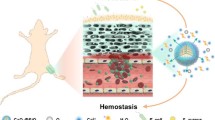
Abstract
Hemostatic materials with antibacterial properties become increasingly significant in military and civilian trauma care. In recent years, some advanced inorganic hemostatic materials (e.g. QuikClot, Combat Gauze, and WoundStat) have been reported to have their own drawbacks and side effects. In the study, the mesoporous silica nanoparticles (MSN) doped with calcium and gallium (Ga0.5CaMSS) was explored for hemorrhage control together with good antibacterial properties. Transmission electron microscopy (TEM), the energy-dispersive X-ray spectrometer (EDS), Nitrogen gas adsorption-desorption measurements, X-Ray diffraction measurements (XRD) and the Fourier transform infrared (FTIR) spectra were used to characterize the coagulation-promoting surface chemistry, morphology and porous structure of the samples. The clotting blood tests (CBT), activated partial thromboplastin time (APTT) tests, prothrombin time (PT) tests, and thromboelastograph (TEG) analysis showed the much better hemostatic properties of the Ga0.5CaMSN compared with the control and blank MSN. Besides, Ga0.5CaMSN was found to have obvious antibacterial effect against both S. aureus and E. coli. Furthermore, the perfect biocompatibility of all samples was proved by hemolysis assay study and cytotoxicity test. Associated with the blood coagulation tests and antibacterial tests, it could be inferred that the stimulation of the blood coagulation and antibacterial activity of Ga0.5CaMSN were attributed to the incorporation of calcium ions and gallium ions. The Ga0.5CaMSN developed here can be a potent candidate for emergency treatment to control hemorrhage and wound infection pre-hospital.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Gordy SD, Rhee P, Schreiber MA (2011) Military applications of novel hemostatic devices[J]. Expert review of medical devices 8(1):41–47
Wei S, Lixin S, Jiewei L et al (2014) Comparison of the drug for infection prevention in combat injuries between the PLA and USA armed force[J]. Journal of Pharmaceutical Practice 01:75–77
Pfeifer R, Tarkin IS, Rocos B, Pape HC (2009) Patterns of mortality and causes of death in polytrauma patients—has anything changed?[J]. Injury 40(9):907–911
Kheirabadi BS, Scherer MR, Estep JS, Dubick MA, Holcomb JB (2009) Determination of efficacy of new hemostatic dressings in a model of extremity arterial hemorrhage in swine[J]. J Trauma Acute Care Surg 67(3):450–460
Kheirabadi BS, Mace JE, Terrazas IB, Fedyk CG, Estep JS, Dubick MA, Blackbourne LH (2010) Safety evaluation of new hemostatic agents, smectite granules, and kaolin-coated gauze in a vascular injury wound model in swine[J]. J Trauma Acute Care Surg 68(2):269–278
Kheirabadi BS, Edens JW, Terrazas IB, Estep JS, Klemcke HG, Dubick MA, Holcomb JB (2009) Comparison of new hemostatic granules/powders with currently deployed hemostatic products in a lethal model of extremity arterial hemorrhage in swine[J]. J Trauma Acute Care Surg 66(2):316–328
Littlejohn L, Bennett BL, Drew B (2015) Application of current hemorrhage control techniques for backcountry care: part two, hemostatic dressings and other adjuncts[J]. Wilderness & environmental medicine 26(2):246–254
Rhee P, Brown C, Martin M, et al. QuikClot use in trauma for hemorrhage control: case series of 103 documented uses[J]. Journal of Trauma and Acute Care Surgery, 2008, 64(4): 1093-1099
McManus J, Hurtado T, Pusateri A et al (2007) A case series describing thermal injury resulting from zeolite use for hemorrhage control in combat operations[J]. Prehospital Emergency Care 11(1):67–71
Plurad D, Chandrasoma S, Best C, Rhee P (2009) A complication of intracorporeal use of QuikClot for pelvic hemorrhage[J]. J Trauma Acute Care Surg 66(5):1482–1484
Wright JK, Kalns J, Wolf EA, Traweek F, Schwarz S, Loeffler CAK, Snyder W, Yantis Jr LD, Eggers J (2004) Thermal injury resulting from application of a granular mineral hemostatic agent[J]. J Trauma Acute Care Surg 57(2):224–230
Bennett BL, Littlejohn L Review of new topical hemostatic dressings for combat casualty care[J]. Military Medicine 179(5):497–514
Li F, Tian F, Liu CJ et al (2013) Advance in emergengcy hemostatic materials[J]. Materials Review 27(3):70–73
Clay JG, Grayson JK, Zierold D (2010) Comparative testing of new hemostatic agents in a swine model of extremity arterial and venous hemorrhage[J]. Mil Med 175(4):280–284
Littlejohn LF, Devlin JJ, Kircher SS, Lueken R, Melia MR, Johnson AS (2011) Comparison of Celox-a, ChitoFlex, WoundStat, and combat gauze hemostatic agents versus standard gauze dressing in control of hemorrhage in a swine model of penetrating trauma[J]. Acad Emerg Med 18(4):340–350
Wang C, Zhou H, Niu H, Ma X, Yuan Y, Hong H, Liu C (2018) Tannic acid-loaded mesoporous silica for rapid hemostasis and antibacterial activity[J]. Biomaterials science 6(12):3318–3331
Nie W, Dai X, Li D, McCoul D, Gillispie GJ, Zhang Y, Yu B, He C (2018) One-pot synthesis of silver nanoparticle incorporated mesoporous silica granules for hemorrhage control and antibacterial treatment[J]. ACS Biomaterials Science & Engineering 4(10):3588–3599
Zhou H, Wang C, Niu H, Duan B, Ma X, Hong H, Yuan Y, Liu C (2018) A novel droplet-fabricated mesoporous silica-based nanohybrid granules for hemorrhage control[J]. J Biomed Nanotechnol 14(4):649–661
Doolittle RF (2017) The conversion of fibrinogen to fibrin: a brief history of some key events[J]. Matrix Biol 60:5–7
Lebrasseur N (2003) Calcium for strong clotting. J Cell Biol 160(7):980.2
Kaneko Y, Thoendel M, Olakanmi O, Britigan BE, Singh PK (2007) The transition metal gallium disrupts Pseudomonas aeruginosa iron metabolism and has antimicrobial and antibiofilm activity[J]. J Clin Invest 117(4):877–888
Zeimaran E, Pourshahrestani S, Djordjevic I, Pingguan-Murphy B, Kadri NA, Towler MR (2015) Bioactive glass reinforced elastomer composites for skeletal regeneration: a review[J]. Mater Sci Eng C 53:175–188
Arnold CE, Bordin A, Lawhon SD, Libal MC, Bernstein LR, Cohen ND (2012) Antimicrobial activity of gallium maltolate against Staphylococcus aureus and methicillin-resistant S. aureus and Staphylococcus pseudintermedius: an in vitro study[J]. Vet Microbiol 155(2–4):389–394
Lyu YH, Li X, Liu Z et al (2018) Antimicrobial activity of gallium[J]. Chin J Antibiot 43(4):394–400
Xu Z, Zhao X, Chen X, Chen Z, Xia Z (2017) Antimicrobial effect of gallium nitrate against bacteria encountered in burn wound infections[J]. RSC Adv 7(82):52266–52273
Goodley PH, Rogosnitzky M (2011) The effect of gallium nitrate on arresting blood flow from a wound[J]. Case Rep Med 2011:1–3
Rogosnitzky M. Method of wound hemostasis using liquid gallium nitrate: U.S. Patent 8,652,509[P]. 2014-2-18
Goncalves J, Wasif N, Esposito D, Coico JM, Schwartz B, Higgins PJ, Bockman RS, Staiano-Coico L (2002) Gallium nitrate accelerates partial thickness wound repair and alters keratinocyte integrin expression to favor a motile phenotype[J]. J Surg Res 103(2):134–140
Bauters A, Holt DJ, Zerbib P, Rogosnitzky M (2013) Gallium nitrate induces fibrinogen flocculation: an explanation for its hemostatic effect?[J]. BioMetals 26(6):935–939
Chen Z, Li F, Liu C, Guan J, Hu X, du G, Yao X, Wu J, Tian F (2016) Blood clot initiation by mesoporous silica nanoparticles: dependence on pore size or particle size?[J]. J Mater Chem B 4(44):7146–7154
Zhang Y, Zhi Z, Jiang T, Zhang J, Wang Z, Wang S (2010) Spherical mesoporous silica nanoparticles for loading and release of the poorly water-soluble drug telmisartan[J]. J Control Release 145(3):257–263
Nandiyanto ABD, Kim SG, Iskandar F, Okuyama K (2009) Synthesis of spherical mesoporous silica nanoparticles with nanometer-size controllable pores and outer diameters[J]. Microporous Mesoporous Mater 120(3):447–453
Wu S, Huang Z, Yue J, Liu D, Wang T, Ezanno P, Ruan C, Zhao X, Lu WW, Pan H (2015) The efficient hemostatic effect of Antarctic krill chitosan is related to its hydration property[J]. Carbohydr Polym 132:295–303
Zhang Y, Guan J, Wu J, Ding S, Yang J, Zhang J, Dong A, Deng L (2019) N-alkylated chitosan/graphene oxide porous sponge for rapid and effective hemostasis in emergency situations[J]. Carbohydr Polym 219:405–413
Zhao X, Guo B, Wu H, Liang Y, Ma PX (2018) Injectable antibacterial conductive nanocomposite cryogels with rapid shape recovery for noncompressible hemorrhage and wound healing[J]. Nat Commun 9(1):2784
Yoshida T, Yoshioka Y, Morishita Y, Aoyama M, Tochigi S, Hirai T, Tanaka K, Nagano K, Kamada H, Tsunoda SI, Nabeshi H, Yoshikawa T, Higashisaka K, Tsutsumi Y (2015) Protein corona changes mediated by surface modification of amorphous silica nanoparticles suppress acute toxicity and activation of intrinsic coagulation cascade in mice[J]. Nanotechnology 26(24):245101
Lee DW, Powers K, Baney R (2004) Physicochemical properties and blood compatibility of acylated chitosan nanoparticles[J]. Carbohydr Polym 58(4):371–377
Acknowledgements
The authors gratefully acknowledge the support for this work from the Military Medical Innovation Project [No. 18CXZ043], Natural Sciences Foundation of China [No. 51502345], Tianjin Science and Technology Major Project Program [18ZXJMTG00070] and Tianjin Sciences Foundation [16JCQNJC03100].
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ding, S., Wei, X., Yang, K. et al. Ca-Ga Double Doping Strategy to Fabricate Hemostatic Mesoporous Silica Nanoparticles (MSN) with Antibacterial Activity. Silicon 13, 4033–4045 (2021). https://doi.org/10.1007/s12633-020-00692-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12633-020-00692-y