Abstract
Prenatal and early life exposure of chlorpyrifos (CPF), a widely used pesticide, is known to cause neuronal deficits and Parkinson’s disease (PD). However, data about the effect of its exposure at adult stages on PD-like symptoms and associated bone loss is scanty. In the present study, we investigated the impact of CPF on the behavioral alterations seen in PD using adult Swiss albino mice. PD is often associated with bone loss. Hence, skeletal changes were also evaluated using micro-computed tomography and histology. MPTP was used as a positive control. Cell culture studies using MC3T3E-1, SHSY5Y, and primary osteoclast cultures were done to understand the cellular mechanism for the behavioral and skeletal changes. Our results showed that CPF treatment leads to PD-like symptoms due to the loss of dopaminergic neurons. Moreover, CPF has a deleterious effect on the trabecular bone through both indirect changes in circulating factors and direct stimulation of multinucleate osteoclast cell formation. The impact on the bone mass was even stronger than MPTP. In conclusion, this is the first report demonstrating that CPF induces parkinsonian features in adult Swiss albino mice and it is accompanied by loss of trabecular bone.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Chlorpyrifos (CPF) is a pesticide, which comes under the organophosphothionate (OPT) group of chemicals (Eaton et al. 2008). During World War II, OPTs were first introduced as nerve gas chemicals to attack the neurotransmitters in the body. However, later this group of molecules was repurposed as insecticides (Trasande 2017). CPF is one of the most commonly used OPT insecticides for agricultural and domestic applications, and its contamination is detectable in crops, fruits, and vegetables (Konradsen 2007; Lozowicka 2015). Numerous human and animal studies have reported that CPF exposures cause hepatotoxicity, neurotoxicity, and endocrine disruption (Eaton et al. 2008; Trasande 2017). Indoor residential and some agricultural uses of CPF were prohibited in 2000–2001 after the US environmental protection agency (EPA) found that the exposure levels faced by the children were unsafe (Kim et al. 2013; Mojsak et al. 2018; Rauh 2018). In 2016, EPA put a complete ban on the use of CPF for all agricultural, gardening, and domestic purposes (Rauh 2018). However, this ban was not implemented as in 2017 this plan was scrapped by the new administration of EPA (Rauh 2018). Moreover, most of the other parts of the world are still using it as an insecticide due to its low cost and easy availability.
Owing to its genesis, the most prominent effect of CPF is neuronal damage. Several epidemiological studies have revealed that developmental CPF exposure primes to cognitive impairment (Engel et al. 2011; Grandjean and Landrigan 2014). Besides, CPF’s fetal and neonatal exposure leads to behavioral changes due to alterations in the developing rodent brain (Aldridge et al. 2005; Venerosi et al. 2015) along with cholinergic impairment and long-term dopaminergic neuronal damage (Dam et al. 2000). Some of the reports have also suggested that developmental exposure of CPF may cause Parkinson’s disease (PD)-like symptoms in rodents. PD is a neurodegenerative disorder with a characteristic dopaminergic neuronal loss in the ventrolateral and caudal portions of the substantia nigra pars compacta (SNpc) and a decline in the levels of dopamine (DA) in the striatum (Kalia and Lang 2015; Opara et al. 2017; Schneider et al. 2017; Titova et al. 2017a; Titova et al. 2017b). The disease has both motor and non-motor phenotypes. The motor phenotype includes trouble in movement, resting tremor, postural instability, and rigidity with impaired speech and dementia at advanced stages. There is a broad spectrum of non-motor symptoms associated with PD-like cognitive impairment, mood disorders, sleep difficulties, loss of sense of smell, constipation, etc. (Cecchini et al. 2019; Jiang 2019) One of the most common non-motor features of PD is osteoporosis, which results in low bone mass leading to fracture (Choi et al. 2017; Cummings and Eastell 2016). Data suggest that 91% of women and 61% of men with PD also have osteoporosis (Watts 2014). Osteoporosis is more frightening in PD patients as there are higher chances of them falling due to impaired movement and postural instability (Metta et al. 2017; Sleeman et al. 2016).
Most of the previous studies with CPF exposures were directed towards its developmental toxicity demonstrating the loss of dopaminergic neurons in the striatum (Basaure et al. 2018; Silver et al. 2017). Only limited studies focused on the PD-associated behavioral changes when CPF exposures happened in the later stages of life. Besides, there is no study investigating the PD-associated bone loss after CPF exposure. Recently, in our studies, we have shown that the MPTP mouse model of PD not only displays the behavioral symptoms of the disease but also shows deterioration of bone. We have also demonstrated that this effect on bone mass was due to the endocrine changes in the serum of the animals (Ali et al. 2019). In the present study, we are investigating the effect of CPF exposures on the behavioral aspects of PD when given to the adult mice. Taking the lead from our previous study, we have also evaluated its effect on the skeleton and whether the result is due to the direct exposure of CPF to bone cells or indirectly through the changes in the circulating factors. MPTP is used as a positive control throughout the study. Until now, there was no study evaluating the effect of CPF exposure on bone mass or bone cells. Conclusively, the present study provides novel data on the effects of CPF exposures on dopaminergic neurotoxicity in the striatum of adult mice along with its predisposition to the osteoporotic conditions.
Materials and Methods
CPF, 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine and 1-methyl-4-phenylpyridinium iodide (MPTP and MPP+), cell culture mediums, fine chemicals (α-MEM, FBS), sodium pyruvate, non-essential amino acid, antimycotic antibiotic solution (100×), sodium bicarbonate, and Hams F-12 were purchased either from Invitrogen (Carlsbad, CA, USA) or Sigma-Aldrich (St. Louis, MO, USA).
Pesticide Solution
CPF was dissolved in groundnut oil and administered orally at of 3 and 6 mg/kg body weight doses. MPTP was dissolved in normal saline and administered intraperitoneally at a dose of 20 mg/kg body weight of mice.
Animals and Care
Male Swiss albino mice (8 weeks old and 25–30 g) were provided standard conditions of 12:12-h light/dark cycles, 50% ± 5% humidity, and temperature of 25 ± 2 °C, and housed in polypropylene cages. Pellet diet was provided ad libitum with free access to water. All animal procedures were done following the guidelines of the Institute Animal Ethics Committee.
Study Overview
There were four groups into which 40 male Swiss albino mice were distributed: Control, MPTP, 3 and 6 mg/kg/day doses of CPF (1/20 and 1/10 of the LD50, respectively). The MPTP treatment included four I.P. injections at a total dose of 20 mg/kg b.wt. with 2 h interval on day 1 (Jackson-Lewis and Przedborski 2007) followed by animal maintenance on chow diet for 8 weeks. Acute oral LD50 of CPF in mice was 60 mg/kg and sublethal doses not exceeding 1/3 of the LD50 were selected based on previous studies (El-Sebae et al. 1978). Mice received oral gavage of CPF. Analysis was done for neurobehavioral deficits after CPF and MPTP treatments at 4 and 8 weeks. After which, they were sacrificed, and various organs such as the brain, bones, and serum were harvested. Dissected femurs and tibias were stored at 4 °C until the measurement of various bone parameters was performed.
Neurobehavioral Studies
Block Test
This behavioral assay was done in accordance with previously published protocols (Ali and Rajini 2016; Sanberg et al. 1988). Time taken by the mouse to recover from a cataleptic posture, i.e., from an awkward to its normal posture when holding a 3-cm wooden block by its forelimb was recorded. A score of 0.1 was awarded on failing to revert to its normal posture. The average score of 10 trials was considered as the catalepsy score.
Rotarod Test
Motor coordination and learning test were followed using a previously published method (Deacon 2013). Briefly, mice were made to run on a digitalized treadmill (the rotarod apparatus, Orchid Scientifics, Nasik, India) at an initial speed of 4 rpm that was constantly accelerated at a rate of 20 rpm/min. Time at which they fell off the rotarod was noted. A retrial was given if the mouse fell off before 10 s and a baseline score of 4 rpm was given if it failed to grip the rod within 10 s for three consecutive trials. Ten such tests were given to each animal per day.
Gait Analysis
For gait analysis, fore and hind paws of animals were wetted with color and were allowed to run on a strip of a white sheet of paper measuring 42 × 4.5 cm in length. For stride length (SL), the distance between two consecutive prints of the same paw was measured.
DA Content
Striatal DA was measured using a previously published protocol (Gruntenko et al. 2005). In short, we homogenized striatal samples in 100 mM PBS buffer with 0.1% Tween-20 pH 8 and centrifuged them at 4 °C and 14,000 rpm for 30 min. DA levels were estimated using high-performance liquid chromatography (HPLC) with the following specifications: C18 reverse phase column, isocratic pump, with a flow rate of 1 mL/min and detection at 280 nm. The mobile phase was made with 0.2% trifluoroacetic acid in 30% methanol. A 20-μL of sample was injected. DA concentration was expressed as μg/mg protein.
Acetylcholinesterase Activity
Acetylcholinesterase (AChE) activity was measured following a previously published method (Ellman et al. 1961). Briefly, to an amount of phosphate buffer (100 mM, pH 8), we added acetylthiocholine iodide (0.01 mL of 0.1 M). To this, a suitable amount of striatal/cell supernatant stock and dithionitrobenzene were also added. The rate of change in absorbance for the solution was examined over 5 min at 420 nm in a microplate reader, and the results were expressed as units/mg protein.
Bone Parameter Measurement Through Micro-computed Tomography
Bone analysis was done using Sky Scan 1076 micro-computed tomography (μ-CT) scanner (Aartselaar, Belgium) (Sharan et al. 2011). Briefly, the bone samples were scanned at of 9-μm resolution trailed by reconstruction by recon software. The trabecular bone analysis was done by drawing regions of interest of 350 secondary spongiosa bone slides after excluding all primary spongiosa. For cortical bone analysis, 450 following image slides from growth plate were discarded, 100 following image slides were selected for analysis. BMD of the femur and tibia were measured from the volume of interest made for the cortical and trabecular region using hydroxyapatite phantom rods of 4 mm in diameter with known BMD taken as reference (Bachagol et al. 2018).
Quantitative Real-Time Polymer Chain Reaction
RNA from the bones of CPF (6 mg/kg/day dose), MPTP, and vehicle-treated mice were isolated through Trizol method (Invitrogen) following previously published protocols (Khan et al. 2012; Lewis et al. 2017). One microgram of RNA from each was used for cDNA synthesis using a cDNA synthesis kit (Applied Biosystems). Using specific primers, relative expression of transcripts for all genes was ascertained using SYBR Green chemistry. β-Actin was taken as an internal control (Supplementary Table 1).
Bone Histology
Bone histology was done with third lumbar vertebral samples. Calcein double fluorochrome labeling on the bone samples was performed for histomorphometric study using a previously published method (Roman-Garcia et al. 2014). Briefly, bones were embedded in an acrylic material, and 5 μm sections were sliced from lumber three vertebra using Leica microtome. For the determination of bone formation rate/bone surface (BFR/BS) and mineral appositional rate (MAR), photographs were taken under fluorescent microscope aided with appropriate filters. BFR/BS and MAR were calculated according to a previously published method (Sharan et al. 2011). Osteomeasure Analysis System (Osteometrics, Atlanta, GA, USA) was used to analyze undecalcified sections stained with tartrate-resistant acid phosphatase (TRAP) or Von Kossa/Von Gieson following previously published protocols (Lewis et al. 2017; Sharan et al. 2017).
Cell Culture
MC-3T3-E-1 preosteoblast and SH-SY5Y human neuroblastoma cell lines were procured from American Type Culture Collection (ATCC, USA). MC-3T3-E-1 cells were grown in α-MEM, and SH-SY5Y cells in Hams F-12 media supplemented with heat-inactivated fetal bovine serum, penicillin (10 IU/mL), streptomycin, (10 mg/mL), 2 mM glutamine, 1 mM sodium pyruvate, and 0.1 mM non-essential amino acids.
Dopaminergic Differentiation of SH-SY5Y Cells
Seven thousand SH-SY5Y cells/well were seeded on a 96-well plate that was grown for 24 h at 37 °C with 5% CO2 after which they were differentiated. Later, the cells were treated with 10 μM retinoic acid (RA) per well for 3 days, followed by 80 nM 12-O-tetradecanoyl-phorbol-13-acetate (TPA) per well for another 3 days (Presgraves et al. 2004). On the 7th day, MPP+ and CPF treatments with concentrations from 1 to 100 μM were given to the cells, and various assays were performed as mentioned below.
MTT Assay
Five thousand MC3T3-E1 cells/well and 10,000 SH-SY5Y cells/well were plated in a 96-well plate and were allowed to grow for 24 h. They were further treated with varying concentrations of CPF and MPP+ (1–100 μM) in 0.5% FBS. After 24 h, MTT (5 mg/mL) was added to the cells for 4 h. The formazan crystals formed were dissolved in 50 μL of DMSO. The absorbance was read at 560 nm.
Bromodeoxyuridine Cell Proliferation Assay
For bromodeoxyuridine (BrdU) assay, 4000 MC-3T3-E-1 cells were plated on a 96-well plate and were exposed to CPF and MPP+ (1–100 μM concentrations). BrdU ELISA from Roche (Indianapolis, IN, USA) was used to analyze cell proliferation following the manufacturer’s instructions.
Alkaline Phosphatase Assay
Alkaline phosphatase (ALP) activity was measured using previously published protocols (Khan et al. 2013; Sharan et al. 2017). Briefly, CPF and MPP+ (at concentrations 1–100 μM) treatment were given to MC3T3-E-1 cells for 48 h under osteoblast differentiating conditions (α-MEM medium with 10% FBS, 10 mM β-glycerophosphate, and 50 mg/mL of ascorbic acid). Total ALP activity was measured using p-nitrophenyl phosphate (PNPP) as a substrate, and absorbance was read at 405 nm.
In Vitro Mineralization Assay
Twenty-five thousand to 30,000 MC3T3-E1 cells/well were seeded in 12-well plate and were grown in the presence of CPF and MPP+ for 30 days in osteoblast differentiation medium. The media were changed every alternate day along with the treatments. After the end of the experiment, cells were washed in PBS and fixed for 30 min with 4% formalin after which they were stained with 40 mM Alizarin red stain for 1 h to detect calcification. Extraction was done with 10% cetylpyridinium chloride and read calorimetrically at 570 nm (Sharan et al. 2017).
Differentiation of Osteoclasts
Bone marrow cell (BMC) cultures were exposed to osteoclast differentiation medium comprising 30 ng/mL of receptor activator of nuclear factor kB ligand (RANKL) and 50 ng/mL of monocyte/macrophage colony-stimulating factor (M-CSF). Osteoclast differentiation was quantified through histochemical staining for TRAP (Siddiqui et al. 2010; Swarnkar et al. 2011). Treatment was done throughout the differentiation.
Statistical Analysis
Mean and standard error (SE) values were determined for all the parameters, and the results were expressed as mean ± SE. The data were analyzed employing one-way analysis of variance (ANOVA) followed by post hoc analysis (Tukey test) for the comparison of means to determine the significance of difference among the groups. “P” values below 0.05 were considered statistically different.
Results
Effect of CPF on the Behavioral Symptoms of PD
Male Swiss albino mice were divided into four groups namely vehicle, MPTP, and two doses of CPF (3 and 6 mg/kg/day). MPTP injections were administered four times with 2-h interval on the first day and mice were maintained on chow diet for 8 weeks. CPF treatments were given daily for 8 weeks by oral gavage. Behavioral studies were done 4 weeks and 8 weeks after the treatment to evaluate the potential of CPF to cause the development of PD-like symptoms. MPTP was used as a positive control.
Treatment-induced catalepsy in mice was analyzed through the block test. While a lower dose of CPF did not affect catalepsy score after 4 weeks of treatment, and symptoms were apparent nearing 8 weeks of treatment. The higher dose of 6 mg/kg/day of CPF however showed an increased cataleptic score even after 4 weeks of treatment which persisted till 8 weeks when compared to control (Fig. 1a, b). MPTP cataleptic scores were higher as compared to CPF at both 4 and 8 weeks after treatment.
CPF increased neurobehavioral deficits similar to MPTP and decreased neurochemical parameters in the striatum as compared to control. Vehicle or CPF at 3 and 6 mg/kg/day doses were administered orally to the animals. MPTP injections were given on day 1. All the groups were analyzed at 4 and 8 weeks for a, b catalepsy score by block test, c, d total time taken to remain balanced on the rotating drum at consecutive rpm by rotarod test, e, f time taken to remain balanced on the rotating drum at static rpm by rotarod test, and g, h stride length by gait analysis. i Dopamine content and j AChE activity in the striatum were measured after 8 weeks. Each group contains ten animals. Data are presented as mean ± S.E.M. (*P < 0.05, **P < 0.01; ***P < 0.001)
We further subjected the mice to the rotarod test to assess their motor coordination and learning ability. Results of the study showed that CPF exposures at both the doses had a significant decrease in their motor coordination comparably for both 4 and 8 weeks of treatment as compared to control when tested over the accelerating drum (Fig. 1c, d). The effect was even comparable to the MPTP group. However, the analysis of time taken to remain balanced on the rotating drum at static rpm showed a significant decrease at 4 weeks and 8 weeks only with 6 mg/kg/day dose of CPF when compared with control. The effect at this dose was even comparable to that of MPTP treatment (Fig. 1e, f).
Finally, both CPF and MPTP treatments were studied for their effect on the parameters of gait impairment as it is a cardinal clinical finding in patients with PD. CPF treatment at both doses caused a significant decrease in the stride length of forelimb and hind limb after 4 and 8 weeks of treatment. The results were comparable to the MPTP group (Fig. 1g, h).
Effect of CPF Treatment on DA Content and AChE Activity in the Striatum
Striatal DA levels were measured in the mice after 8 weeks of CPF and MPTP treatment. A significant decrease in striatal DA levels was observed in both the doses of CPF-treated groups as compared to the control (Fig. 1i). Next, we measured the AChE activity calorimetrically in the striatal homogenates for CPF and MPTP-treated mice. Similar to DA, the results showed a significant reduction of the striatal AChE activity at both the doses of CPF when compared to control. The effect at both the doses was comparable to MPTP treatments (Fig. 1j).
Effect of CPF on SH-SY5Y Neuroblastoma Cell Lines
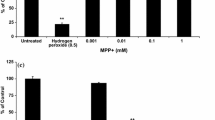
Dopaminergic neuron toxicity was further analyzed for both 1-methyl-4-phenylpyridinium ion (MPP+) (the active toxic metabolite of MPTP) and CPF using neuroblastoma cell line. In vitro treatment of MPP+ and CPF to undifferentiated SH-SY5Y cells had no cytotoxic effect at 1–100 μM concentrations (Fig. 2a). However, when SH-SY5Y cells were differentiated towards a dopaminergic lineage, MPP+ (~ 37%) and CPF (~ 41%) treatments both showed cytotoxicity at 100 μM concentration (Fig. 2b). Moreover, we also found a significant decrease in the AChE activity of the differentiated SH-SY5Y cells when treated with both MPP+ and CPF at 10 and 100 μM concentrations (Fig. 2c).
CPF and MPP+ decrease SH-SY5Y cell viability differentiated towards dopaminergic neurons. a MTT assay on undifferentiated SH-SY5Y cells treated with varying concentrations of CPF and MPP+. b MTT assay on SH-SY5Y cells differentiated towards dopaminergic phenotype and treated with a 100-μM concentration of CPF and MPP+ for 24 h. c AChE activity of SH-SY5Y cells differentiated to dopaminergic phenotype and treated with CPF and MPP+ at 100 μM concentration. Data are presented as mean ± S.E.M. (*P < 0.05, **P < 0.01; ***P < 0.001 as compared to vehicle)
Effect of CPF and MPTP on Trabecular Bone
We next analyzed the trabecular bone structure at femur distal metaphysis and tibia metaphysis of the control and treatment group mice by μ-CT after 8 weeks of treatment with MPTP and CPF. As shown in Fig. 3a, the reconstructed 3D μ-CT image at femur trabeculae revealed a gross depletion of trabecular structure in MPTP and both the doses of CPF as compared to the control. Furthermore, CPF treatment at both doses resulted in decreased trabecular bone volume/tissue volume (BV/TV) (Fig. 3b), trabecular bone mineral density (BMD) (Fig. 3c), trabecular number (Tb.N) (Fig. 3d), and connection density (Conn. Den) (Fig. 3e) when compared to control. The effect was even comparable to the MPTP group. Femur trabecular separation (Tb.Sp) (Fig. 3f) was only increased in 6 mg/kg/day dose of CPF when compared to control, whereas the lower dose of CPF and MPTP did not affect (Fig. 3f). Structure model index (SMI) (Fig. 3g) which is inversely proportional to the quality of bone was significantly increased in MPTP and both the doses of CPF group as compared to the control.
CPF and MPTP treatment cause loss of bone microarchitecture. a Representative reconstructed 3-D μ-CT images, b BV/TV (%), c BMD (g cm−3), d Tb.N (mm−1), e connection density (mm−3), f Tb.Sp (mm), and g SMI at trabecular region of femur distal metaphysis of the vehicle, CPF (3 and 6 mg/kg/day), and MPTP-treated mice. h Representative reconstructed 3-D μ-CT images, i BV/TV (%), j BMD (g cm−3), k Tb.N (mm−1), l connection density (mm−3), m Tb.Sp (mm), and n SMI at trabecular tibia metaphysis of the vehicle, CPF, and MPTP-treated mice. Each group contains 10 animals. Data are presented as mean ± S.E.M. (*P < 0.05, **P < 0.01; ***P < 0.001 as compared to vehicle)
Similarly, in the tibia, CPF (at both 3 and 6 mg/kg/day doses) and MPTP treatments resulted in a gross decrease of trabecular bone structure (Fig. 3h) with a decreased BV/TV (Fig. 3i), BMD (Fig. 3j), Tb.N (Fig. 3k), and Conn. Den (Fig. 3l). Similar to the femur, Tb.Sp (Fig. 3m) was only increased in 6 mg/kg/day dose of CPF whereas SMI was increased at both the doses of CPF and MPTP (Fig. 3n) when compared to control.
Effect of CPF and MPTP on Vertebral Histology
The histological analysis of the third lumbar vertebra of the control and treated mice was performed. The results showed a significant decrease in the in BV/TV (%) of CPF (at both 3 and 6 mg/kg/day dose) treated mice compared to control, where the values at higher dose were even significantly lower than MPTP bones. MAR and BFR/BS also had a significant decrease at both the doses of CPF showing a decline in bone formation. Consistent to that both osteoblasts per trabecular area (N.Ob/T.Ar) and the number of osteocytes per trabecular area (N.Os/T.Ar) were decreased in CPF and MPTP-treated groups when compared control. Analysis of osteoclast activity parameters revealed an increase in the number of osteoclast per trabecular area (N.Oc/T.Ar) and osteoclast surface per bone surface (Oc.S/BS) in the MPTP and CPF-treated mice compared to control (Fig. 4a). Interestingly, the Oc.S/BS and N.Oc/T.Ar were significantly higher in 6 mg/kg/day dose of CPF as compared to MPTP.
CPF treatment decreases vertebral bone mass more than MPTP with decreased osteoblastogenesis and increased osteoclastogenesis. a Representative von Kossa staining for lumbar 4 vertebrae from CPF (3 and 6 mg/kg/day), MPTP, and vehicle-treated mice with measurement of bone volume/tissue volume percentage (BV/TV %), measurements for mineral apposition rate (MAR) and bone formation rate per bone surface (BFR/BS), analysis of osteoblast numbers per total area (N.Ob/T.Ar), osteocyte number per total area (N.Os/T.Ar), analysis of osteoclast numbers per total area (N.Oc/T.Ar), and osteoclast surface per bone surface (Oc.S/BS). b–e qPCR analysis of long bone from control, CPF (6 mg/kg/day), and MPTP-treated mice showed a decreased mRNA expression levels for bRunx2, cAtf-4, dCol1a1 and increased levels for eTrap. Data are presented as mean ± S.E.M for n = 6 (*P < 0.05, **P < 0.01; ***P < 0.001 and ^P < 0.05 as compared to MPTP)
Effect of CPF on the mRNA Expression of the Markers of Bone Formation and Resorption
The quantitative real-time PCR analysis of the bone formation markers from the tibia of control and treatment group mice showed a significant decrease in mRNA expression levels of osteoblast early differentiation marker Runx2, Col1a1, and late differentiation marker Atf-4 in the 6 mg/kg/day dose of the CPF group when compared to control. The expression was even comparable to MPTP-treated mice bones (Fig. 4b–d). Whereas, the expression levels of Trap, the osteoclast differentiation marker, were significantly increased in the CPF treatment group (Fig. 4e). Interestingly, the expression levels of Trap in CPF-treated bones were even significantly higher than the MPTP group.
Effect of CPF and MPTP-Treated Mice Serum on Bone Cells
An ex vivo model of toxicity of MPTP and CPF was also set up to model PD physiology. To clear out the CPF and its metabolite from serum, the treatment was stopped 3 days before the sacrifice. Serum that was collected from both MPTP and CPF (6 mg/kg/day) treated mice was exposed to bone cells (to a final volume of 1% mice serum in the medium). MTT assay with MC3T3-E1 preosteoblast cells showed a significant decrease in cell viability when treated with MPTP and CPF-treated mice serum as compared to control mice serum treatment (Fig. 5a). Similarly, proliferation (Fig. 5b) and differentiation (Fig. 5c) of the cells also decreased in the presence of both MPTP and CPF-treated mice serum.
Ex vivo treatment of CPF and MPTP-treated mice serum decreases osteoblastogenesis and increases osteoclastogenesis. a MTT assay, b BrdU ELISA, and c ALP assay of cells treated with CPF and MPTP-treated mice sera on MC3T3-E1 cells as compared to vehicle-treated mice sera. d Representative picture and e analysis of the number of TRAP-positive multinucleated osteoclasts from primary osteoclast culture treated with mice serum for 6 days. Data are presented as mean ± S.E.M. (*P < 0.05, **P < 0.01; ***P < 0.001)
MPTP and CPF-treated mice serum was also treated with primary bone marrow cells to study their potential for osteoclast differentiation. The results showed a ~threefold increase in the number of multinucleated TRAP-positive osteoclast cells in both MPTP and CPF-treated mice serum culture when they were compared to the control group (Fig. 5d).
Effect of CPF on MC3T3-E1 Preosteoblastic Cell Lines
To evaluate whether the decrease in bone mass due to CPF treatment was caused by its direct effect on bone-forming osteoblast cells, we treated MC3T3-E1 preosteoblastic cells with MPP+ and CPF.
Our results showed that treatment of MPP+ and CPF at varying concentrations (1–100 μM) did not affect the osteoblast cell viability (Fig. 6a), differentiation (Fig. 6b), and matrix mineralization (Fig. 6c).
CPF treatment shows no effect on osteoblast cells but increases osteoclastogenesis. a MTT assay: MC3T3-E1 preosteoblast cells treated with varying concentrations of CPF and MPP+ for 24 h. b Alkaline phosphatase (ALP) assay: Cells treated with varying concentrations of CPF and MPP+ for 48 h followed by ALP activity assay. c MC3T3-E1 cells treated with CPF and MPP+ for 21 days followed by alizarin red staining and extraction. d Primary osteoclast culture treated with CPF and MPP+ for 6 days in osteoclast differentiation media and analysis of the number of multinucleated TRAP-positive cells after CPF treatment. Data are presented as mean ± S.E.M. (*P < 0.05, **P < 0.01; ***P < 0.001)
Effect of CPF on Primary Osteoclast Differentiation
Effect of MPP+ and CPF treatments on bone marrow-derived primary osteoclast cell differentiation was also investigated. Our results showed that while MPP+ treatment had no significant effect on the formation of multinucleated TRAP-positive cells (Fig. 6d), CPF at 100 μM concentration significantly increased (~two times) the number of multinucleated TRAP-positive osteoclast cells when compared to control and MPTP (Fig. 6d).
Discussion
Despite a complete ban over the use of CPF by EPA (Clevelan et al. 2001), it is still the widely used pesticide in most parts of the world. Although there is evidence that fetal and early life exposure to CPF leads to persistent neurological deficits in humans and rodents (Eaton et al. 2008; Engel et al. 2011), there is no data in conjunction to the bone health, as osteoporosis is the most common non-motor symptom of PD (Dobson et al. 2013; Schneider et al. 2008). Moreover, the knowledge about the neurotoxic effect of CPF exposure leading to PD at adult stages remains obscure. These limitations highlight the need to implement further investigations in this direction. Therefore, in the present study, using an animal model, we have evaluated the neurotoxic effect of CPF in the adult brain and the consequent behavioral symptoms of PD. Moreover, we have also reported for the first time its influence over the adult bone microarchitecture and associated histomorphometric changes. The results obtained were compared with MPTP treatment groups as it is an established model for PD and bone loss.
PD pathogenesis has been shown to affect catalepsy, motor coordination, and gait negatively (Beitz 2014). In the present study, to examine the effect of CPF treatment on PD symptoms, adult male Swiss albino mice were treated with CPF at 3 and 6 mg/kg/day. Mice were subjected to several behavioral tests after 4 and 8 weeks of treatment to check whether PD-like symptoms have developed or not. While 6 mg/kg/day dose of CPF resulted in catalepsy, motor deficit, and gait impairment even after 4 weeks of treatment, 3 mg/kg/day dose had a limited effect. These changes were further enhanced after 8 weeks at both doses. Interestingly, 6 mg/kg/day dose of CPF showed effect comparable to MPTP except for the catalepsy score. Further, reduced levels of striatal DA and AChE activity, characteristic features of PD, were also observed at both the doses of CPF with 6 mg/kg/day showing a greater loss. The effect at both the doses was even comparable to MPTP, indicating the severity of the damage. Next to understand whether CPF causes loss of neurons by targeting undifferentiated neuronal cells or the differentiated dopaminergic neurons, we used SHY5Y cell line. Treatment of CPF, like MPP+ (the toxic metabolite of MPTP), did not affect undifferentiated cells. However, it resulted in the decreased viability of dopaminergic neurons and their reduced AChE activity. This result suggested that similar to MPTP, CPF targets the differentiated dopaminergic neurons. These results were consistent with the earlier studies where it had been shown that developmental and early age exposure of CPF causes PD-like symptoms (Dam et al. 2000; Venerosi et al. 2015).
In recent years, understanding of the neuroendocrine regulation of bone mass has gained tremendous importance. Several pieces of evidence suggest that factors originating from the brain regulate skeleton maturation and homeostasis (Fukuda and Takeda 2015; Huang et al. 2019; Sharan et al. 2017; Sharan and Yadav 2014). In neurodegenerative diseases like PD, clinicians have observed that bone loss is a common phenomenon. Data suggest that there is a strong association between PD and osteoporosis (Ali et al. 2019; Cheng et al. 2019). In our recent study using a mouse model of PD, we have also shown that PD results in the trabecular bone loss in rodents due to decreased bone-forming osteoblast cells and increased bone-resorbing osteoclast cells (Ali et al. 2019). Results of the present study showed the occurrence of PD-like symptoms with CPF treatment. Hence, next, we evaluated whether the loss of the dopaminergic neurons is also accompanied by a decline in bone mass in CPF-treated mice or not. Results of our study showed that both the doses of CPF caused decreased BV/TV and BMD of femur and tibia. Interestingly, both lower and higher doses of CPF had a similar and comparable decline in these parameters when compared to MPTP. Besides, connection density and SMI were also negatively affected by CPF, and there was no significant difference between CPF and MPTP group. Although there was a comparable decrease in tibial Tb.N with MPTP in CPF group, in femur only CPF (at both the doses) decreased the Tb.N. Moreover, Tb.Sp. was only increased by 6 mg/kg/day dose of CPF. These results indicated a more robust effect of CPF on the bone as compared to MPTP. Histological and histomorphometrical analysis of lumber three vertebrae of mice revealed that CPF groups had higher bone loss in vertebra as compared to MPTP (lower BV/TV). There was a comparable decrease in bone formation parameters, osteoblast, and osteocyte numbers in CPF and MPTP groups. However, there was a stronger effect on osteoclastogenesis in CPF groups as compared to MPTP groups, where there was a greater increase in osteoclast number and surface to the bone surface in the CPF group. Moreover, while there was a comparable decline in gene expression of osteogenic genes between CPF and MPTP bones, the expression of Trap (an osteoclastogenic marker) was higher in the CPF group as compared to MPTP.
In our previous study, we have shown that in the MPTP mouse model of PD, bone loss happens due to the paracrine changes in the circulating neuroendocrine factors (Ali et al. 2019). In this study where the PD symptoms were weaker than MPTP but the bone loss was higher with increased osteoclast markers, there was a possibility that CPF had both indirect (through circulating factors) and direct (through osteoclast) effect on bone cells. To further test this, we first evaluated whether the changes in circulating factors are indirectly responsible for the greater decrease in bone mass of the CPF group than the MPTP-treated mice. For that, we did the ex vivo treatment of the bone cells with the serum obtained from the control and treatment group mice. Our results demonstrated that the CPF-treated mice serum was equally cytotoxic to the osteoblast cells when compared to MPTP. Moreover, both MPTP and CPF-treated mice serum had a comparable negative effect on the proliferation and differentiation of osteoblast cells. Besides, both the groups’ sera treatment equally enhanced the formation of multinucleated osteoclast cells. These results suggested that both MPTP and CPF affected the circulating factors in more or less the same way. Next, to find out the possibility of the direct modulation of bone cells activity by CPF, we treated osteoblast and osteoclast cells with varying concentrations of CPF and MPP+. Although both CPF and MPP+ did not affect osteoblast cell viability, proliferation, differentiation, and mineralization, only CPF treatment led to an increase in osteoclastogenesis when treated to differentiation osteoclast cells. These results suggested that unlike MPTP, CPF has a deleterious effect on bone both indirectly through changes in circulating factors and directly through the stimulation of multinucleate osteoclast cell formation.
In conclusion, our study reveals that CPF, when given to adult mice, induces PD-like behavioral changes along with the loss of DA and AChE activity in the striatum. Furthermore, the loss of dopaminergic neurons is accompanied by the microarchitectural deterioration of the trabecular bone even at a 1/20th time dose of the LD50 of CPF. We have also shown that the effect is both due to the changes in the circulating factors in the serum (leading to the decreased bone formation and increased bone resorption) and direct stimulation of osteoclastogenesis (leading to further increase in bone resorption) by CPF (Fig. 7).
References
Aldridge JE, Meyer A, Seidler FJ, Slotkin TA (2005) Alterations in central nervous system serotonergic and dopaminergic synaptic activity in adulthood after prenatal or neonatal chlorpyrifos exposure. Environ Health Perspect 113:1027–1031. https://doi.org/10.1289/ehp.7968
Ali SJ, Rajini PS (2016) Effect of monocrotophos, an organophosphorus insecticide, on the striatal dopaminergic system in a mouse model of Parkinson’s disease. Toxicol Ind Health 32:1153–1165. https://doi.org/10.1177/0748233714547733
Ali SJ, Ellur G, Khan MT, Sharan K (2019) Bone loss in MPTP mouse model of Parkinson’s disease is triggered by decreased osteoblastogenesis and increased osteoclastogenesis. Toxicol Appl Pharmacol 363:154–163. https://doi.org/10.1016/j.taap.2018.12.003
Bachagol D, Joseph GS, Ellur G, Patel K, Aruna P et al (2018) Stimulation of liver IGF-1 expression promotes peak bone mass achievement in growing rats: a study with pomegranate seed oil. J Nutr Biochem 52:18–26. https://doi.org/10.1016/j.jnutbio.2017.09.023
Basaure P, Guardia-Escote L, Cabre M, Peris-Sampedro F, Sanchez-Santed F et al (2018) Postnatal chlorpyrifos exposure and apolipoprotein E (APOE) genotype differentially affect cholinergic expression and developmental parameters in transgenic mice. Food Chem Toxicol 118:42–52. https://doi.org/10.1016/j.fct.2018.04.065
Beitz JM (2014) Parkinson’s disease: a review. Front Biosci (Schol Ed) 6:65–74
Cecchini MP, Federico A, Zanini A, Mantovani E, Masala C et al. (2019) Olfaction and taste in Parkinson’s disease: the association with mild cognitive impairment and the single cognitive domain dysfunction. https://doi.org/10.1007/s00702-019-01996-z
Cheng WW, Zhu Q, Zhang HY (2019) Mineral nutrition and the risk of chronic diseases: a Mendelian randomization study. Nutrients 11. https://doi.org/10.3390/nu11020378
Choi SM, Kim BC, Jung HJ, Yoon GJ, Kang KW et al (2017) The association of musculoskeletal pain with bone mineral density in patients with Parkinson’s disease. Eur Neurol 77:123–129. https://doi.org/10.1159/000455009
Clevelan CB, Oliver GR, Chen B, Mattsson J (2001) Risk assessment under FQPA: case study with chlorpyrifos. Neurotoxicology 22:699–706
Cummings SR, Eastell R (2016) Risk and prevention of fracture in patients with major medical illnesses: a mini-review. J Bone Miner Res Off J Am Soc Bone Miner Res 31:2069–2072. https://doi.org/10.1002/jbmr.3030
Dam K, Seidler FJ, Slotkin TA (2000) Chlorpyrifos exposure during a critical neonatal period elicits gender-selective deficits in the development of coordination skills and locomotor activity. Brain Res Dev Brain Res 121:179–187
Deacon RM (2013) Measuring the strength of mice. J Vis Exp. https://doi.org/10.3791/2610
Dobson R, Yarnall A, Noyce AJ, Giovannoni G (2013) Bone health in chronic neurological diseases: a focus on multiple sclerosis and parkinsonian syndromes. Pract Neurol 13:70–79. https://doi.org/10.1136/practneurol-2012-000435
Eaton DL, Daroff RB, Autrup H, Bridges J, Buffler P et al (2008) Review of the toxicology of chlorpyrifos with an emphasis on human exposure and neurodevelopment. Crit Rev Toxicol 38(Suppl 2):1–125. https://doi.org/10.1080/10408440802272158
Ellman GL, Courtney KD, Andres V Jr, Feather-Stone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95
El-Sebae AH, Ahmed NS, Soliman SA (1978) Effect of pre-exposure on acute toxicity of organophosphorus insecticides to white mice. J Environ Sci Health B 13:11–24. https://doi.org/10.1080/03601237809372074
Engel SM, Wetmur J, Chen J, Zhu C, Barr DB et al (2011) Prenatal exposure to organophosphates, paraoxonase 1, and cognitive development in childhood. Environ Health Perspect 119:1182–1188. https://doi.org/10.1289/ehp.1003183
Fukuda T, Takeda S (2015) Frontiers in live bone imaging researches. Functional cross talk between bone and nervous system. Clin Calcium 25:891–898
Grandjean P, Landrigan PJ (2014) Neurobehavioural effects of developmental toxicity. Lancet Neurol 13:330–338. https://doi.org/10.1016/s1474-4422(13)70278-3
Gruntenko NE, Karpova EK, Alekseev AA, Chentsova NA, Saprykina ZV et al (2005) Effects of dopamine on juvenile hormone metabolism and fitness in Drosophila virilis. J Insect Physiol 51:959–968. https://doi.org/10.1016/j.jinsphys.2005.04.010
Huang S, Li Z, Liu Y, Gao D, Zhang X et al (2019) Neural regulation of bone remodeling: identifying novel neural molecules and pathways between brain and bone. J Cell Physiol 234:5466–5477. https://doi.org/10.1002/jcp.26502
Jackson-Lewis V, Przedborski S (2007) Protocol for the MPTP mouse model of Parkinson’s disease. Nat Protoc 2:141–151. https://doi.org/10.1038/nprot.2006.342
Jiang P (2019) Parkinson’s disease is associated with dysregulations of a dopamine-modulated gene network relevant to sleep and affective neurobehaviors in the striatum. Sci Rep 18:4808. https://doi.org/10.1038/s41598-019-41248-4
Kalia LV, Lang AE (2015) Parkinson’s disease. Lancet (London, England) 386:896–912. https://doi.org/10.1016/s0140-6736(14)61393-3
Khan K, Singh A, Mittal M, Sharan K, Singh N et al (2012) [6]-Gingerol induces bone loss in ovary intact adult mice and augments osteoclast function via the transient receptor potential vanilloid 1 channel. Mol Nutr Food Res 56:1860–1873. https://doi.org/10.1002/mnfr.201200200
Khan MP, Mishra JS, Sharan K, Yadav M, Singh AK et al (2013) A novel flavonoid C-glucoside from Ulmus wallichiana preserves bone mineral density, microarchitecture and biomechanical properties in the presence of glucocorticoid by promoting osteoblast survival: a comparative study with human parathyroid hormone. Phytomedicine 20:1256–1266. https://doi.org/10.1016/j.phymed.2013.07.007
Kim HH, Lim YW, Yang JY, Shin DC, Ham HS et al (2013) Health risk assessment of exposure to chlorpyrifos and dichlorvos in children at childcare facilities. Sci Total Environ 444:441–450. https://doi.org/10.1016/j.scitotenv.2012.11.102
Konradsen F (2007) Acute pesticide poisoning—a global public health problem. Dan Med Bull 54:58–59
Lewis KE, Sharan K, Takumi T, Yadav VK (2017) Skeletal site-specific changes in bone mass in a genetic mouse model for human 15q11-13 duplication seen in autism. Sci Rep 7:9902. https://doi.org/10.1038/s41598-017-09921-8
Lozowicka B (2015) Health risk for children and adults consuming apples with pesticide residue. Sci Total Environ 502:184–198. https://doi.org/10.1016/j.scitotenv.2014.09.026
Metta V, Sanchez TC, Padmakumar C (2017) Osteoporosis: a hidden nonmotor face of Parkinson’s disease. Int Rev Neurobiol 134:877–890. https://doi.org/10.1016/bs.irn.2017.05.034
Mojsak P, Lozowicka B, Kaczynski P (2018) Estimating acute and chronic exposure of children and adults to chlorpyrifos in fruit and vegetables based on the new, lower toxicology data. Ecotoxicol Environ Saf 159:182–189. https://doi.org/10.1016/j.ecoenv.2018.05.006
Opara J, Malecki A, Malecka E, Socha T (2017) Motor assessment in Parkinson’s disease. Ann Agric Environ Med 24:411–415. https://doi.org/10.5604/12321966.1232774
Presgraves SP, Ahmed T, Borwege S, Joyce JN (2004) Terminally differentiated SH-SY5Y cells provide a model system for studying neuroprotective effects of dopamine agonists. Neurotox Res 5:579–598
Rauh VA (2018) Polluting developing brains—EPA failure on chlorpyrifos. N Engl J Med 378:1171–1174. https://doi.org/10.1056/NEJMp1716809
Roman-Garcia P, Quiros-Gonzalez I, Mottram L, Lieben L, Sharan K et al (2014) Vitamin B(1)(2)-dependent taurine synthesis regulates growth and bone mass. J Clin Invest 124:2988–3002. https://doi.org/10.1172/jci72606
Sanberg PR, Bunsey MD, Giordano M, Norman AB (1988) The catalepsy test: its ups and downs. Behav Neurosci 102:748–759
Schneider JL, Fink HA, Ewing SK, Ensrud KE, Cummings SR (2008) The association of Parkinson’s disease with bone mineral density and fracture in older women. Osteoporos Int 19:1093–1097. https://doi.org/10.1007/s00198-008-0583-5
Schneider RB, Iourinets J, Richard IH (2017) Parkinson’s disease psychosis: presentation, diagnosis and management. Neurodegener Dis Manag 7:365–376. https://doi.org/10.2217/nmt-2017-0028
Sharan K, Yadav VK (2014) Hypothalamic control of bone metabolism. Best Pract Res Clin Endocrinol Metab 28:713–723. https://doi.org/10.1016/j.beem.2014.04.003
Sharan K, Mishra JS, Swarnkar G, Siddiqui JA, Khan K et al (2011) A novel quercetin analogue from a medicinal plant promotes peak bone mass achievement and bone healing after injury and exerts an anabolic effect on osteoporotic bone: the role of aryl hydrocarbon receptor as a mediator of osteogenic action. J Bone Miner Res 26:2096–2111. https://doi.org/10.1002/jbmr.434
Sharan K, Lewis K, Furukawa T, Yadav VK (2017) Regulation of bone mass through pineal-derived melatonin-MT2 pathway. J Pineal Res. https://doi.org/10.1111/jpi.12423
Siddiqui JA, Swarnkar G, Sharan K, Chakravarti B, Sharma G et al (2010) 8,8″-Biapigeninyl stimulates osteoblast functions and inhibits osteoclast and adipocyte functions: osteoprotective action of 8,8″-biapigeninyl in ovariectomized mice. Mol Cell Endocrinol 323:256–267. https://doi.org/10.1016/j.mce.2010.03.024
Silver MK, Shao J, Zhu B, Chen M, Xia Y et al (2017) Prenatal naled and chlorpyrifos exposure is associated with deficits in infant motor function in a cohort of Chinese infants. Environ Int 106:248–256. https://doi.org/10.1016/j.envint.2017.05.015
Sleeman I, Che ZC, Counsell C (2016) Risk of fracture amongst patients with Parkinson’s disease and other forms of parkinsonism. Parkinsonism Relat Disord 29:60–65. https://doi.org/10.1016/j.parkreldis.2016.05.026
Swarnkar G, Sharan K, Siddiqui JA, Chakravarti B, Rawat P et al (2011) A novel flavonoid isolated from the steam-bark of Ulmus wallichiana planchon stimulates osteoblast function and inhibits osteoclast and adipocyte differentiation. Eur J Pharmacol 658:65–73. https://doi.org/10.1016/j.ejphar.2011.02.032
Titova N, Martinez-Martin P, Katunina E, Chaudhuri KR (2017a) Advanced Parkinson’s or “complex phase” Parkinson’s disease? Re-evaluation is needed. J Neural Transm (Vienna) 124:1529–1537. https://doi.org/10.1007/s00702-017-1799-3
Titova N, Padmakumar C, Lewis SJG, Chaudhuri KR (2017b) Parkinson’s: a syndrome rather than a disease? J Neural Transm (Vienna) 124:907–914. https://doi.org/10.1007/s00702-016-1667-6
Trasande L (2017) When enough data are not enough to enact policy: the failure to ban chlorpyrifos. PLoS Biol 15:e2003671. https://doi.org/10.1371/journal.pbio.2003671
Venerosi A, Tait S, Stecca L, Chiarotti F, De Felice A et al (2015) Effects of maternal chlorpyrifos diet on social investigation and brain neuroendocrine markers in the offspring—a mouse study. Environ Health 14:32. https://doi.org/10.1186/s12940-015-0019-6
Watts NB (2014) Insights from the global longitudinal study of osteoporosis in women (GLOW). Nat Rev Endocrinol 10:412–422. https://doi.org/10.1038/nrendo.2014.55
Acknowledgments
This study was supported by CSIR-Central Food Technological Research Institute, Mysore, India, and SERB N-PDF grant. Funding from the Science and Engineering Research Board (SERB), Government of India (K.S.), and the Department of Biotechnology, Government of India (K.S.), is acknowledged. Research fellowship grants from the SERB (S.J.A.), Department of Science and Technology (G.E.), and Council of Scientific and Industrial Research (K.P), Government of India, are also acknowledged. We thank Dr. Naibedya Chattopadhyay (CSIR-Central Drug Research Institute, Lucknow, India) for the μ-CT facility.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Supplementary table 1
(DOCX 12 kb)
Rights and permissions
About this article
Cite this article
Ali, S.J., Ellur, G., Patel, K. et al. Chlorpyrifos Exposure Induces Parkinsonian Symptoms and Associated Bone Loss in Adult Swiss Albino Mice. Neurotox Res 36, 700–711 (2019). https://doi.org/10.1007/s12640-019-00092-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-019-00092-0