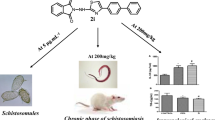
Abstract
The present work aimed to study the effect of phenyl vinyl sulphone (PVS), a CPI, on different stages of Schistosoma (S.) mansoni in an in vitro culture study and in experimentally infected mice, compared to PZQ. As regards the in vitro study, different concentrations of PVS (1, 2, 4, 6, 8 and 10 µg/ml) and PZQ (1 µg/ml) were assessed by % worm mortality for schistosomula and adults, and hemoglobin degradation by schistosomula. In vivo study included 8 groups of mice. Intraperitoneal PVS, subgroup (a), and oral PZQ, subgroup (b), were assessed at different durations post infection (pi); at 1, 3, 5 and 7 weeks pi (groups I, II, III and IV, respectively). Infection, PVS, PZQ, and normal control groups (groups V–VIII) were included. The anti-schistosomal effects of PVS were assessed by parasitological, histopathological and haematological parameters. In in vitro study, PVS had a schistosomicidal effect in a concentration and time dependent manner, PVS showed 100% schistosomula mortality at day 2 and 92% adult worm mortality at day 5. Furthermore, PVS decreased hemoglobin degradation by schistosomula. In in vivo study, PVS showed a decrease in total worm burden and tissue egg load in intestine and liver with an increase in number of dead ova in intestine of mice. Furthermore, PVS resulted in a decrease in number, size and cellularity of hepatic granulomas and an increase in hemoglobin concentration.PVS was better than PZQ in reducing each of tissue egg count in intestine at 5 and 7 weeks pi, and hepatic granuloma size at 3, 5 and 7 weeks pi. These results suggest that PVS can be a promising chemotherapeutic agent in Schistosoma mansoni infection.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Schistosomiasis is a common disease in endemic areas of Sub-Saharan Africa, South America and Asia (Hinz et al. 2017). Praziquantel (PZQ) is the only drug of choice for treatment of human schistosomiasis due to its high efficacy, simple administration and competitive cost (Wang et al. 2012). Although PZQ is highly effective in killing adult worms, yet it has a poor efficacy against larvae and immature worms (Caffrey 2007, Danso-Appiah et al. 2013). Moreover, there have been stronger evidences of PZQ-resistance by Schistosoma (S.) mansoni (Pinto-Almeida et al. 2016).
The gut of schistosomula and adult worms of S. mansoni synthesize and secrete different cathepsins belonging to the cysteine protease family, that are involved in host hemoglobin degradation; a major source for the schistosome metabolism, growth, and in parasite development and reproduction (Correnti et al. 2005). These enzymes may function in membrane biogenesis or in intracellular protein turnover, besides their roles in the hemoglobin degradation (Dalton et al. 1995). Phenyl vinyl sulfone (PVS) was chosen as an example of the vinyl sulfones to study its effect on S. mansoni using in vitro culture technique, which was not studied before, as PVS effect on in vitro culture was studies with Fasciola only (Helmy et al. 2008). As well as in vivo parameters through different durations to assess its effect on both acute and chronic schistosomiasis.
The aim of the current work is to study the effect of a cysteine protease inhibitor; Phenyl vinyl sulfone (PVS) on different stages of Schistosoma mansoni in an in vitro culture study, and on the parasite subjected to PVS at different durations of Schistosoma mansoni in experimentally-infected mice, in comparison to the conventionally employed drug; praziquantel (PZQ).
Materials and methods
Parasite
Schistosoma mansoni cercariae were obtained from laboratory-bred infected Biomphalaria alexandrina snails in Schistosome Biological Supply Program (SBSP),Theodor Bilharz research institute (TBRI), Giza, Egypt.
Drugs
Phenyl vinyl sulphone (PVS) (Sigma, USA) and Praziquantel (PZQ) (EPICO, Egypt).
In vitro study
Ten Schistosoma mansoni schistosomula and 6 adult worms (3 males and 3 females) were maintained, separately, in RPMI-1640 medium (Neves et al. 2011). One mg of each of PVS and PZQ was dissolved in Dimethyl sulphoxide (DMSO)then diluted in 1 ml medium to make a working solution (Botros et al. 2009). Different concentrations of PVS (1, 2, 4, 6, 8 and 10 µg/ml) were applied to the wells in duplicates. They were assessed by dose response studies in the form of % worm mortality for schistosomula and adults (schistosomula and adult worms showing no signs of motility for 1 min, were considered dead), which were monitored every 24 h (hrs) for 96 h under inverted microscope ×30 (Abdulla et al. 2009; Botros et al. 2009)and growth inhibition studies in the form of hemoglobin degradation (in the form of black gut) by schistosomula, where the schistosomula were monitored every 24 h for an 8-day period (Wasilewski et al. 1996). Positive and negative controls (PZQ at 1 μg/ml and DMSO, respectively) were also studied (Neves et al. 2011).
In vivo study
The study was performed on 72 female Swiss albino mice, 6–8 weeks old, weighing 20–25 g, obtained and housed, fulfilling the animal ethical considerations at the SBSP, Theodor Bilharz research institute, Giza, Egypt. Mice were classified into the following groups:
- Group I:
-
[Infected mice treated 1 week post-infection (pi)] included 12 mice equally divided into: subgroup (a): Infected-PVS treated mice, and subgroup (b): Infected-PZQ treated mice.
- Group II:
-
(Infected -mice treated 3 weeks pi) included 12 mice equally divided into: subgroup (a): Infected-PVS treated mice, and subgroup (b): Infected-PZQ treated mice.
- Group III:
-
(Infected mice treated 5 weeks pi) included 12 mice equally divided into: sub group (a): Infected-PVS treated mice, and subgroup (b): Infected-PZQ treated mice.
- Group IV:
-
(Infected mice treated 7 weeks pi)included 12 mice equally divided into: subgroup (a): Infected-PVS treated mice, and subgroup (b): Infected-PZQ treated mice.
- Group V:
-
(Infection control):6 infected non-treated mice.
- Group VI:
-
(PVS control):6 non-infected PVS treated mice.
- Group VII:
-
(PZQ control):6 non-infected PZQ treated mice.
- Group VIII:
-
(Normal control): 6 non-infected non-treated apparently healthy mice.
Mice were infected by 60 ± 10 S. mansoni cercariae by the subcutaneous injection route (Peters and Warren 1969) at SBSP.
Each drug was dissolved in its suitable solvent; PVS powder was freshly prepared by being dissolved in sterile distilled water at a concentration of 5 mg/ml and heated at 60 °C till completely dissolved. A 600 mg tablet of PZQ was ground and suspended in 2% Cremophor El. Then each drug was administered through its suitable route according to its pharmacokinetics; the administered dose of PVS was 50 mg/kg/mouse, once daily by intraperitoneal injection for 7 consecutive days (Abdulla et al. 2007). While, PZQ was freshly prepared before oral administration at a dose of 500 mg/kg/mouse, once daily for two consecutive days using nasogastric feeding tube (Keiser et al. 2006; Abaza et al. 2013). All mice were sacrificed at 10 weeks pi.
The anti-schistosomal effects of PVS were assessed by:
-
1.
Parasitological parameters Small intestine and liver from each mouse were examined for total worm burden by sucking the worms from their tissues (Smithers and Terry 1965),tissue egg load in small intestine and liver (Cheever 1968) and oogram pattern (Pellegrino and Faria 1965) where ova were classified according to their stages of development into: mature (fully developed miracidia), immature (small embryos occupying 1/3, 1/2, 2/3 or the whole space of the egg shell)or dead (eggs appear semitransparent, granular and dark showing a clear longitudinal half).
-
2.
Histopathological parameters Part of liver was embedded in paraffin blocks to prepare slides from sections of 5 µm thickness followed by staining with Hematoxylin and Eosin (Hx and E) stain for detection of egg-induced granulomatous cellular reaction (Druray and Wallington 1980).Microscopic examination of previously prepared liver sections stained with Hx and E stain to determine the granuloma number and size, according to the following criteria (Jacobs et al.1997):
-
1.
Single egg granulomas were selected for diameter measurements.
-
2.
The greatest diameter and its perpendicular diameter were measured, by using a trinocular microscope with mounted camera, and the mean of both diameters was considered granuloma diameter.
-
3.
The mean size of granulomas for each group was calculated.
-
4.
Mean number of granulomas/low power field (×10 objective) was calculated in each group.
-
1.
-
3.
Haematological parameter Detection of hemoglobin concentration for each mouse followed by the detection of mean hemoglobin concentration for each group (Feldmann et al. 2000).
-
4.
Statistical study The collected data were introduced to a personal computer using Statistical Package for Social Science (SPSS 18.0.1 for Windows; SPSS Inc, Chicago, IL). Descriptive statistics included: mean, standard deviation (±SD) for parametric numerical data, percentage of non-numerical data. Analytical statistics was done using the Mann–Whitney Test (U test) to assess the statistical significance of the difference of a variable between two study groups. p value less than or equal 0.05 was considered significant.IC50 (Half maximal inhibitory concentration) was also calculated (Sebaugh 2011).
Results
In vitro study
Dose–response studies
10 Schistosoma mansoni schistosomula and 6 adult worms (3 males and 3 females) were maintained were subjected to different concentrations of PVS (1, 2, 4, 6, 8 and 10 µg/ml) then all schistosomula and adult worms in all wells were counted (Neves et al. 2011). The 10 µg/ml dose of PVS resulted in 90% schistosomula mortality by day 1 which was higher than that by PZQ (80%), reaching 100% mortality rate by day 2, the IC50 of PVS was 7.5 µg/ml at day 1, 4 µg/ml at day 2 and 2.5 µg/ml at day 3 (Fig. 1; Table 1). Moreover, PVS resulted in 92% adult worms mortality by day 5 reaching 100% by day 6, the IC50 of PVS was 6 µg/ml at day 4 and 3.9 µg/ml at day 5 (Fig. 2; Table 2).
Growth inhibition studies
In the current study, the effect of different concentrations of PVS on S. mansoni schistosomula hemoglobin degradation in vitro was studied. Results showed concentration dependent arrest of hemoglobin degradation in schistosomula treated with PVS as determined by lack of black pigmentation of the gut (Fig. 3).
In vivo study
Parasitological parameters
Total worm burden As regards the in vivo study in the present work, PVS decreased total worm burden when given at 1, 3, 5 and 7 wks pi with non-significant difference (p > 0.05) when compared to infection control. A significant decrease (p ≤ 0.05) in total worm burden was recorded in mice treated with PZQ at 5 and 7 wks pi compared to infection control (Fig. 4).
Tissue egg count/gm intestine and liver In the current work, PVS given at 1, 3, 5 and 7 wks pi showed significant decrease (p ≤ 0.05) in count of eggs/gm intestine and liver when compared to infection control (Figs. 5 and 6).
Bar chart illustrating mean egg count/gm intestine among different study groups. A decrease in mean egg count/gm intestine in subgroup a of groups I, II, III and IV compared to group V was recorded. Subgroup a in each of group III and IV showed decrease in mean egg count than subgroup b of same groups
Oogram pattern In the present study, the effect of PVS on the oogram pattern of S. mansoni eggs was assessed. At 1, 3 and 5 wks pi, PVS showed an oogram pattern similar to that of infection control. Moreover, at 7 wks pi, PVS showed a significant increase (p ≤ 0.05) in mean number of dead ova compared to infection control (Table 3).
Histopathological parameter
In the current study giving each of PVS and PZQ at early durations of infection (at 1 and 3 wks pi) resulted in a nearly similar histopathological picture as infection control (Figs. 7, 8 and 9). There was cloudy swelling in liver cells with cellular granuloma made up of histiocytes, lymphocytes and scattered fibroblasts with bilharzial pigment in Von-Kupffer cells. Also, at 1 and 3 wks pi there was a recorded significant decrease (p ≤ 0.05) in mean hepatic granuloma size when using each of PVS and PZQ compared to infection control, with PVS resulting in smaller granuloma size than PZQ when each was given at 3 wks pi (p ≤ 0.05). Moreover, there was a recorded significant decrease (p ≤ 0.05) in mean hepatic granuloma number at 3 wks pi using each of PVS and PZQ compared to infection control (Tables 4 and 5).
Hepatic granuloma in groups of study. a Subgroup IIa showing a single dead partially calcified S. mansoni ova in the center of a well circumscribed granuloma made up of fibroblasts with lymphocytes mainly seen at the periphery. Perigranulomatous tissue shows expansion of portal tracts by lymphocytic infiltrate along with bile duct injury and focal interface hepatocytes (Hx and E stain ×400). b Subgroup IIb showing a single dead partially calcified S. mansoni ova in the center of a partially circumscribed granuloma made up of histiocytes and fibroblasts with intervening collagen, lymphocytes mainly seen at the periphery (Hx and E stain ×400). c Infection control group (V) showing granuloma made up of histiocytes and few fibroblasts with lymphocytes mainly the periphery, a viable S. mansoni ova is seen, perigranulomatous tissue shows lymphocytic infiltrate along with bilharzial pigment in Von-Kupffer cells (Hx and E stain ×400). d Normal control group (VI) (Hx and E stain ×400)
-
1.
Histopathological examination of liver tissue (Fig. 7):
-
2.
Hepatic granuloma number and size.
-
a.
Hepatic granuloma number.
-
b.
Hepatic granuloma size.
-
a.
Hematological parameter
In the present work, the study of the effect of PVS on hemoglobin concentration in S. mansoni infected mice revealed that PVS increased mean hemoglobin concentration when given at different durations of infection resulting in a significant (p ≤ 0.05) increase in mean hemoglobin concentration when given at each of1and 5wk pi compared to infection control.PVS given at different durations post infection showed non-significant difference (p > 0.05) in mean hemoglobin concentration when compared to PZQ treated- mice (Table 6).
Discussion
The present work was done to study the effect of different concentrations of PVS, a CPI, on S. mansoni schistosomula and adults in an in vitro study and on the parasite subjected to PVS at different durations of experimental S. mansoni infection.
Currently, as regards in vitro study, the S. mansoni schistosomula and adult worms were subjected to different concentrations of PVS (1, 2, 4, 6, 8 and 10 µg/ml) (Neves et al. 2011). The 10 µg/ml dose of PVS resulted in 90% schistosomula mortality by day 1 which was higher than that by PZQ (80%), reaching 100% mortality rate by day 2, the half maximal inhibitory concentration (IC50) of PVS was 7.5 µg/ml at day 1,4 µg/ml at day 2 and 2.5 µg/ml at day 3 (Fig. 1; Table 1). Moreover, PVS resulted in 92% adult worms mortality by day 5 reaching 100% by day 6, the IC50 of PVS was 6 µg/ml at day 4 and 3.9 µg/ml at day 5 (Fig. 2; Table 2). This goes well with the results of Fahmy and Helmy (2007) who reported that 300 ppm PVS caused immediate death of S. mansoni adults. Similarly, Farid et al. (2013b) studied the effect of other CPIs on the viability of S. mansoni adults and their results showed that death started 1 h after exposure to 500 ppm of FMK, VS and sodium-nitro-prussid with percent worm mortality 75, 70 and 60%, respectively and reaching 100% mortality after 3 h. The death of PZQ treated worms started from day 2 with 100% worm mortality. This goes well with previous studies which reported that PZQ exerts schistosomicidal activity in vitro on schistosomula and adults of S. mansoni (Cioliand Pica-Mattoccia 2002; Neves et al. 2011).
In the current study, the effect of different concentrations of PVS on S. mansoni schistosomula hemoglobin degradation in vitro was studied. Results showed concentration dependent arrest of hemoglobin degradation in schistosomula treated with PVS as determined by lack of black pigmentation of the gut (Fig. 3). These results were in context with Wasilewski et al. (1996) who revealed that CPIs produced a dramatic arrest of hemoglobin degradation in vitro by schistosomula suggesting that the death of schistosomula in their study, was due to inhibition of CPs and not due to toxic effect of the inhibitor. Also, Shenai et al. (2003) tested the effect of VSs on Plasmodium falciparum and showed that inhibition of parasite development was accompanied by the appearance of darkly stained, swollen food vacuoles, which are indicative of a block in hemoglobin hydrolysis and blocking of food vacuole hemoglobinases.
As regards the in vivo study in the present work, a significant decrease (p ≤ 0.05) in total worm burden was recorded in mice treated with PZQ at 5 and 7 wks pi compared to infection control (Fig. 4) which goes well with the known potency of PZQ against schistosome adult worms (Xiao et al. 1985, Issa 2007). PVS given at 1 and 3 wks pi showed a non-significant decrease (p > 0.05) in total number of worms when compared to PZQ treated-mice at the same durations, which is on the contrary to the results of Farid et al. (2013b) using other CPIs at 3 wks pi. However, at 5 and 7 weeks pi the PZQ treated-mice showed significant decrease (p ≤ 0.05) in total number of worms compared to PVS given at the same durations. These results are similar to another study where PZQ and PVS were given to mice at 6 wks pi (Abaza et al. 2013).
In the current work, PVS given at 1, 3, 5 and 7 wks pi showed significant decrease (p ≤ 0.05) in count of eggs/gm intestine and liver when compared to infection control (Figs. 5 and 6). In this context, Abaza et al. (2013) also found that PVS treated mice showed decrease in mean tissue egg load/gm liver and intestine in comparison to infected non treated control mice in their study.
In the present study, the effect of PVS on the oogram pattern of S. mansoni eggs was assessed (Table 3). At 1, 3 and 5 wks pi, PVS showed an oogram pattern similar to that of infection control. To the contrary of Farid et al. (2013a) who by studying 3 CPIs; VS, FMK and sodium nitroprussid given at 3 wks and 5 wks pi reported an increase in mean number of dead ova in comparison to infected non treated mice, in their study. Moreover, at 7 wks pi, PVS showed a significant increase (p ≤ 0.05) in mean number of dead ova compared to infection control. These results were in context with Abaza et al. (2013) who revealed that PVS given at 6 wks pi showed a decrease in mean number of immature ova and increase in mean number of dead ova in comparison to infected non treated group in their study.
In the current study giving each of PVS and PZQ at early durations of infection (at 1 and 3 wks pi) resulted in a nearly similar histopathological picture as infection control (Figs. 7, 8 and 9) (Table 4 and 5). There was cloudy swelling in liver cells with cellular granuloma made up of histiocytes, lymphocytes and scattered fibroblasts with bilharzial pigment in Von-Kupffer cells. Also, at 1 and 3 wks pi there was a recorded significant decrease (p ≤ 0.05) in mean hepatic granuloma size when using each of PVS and PZQ compared to infection control, with PVS resulting in smaller granuloma size than PZQ when each was given at 3 wks pi (p ≤ 0.05). Moreover, there was a recorded significant decrease (p ≤ 0.05) in mean hepatic granuloma number at 3 wks pi using each of PVS and PZQ compared to infection control (Tables 4 and 5). The above mentioned findings go well with the results of Farid et al. (2013a) who found that VS, a CPI, given to mice at 3 wks pi, showed similar histopathological picture with a decrease in hepatic granuloma size and number compared to infection control in their study. They deduced that their detected VS effect can be attributed to a mild suppression of the host immune system. The results of histopathological examination using PZQ in the present study goes well with those of El Beshbishi et al. (2013) when using PZQ 3 and6 wks pi. The decreased granuloma size in PZQ treated mice, in the present study, can be attributed to the fact that hepatic granulomas shrink progressively after using a curable drug (Morsy 2009).
In the present work, the study of the effect of PVS on hemoglobin concentration in S. mansoni infected mice revealed that PVS increased mean hemoglobin concentration when given at different durations of infection resulting in a significant (p ≤ 0.05) increase in mean hemoglobin concentration when given at each of1and 5wk pi compared to infection control (Table 6). The recorded increase in hemoglobin concentration induced by PVS probably reflects the inhibitory effect of PVS on the S. mansoni CPs. Vermiere et al. (2012) studied the effect of K11777, a CPI, against Ancylostoma caninum infected hamsters and reported that it reversed a severe decrease in blood hemoglobin levels. In the present work, PZQ induced a significant increase (p ≤ 0.05) in mean hemoglobin concentration when given at different durations post infection compared to infection control, which goes well with Tohon et al. (2008) who reported that the use of PZQ was followed by a significant reduction of anemia. Meanwhile, PVS given at different durations post infection showed non-significant difference (p > 0.05) in mean hemoglobin concentration when compared to PZQ treated- mice.
It is concluded from the present study that PVS has a schistosomicidal effect in a concentration and time dependent manner with a possible anti-immonopathological role during Schistosoma mansoni infection. PVS was effective in killing S. mansoni schistosomula and adult worms in in vitro study. Moreover, PVS decreased hemoglobin degradation by schistosomula. As regards in vivo study, PVS showed a decrease in total worm burden and tissue egg load in intestine and liver with an increase in number of dead ova in intestine and resulted in a decrease in number, size and cellularity of hepatic granulomas. Furthermore, PVS was better than PZQ in reducing each of tissue egg count in intestine, and hepatic granuloma size.
References
Abaza SA, El-Moamly AA, Ismail OA, Alabbassy MM (2013) Cysteine protease inhibitors (Phenyl vinyl sulfone and valproic acid) in treatment of schistosomiasis mansoni-infected mice: an experimental study to evaluate their role in comparison to praziquantel. PUJ 6(1):99–108
Abdulla M, Lim KC, Sajid M, McKerrow JH, Caffrey CR (2007) Schistosomiasis mansoni: novel chemotherapy using a cysteine protease inhibitor. PLoS Med 4(1):130–138
Abdulla MH, Ruelas DS, Wolff B, Snedecor J, Lim KC, Fengyun X, Adam RR, Janice W, McKerrow JH, Caffrey CR (2009) Drug discovery for schistosomiasis: hit and lead compounds identified in a library of known drugs by medium-through put phenotypic screening. PLoS Negl Trop Dis 3(7):e478
Botros S, William S, Beadle LR, Valiaeva N, Hostetler KY (2009) Anti-schistosomal activity of hexadecyloxypropyl cyclic 9-(S)-[3-Hydroxy-2-(phosphonomethoxy) propyl] adenine and other alkoxyalkyl esters of acyclic nucleoside phosphonates assessed by schistosome worm killing in vitro. Antimicrob A Chemother 53(12):5284–5287
Caffrey CR (2007) Chemotherapy of schistosomiasis: present and future. Curr Opin Chem Biol 11:433–439
Cheever AW (1968) Conditions affecting the accuracy of potassium hydroxide digestion techniques for counting Schistosoma mansoni eggs in tissues. Bull WHO 39:328–331
Cioli D, Pica-Mattoccia L (2002) Praziquantel. Parasitol Res 90(1):53–59
Correnti JM, Brindley PJ, Pearce EJ (2005) Long-term suppression of cathepsin B levels by RNA interference retards schistosome growth. Mol Biochem Parasitol 143(2):209–215
Dalton JP, Smith AM, Clough KA, Brindley PJ (1995) Digestion of haemoglobin by schistosomes: 35 years on. Parasitol Today 11:299–303
Danso-Appiah A, Olliaro PL, Donegan S (2013) Drugs for treating Schistosoma mansoni infection. Coranche Database Syst Rev 2:CD000528
Druray RA, Wallington EA (1980) Carletonʼs histological technique, 5th edn. Oxford University Press, Oxford, New York, pp 195–210
El Beshbishi SN, Taman A, El Malky M, Azab MS, El Hawary AK, El Tanatwy DA (2013) First insight into the effect of single oral dose therapy with artemisinin–naphthoquine phosphate combination in a mouse model of Schistosoma mansoni infection. Int J Parasitol 43:521–530
Fahmy ZH, Helmy AM (2007) Assessment of cysteine protease in schistosomiasis and haematolgical parasitogical role of its inhibitor in the control of the disease. New Egypt J Med 39:1–7
Farid A, Ismail A, Rabee I, Zalat R, El Amir A (2013a) Study of the efficacy of cysteine protease inhibitors alone or combined with praziquantel as chemotherapy for mice schistosomiasis mansoni. Int J Med Health Biomed Bioeng Pharm Eng 7(12):849–856
Farid A, Malek AA, Rabie I, Helmy A, El Amir AM (2013b) Overview on cysteine protease inhibitors as chemotherapy for Schistosomiasis mansoni in mice and also its effect on the parasitological and immunological profile. Pak J Biol Sci 16(24):1849–1861
Feldman BF, Zinkl JG, Jain NC (2000) Hematology of the mouse (Mus musculus). In: Lippincott W, Wilkins (eds) Schalm’s veterinary hematology, 5th edn. Philadelphia, London, pp 1210–1224
Helmy MM, Fahmy ZH, Sabry HY (2008) Fasciola gigantica: evaluation of the effect of phenyl vinyl sulfone in vitro. Exp Parasitol 119(1):125–134
Hinz R, Schwarz NG, Hahn A, Frickmann H (2017) Serological approaches for the diagnosis of schistosomiasis—a review. Mol Cell Probes 31:2–21. doi:10.1016/j.mcp.2016.12.003
Issa RM (2007) Schistosoma mansoni: the prophylactic and curative effects of propolis in experimentally-infected mice. Rawal Med J 32:94–98
Jacobs W, Bogers J, Deelder A, Wery M, Van Marck E (1997) Adult Schistosoma mansoni worms positively modulate soluble egg antigen-induced inflammatory hepatic granuloma formation in vivo. Stereological analysis and immunophenotyping of extracellular matrix proteins, adhesion molecules, and chemokines. Am J Pathol 150:2033–2045
Keiser J, El Ela NA, El Koumy E, El Lakany N, Diab T, Chollet J, Utzinger J, Barakat R (2006) Triclabendazole and its two main metabolites lack activity against Schistosoma mansoni in mouse model. Am J Trop Med Hyg 75(2):287–291
Morsy GH (2009) Parasitological and histopathological studies on schistosomiasis mansoni infected mice and treated with praziquantel and/or oltipraz. J Egypt Soc Parasitol 39(2):687–701
Neves JKAL, Sarinho S, De Melo CML, Pereira VRA, De Lima MCA, Pitta IR, Albuquerque MCPA, Galdino SL (2011) Immunological studies and in vitro schistosomicide action of new imidazolidine derivatives. J Ven Anim Toxicol Trop Dis 17(3):277–286
Pellegrino J, Faria J (1965) The oogram method for the screening of drugs in schistosomiasis mansoni. Am J Trop Med Hyg 14:363–369
Peters PA, Warren KS (1969) A rapid method of infecting mice and other laboratory animals with Schistosoma mansoni: subcutaneous injection. J Parasitol 55(3):558
Pinto-Almeida A, Mendes T, de Oliveira RN, CorrêaSde A, Allegretti SM, Belo S, Tomás A, Anibal Fde F, Carrilho E, Afonso A (2016) Morphological characteristics of Schistosoma mansoni PZQ-resistant and -susceptible strains are different in presence of Praziquantel. Front Microbiol 26(7):594
Sebaugh JL (2011) Guidelines for accurate EC50/IC50 estimation. Pharm Stat 10(2):128–134
Shenai BR, Lee BJ, Alvarez-Hernandez A, Chong PY, Emal CD, Neitz RJ, Roush WR, Rosenthal PJ (2003) Structure activity relationships for inhibition of cysteine protease activity and development of Plasmodium falciparum by Peptidyl Vinyl Sulfones. Antimicrob Agents Chemother 47(1):154–160
Smithers SR, Terry RJ (1965) Infection of laboratory hosts with cercariae of Schistosoma mansoni and the recovery of adult worms. Parasitology 55:695–700
Tohon ZB, Mainassara HB, Garba A, Mahamane AE, Bosqué-Oliva E, Ibrahim M, Duchemin J, Chanteau S, Boisie P (2008) Controlling schistosomiasis: significant decrease of anaemia prevalence one year after a single dose of praziquantel in Nigerien schoolchildren. PLoS Negl Trop Dis 2(5):e241
Vermeire JJ, Lantz LD, Caffrey CR (2012) Cure of hookworm infection with a cysteine protease inhibitor. PLoS Negl Trop Dis 6(7):e1680
Wang W, Wang L, Liang YS (2012) Susceptibility or resistance of praziquantel in human schistosomiasis: a review. Parasitol Res 111(5):1871–1877
Wasilewski MM, Limb KC, Jennifer P, McKerrow JH (1996) Cysteine protease inhibitors block schistosome hemoglobin degradation in vitro and decrease worm burden and egg production in vivo. Mol Biochem Parasitol 81:179–189
Xiao SH, Catto BA, Webster LT Jr (1985) Effects of praziquantel on different developmental stages of Schistosoma mansoni in vitro and in vivo. J Infect Dis 151(6):1130–1137
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
We wish to confirm that there are no known conflicts of interest associated with this publication and there has been no significant financial support for this work that could have influenced its outcome. We confirm that the manuscript has been read and approved by all named authors and that there are no other persons who satisfied the criteria for authorship but are not listed. We further confirm that the order of authors listed in the manuscript has been approved by all of us.
Ethical approval
The study was approved by the Research Ethics Committee, Faculty of Medicine, Ain Shams University. All the animal experiments were performed according to the rules and regulation of the Animal Ethics rules, Ain-Shams University, Cairo, Egypt.
Rights and permissions
About this article
Cite this article
Mahmoud, M.S.ED., Ibrahim, A.N., Badawy, A.F. et al. Effect of phenyl vinyl sulphone cysteine protease inhibitor on Schistosoma mansoni: in vitro and in vivo experimental studies. J Parasit Dis 41, 1049–1058 (2017). https://doi.org/10.1007/s12639-017-0933-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12639-017-0933-3