intracellular interaction and metabolic fate of arsenite and arsenate in mice and rabbits. Chem Biol Interact 47:29–44
Van Geen A, Ahmed KM, Seddique AA, Shamsudduha M (2003) Community wells to mitigate the current arsenic crisis in Bangladesh. Bull World Health Organ 82:632–638
Wang S, Mulligan CN (2006a) Occurrence of arsenic contamination in Canada: sources, behavior and distribution. Sci Total Environ 366(2–3):701–721
Wang S, Mulligan CN (2006b) Effect of natural organic matter on arsenic release from soils and sediments into groundwater. Environ Geochem Health 28:197–214
WHO (1999) Arsenic in Drinking Water, Geneva, p 210
WHO (2001) Environmental health criteria 224. arsenic and arsenic compounds, 2nd edn. World Health Organization, Geneva
Xia Y, Liu J (2004) An overview on chronic arsenism via drinking water in PR China. Toxicology 198(1–3):25–29
Yang L, Wng W, Hou W, Williams WP, Peterson PJ (2002) Arsenism clinical stages and their relation with hair arsenic concentration of residents of Bayimnaodo rural district, inner Mongolia, China. J Environ Geochem Health 24:337–348
Acknowledgments
The authors wish to express their gratitude to Mr. Lahijanzadeh of Esfahan environmental protection office for financially supporting this research. We would also like to extend our thanks to the members of the Research Committee of Shiraz University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Keshavarzi, B., Moore, F., Rastmanesh, F. et al. Arsenic in the Muteh gold mining district, Isfahan, Iran. Environ Earth Sci 67, 959–970 (2012). https://doi.org/10.1007/s12665-012-1532-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12665-012-1532-3