Abstract
Introduction
Oral Squamous Cell Carcinomas have been considered as the most prevalent malignancies in the head and neck region and are frequently undiagnosed until symptomatic with an advanced stage of disease. So there is an urgent need to device methods for the detection of oral premalignant lesions and oral cancer at an early stage in order to improve the survival rate for patients. A number of tests have been done for the detection of oral cancer which include oral brush biopsy, the Vizilite, oral autofluorescence including chemiluscence, photodynamic detection, toluidine blue staining, methylene blue staining, incisional biopsy and many more.
Material
The article reviews various diagnostic modalities available at present for detection of squamous cell carcinomas and oral epithelial dysplasias based on advanced PUBMED search of the English language literature from the year 1972 to present in order to help us select the most suitable among them fulfilling the desired criteria of being non-invasive, highly specific and sensitive, economically viable, having a scope to be used for mass screening, easy to process, having low inter examiner variability and possibly not requiring high expertise to conduct and interpret the results.
Conclusion
After reviewing various diagnostic modalities, we conclude that toluidine blue staining emerges as a clear winner among all these and it can act as a valuable adjunct to incisional biopsy in detection of oral cancer and may not substitute it except in certain circumstances when its results are carefully correlated with the patient history and clinical characteristics of the mucosal disorder, considering the fact that incisional biopsy has been reported to cause dissemination of cancer cells in the circulation there by increasing the possibility of metastasis. We must emphasize that toludine blue is a screening modality and not a diagnostic procedure like biopsy and hence cannot replace a confirmatory biopsy as a whole
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Squamous cell carcinoma (SCC) is the most prevalent malignancy in the head and neck, the oral cavity, and pharynx [1–5]. Approximately 300,000 new oral cavity cancer cases and 68,000 deaths worldwide are expected annually [6].
It is defined as “a malignant epithelial neoplasm exhibiting squamous differenciation as characterized by the formation of keratin and/or the presence of intercellular bridges”.
Clinically, oral squamous cell carcinomas (OSCCs) may appear as red, white, or mixed patches; a mass with or without ulceration, which may develop in an area of clinically normal mucosa or arise from an oral premalignant lesion [1, 2]. The most common sites of OSCC are the lower lip, the lateral border of the tongue, and the floor of the mouth [2], which contain relatively thin epithelium, minimal keratinization and, thus, may be more susceptible to environmental carcinogens.
Even though the oral cavity is readily accessible for examination by inspection and palpation, oral squamous cell carcinoma (OSCC) is frequently not diagnosed until symptomatic with an advanced stage of disease [7–11].
Approximately two thirds of OSCCs are diagnosed at stage III or IV of disease with spread to adjacent tissues and regional lymph nodes, leading to an overall poor 5-year survival rate [12–15]. Thus, there is a pressing need for early detection of oral premalignant lesions (OPLs) and OSCC.
Mouth cancer is a major health problem in many parts of the world. While its incidence is relatively low in most western countries, there are some important exceptions to this trend. Significant geographic variation is noted in the incidence of mouth cancer, with high rates reported for the Indian subcontinent and parts of Asia (male incidence rates in excess of 10 per 100,000 per annum). It is also noted that as with other forms of oral cancer, the majority of population-based data for mouth cancer comes from the western world with a paucity of reliable data from the so-called developing countries [16].
In India, oral cancer constituted 9.8 % of an estimated 644,600 incident cancer cases in 1992, ranks first among all cancer cases in males and is the third most common among females with the incidence rate from 7 to 17/100,000 persons/year; incidence rate being higher than the western rate of 3–4/100,000/year [17].
The incidence of oral cancer has risen gradually over the past 40 years. Thus, early detection of asymptomatic oral premalignant lesions (OPMLs), including oral submucous fibrosis (OSF), homogenous leukoplakia, non-homogenous leukoplakia, erythroplakia, etc. is imperative before their transformation into malignancies [18].
A number of tests have been done for the detection of oral cancer which include brush biopsy, the Vizilite, oral autofluorescence including chemiluscence, photodynamic detection, toluidine blue staining, methylene blue staining, incisional biopsy, etc.
In this review, we will discuss about the reliability of toluidine blue in detecting premalignant and malignant lesions and conditions of the oral cavity.
Various Methods for Early Cancer Detection
Carcinoma in an early stage of development is hard to detect clinically because the lesion may not be palpable and colour of the lesional tissue is not necessarily different from the colour of surrounding mucosa. In order to improve the efficacy of the diagnosis, techniques are being developed to complement clinical examination and to facilitate the identification of initial carcinomas.
Brush Biopsy
Dysplastic or immature epithelial cells arise, of course, from the bottom of the squamous epithelium and should not be expected to be found by scraping a thick surface layer of keratin [19].
This technique involves screwing a bristle-covered wire (the “brush”) through the thick surface keratin to the basal layer of the epithelium [20]. This relatively painless procedure captures the deeper epithelial cells on the bristles and the entire brush is sent to a pathology lab, where the cells are removed and plated on a microscopic slide. From that point on, the process is the same as a routine pap smear.
Computer-associated optical scanner compares the size of each individual cell with the size of its nucleus. Large, dark nuclei are found in dysplastic or immature cells, as are abnormal nuclear shapes (pleomorphism).
Recently, liquid-based cytology (LBC) has become a principle methodology in cytopathology replacing conventional smears, owing to better cell recovery and morphologic preservation. It is a good tool in an experienced, knowledgeable hand, with very few false positive or negative results when used appropriately. However, it is not a good screening procedure and no studies have correlated normal mucosa with brush biopsy results. Perhaps more significantly, the brush biopsy is not a true diagnostic tool and cannot, therefore, provide a definitive diagnosis—an incisional biopsy is always needed for that [21].
Hohlweg-Majert et al. did a study to evaluate the advantage of computer-assisted analysis of the oral brush biopsy compared with synchronous scalpel biopsy in the early detection of oral lesions. In this prospective, randomized, controlled study, brush and scalpel biopsies were performed on 75 patients. Six patients had to be excluded due to inadequate results, and 43 were shown to have dysplastic epithelium, 15 carcinoma, and 11 suspicious lesions. Therefore, the sensitivity for the detection of abnormal cells by means of OralCDx was 52 %, specificity 29 %, and the positive predictive value 63 %. According to their results, the use of oral brush biopsy as a standardized, minimally invasive method of screening oral lesions should be reconsidered [22].
Reddy et al. did a study to assess the diagnostic accuracy of brush biopsy when compared to histopathology in a group of patients with features of potential malignancy.
In this study, 60 cases of clinically diagnosed leukoplakia were selected and subjected to histopathology and brush biopsy. Results showed that, of 16 dysplasia cases confirmed by histopathology, only 12 were positively reported in oral brush biopsy. In 44 cases, the reports are same for histopathology and brush biopsy. The sensitivity of oral brush biopsy is 43.5 % and specificity is 81.25 % with a positive predictive value of 58.3 %. The results showed that oral brush biopsy with molecular markers like tenascin and keratins can be an accurate diagnostic test [23].
Advantages
-
(1)
The biopsy brush penetrates to the basement membrane, removing tissue from all three epithelial layers of the oral mucosa [24].
-
(2)
The oral brush biopsy does not require topical or local anesthetic and causes minimal bleeding and pain [24].
-
(3)
It can be used as an adjunct to the clinical examination and to identify a disease at an early and curable stage. It is simple to perform, office-based, painless test; and can be integrated into the daily routine of practice [24].
-
(4)
It is useful in those situations when a patient refuses to have a biopsy performed or when medically compromised patients would be exposed to unnecessary surgical risks [25].
Disadvantages
-
(1)
It does not maintain the architecture of the cells relationship to each other [24].
-
(2)
When a positive result is returned by the brush biopsy pathology, it cannot be used as the conclusive determination of malignancy, and a conventional, gold standard, incisional or punch biopsy must be performed [24].
The ViziLite: Highlighting the Keratin
It is based on the ability of acetic acid to enhance regions of thickened surface keratin. In the oral environment it makes the keratin more white and, therefore, more visible to the naked eye. A thin leukoplakia which might otherwise have been missed could be detected after a minute of contact with acetic acid. The ViziLite(R) system takes advantage of this and adds bright blue light to even further enhance keratin detection [26–28]. Dysplasia, of course, begins in the lowest layers of the epithelium and so reflected light will identify such cells only if they are associated with surface hyperkeratosis, e.g. leukoplakia. With this caveat, however, it does well, with a very high ability to enhance identification of keratotic patches [26, 29, 30].
The light is derived from either chemical tubes (chemiluminescence) or a laser and recently, toluidine blue has been added to the kit (ViziLite Plus(R)) for identification of superficial nuclear abnormalities.
The manufacturer claims that “light from ViziLite(R) is absorbed by normal tissue and reflected by dysplastic tissue, which will appear white.” [21]
Ram et al. did a study in which forty-six patients with a previous history of oral cancer or premalignancy were examined by ViziLite. Out of a total of 46 acetowhite lesions, 31 received scalpel biopsy. The sensitivity and specificity was reported as 100 and 14 % respectively [31].
Advantages
-
(1)
This system is valuable in finding hyperkeratotic patches that may have been missed with routine visual inspection.
-
(2)
Vizilite is a painless, effective, fast and a life saving procedure.
-
(3)
Vizilite hopes to make early detection of oral cancer in patients who do not show symptoms [32].
Disadvantages
-
(1)
As with other adjunctive diagnostic technologies, the ViziLite(R) exam has disadvantages. It seems to have a high proportion of false positive and false negative tests, relative to identification of dysplastic cells rather than hyperkeratosis [28, 30].
-
(2)
It is best performed in a completely dark room, which is often difficult in today’s dental office [31].
-
(3)
A number of studies have been done which show that Vizilite does not aid in the identification of oral lesions [28, 31, 33].
Oral Autofluorescence: When the Mucosa Does Not Glow
Autofluorescence originates from a variety of fluorophores in the oral cavity, and is sensitive to alterations in both tissue morphology and biochemistry associated with neoplasia [34, 35].
Two optical devices, the VELScope(R) (LED Dental, Inc. White Rock, BC, Canada) introduced 3 years ago, and the new Identafi(R) 3000 Ultra (Trimira, LLC, Houston, Texas), take advantage of the fact that, to a certain degree, we all glow. Each of our cells contain molecules capable of self-fluorescence, especially when activated (excited) by specific light waves. In humans, these fluorescing products are numerous: tryptophan, porphyrins, collagen cross-links, elastin, NADH (nicotinamide adenine dinucleotide), and flavins (FAD, flavin adenine dinucleotide) [36].
This fluorescent signaling has been used to assess the metabolic state of tissues and to identify primitive/dysplastic cells.
The amount of fluorescence given off from living tissues is very slight; certainly not capable of being seen under normal conditions. However, if violet or blue light is used in a darkened room and the clinician peers through an eyepiece or pair of glasses which filter out virtually all reflected light and only allows transmission of light of the wavelength(s) of the fluorescing tissues, the autofluorescence is easily seen. The wavelengths which excite the greatest fluorescence in oral mucosa range from 400 to 460 nm, i.e. violet and blue light.
An immature or dysplastic epithelial cell has much less NADH and FAD activity than a normal cell and so mucosal areas with such cells will not fluoresce, thereby appearing black (actually blackish-green or blackish-blue) through the eyepiece or glasses [37]. Additionally, data also suggests that the cross-links in subepithelial collagen fibers beneath dysplastic cells also lose fluorescent activity, contributing to the “black spot” seen through the filter.
A number of recent studies have suggested that the VELscope can be used as an adjunct to visual examination to improve the detection of oral neoplasia [38–43].
Two types of imaging in autofluorescense:
-
1.
Wide-field autofluorescence imaging—The VELscope® is a commercially available device to visualize loss of tissue autofluorescence associated with precancer and cancer in the oral cavity.
-
2.
Benign lesions, such as inflammation, are also associated with loss of stromal autofluorescence, which may limit diagnostic specificity especially in low-risk populations.
-
3.
High-resolution imaging—High-resolution imaging may provide a tool to discriminate benign changes, such as inflammation, from neoplasia with better specificity than wide-field imaging [44].
Roblyer et al. used the multispectral digital microscope to select optimal wavelengths to distinguish neoplastic from non-neoplastic oral mucosa [37]. Results showed that the normalized red-to-green fluorescence intensity ratio at 405 nm excitation provided the best discrimination between neoplastic and non-neoplastic areas. A quantitative algorithm, based on the red-to-green fluorescence intensity ratio from regions of interest, could discriminate normal tissue from dysplasia and cancer in a high-prevalence population, with a sensitivity of 95.9 % and specificity of 96.2 % in a training set of 46 subjects, and with a sensitivity of 100 % and specificity of 91.4 % in a validation set of 21 subjects.
Advantages
-
(1)
The beauty of the self-fluorescence test is that the light used to excite the oral cells penetrates to the deepest part of the epithelium and so easily reaches dysplastic cells in the lower regions of the epithelium, as well as the subepithelial collagen fibers [21].
-
(2)
VELscope can assist clinicians in detecting oral lesions that are occult under white light examination and in more effectively identifying which regions to biopsy [45, 46].
Disadvantages
-
(1)
Direct visual inspection of tissue autofluorescence has shown encouraging results in high-prevalence populations, but the technique requires subjective interpretation and depends on the visual recognition skills of the examiner.
-
(2)
Benign lesions such as inflammation may give rise to false-positive results especially on wide field imaging.
Photodynamic Detection
Photodynamic detection is a non-invasive technique for detection of fluorescence in tissues that arises from a photosensitiser that has been taken, and the exploitation of differences in the fluorescence spectrum between types of tissues for the detection of cancer. One such photosensitiser is 5-aminolaevulinic acid (ALA), which metabolises within tissue to form the photosensitiser protoporphyrin IX (PpIX), and owes its popularity to the selectivity with which PpIX is formed within cancerous tissue. A compact spectroscopic instrument is used to exite and take in vivo measurements of fluorescence in the oral cavity.
Therefore, the patients having oral cancer are expected to have higher PpIX fluorescence and more widely dispersed spectra in spectroscopic device than those without oral cancer which has been attributed to differences in structure between healthy and cancerous tissues [47].
Advantages
-
(1)
User-friendly device that allows the clinician to distinguish quickly between cancerous and normal tissue by examination of the fluorescence spectrum that arises from a single point in tissue, and so reduce the number of actual biopsy specimens necessary.
-
(2)
It may also prompt more relevant early biopsy specimens that could help improve the prognosis of oral cancer [47].
Disadvantages
-
(1)
It is not possible to claim that the device is able to distinguish between dysplasia and inflammation, and between dysplasia and early cancer [47].
Toluidine Blue
Toluidine Blue (TB) has been known for various medical applications since its discovery by William Henry Perkin in 1856, after which it was primarily used by the dye industry. Also known as methylanaline or aminotoluene, it basically has 3 isoforms, namely, ortho-toluidine, para-toluidine, and meta-toluidine. Toluidine blue has been extensively used as a vital stain for mucosal lesions and also has found applications in tissue sections to specifically stain certain components owing to its metachromatic property [48].
Toluidine Blue has been used as a vital stain to highlight potentially malignant oral lesions and may identify early lesions, which could be missed out on clinical examination. Moreover, it can outline the full extent of dysplastic epithelium or carcinoma prior to excisions [49].
Toluidine Blue (also known as tolonium chloride) is an acidophilic metachromatic dye that selectively stains acidic tissue components (sulfates, carboxylates, and phosphate radicals) [50]. In a solution, toluidine blue takes on blue-violet colour [51].
It is a member of the thiazine group and is partially soluble in both water and alcohol [51].
In vivo, Toluidine Blue stains deoxyribonucleic and nucleic acids and may be retained in intracellular spaces of dysplastic epithelium [53–56].
The test is based on the fact that dysplastic cells may contain quantitatively more nucleic acids and a dysplastic epithelium also has some loss of cohesion. These features facilitate the penetration of toluidine blue through the epithelium and retention of the dye in cancer cells, which are replicating in vivo, whereas normal mucosa fail to retain the dye [57].
Dysplastic and malignant tissues may retain Toluidine Blue due to the loss of tumor suppressor genes that predict progression of Oral Premalignant Lesions to oral squamous cell carcinoma (OSCC) or may represent OSCC at diagnosis [11, 58–60].
Neibel, Chomet, Shedd and co-workers were the first to report vital application of Toluidine Blue for the detection of premalignant and malignant lesions of the oral cavity. They confirmed the property of Toluidine Blue to verify clinically suspicious lesions as neoplastic, to delineate margins of premalignant and malignant growth, and to detect unnoticed or satellite tumors [61].
Toluidine Blue is generally prepared in 1 % concentration for oral application. A 100 mL of 1 % Toluidine Blue consists of 1 gm Toluidine Blue powder, 10 mL of 1 % acetic acid, 4.19 mL absolute alcohol, and 86 mL distilled water to make up 100 mL. The pH is usually regulated to 4.5 [62].
The technique of application usually involves rinsing of the mouth twice with water for 20 s to remove debris. And 1 % acetic acid is then applied for 20 s to remove ropey saliva. This is followed by 1 % Toluidine Blue application for 20 s either with cotton swab when a mucosal lesion is seen or given as rinse when no obvious lesion is detected. Again, 2 rinses with 1 % acetic acid were performed to reduce the extent of mechanically retained stain. Finally the mouth is rinsed with water [68]. The interpretation is based on the color; a dark blue (royal or navy) stain is considered positive, light blue staining is doubtful and when no color is observed, it is interpreted as negative stain.
Under normal conditions, nucleated scales covering the papillae on the dorsum of the tongue as well as the pores of seromucinous glands in hard palate are frequently stained with Toluidine Blue [63].
In 1989, meta-analysis of available data assessing the effectiveness of Toluidine Blue application in identification of oral squamous cell carcinoma determined sensitivity ranging from 93.5 to 97.8 % and specificity ranging from 73.3 to 92.9 % [7].
The application of TB in 81 lesions of which 48 lesions were considered clinically suspicious and 33 were clinically benign showed that 28 cases had no stain, 20 had equivocal stain and 33 had positive stain. On biopsy of these lesions 54 were nonmalignant and 27 were carcinomas. The study found 100 % sensitivity and 52 % specificity [8].
Advantages
-
1.
It is a simple, rapid, inexpensive and less technique sensitive method which can be used for mass screening of oral cancer especially in a very large population [64].
-
2.
In patients who are unwilling to undergo an incisional biopsy, toluidine blue along with clinical examination may serve the purpose of oral cancer screening.
-
3.
Toluidine Blue may provide information regarding lesion margins, accelerate the decision to biopsy, guide biopsy site selection and treatment of oral premalignant and malignant lesions.
-
4.
Studies have demonstrated that toluidine blue has a high sensitivity in its detection of malignant oral lesions; values vary from 84 to 100 % [65].
Disadvantages
-
1.
The disadvantages of toluidine blue include the risk of obtaining a false negative reaction in a case where the patient is not followed up adequately [66].
-
2.
Binding of Toluidine Blue to the nucleic acids may also occur in mucosal ulcerations, granulation tissue [54, 66, 67] and in inflammatory lesions that can contribute to false positive outcomes [9, 31].
-
3.
Toluidine blue is hazardous if swallowed, and was shown to have toxicity to fibroblasts [69].
Methylene Blue
Methylene Blue has also been used as a diagnostic test for the detection of oral cancer. Methylene Blue, has a similar chemical structure and exhibits similar physicochemical properties to toluidine blue.
The exact mechanism for the uptake of methylene blue dye in epithelial cells is still not very clear, but it resembles toluidine blue dye in its acidophilic characteristic and may penetrate into cells with an abnormal increase in nucleic acid, thus resulting in different uptake between normal and highly dysplastic/malignant cells [69].
Its application has been more linked with detecting some gastrointestinal abnormalities such as Barrett’s esophagus [70–72], gastric cancer [73], prostate cancers [74, 75], and also bladder cancer.
Chen et al. did a study which involved the examination of fifty-eight patients suspected of having oral malignant or precancerous lesions by methylene blue staining. The results of methylene blue uptake were compared with a simultaneous biopsy of these lesions. The pathologically confirmed precancers and cancers were the positive targets of this screening, while benign epithelial lesions were sorted as negative subjects of screening. The results revealed sensitivity of 90 %, specificity of 69 %, positive predictive value of 74 %, and negative predictive value of 87 % [69].
Advantages
-
1.
It is less toxic to the human body as compared to toluidine blue and is inexpensive [69].
-
2.
Methylene blue is a commonly used stain that helps us see microscopic life in brilliant color [76].
Disadvantages
-
1.
The application of this material in detecting oral lesions has so far not been addressed in many studies [69].
-
2.
The exact mechanism for the uptake of methylene blue dye in epithelial cells is still not very clear [69].
Incisional Biopsy: The Gold Standard
There are oral lesions whose diagnosis can be made relying on data gathered during the history, physical examination, and/or other non-invasive methods but there are others where histopathological studies are needed to confirm the presumed clinical diagnosis.
Incisional biopsy has been considered as a gold standard for the detection of oral premalignant and malignant lesions which consists of removal of a representative sample of the lesion and normal adjacent tissue in order to make a definitive diagnosis before treatment.
The aim of the biopsy is to:
-
define a lesion on the basis of its histopathological aspect;
-
to establish a prognosis in malignant or premalignant lesions;
-
facilitate the prescription of specific treatment;
-
contribute to the assessment of the efficacy of the treatment;
Advantages
-
1.
When a suspicious lesion is identified, a conventional biopsy using a scalpel or small biopsy forceps remains the best and most accurate means of assessing it [77].
Disadvantages
-
1.
The most common risk associated with the procedure is bleeding. A hematoma, or a pocket of blood, can form and collect at the site of the biopsy. This can be uncomfortable but should resolve over the following week.
-
2.
The armamentarium required in such a procedure is much more than used for simple non-invasive staining procedures like toluidine blue etc.
-
3.
Incisional biopsy is not feasible for mass screening in a large population like in India.
-
4.
The standard biopsy techniques may require modification in some patients, including those with conditions that preclude the safe use of local anesthetic and those with severe bleeding diatheses or coagulopathies.
-
5.
A number of studies have documented the dissemination of cancer cells into the circulation resulting in an increased risk of metastasis after the incisional biopsy [78, 79].
Revisiting Incisional Biopsy
Incisional biopsy of oral carcinomas has long been recognized as a very useful method for establishing a firm diagnosis and for directing management of specific lesions. However, a number of clinicians are concerned that this procedure may spread cancer cells beyond the borders of tumor into the surrounding normal tissue; thus, promoting local spread as well as increasing the potential for metastasis. It has been indicated that surgical trauma inflicted on malignant tumors may increase their metastatic potency [80, 81]. The major physical barriers to the migration of tumor cells during this process are connective tissue and basement membrane. The surgical intervention may destroy these barriers and facilitate invasion of vascular system at the site of the injury.
A number of animal studies have been done which revealed that an incision into the oral cancer significantly increased the risk of regional lymph node metastasis [81–83]. Recently, it has been reported that there is an increased frequency of neck metastasis from stage I and II SCCs after incisional biopsy [84, 85]. It is still unclear, however, whether the biopsy procedure would affect the final outcome of this malignancy [86, 87].
Kusukawa et al. did a study to examine whether cancer cell dissemination results from incisional biopsy. In this squamous cell carcinoma cells were tried to be detected before and after incisional biopsy by means of cytokeratin 19(CK19), reverse-transcriptase polymerase chain reaction (RT-PCR). The study population consisted of 20 patients with oral SCC in which 10 were given incisional biopsy followed by radical excision and 10 were treated by excisional biopsy alone. 2(20 %) out of 10 patients from the incisional biopsy group were positive for CK19 transcripts in their peripheral blood drained 15 min after incision. In contrast, CK19 transcript was not detected in excisional biopsy group. Therefore this study demonstrated that surgical invasiveness for oral cancer, including incisional biopsy, causes dissemination of cancer cells into circulation resulting in increased risk of metastasis [78].
Ramani et al. utililizing reverse transcriptase polymerase chain reaction (RT-PCR) to detect disseminated cancer cells after incisional biopsy among OSCC patients failed to support the argument of cancer spread by incisional biopsy although only 10 patients were studied [88].
Frydrych et al. did a study aimed to investigate the five-year survival and recurrence of oral SCC after incisional and excisional biopsy in total population data available from the Western Australian Cancer Registry (WACR). No association was found between biopsy type and five-year survival or recurrence amongst individuals with Stage I or II disease adding to the evidence that incisional biopsy of oral SCC can be a safe procedure [89].
Dyavanagoudar et al. carried out a study to examine whether cancer cell dissemination results from incisional biopsy by trying to detect oral squamous cell carcinoma (OSCC) cells in the peripheral blood sample before and after incision biopsy by CK19 RT-PCR. The study group consisted of 25 OSCC patients and the control group consisted of five patients with oral submucous fibrosis and five with leukoplakia. Five ml of blood collected before and twice (15 and 30 min) after incision were used for CK19 RT-PCR. Four (16 %) of 25 cases of OSCC were positive for CK19 transcripts in their peripheral blood drained 15 min after incision. They concluded that surgical invasion, in the form of incisional biopsy, causes dissemination of cancer cells into circulation, resulting in increased risk of metastasis [79].
Craig has also reported a similar percentage (48 %) of lymph node metastasis following a longer period of observation. It is quite interesting in the present study that the incisional biopsy in the same model of experimental oral carcinogenesis showed no neck lymph node metastasis at 3 weeks following the biopsy of the carcinomas, but four out of the ten animals exhibited lymph node metastasis at 5 weeks postbiopsy [90].
It has been reported that, for small and localized tumors, excisional biopsy is thought to be effective in preventing the spread of cancer cells by incisional biopsy. And also the incidence of neck metastasis in stage I and stage II SCCs treated with excisional biopsy is significantly lower than that in tumors excised following incisional biopsy [84, 85].
Therefore, after reviewing all the diagnostic modalities available at present for detection of squamous cell carcinomas and oral epithelial dysplasias, we are standing at the crossroads where we are still looking for one diagnostic modality which is non-invasive, highly specific and sensitive, economically viable, has a scope to be used on mass screening of population, is easy to process and takes less time for processing, has low inter examiner variability and possibly would not require high expertise to conduct and interpret the results and above all does not cause any dissemination of squamous cell carcinoma cells to other parts of the body.
After reviewing all the diagnostic modalities we have narrowed down to toluidine blue, which meets the above mentioned criteria. Various studies have been done to assess the reliability of toluidine blue as a diagnostic modality in the detection of oral cancer.
Reliability of Toludine Blue Staining in Detection of Oral Squamous Cell Carcinomas
In India, with such a large population where oral cancer is the most common cancer in males, detection of oral cancer and potential malignant lesions/precancer may be cumbersome and may not be accessible to the entire population. So there is a need for a test which is easily accessible even in remote parts of our country, cheap so that even poor can afford it, less technique sensitive so that even a common man with a little training can perform the test, with a high sensitivity and specificity, and minimal morbidity or adverse effects associated with it.
Several studies have been performed over the years to determine the reliability of in vivo TB staining in terms of their sensitivities, specificities, positive predictive values, negative predictive values etc.
Warnakulasuriya et al. in 1996 evaluated the efficacy of 1 % toluidine blue in the identification of oral malignancies and potentially malignant oral lesions among a group of Asian patients (n = 102) with undiagnosed oral mucosal lesions and conditions (n = 145) and they utilized a ready to use kit of 1 % toluidine blue (Orascan). Eighteen oral carcinomas all retained the dye and there were no false negatives, yielding a test sensitivity of l00 %. Eight of 39 oral epithelial dysplasias were toluidine blue-negative, giving a false negative rate of 20.5 % and a sensitivity of 79.5 % for oral epithelial dysplasias. The specificity of the technique was low (62 %). Five dysplastic lesions were detected solely by the kit and this suggests that the method is valuable for surveillance of high-risk subjects in addition to its remarkable sensitivity in the detection of invasive carcinoma [91].
Rodriguez et al. in 2011 did a study to evaluate the efficacy of the toluidine blue (TB) test as a diagnostic tool in the detection of malignant and dysplastic lesions of the oral cavity which included 160 patients with oral mucosal disorders. The sensitivity and specificity for the detection of malignant or dysplastic lesions by this test were 65.5 and 73.3 %, respectively. Overall, the detection rate with TB (sensitivity) was slightly lower compared with those reported by other authors but the specificity was comparable to several reports. Positive predictive value (35.2 %) was also lower than previous studies, whereas negative predictive value (90.6 %) was similar. They concluded that the simplicity of the test procedure and the validity of derived values suggest TB staining can be a valuable adjunct to the diagnostic process, as long as it is carefully correlated with the clinical characteristics of the mucosal disorder and histopathological diagnosis [57].
Gupta et al. carried out a study to evaluate the usefulness of toluidine blue and brush biopsy in precancerous oral lesions and squamous cell carcinoma. Ninety-six patients with suspicious oral lesions who attended the outpatient clinics of otorhinolaryngology were screened with in vivo toluidine blue staining and oral brush biopsy. Oral brush biopsy showed high specificity and sensitivity. Toluidine blue staining was highly sensitive and moderately specific for malignant lesions but less sensitive for premalignant lesions. They concluded that early detection of oral carcinoma is possible even at the precancerous stages by using noninvasive, painless and outpatient procedures, such as in vivo toluidine blue staining and brush biopsy [92].
Zhang et al. did a study where they monitored OPLs from 100 patients without any history of oral cancer for an average of 44 months in order to evaluate the association of toluidine blue status with clinicopathologic risk factors, molecular patterns (microsatellite analysis on seven chromosome arms: 3p, 9p, 4q, 8p, 11q, 13q, and 17p) and outcome. Toluidine blue-positive staining correlated with clinicopathologic risk factors and high-risk molecular risk patterns. Significantly, a greater than sixfold elevation in cancer risk was observed for toluidine blue-positive lesions, with positive retention of the dye present in 12 of the 15 lesions that later progressed to cancer (p = 0.0008). This association of toluidine blue status with risk factors and outcome was evident even when the analysis was restricted to OPLs with low-grade or no dysplasia. Our results suggest the potential use of toluidine blue in identifying high-risk OPLs [60].
A pilot-study is presented to evaluate the use of Toluidine blue as a screening agent of premalignant and malignant oral lesions. Test results in 20 patients showed that Toluidine blue is effective in staining malignant lesions and not so in premalignant ones. The staining technique appears to be a worthwhile and simple diagnostic aid to support clinical evidence of malignancy. A biopsy remains mandatory in reaching the final diagnosis [93].
Silverman et al. did a study in which 132 patients suspected of having malignant or precancerous oral lesions were studied by comparing toluidine blue dye uptake clinically with a simultaneous biopsy. The histopathologic diagnosis confirmed 57 squamous carcinomas, 42 epithelial dysplasias, and 33 benign mucosal changes. Overall accuracy of the toluidine blue uptake was 91 %. In the dysplastic and malignant lesions, the false negatives were 2 %, and there were 30 % false positives in the benign lesions. It was concluded that toluidine blue staining is a useful adjunct to careful examination, clinical judgement, and biopsy [94].
Awan et al. did a study to determine the effectiveness of toluidine blue in detecting leukoplakia and erythroplakia and its accuracy in identifying cases with oral epithelial dysplasia. Ninety-two patients attending two oral medicine clinics in London, presenting with white and red patches of the oral mucosa, were investigated by the application of toluidine blue. Eighty-two patients were clinically diagnosed as OPMDs and 10 were frictional keratosis. A surgical biopsy was performed to assess epithelial dysplasia. Of 64 oral leukoplakias, 34 (53.1 %) were positive for toluidine blue and among nine erythroplakias seven stained positive. Of 41 oral dysplasia cases, a little more than half of the lesions (n = 23) were stain positive, an estimated sensitivity of 56.1 %. Toluidine Blue test had a higher sensitivity for detecting higher-grade dysplastic lesions (5/8 moderate dysplasia, sensitivity 62.5 %; 5/7 severe dysplasia; sensitivity 71.4 %) compared with lower grades of dysplasia, but the differences were not significant (p = 0.60). It was concluded that the test has the potential to detect OPMDs and yielded a sensitivity of 56.1 % and specificity of 56.9 % to detect oral epithelial dysplasia [95].
Onofre et al. evaluated the TB staining in premalignancies, and superficial oral ulceration suggesting malignancy. The study showed 100 % sensitivity in the detection of in situ and invasive carcinoma and no false-negative results occurred. The lesions that were diagnosed as dysplasia did not retain stain, and thus gave false-negative results. Staining specificity was 65 % because the inflammatory lesions were eliminated for the first time and re-stained after 14 days. In lesions without epithelial dysplasia or atypical cells, false positivity was 35 % [65].
Hegde et al. found a sensitivity of 97.29 % and specificity of 62.5 %. False positivity of 7.69 % and false negativity of 16.67 % was noticed. The authors suggested that specificity was reduced because of retention of the dye in some benign lesions [63].
Vahidy reported a sensitivity of 86 % and a specificity of 76 % after excluding numerous doubtful lesions where staining properties could not be readily judged as either positive or negative [96].
Allegra et al. did a study which focused on 45 oral mucosal lesions in 32 patients (13 female, 19 male). In 9 cases, multiple biopsies were collected. Of the 45 lesions examined, 26 (57.0 %) were defined clinically benign, while 19 (42.3 %) were defined as suspected lesions (premalignant or malignant). According to the clinical examination, the sensitivity was 53 % (16/30) and for toluidine blue staining 96.2 % (26/27) (p = 0.0007). The specificity was 80 % (12/15) for clinical examination and 77.7 % (14/15) for toluidine blue staining (p = 0.79). In conclusion toluidine blue stain has been shown to be a reliable aid when clinical examination is unable to differentiate lesions at high risk of progression and then it improves early diagnosis for oral cavity and oropharyngeal cancer [97].
Upadhyay et al. did a study which attempted to evaluate the efficacy of toluidine blue vital dye for detection of PMOL. The study included 47 biopsies (TBP:35 and TBN:12), of which 23 cases were confirmed as dysplastic (TBP = 17 and TBN = 6), 7 as hyperkeratosis (TBP = 4 and TBN = 3), 8 as epithelial hyperplasia (TBP = 6 and TBN = 3) and 5 as other benign lesions (TBP = 4 and TBN = 1). The validity test revealed a sensitivity of 73.9 % and specificity of 30 %. The positive predictive value was 54.8 % and negative predictive value 50 %. The study intended to highlight the false negative result (26.1 %) which was mainly attributed to mild dysplasia and the false positive (32.6 %) which included hyperkeratosis, hyperplasia, lichen planus and traumatic ulcer. The study concluded that toluidine blue staining should not blindly direct the clinician’s opinion, and strongly discouraged the use of toluidine blue as a screening test so that interpretation of screening test is done with caution [98].
Case Example
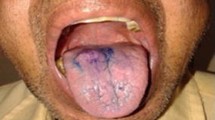
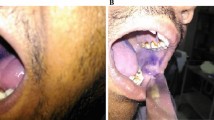
A 45 year old patient reported with a lesion present in the left lower vestibule for the past 2 months which was enlarging rapidly (Fig. 1a). He gave a history of tobacco chewing for the past 3–4 years. On neck examination none of the lymph node groups were found to be fixed but left submandibular lymph nodes were tender. There was a level of ambiguity as the patient’s history and clinical examination were not going in favour of squamous cell carcinoma, so we decided to go for toluidine blue staining of the lesion. Toluidine blue staining was done and it was retained (Fig. 1c) suggesting the possibility of dysplastic chances. So an incisional biopsy was performed from the areas that retained the maximum stain in order to have an accurate diagnosis of the lesion and histopathological examination confirmed it to be well differentiated squamous cell carcinoma (Fig. 1d).
a Initial presentation of the lesion in the left lower buccal vestibule. b Toluidine blue staining being performed. c Retention of the toluidine blue seen suggesting possible dysplastic changes. Histopathological examination confirmed squamous cell carcinoma. d H&E stained section of well differentiated squamous cell carcinoma of the patient
Conclusion
In a country like ours, where oral cancer is one of the most common cancers, toluidine blue staining can be very effective for the early screening of oral cancer. It is an inexpensive, highly sensitive, easy to perform method with low inter examiner variability and does not require high expertise to conduct and interpret the results. It can be used for mass screening of oral cancer as incisional biopsy is not feasible to be carried out in large groups of people. More resources, finances and expertise is required to carry out the biopsy procedure which is more cumbersome and requires more time for the outcome. Also when we take into account the psychological status of the patient after incisional biopsy, it is more traumatic for the patient to wait for days being confounded about the outcome of the biopsy report whereas in toluidine blue staining the result arrives immediately and the patient does not have to go through that waiting period for the report and act according to the report as soon as it arrives.
We must conclude by saying that the simplicity of the test procedure and the validity of derived values suggest Toluidine Blue staining can be a valuable adjunct to Incisional Biopsy in detection of oral cancer and may not substitute it except in certain circumstances when its results are carefully correlated with the patient history and clinical characteristics of the mucosal disorder. It is considered as a much safer procedure than incisional biopsy, as incisional biopsy has been reported to cause dissemination of cancer cells in the circulation which may result in metastasis. Along with all this, we must specify that toluidine blue is a screening modality and not a diagnostic procedure like biopsy and hence cannot replace a confirmatory biopsy as a whole.
It will not be correct to state that toluidine blue does not have any shortcomings but evaluating as a whole and comparing with other modalities it clearly emerges as a winner based on evidence. However, more detailed studies with large study samples are needed to investigate the reliability of toluidine blue staining and other screening methods in detection of oral cancer so that a more accurate assessment can be done.
References
Enwonwu CO, Phillips RS, Ibrahim CD, Danfillo IS (2004) Nutrition and oral health in Africa. Int Dent J 54:344–351
Epstein JB, Zhang L, Rosin M (2002) Advances in the diagnosis of oral premalignant and malignant lesions. J Can Dent Assoc 68(10):617–621
Gray MGL, Burls A, Elley K (2000) The clinical effectiveness of toluidine blue dye as an adjunct to oral cancer screening in general dental practice. A West Midlands Development and Evaluation Service Report. http://www.pcpoh.bham.ac.uk/publichealth/wmhtac/pdf/toludine_blue.pdf
Johnson N, Franceschi S, Ferlay J, et al. (2005) Squamous cell carcinoma: tumours of the oral cavity and oropharynx (Chapter 4). In: Barnes L, Eveson JW, Reichart P, Sidransky D (eds) World Health Organization classification of tumours, pathology and genetics of head and neck tumours. Lyon: IARC publication. http://www.iarc.fr/IARCPress/pdfs/bb9/bb9-chap4.pdf.4. American Cancer Society. Facts and figures 2007. http://www.cancer.org/downloads/STT/2008CAFFfinalsecured.pdf
Bettendorf O, Piffkò J, Bankfalvi A (2004) Prognostic and predictive factors in oral squamous cell cancer: important tools for planning individual therapy? Oral Oncol 40:110–119
American Cancer Society. Facts and figures 2007. http://www.cancer.org/downloads/STT/2008CAFFfinalsecured.pdf
Rosenberg D, Cretin S (1989) Use of meta-analysis to evaluate tolonium chloride in oral cancer screening. Oral Surg Oral Med Oral Pathol 67:621–627
Mashberg A, Samit A (1995) Early diagnosis of asymptomatic oral and oropharyngeal squamous cancers. CA Cancer J Clin 45:328–351
Epstein JB, Oakley C, Millner A et al (1997) The utility of toluidine blue application as a diagnostic aid in patients previously treated for upper oropharyngeal carcinoma. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 83:537–547
Scuibba J (1999) Improving detection of precancerous and cancerous oral lesions. J Am Dent Assoc 130:1445–1457
Epstein JB, Zhang L, Poh C et al (2003) Increased allelic loss in toluidine blue positive oral malignant lesions. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 95:45–50
National Cancer Institute (2003) SEER cancer statistics review 1975–2003
Tsantoulis PK, Kastrinakis NG, Tourvas AD et al (2007) Advances in the biology of oral cancer. Oral Oncol 43:523–534
Yeole BB, Ramanakumar AV, Sankaranarayanan R (2003) Survival from oral cancer in Mumbai (Bombay), India. Cancer Causes Control 14:945–952
Acha A, Ruesga MT, Rodríguez MJ, Martínez de Pancorbo MA, Aguirre JM (2005) Applications of the oral scraped (exfoliative) cytology in oral cancer and precancer. Med Oral Patol Oral Cir Bucal 10:95–102
Moore SR, Johnson NW, Pierce AM, Wilson DF (2000) The epidemiology of mouth cancer: a review of global incidence. Oral Dis 6(2):65–74
Shafer WG, Hine MK, Levy BM (2008) Shafer’s textbook of oral pathology, 5th edn. Elsevier India, New Delhi
Su WW-Y, Yen AM-F, Chiu SY-H, Chen TH-H (2010) A community-based RCT for oral cancer screening with toluidine blue. J Dent Res 89(9):933–937
Bouquot JE, Farthing PM, Speight PM (2006) The pathology of oral cancer and precancer revisited. Curr Diag Path 12:11–21
Sciubba JJ (2002) Improving detection of precancerous and cancerous oral lesions. Computer assisted analysis of the oral brush biopsy. US Collaborative OralCDx Study Group. J Am Dent Assoc 133:272
Bouquot JE, Suarez P, Vigneswaran N (2010) Oral precancer and early cancer detection in the dental office—review of new technologies. J Implant Adv Clin Dent 2:47–63
Hohlweg-Majert B, Deppe H, Metzger MC, Schumm S, Hoefler H, Kesting MR, Hölzle F, Wolff KD (2009) Sensitivity and specificity of oral brush biopsy. Cancer Invest 27(3):293–297
Reddy SG, Kanala S, Chigurupat A, Kumar SR, Poosarla CS, Reddy BVR (2012) The sensitivity and specificity of computerized brush biopsy and scalpel biopsy in diagnosing oral premalignant lesions: a comparative study. J Oral Maxillofac Pathol 16(3):349–353
Oral Brush Biopsy in the Early Detection of Oral Precancers and Cancers. Cytology in diagnosis oral lesions from PubMed NIH.Gov 2006
Babshet M, Nandimath K, Pervatikar SK, Naikmasur VG (2011) Efficacy of oral brush cytology in the evaluation of the oral premalignant, malignant lesions. J Cytol 28(4):165–172
Kerr AR, Sirois DA, Epstein JB (2006) Clinical evaluation of chemiluminescent lighting: an adjunct for oral mucosal examinations. J Clin Dent 17:59–63
Pierre M, Lane PM, Whitehead TGP, Zeng H et al (2006) Simple device for the direct visualization of oral-cavity. J Biomed Optics 11:024–026
Oh ES, Laskin DM (2007) Efficacy of the ViziLite system in the identification of oral lesions. J Oral Maxillofac Surg 65:424–426
Trullenque-Eriksson A, Muñoz-Corcuera M, Campo-Trapero J, Cano-Sánchez J, Bascones-Martínez A (2009) Analysis of new diagnostic methods in suspicious lesions of the oral mucosa. Med Oral Patol Oral Cir Bucal 14:210–216
Patton LL, Epstein JB, Kerr AR (2008) Adjunctive techniques for oral cancer examination and lesion diagnosis: a systematic review of the literature. J Am Dent Assoc 139:896–905
Ram S, Siar CH (2005) Chemiluminescence as a diagnostic aid in the detection of oral cancer and potentially malignant epithelial lesions. Int J Oral Maxillofac Surg 34(5):521–527
Sambandham T, Masthan KMK, Kumar MS, Jha A (2013) The application of Vizilite in oral cancer. J Clin Diagn Res 7(1):185–186
Farah CS, McCullough MJ (2007) A pilot case control study on the efficacy of acetic acid wash and chemiluminescent illumination (ViziLite™) in the visualisation of oral mucosal white lesions. Oral Oncol 43:820–824
De Veld DC, Witjes MJ, Sterenborg HJ, Roodenburg JL (2005) The status of in vivo autofluorescence spectroscopy and imaging for oral oncology. Oral Oncol 41:117–131
Roblyer D, Richards-Kortum R, Sokolov K et al (2008) Multispectral optical imaging device for in vivo detection of oral neoplasia. J Biomed Opt 13(2):024019
Skala MC, Riching KM, Gendron-Fitzpatrick A, Eickhoff J, Eliceiri KW, White JG, Ramanujam N (2007) In vivo multiphoton microscopy of NADH and FAD redox states, fluorescence lifetimes, and cellular morphology in precancerous epithelia. PNAS 104:19494–19499
Roblyer D, Kurachi C, Stepanek V, Williams MD, El-Naggar AK, Lee JJ, Gillenwater AM, Richards-Kortum R (2009) Objective detection and delineation of oral neoplasia using autofluorescence imaging. Cancer Prev Res (Phila Pa) 2:423–431
Poh CF, Williams PM, Zhang L, Rosin MP (2006) Heads up!—A call for dentists to screen for oral cancer. J Can Dent Assoc 72(5):413–416
Westra WH, Sidransky D (2006) Fluorescence visualization in oral neoplasia: shedding light on an old problem. Clin Cancer Res 12(22):6594–6597
Laronde DM, Poh CF, Williams PM et al (2007) A magic wand for the community dental office? Observations from the British Columbia Oral Cancer Prevention Program. J Can Dent Assoc 73(7):607–609
Rosin MP, Poh CF, Guillard M, Williams PM, Zhang L, MacAulay C (2007) Visualization and other emerging technologies as change makers for oral cancer prevention. Ann NY Acad Sci 1098:167–183
Williams PM, Poh CF, Hovan AJ, Ng S, Rosin MP (2008) Evaluation of a suspicious oral mucosal lesion. J Can Dent Assoc 74(3):275–280
Rosin MP, Poh CF, Elwood JM et al (2008) New hope for an oral cancer solution: together we can make a difference. J Can Dent Assoc 74(3):261–266
Shin D, Vigneswaran N, Gillenwate A, Richards-Kortum R (2010) Advances in fluorescence imaging techniques to detect oral cancer and its precursors. Future Oncol 6(7):1143–1154
Poh CF, Ng SP, Williams PM et al (2007) Direct fluorescence visualization of clinically occult high-risk oral premalignant disease using a simple hand-held device. Head Neck 29(1):71–76
Kois JC, Truelove E (2006) Detecting oral cancer: a new technique and case reports. Dent Today 25(10):94, 96–97
Dwyer MO, Day A, Padgett M, Ogden GR, Mclaren S, Goodman CR (2008) Detection of mucosal abnormalities in patients with oral cancer using photodynamic technique: a pilot study. Br J Oral Maxillofac Surg 46(1):6–10
Sridharan G, Shankar AA (2012) Toludine blue: a review of its chemistry and clinical utility. J Oral Maxillofac Pathol 6:251–255
Lingen MW, Kalmar JR, Karrison T, Speight PM (2008) Critical evaluation of diagnostic aids for the detection of oral cancer. Oral Oncol 44:10–22
Epstein JB, Scully C, Spinelli J (1992) Toluidine blue and Lugol’s iodine application in the assessment of oral malignant disease and lesions at risk of malignancy. J Oral Pathol Med 21:160–163
Lopez ML, del Valle MO, Cueto A (1994) Knowledge of the European code against cancer in sixth–form pupils and teachers in Asturias (Spain). Eur J Cancer Prev 3:207–213
Gandolfo S, Pentenero M, Broccoletti R, Pagano M, Carrozzo M, Scully C (2006) Toluidine blue uptake in potentially malignant oral lesions in vivo: clinical and histological assessment. Oral Oncol 42(1):89–95
Patton LL (2003) The effectiveness of community-based visual screening and utility of adjunctive diagnostic aids in the early detection of oral cancer. Oral Oncol 39:708–723
Missmann M, Jank Siegfried J, Laimer K, Gassner R (2006) A reason for the use of toluidine blue staining in the presurgical management of patients with oral squamous cell carcinomas. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 102:741–743
Mashberg A (1983) Final evaluation of tolonium chloride rinse for screening of highrisk patients with asymptomatic squamous carcinoma. J Am Dent Assoc 106:319–323
Martin LC, Kerawala CJ, Reed M (1998) The application of toluidine blue as a diagnostic adjunct in the detection of epithelial dysplasia. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 85:444–446
Rodriguez PC, Lapiedra RC, Gomez GE, Martinez SL, Warnakulasuriya S (2011) The use of toludine blue in the detection of premalignant and malignant oral lesions. J Oral Pathol Med 40:300–304
Guo Z, Yamaguchi K, Sanchez-Cespedes M et al (2001) Allelic losses in Ora Test-directed biopsies of patients with prior upper aerodigestive tract malignancies. Clin Cancer Res 7:1963–1968
Epstein JB, Feldman R, Dolor RJ, Porter SR (2003) The utility of tolonium chloride rinse in the diagnosis of recurrent or second primary cancers in patients with prior upper aerodigestive tract cancer. Head Neck 25:911–921
Zhang L, Williams M, Poh CF et al (2005) Toluidine blue staining identifies high-risk primary oral premalignant lesions with poor outcome. Cancer Res 65:8017–8021
Miller RL, Simms BW, Gould AR (1988) Toluidine blue staining for detection of oral premalignant lesions and carcinomas. J Oral Pathol Med 17:73–78
Mashberg A (1980) Reevaluation of toluidine blue application as a diagnostic adjunct in the detection of asymptomatic oral squamous cell carcinoma: a continuing prospective study of oral cancer III. Cancer 46:758–763
Hegde CM, Kamath PN, Sridharan S, Dannana NK, Raju RM (2006) Supravital staining: its role in detecting early malignancies. Indian J Otolaryngol Head Neck Surg 58:31–34
Epstein JB, Guneri P (2009) The adjunctive role of toluidine blue in detection of oral premalignant and malignant lesions. Curr Opin Otolaryngol Head Neck Surg 17:79–87
Onofre MA, Sposto MR, Navarro CM (2001) Reliability of Toluidine blue application in the detection of oral epithelial dysplasia and in situ and invasive squamous cell carcinoma. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 91:535–540
The Oral Cancer Foundation. Early Detection, Diagnosis and Staging. The Oral Cancer Foundation site. Available at: http://www.oralcancerfoundation.org/cdc/cdc_chapter5.php
Patton LL, Elter JR, Southerland JH, Strauss RP (2005) Knowledge of oral cancer risk factors and diagnostic concepts among North Carolina dentists. J Am Dent Assoc 136:602–610
Siddiqui IA, Farooq MU, Siddiqui RA, RAfi SMT (2006) Role of toluidine blue in early detection of oral cancer. Pak J Med Sci 22:184–187
Chen YW, Lin JS, Wu CH, Lui MT, Kao SY, Fong Y (2007) Application of in vivo stain of methylene blue as a diagnostic aid in the early detection and screening of oral squamous cell carcinoma and precancer lesions. J Chin Med Assoc 70(11):497–503
Canto MI, Setrakian S, Willis JE, Chak A, Petras RE, Sivak MV (2001) Methylene blue staining of dysplastic and nondysplastic Barrett’s esophagus: an in vivo and ex vivo study. Endoscopy 33:391–400
Canto MI, Setrakian S, Willis J, Chak A, Petras R, Powe NR, Sivak MV Jr (2000) Methylene blue-directed biopsies improve detection of intestinal metaplasia and dysplasia in Barrett’s esophagus. Gastrointest Endosc 51:560–568
Canto MI, Setrakian S, Petras RE, Blades E, Chak A, Sivak MV Jr (1996) Methylene blue selectively stains intestinal metaplasia in Barrett’s esophagus. Gastrointest Endosc 44:1–7
Suzuki S, Murakami H, Suzuki H, Sakakibara N, Endo M, Nakayama K (1979) An endoscopic staining method for detection and operation of early gastric cancer. Int Adv Surg Oncol 2:223–241
Mufti GR, Shah PJ, Parkinson MC, Riddle PR (1990) Diagnosis of clinically occult bladder cancer by in vivo staining with methylene blue. Br J Urol 65:173–175
Gill WB, Huffman JL, Lyon ES, Bagley DH, Schoenberg HW, Straus FH 2nd (1984) Selective surface staining of bladder tumors by intravesical methylene blue with enhanced endoscopic identification. Cancer 53:2724–2727
Riaz A, Shreedhar B, Kamboj M, Riaz SN (2013) Methylene blue as an early diagnostic marker for oral precancer and cancer. Springer Plus 2:95
Neville BW, Day TA (2002) Oral cancer and precancerous lesion. CA Cancer J Clin 52:195–215
Kusukawa J, Suefuji Y, Ryu F, Noguchi R, Iwamoto O, Kameyama T (2009) Dissemination of cancer cells into circulation occurs by incisional biopsy of oral squamous cell carcinoma. J Oral Pathol Med 29:303–307
Dyavanagoudar S, Kale A, Bhatt K, Hallikerimath S (2008) Reverse transcriptase polymerase chain reaction study to evaluate dissemination of cancer cells into circulation after incision biopsy in oral squamous cell carcinoma. Indian J Dent Res 19(4):315–319
Peters LJ (1975) A study of the influence of various diagnostic and therapeutic procedures applied to a murine squamous carcinoma on its metastatic behaviour. Br J Cancer 32:355–372
Safour IM, Wood NK, Tsiklakis K, Doemling DB, Joseph G (1984) Incisional biopsy and seeding in hamster cheek pouch carcinoma. J Dent Res 63:1116–1120
Ohtake K, Shingaki S, Nakajima T (1990) Effects of incision and irradiation on regional lymph node metastasis in carcinoma of the hamster tongue. Oral Surg Oral Med Oral Pathol 70:62–69
Eriksson O, Hagmar B, Ryd W (1984) Effects of fine-needle aspiration and other biopsy procedures on tumor dissemination in mice. Cancer 54:73–78
Kusukawa J, Kameyama T (1995) Clinical evaluation of excisional biopsy for stage I and II squamous cell carcinoma of the oral cavity. J Jpn Soc Cancer Ther 30:1817–1827
Kusukawa J, Nakamura Y, Kameyama T (1998) Evaluation of excisional biopsy for stage I and II squamous cell carcinoma of the oral cavity. Int J Clin Oncol 3:311–316
Cai WM, Zhang HX, Hu YH, Gu XZ (1983) Influence of biopsy on the prognosis of nasopharyngeal carcinoma—a critical study of biopsy from the nasopharynx and cervical lymph node of 649 patients. Int J Radiol Oncol Biol Phys 9:1439–1444
Gandolfo S, Carbone M, Carrozzo M, Scamuzzi S (1993) Biopsy technics in oral oncology: excisional or incisional biopsy? A crical review of the literature and the authors’ personal contribution. Minerva Stomatol 42:69–75
Ramani P, Thomas G, Ahmed S (2005) Use of Rt-PCR in detecting disseminated cancer cells after incisional biopsy among oral squamous cell carcinoma patients. J Cancer Res Ther 1:92–97
Frydrych AM, Parsons R, Threlfall T, Austin N, Davies GR, Booth D (2010) Oral cavity squamous cell carcinoma survival by biopsy type: a cancer registry study. Aust Dent J 55:378–384
Hellman K, Hilgard P, Eccles S (1980) Metastasis: clinical and experimental aspects. Martinus Nijhoff, The Hague, pp 50–54
Warnakulasuriya KA, Johnson NW (1996) Sensitivity and specificity of OraScan toludine blue mouthrinse in the detection of oral cancer and precancer. J Oral Pathol Med 25:97–103
Gupta A, Singh M, Ibrahim R, Mehrotra R (2007) Utility of toluidine blue staining and brush biopsy in precancerous and cancerous oral lesions. Acta Cytol 51(5):788–794
Wiersma AC, van der Waal I (1990) Toluidine blue staining for detection of oral squamous carcinoma. Ned Tijdschr Tandheelkd 97(10):431–432
Silverman S Jr, Migliorati C, Barbosa J (1984) Toluidine blue staining in the detection of oral precancerous and malignant lesions. Oral Surg Oral Med Oral Pathol 57(4):379–382
Awan Kh, Yang Y, Morgan P, Warnakulasuriya S (2012) Utility of toluidine blue as a diagnostic adjunct in the detection of potentially malignant disorders of the oral cavity—a clinical and histological assessment. Oral Dis 18(8):728–733
Vahidy NA. Zaidi SHM, Jafarey NA (1972) Toluidine blue test for detection of carcinoma of the oral cavity: an evaluation. J Surg Oncol 434
Allegra E, Lombardo N, Puzzo L, Garozzo A (2009) The usefulness of toluidine staining as a diagnostic tool for precancerous and cancerous oropharyngeal and oral cavity lesions. Acta Otorhinolaryngol Ital 29(4):187–190
Upadhyay J, Rao NN, Upadhyay RB, Agarwal P (2011) Reliability of toluidine blue vital staining in detection of potentially malignant oral lesions—time to reconsider. Asian Pacific J Cancer Prev 12:1757–1760
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chhabra, N., Chhabra, S. & Sapra, N. Diagnostic Modalities for Squamous Cell Carcinoma: An Extensive Review of Literature-Considering Toluidine Blue as a Useful Adjunct. J. Maxillofac. Oral Surg. 14, 188–200 (2015). https://doi.org/10.1007/s12663-014-0660-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12663-014-0660-6