Abstract
Purpose
This study was designed to determine the most effective peripheral nerve block supplement to standard anesthesia management for hip fracture patients.
Methods
We systematically reviewed randomized controlled trials (RCTs) published from 1990 to 2010 and conducted multiple treatment comparisons using direct and indirect evidence for two outcomes, i.e., acute pain intensity and delirium. We combined trials by type of injection (regardless of time of insertion during the perioperative phase, use of nerve block catheter, local anesthetic type, additives, or duration of treatment).
Results
Twenty-one RCTs comprising 1,422 participants were included. In most cases, the trials were conducted in Europe; they excluded patients with cognitive impairment, and they were unclear or at high risk of bias. The combination of obturator and lateral femoral cutaneous nerve blockade had the highest probability of being the most effective against acute postoperative pain. Fascia iliaca blockade had the highest probability of being the most effective against delirium. There was no difference in outcomes among other nerve blocks.
Conclusion
Not all nerve blocks are equally effective in improving outcomes after hip fracture. Multiple treatment comparison, a tool to compare the effectiveness of multiple treatments simultaneously, provides useful guidance to anesthesia providers seeking effective treatment when faced with a body of RCTs wherein each investigates one treatment. More RCTs comparing multiple nerve blocks in hip fracture are needed.
Résumé
Objectif
Cette étude a pour but de déterminer quel bloc nerveux périphérique, en supplément de la prise en charge anesthésique standard, est le plus efficace pour traiter les patients opérés suite à une fracture de la hanche.
Méthode
Nous avons effectué une revue méthodique des études randomisées contrôlées (ERC) publiées entre 1990 et 2010. Nous avons réalisé des comparaisons de plusieurs traitements (CPT) en nous fondant sur les données probantes directes et indirectes concernant deux critères d’évaluation : l’intensité de la douleur aiguë et le delirium. Nous avons combiné les études par type d’injection, indépendamment du moment d’insertion [du cathéter] en phase périopératoire, utilisation d’un cathéter pour le bloc nerveux, type d’anesthésique local, additifs et durée du traitement.
Résultats
21 ERC, comprenant 1422 participants, ont été incluses. La plupart des études ont été réalisées en Europe, exclu les patients souffrant de troubles cognitifs, et étaient peu claires ou présentaient un fort risque de biais. La combinaison de blocs des nerfs obturateur et fémoral cutané latéral a présenté la plus forte probabilité d’être le traitement le plus efficace pour contrer la douleur postopératoire aiguë. Le bloc du nerf iliofascial présentait la plus forte probabilité d’être le plus efficace contre le delirium. Aucune différence au niveau des critères d’évaluation n’a été observée avec les autres blocs nerveux.
Conclusion
Les blocs nerveux n’ont pas tous la même efficacité pour améliorer le pronostic après une fracture de la hanche. Les CPT, un outil pour comparer simultanément l’efficacité de plusieurs traitements, fournissent un cadre utile aux professionnels en anesthésie à la recherche d’un traitement efficace lorsqu’ils font face à un ensemble d’ERC qui se concentrent chacune sur un traitement. Il est nécessaire de réaliser davantage d’ERC comparant plusieurs blocs nerveux pour les fractures de la hanche.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hip fractures in elderly people are almost always fixed surgically.1 A variety of anesthetic techniques can be used for these procedures. In a recent comparative effectiveness review that examined all available strategies (anesthesia- and non-anesthesia-based) to decrease pain in hip fracture, we found that important outcomes, such as survival and major morbidity, are not affected by the choice of anesthetic technique.2,3 Nevertheless, we did find evidence that the use of supplemental peripheral nerve blockade was generally associated with reductions in postoperative pain intensity, delirium, and length of hospital stay (Fig. 1).
The hip joint and adjacent structures are amenable to nerve blockade by a number of different approaches and techniques. While our findings were consistent with an earlier review which indicated that nerve blocks reduce perioperative pain after hip fracture surgery,4 it is not known which, if any, of these methods is most effective.
By combining the results of randomized controlled trials (RCTs), conventional meta-analysis allows inferences to be made that might be invisible in the individual source studies because of a lack of statistical power. The vast majority of anesthesia trials, as in other areas of medicine, compare one or two types of active treatment with a reference treatment, placebo, or no treatment. While this method yields robust data about the absolute performance of each treatment, it is not methodologically valid simply to collect the aggregate effect sizes of each block and determine the best treatment by choosing the biggest number, because this approach would ignore any data we have from direct comparisons.5
How then can we determine best practice? The difficulty of performing an omnibus multi-armed RCT to find the single best option becomes obvious if, for the sake of discussion, we accept that there are (at least) seven different anatomic approaches to the nerves innervating the hip, three different technical methods of nerve location, catheter vs single-shot options, and several different local anesthetics that can be administered in any number of concentrations and doses, with many different additives, and for a variety of durations. Such a trial would be prohibitively large, complex, and costly.
Multiple treatment comparison (MTC) is a recent development in evidence synthesis that provides inferences on the comparative effectiveness of interventions that may never have been directly evaluated in clinical trials.6 This analytic method, while computationally complex, is conceptually simple. If there are three treatments, A, B and C, the relative effectiveness of A vs C can be inferred even if the only available direct comparisons are A vs B and B vs C. Multiple treatment comparisons have previously been used in cardiology,6 pediatric emergency medicine,7 and respirology.8
We applied MTC to the RCT data obtained during our previous comparative effectiveness review on pain management intervention in hip fracture patients. Our specific aim was to determine whether there is evidence that one or more nerve block approaches are more effective than others when used as a supplement to standard care for hip fracture.
Methods
We followed an a priori research protocol using recognized methodological approaches for conducting systematic reviews (http://effectivehealthcare.ahrq.gov/search-for-guides-reviews-and-reports/?pageaction=displayproduct&productID=368).
Search strategies
We searched 25 electronic databases (including MEDLINE, CINAHL, EMBASE, the Cochrane Database of Systematic Reviews, and Web of Science) and clinical trials registered from January 1990 to December 2010. We conducted hand searches of scientific meeting proceedings and the reference lists of reviews and included studies for RCTs and nonrandomized controlled trials (NRCTs) that a) were published from 1990 to 2010; b) focused on adults aged ≥ 50 yr who were admitted to hospital with acute hip fracture due to low-energy trauma; and c) examined the use of any peripheral nerve block. In order to preserve as much statistical power as possible, we made no analytic distinction between studies in which blocks were placed before, at the time of, or immediately after surgery, or between studies that used single-shot, catheter based, or intermittent injection techniques. No language restrictions were applied to the articles searched. A detailed description of our search strategy is available in the full report.3
Study selection
Two reviewers independently screened titles, abstracts, and the full text of potentially relevant articles. They extracted data, assessed methodological quality, and rated the body of evidence. Discrepancies were resolved by consensus or by third-party adjudication. We extracted study characteristics, inclusion and exclusion criteria, participant characteristics, interventions, and outcomes. We used the Cochrane Collaboration’s tool9 to assess risk of bias. Potential publication bias was explored graphically through funnel plots for comparisons for which meta-analyses were conducted and when there were at least ten studies in the analysis. Additionally, if bias was suspected, publication bias was quantitatively assessed using the Begg adjusted rank correlation test and the Egger regression asymmetry test.10
We selected the following outcomes of interest a priori: acute postoperative pain intensity, delirium, other adverse events, myocardial infarction, stroke, renal failure, and 30-day mortality.
Analysis
We conducted a MTC using a Bayesian network model in a single analysis.6,11,12 Results are expressed as standardized mean differences (continuous outcomes) or odds ratios (binary outcomes) with 95% credible intervals (CrIs). Credible intervals are Bayesian versions of confidence intervals and, for the purposes of this report, can be interpreted in the same way as confidence intervals. The analysis also yields the probability that each of the treatments tested is the best. For the outcomes of acute postoperative pain intensity and delirium, we conducted the MTC using a Bayesian network model to compare all interventions simultaneously and to use all available information on treatment effects in a single analysis. The analysis yields the probability that each of the treatments tested is the best, and delivers effect size estimates with narrower confidence intervals than conventional methods. For the one node where we had both direct and indirect evidence for a treatment comparison, we sought evidence of statistical inconsistency.13
Technical details about the conduct of the MTC are given in Appendix A.
Results
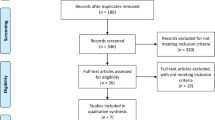
The search strategy identified 9,357 citations. Of these, 21 RCTs comprising 1,422 patients were eligible for inclusion (Fig. 2). One of these studies14 yielded non-analyzable data, leaving 20 studies for analysis. Eighteen pairwise comparisons were possible for acute pain intensity, five for delirium, four for mortality, and two for other adverse events.
Detailed assessment of each included study’s risk of bias is given in Appendix B. Overall, 1 (5%) RCT15 was assessed as low risk of bias, 9 (43%)14,16-23 had high risk of bias, and the remaining 11 (52%)13,24-33 had unclear risk of bias. The risk of bias relating to incomplete outcome data and selective outcome reporting was assessed as low in the majority of trials, (14/21trials [67%] and 18/21 [86%], respectively). Most trials were assessed as being at unclear risk of bias for sequence generation (15/21 trials [71%]), concealment allocation (16/21trials [76%]), and other bias (13/21 trials [62%]).
The Table lists key characteristics of the eligible studies. They were published from 1991 to 2010 and ranged in size from 14-209 participants. The range of mean age reported for participants was from 59-86 yr, with the majority being female (74%). Most studies (n = 15) were conducted in Europe. The most commonly studied approaches were the femoral and the three-in-one block. In 11 trials, electrical nerve stimulation was used to confirm nerve location, while the remainder used clinical landmarking. None of the trials featured ultrasound guidance. In the majority of trials, the comparator treatment was systemic analgesia or standard care. The definition of these varied from study to study but generally featured systemic opioids titrated to effect with or without a scheduled or on-request regimen of non-opioid analgesics.
Fig. 3 shows the number of studies that were examined in each pairwise comparison of acute postoperative pain intensity. Fig. 4 compares the performance of the nerve blocks at reducing acute pain intensity. We were able to compare acute pain intensity between seven nerve block strategies using data from 16 studies comprising 1,089 subjects. In 15 studies, pain was measured on a 10-cm visual analogue scale or an 11-point numeric rating scale,13,16,17,20-25,27,28,30-33 and in one study, a four-point verbal rating scale was used.18 Pain was also measured at a variety of intervals (ranging from hourly to daily). In most cases, this was a single score on the first postoperative day. When faced with a choice, we extracted data for the epoch that showed the most improvement for the intervention arm. For each nerve block, the reduction in pain compared with standard care is presented along with the 95% CrIs for that estimate. Since the constituent studies used a variety of scales to measure pain, the measure is a standardized mean difference (SMD) and should be interpreted in terms of unit effect size. While there is no definitive clinical way to interpret this, the most common interpretation is that an effect of 0.2 is “small”, an effect of 0.5 is “medium”, and an effect of 0.8 is “large”.34 For all but one intervention, the CrIs cross the null line, indicating a non-significant statistical difference between the interventions, each other, or standard care. The exception was the combination of lateral femoral cutaneous and obturator nerve blockade. This treatment was compared with obturator block alone and to fascia iliaca block in two trials13,22 of 165 patients. The estimate for pain intensity change was SMD -2.0 (95% CrI -3.81 to -0.25), and it had the highest probability of being the best choice for pain relief. Fascia iliaca block, studied in three trials22,30,31 comprising 453 patients, was the next best choice on probability grounds, but its effect size for pain relief did not reach statistical significance.
Fig. 5 compares the performance of nerve blocks at reducing postoperative delirium. The criteria used for the diagnosis of delirium were not reported in three studies.15,19,30 In one study,16 the Mini Mental State Examination (MMSE) was used, but the change in MMSE necessary to define delirium was not provided. In one other trial,31 daily MMSE and two other instruments were used to diagnose delirium against the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV) criteria. Fascia iliaca was the only block that had a statistically significant association with reduction in postoperative delirium (Estimate SMD -0.20; 95% CrI: -0.07 to -0.83).
Discussion
We previously described a robust association between the use of peripheral nerve blockade in hip fracture surgery and improvements in acute pain, delirium, and length of hospital stay.2,3 In the current analysis, all available data were compared simultaneously using MTC, and the combination of obturator and lateral femoral cutaneous blockade yielded the best reductions in acute pain. Fascia iliaca blockade was associated with the best reductions in delirium.
The apparent magnitude of the effect of these techniques (SMD = -2.0) on perioperative pain is clinically relevant. In addition, delirium is a significant cause of morbidity and cost following hip fracture, and the results of this analysis show a > 70% reduction in delirium associated with fascia iliaca blockade.35 It should be noted that most of the trials included in this synthesis excluded patients with preoperative cognitive impairment, arguably the group in whom delirium is most likely to occur and hardest to detect.36
Each of the peripheral nerve blocks studied represents something of a clinical compromise because the hip joint transmits sensation via branches of the femoral, obturator, superior gluteal, and sciatic nerves, and the nerve to the quadratus femoris,37 and no single peripheral injection site allows all of these nerves to be reached. In addition, pain following hip surgery is not only generated by the hip joint but also by the soft tissues that are disrupted as part of the surgical approach. This may partially explain why the fascia iliaca block— which blocks the lateral cutaneous nerve of the thigh more reliably than the femoral or three-in-one approach — and the combined obturator/lateral cutaneous nerve of the thigh block performed better than other methods.
There are obvious hazards when comparing studies of different blocks that used local anesthetics of different durations, continuous vs single-shot techniques, and different additives. Since the pain of soft tissue dissection and osteosynthesis does not disappear on the first postoperative day, intuition would suggest that blocks performed with longer-acting agents or blocks that involved continuous or patient-controlled infusions of local anesthetics would be more beneficial for our outcomes of interest. Regrettably, there were inadequate data to compare single-shot and continuous forms of each individual block directly. We accept that we may have missed important nuances by choosing to combine blocks by type, whether performed before, during, or after surgery.
Regional anesthesia is a rapidly evolving area of anesthesia practice, and the fact that ultrasound guidance was not used in any of the included trials may tempt some readers to dismiss this entire analysis as irrelevant. In our view, the relevant question is not whether ultrasound permits safer or more reliable blockade of the chosen target but whether it changes the efficacy of any given block technique more than any other. We would argue that the major nerves to the hip joint are, for the most part, anatomically consistent and amenable to reliable clinical landmarking and show easily visible responses to electrical nerve stimulation. Consequently, while ultrasound will improve the reliability and safety of these blocks, it may end up that it does not create new differences in efficacy between them.
The limitations of this study should be acknowledged. We were unable to review 25 studies from our original comparative effectiveness review searches either because we could not secure a translation or we could not find the original copy. The impact of this shortcoming in this study is mitigated by the fact that only three of those 25 titles appeared to be controlled studies of nerve blocks (one each femoral, lumbar plexus, and three-in-one block), while one other seemed to be a review article about nerve block techniques. Our assessment of the methodological quality on study publications was performed independently using the risk of bias tool, and we did not contact authors to verify the methods used. Although the methods were poorly reported in some trials, they may have been adequately conducted.
One of the main limitations of MTC is the presence of statistical inconsistency, that is, when there is no agreement between direct and indirect evidence. This was not an issue in our analysis as the only node that had both types of evidence showed consistency between the direct and indirect evidence. In MTC, we also assume that unobserved treatment effects are missing at random and that trials in two different comparisons are exchangeable. These assumptions are similar to those made in a standard meta-analysis. As with conventional meta-analysis, this method is designed to respect the randomization of the original studies and the assumptions that are made about the characteristics of those studies. Threats to the validity of an MTC analysis are similar to those in conventional meta-analyses.
Our analysis was also limited by the paucity of available data. More research, particularly studies including patients with cognitive impairment and studies comparing multiple nerve blocks simultaneously would improve our ability to make good therapeutic decisions in this important clinical area. We were further constrained by the lack of standardization in the reporting of pain as an outcome, which made it impossible to render the aggregate changes in pain intensity as clinically meaningful values. Finally, although pain and function are correlated, most studies focused on pain relief and did not evaluate the effects of an intervention on the patient’s ability to mobilize after surgery, a factor linked to recovery levels after hip fracture.38
In conclusion, using MTC, we found that the combination of obturator and lateral femoral cutaneous blockade yielded the best reductions in acute pain following surgery for a hip fracture, while fascia iliaca blockade was associated with the best reductions in delirium. Multiple treatment comparison, a tool to use when simultaneously comparing the effectiveness of multiple treatments, provides a useful guide for anesthesia providers seeking the best treatment when faced with a body of RCTs in which each trial investigates one treatment.
References
Neuman MD, Fleisher LA, Even-Shoshan O, Mi L, Silber JH. Nonoperative care for hip fracture in the elderly: the influence of race, income, and comorbidities. Med Care 2010; 48: 314-20.
Abou-Setta AM, Beaupre LA, Rashiq S, et al. Comparative effectiveness of pain management interventions for hip fracture: a systematic review. Ann Intern Med 2011; 155: 234-45.
Abou-Setta AM, Beaupre LA, Jones CA, et al. Pain Management Interventions for Hip Fracture [Internet]. Rockville (MD): Agency for Healthcare Research and Quality (US); 2011 May. (Comparative Effectiveness Reviews, No. 30.) Available from URL: http://www.ncbi.nlm.nih.gov/books/NBK56670/ (accessed December 2012).
Parker MJ, Griffiths R, Appadu BN. Nerve blocks (subcostal, lateral cutaneous, femoral, triple, psoas) for hip fractures. Cochrane Database Syst Rev 2002; 1: CD001159.
Song F, Loke YK, Walsh T, Glenny AM, Eastwood AJ, Altman DG. Methodological problems in the use of indirect comparisons for evaluating healthcare interventions: survey of published systematic reviews. BMJ 2009; 338: b1147.
Lu G, Ades AE. Combination of direct and indirect evidence in mixed treatment comparisons. Stat Med 2004; 23: 3105-24.
Hartling L, Fernandes RM, Bialy L, et al. Steroids and bronchodilators for acute bronchiolitis in the first two years of life: systematic review and meta-analysis. BMJ 2011; 342: d1714.
Mills EJ, Druyts E, Ghement I, Puhan MA. Pharmacotherapies for chronic obstructive pulmonary disease: a multiple treatment comparison meta-analysis. Clin Epidemiol 2011; 3: 107-29.
Higgins JP, Altman DG, Gotzsche PC, et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011; 343: d5928.
Hayashino Y, Noguchi Y, Fukui T. Systematic evaluation and comparison of statistical tests for publication bias. J Epidemiol 2005; 15: 235-43.
Higgins JP, Whitehead A. Borrowing strength from external trials in a meta-analysis. Stat Med 1996; 15: 2733-49.
Smith TC, Spiegelhalter DJ, Thomas A. Bayesian approaches to random-effects meta-analysis: a comparative study. Stat Med 1995; 14: 2685-99.
Segado Jimenez MI, Bayon Gago M, Arias Delgado J, et al. Efficacy of obturator and femoral cutaneous nerve blocks for postoperative analgesia in hip surgery (Spanish). Rev Esp Anestesiol Reanim 2009; 56: 590-7.
Antonopoulou E, Papaioannou K, Konstantinou G, Papadopoulos D, Karamoulas B, Molas TH. Continuous femoral block in elderly patients with hip fractures. Regional anesthesia and pain medicine. 2006; 31(5 Suppl 1): 92 (abstract).
Foss NB, Kristensen MT, Kristensen BB, Jensen PS, Kehlet H. Effect of postoperative epidural analgesia on rehabilitation and pain after hip fracture surgery: a randomized, double-blind, placebo-controlled trial. Anesthesiology 2005; 102: 1197-204.
de Visme V, Picart F, Le Jouan R, Legrand A, Savry C, Morin V. Combined lumbar and sacral plexus block compared with plain bupivacaine spinal anesthesia for hip fractures in the elderly. Reg Anesth Pain Med 2000; 25: 158-62.
Eyrolle L. Regional anaesthesia for femoral neck fracture surgery: comparison of lumbar plexus block and spinal anaesthesia. Br J Anaesth 1998; 80(Suppl 1): 112 (abstract).
Fletcher AK, Rigby AS, Heyes FL. Three-in-one femoral nerve block as analgesia for fractured neck of femur in the emergency department: a randomized, controlled trial. Ann Emerg Med 2003; 41: 227-33.
Graham CA, Baird K, McGuffie AC. A pilot randomised clinical trial of 3-in-1 femoral nerve block and intravenous morphine as primary analgesia for patients presenting to the emergency department with fractured hip. Hong Kong J Emerg Med 2008; 15: 205-11.
Matot I, Oppenheim-Eden A, Ratrot R, et al. Preoperative cardiac events in elderly patients with hip fracture randomized to epidural or conventional analgesia. Anesthesiology 2003; 98: 156-63.
Scheinin H, Virtanen T, Kentala E, et al. Epidural infusion of bupivacaine and fentanyl reduces perioperative myocardial ischaemia in elderly patients with hip fracture--a randomized controlled trial. Acta Anaesthesiol Scand 2000; 44: 1061-70.
Segado Jiminez MI, Arias Delgado J, Casas Garcia ML, et al. Post-surgical analgesia in hip surgery: a comparison of three techniques (Spanish). Rev Soc Esp Dolor 2010; 17: 259-67.
Tuncer S, Sert OA, Yosunkaya A, Mutlu M, Celik J, Okesli S. Patient-controlled femoral nerve analgesia versus patient-controlled intravenous analgesia for postoperative analgesia after trochanteric fracture repair. Acute Pain 2003; 4: 105-8.
Chudinov A, Berkenstadt H, Salai M, Cahana A, Perel A. Continuous psoas compartment block for anesthesia and perioperative analgesia in patients with hip fractures. Reg Anesth Pain Med 1999; 24: 563-8.
Cuvillon P, Ripart J, Debureaux S, et al. Analgesia after hip fracture repair in elderly patients: the effect of a continuous femoral nerve block: a prospective and randomised study (French). Ann Fr Anesth Reanim 2007; 26: 2-9.
Gille J, Gille M, Gahr R, Wiedemann B. Acute pain management in proximal femoral fractures: femoral nerve block (catheter technique) vs. systemic pain therapy using a clinic internal organisation model (German). Anaesthesist 2006; 55: 414-22.
Haddad FS, Williams RL. Femoral nerve block in extracapsular femoral neck fractures. J Bone Joint Surg Br 1995; 77: 922-3.
Henderson K, Akhtar S, Sandoval M, Siddiqui S, Todd K, Wirtner A. Femoral nerve block for pain management of hip fractures in the emergency department: preliminary results of a randomized, controlled trial. Ann Emerg Med 2008; 52: S164 (abstract).
Hood G, Edbrooke DL, Gerrish SP. Postoperative analgesia after triple nerve block for fractured neck of femur. Anaesthesia 1991; 46: 138-40.
Monzon DG, Vazquez J, Jauregui JR, Iserson KV. Pain treatment in post-traumatic hip fracture in the elderly: regional block vs. systemic non-steroidal analgesics. Int. J Emerg Med 2010; 3: 321-5.
Mouzopoulos G, Vasiliadis G, Lasanianos N, Nikolaras G, Morakis E, Kaminaris M. Fascia iliaca block prophylaxis for hip fracture patients at risk for delirium: a randomized placebo-controlled study. J Orthop Traumatol 2009; 10: 127-33.
Murgue D, Ehret B, Massacrier-Imbert S, et al. Equimolar nitrous oxide/oxygen combined with femoral nerve block for emergency analgesia of femoral neck fractures (French). Journal Européen des Urgences 2006; 19: 9-14.
Turker G, Uckunkaya N, Yavascaoglu B, Yilmazlar A, Ozcelik S. Comparison of the catheter-technique psoas compartment block and the epidural block for analgesia in partial hip replacement surgery. Acta Anaesthesiol Scand 2003; 47: 30-6.
Cohen J. Power Analysis for the Behavioral Sciences. 2nd ed. Hillsdale NJ: Lawrence Erlbaum Associates, Inc.; 1988 .
Robertson BD, Robertson TJ. Postoperative delirium after hip fracture. J Bone Joint Surg Am 2006; 88: 2060-8.
Eastwood EA, Magaziner J, Wang J, et al. Patients with hip fracture: subgroups and their outcomes. J Am Geriatr Soc 2002; 50: 1240-9.
Birnbaum K, Prescher A, Hessler S, Heller KD. The sensory innervation of the hip joint--an anatomical study. Surg Radiol Anat 1997; 19: 371-5.
Penrod JD, Boockvar KS, Litke A, et al. Physical therapy and mobility 2 and 6 months after hip fracture. J Am Geriatr Soc 2004; 52: 1114-20.
Acknowledgements
This project was funded under Contract No. HHSA 290 2007 10021 I from the Agency for Healthcare Research and Quality, U.S. Department of Health and Human Services. The authors of this report are responsible for its content. Statements in the report should not be construed as endorsement by the Agency for Healthcare Research and Quality or the U.S. Department of Health and Human Services.
Funding source
Agency for Healthcare Research and Quality, U.S. Department of Health and Human Services
Conflicts of interests
None declared.
Author information
Authors and Affiliations
Corresponding author
Additional information
Author contributions
Saifee Rashiq, Ben Vandermeer, Ahmed M. Abou-Setta, Lauren A. Beaupre, C. Allyson Jones, and Donna M. Dryden were involved in developing the study concept. Saifee Rashiq and Ahmed M. Abou-Setta were involved in data extraction. Saifee Rashiq drafted the manuscript, finalized the manuscript following review by others, and is the study guarantor. Ben Vandermeer was involved in the statistical analysis and is the guarantor of the integrity of the statistical methods used in the study. Ben Vandermeer, Ahmed M. Abou-Setta, Lauren A. Beaupre, C. Allyson Jones, and Donna M. Dryden contributed to the manuscript and critically reviewed and edited the manuscript.
This article is accompanied by an editorial. Please see Can J Anesth 2013; 60: this issue.
Appendices
Appendix A: Technical details of multiple treatment comparisons conduct
Mean differences or log-odds ratios were modelled using non-informative prior distributions. A normal prior distribution with mean 0 and a large variance (10,000) was used for each of the trial means or log-odds ratios; their between study variance had a uniform prior with range 0-5 (pain score) or 0-1 (delirium). These priors were checked for influence with sensitivity analyses. While these types of random effects analyses can often be very sensitive to the choice of prior for the between study variance, this was not the case in our analysis. Varying the parameters on the uniform prior anywhere from 2-10 had a negligible effect on the final result. Changing the parameters on the priors for the intervention differences also had no noticeable effect.
We carried out Markov chain Monte Carlo simulations using WinBUGS software (Version 1.4; Imperial College & MRC, UK) to obtain simultaneous estimates of all interventions compared with placebo as well as estimates to determine which interventions were the best.6 A burn-in sample of 20,000 iterations was followed by 200,000 iterations used to compute estimates.
APPENDIX B
Detailed risk-of-bias assessment for each included study
Reference | First Author and Year of Publication | Domain | Judgment | Description |
|---|---|---|---|---|
Segado Jiménez 2009 | Adequate sequence generation | Unclear Risk | Reported as a randomized trial without any further details | |
Allocation concealment | Unclear Risk | Not reported | ||
Blinding | Unclear Risk | Not reported | ||
Incomplete outcome data addressed | Low Risk | All patients completed the study and were included in the analyses (intention-to-treat) | ||
Free of selective reporting | Low Risk | Protocol not available, but the outcomes in the methods match those in the results. | ||
Free of other bias | Unclear Risk | Baseline characteristics are balanced, but there is no source of funding declared. | ||
Antonopoulou 2006 | Adequate sequence generation | Unclear Risk | Reported as a randomized trial without any further details | |
Allocation concealment | Unclear Risk | No description of allocation concealment reported | ||
Blinding | Unclear Risk | Not reported | ||
Incomplete outcome data addressed | Unclear Risk | Not enough information provided in the text to make a precise decision | ||
Free of selective reporting | High Risk | Protocol not available, but methods section numerates differing outcomes than are presented in the results | ||
Free of other bias | Unclear Risk | Baseline characteristics are balanced, but there is no source of funding declared | ||
Foss 2005 | Adequate sequence generation | Low Risk | Reported that randomization was performed using a computer-generated randomization list | |
Allocation concealment | Low Risk | Reported that randomization was performed by a third party | ||
Blinding | Low Risk | Reported that it was a double-blind trial and that the epidural cassettes were packed by the local pharmacy and blinded and supplied with a randomization number by a person not affiliated with the project | ||
Incomplete outcome data addressed | Low Risk | Intention-to-treat principle was not used in the analyses with 5/60 (8.33%) patients excluded from the analyses with reasons given | ||
Free of selective reporting | Low Risk | Protocol not available, but the outcomes in the methods match those in the results | ||
Free of other bias | Low Risk | Baseline characteristics are balanced and source of funding declared as governmental | ||
de Visme 2000 | Adequate sequence generation | Unclear Risk | Reported as a randomized trial without any further details | |
Allocation concealment | Low Risk | Randomization was performed in the hospital pharmacy (third party) | ||
Blinding | High Risk | Not reported but also not possible with the study design | ||
Incomplete outcome data addressed | High Risk | Intention-to-treat principle was not used in the analyses with 11/29 (37.93%) of randomized patients excluded from analysis | ||
Free of selective reporting | Low Risk | Protocol not available, but the outcomes in the methods match those in the results | ||
Free of other bias | Low Risk | Baseline characteristics are balanced, and the source of funding was declared to be institutional | ||
Eyrolle 1998 | Adequate sequence generation | Unclear Risk | Reported as a randomized trial without any further details | |
Allocation concealment | Unclear Risk | No description of allocation concealment reported | ||
Blinding | Unclear Risk | Not reported | ||
Incomplete outcome data addressed | Low Risk | All patients completed the study and were included in the analyses (intention-to-treat) | ||
Free of selective reporting | High Risk | Protocol is not available, and the intended outcomes are not clearly described in the methods section | ||
Free of other bias | Unclear Risk | Baseline characteristics are balanced, but there is no source of funding declared | ||
Fletcher 2003 | Adequate sequence generation | Low Risk | Reported that randomization was performed using a random number generator | |
Allocation concealment | Low Risk | Reported the use of sealed opaque envelopes | ||
Blinding | High Risk | Reported that data collectors and outcome assessors were blinded, but patients were not blinded to group allocation | ||
Incomplete outcome data addressed | Low Risk | All patients completed the study and were included in the analyses (intention-to-treat) | ||
Free of selective reporting | High Risk | Protocol not available, but one of the outcomes in the methods is not presented in the results (i.e. time to discharge) | ||
Free of other bias | Unclear Risk | Baseline characteristics are balanced, but there is no source of funding declared | ||
Graham 2008 | Adequate sequence generation | Unclear Risk | Reported the use of numbered, sequential, sealed opaque envelopes with no further details | |
Allocation concealment | Low Risk | Reported that allocation concealment was ensured using numbered, sequential, sealed opaque envelopes | ||
Blinding | High Risk | Reported as an ‘open-label’ trial | ||
Incomplete outcome data addressed | High Risk | Intention-to-treat principle was not used in the analyses with 7/40 (17.50%) of randomized patients excluded from analyses with reasons provided | ||
Free of selective reporting | Low Risk | Protocol not available, but the outcomes in the methods match those in the results | ||
Free of other bias | Unclear Risk | Baseline characteristics are balanced, but there is no source of funding declared | ||
Matot 2003 | Adequate sequence generation | Low Risk | Reported that randomization was performed using random numbers | |
Allocation concealment | Unclear Risk | No description of allocation concealment reported | ||
Blinding | High Risk | Not reported but also not possible with the study design | ||
Incomplete outcome data addressed | Low Risk | All patients completed the study and were included in the analyses (intention-to-treat) | ||
Free of selective reporting | Low Risk | Protocol not available, but the outcomes in the methods match those in the results | ||
Free of other bias | Low Risk | Baseline characteristics are balanced, and the source of funding was declared to be institutional | ||
Scheinin 2000 | Adequate sequence generation | Low Risk | Reported that randomization was performed using permuted blocks with strata | |
Allocation concealment | Unclear Risk | No description of allocation concealment reported | ||
Blinding | High Risk | Reported as an ‘open-label’ trial | ||
Incomplete outcome data addressed | High Risk | Intention-to-treat principle was not used in the analyses with 18/77 (23.38%) of randomized patients excluded from the analyses | ||
Free of selective reporting | Low Risk | Protocol not available, but the outcomes in the methods match those in the results | ||
Free of other bias | High Risk | Baseline characteristics were unbalanced with more males allocated to the paranteral analgesia group, but the source of funding is declared to be governmental and institutional. | ||
Segado Jiménez 2010 | Adequate sequence generation | Unclear Risk | Reported as a randomized trial with no further details regarding sequence generation | |
Allocation concealment | Unclear Risk | No information reported regarding allocation concealment in trial report. | ||
Blinding | Low Risk | Reported as a double-blind trial with medications identical in appearance and packaged uniformly. | ||
Incomplete outcome data addressed | High Risk | Intention-to-treat principle not utilized for analyses, and 42% of participants did not complete the study. | ||
Free of selective reporting | Low Risk | Protocol is not available, but the outcomes in the methods match those in the results | ||
Free of other bias | Low Risk | No significant differences in baseline characteristics or other sources of bias detected | ||
Tuncer 2003 | Adequate sequence generation | Unclear Risk | Reported as a randomized trial without any further details | |
Allocation concealment | Unclear Risk | No description of allocation concealment reported | ||
Blinding | High Risk | Not reported but also not possible with the study design | ||
Incomplete outcome data addressed | Low Risk | All patients completed the study and were included in the analyses (intention-to-treat) | ||
Free of selective reporting | Low Risk | Protocol not available, but the outcomes in the methods match those in the results | ||
Free of other bias | Unclear Risk | Baseline characteristics are balanced, but there is no source of funding declared | ||
Chudinov 1999 | Adequate sequence generation | Unclear Risk | Reported as a randomized trial without any further details | |
Allocation concealment | Unclear Risk | No description of allocation concealment reported | ||
Blinding | Unclear Risk | Not reported | ||
Incomplete outcome data addressed | Low Risk | All patients completed the study and were included in the analyses (intention-to-treat) | ||
Free of selective reporting | Low Risk | Protocol not available, but the outcomes in the methods match those in the results | ||
Free of other bias | Unclear Risk | Baseline characteristics are balanced, but there is no source of funding declared | ||
Cuvillon 2007 | Adequate sequence generation | Unclear Risk | Reported as a randomized trial without any further details | |
Allocation concealment | Unclear Risk | Reported the use of sealed, numbered envelopes with no further details | ||
Blinding | Unclear Risk | Not reported | ||
Incomplete outcome data addressed | Low Risk | All patients completed the study and were included in the analyses (intention-to-treat) | ||
Free of selective reporting | Low Risk | Protocol not available, but the outcomes in the methods match those in the results | ||
Free of other bias | Low Risk | Baseline characteristics are balanced, and the source of funding was declared to be institutional | ||
Gille 2006 | Adequate sequence generation | Unclear Risk | Reported as a randomized trial without any further details | |
Allocation concealment | Unclear Risk | Not reported | ||
Blinding | Unclear Risk | Not reported | ||
Incomplete outcome data addressed | Unclear Risk | Not clear if all patients completed the trial and were included in the analyses | ||
Free of selective reporting | Low Risk | Protocol not available, but the outcomes in the methods match those in the results | ||
Free of other bias | Low Risk | Baseline characteristics are balanced, and the source of funding was declared to be institutional | ||
Haddad 1995 | Adequate sequence generation | Unclear Risk | Reported as randomized by using sealed envelopes with no further details | |
Allocation concealment | Unclear Risk | Reported the use of a sealed envelope technique with no further details | ||
Blinding | Low Risk | Reported that the staff who monitored the patients and provided rescue analgesia were unaware of the patients’ allocation | ||
Incomplete outcome data addressed | Low Risk | Intention-to-treat principle was not used with 5/50 (10.00%) of randomized patients excluded with reasons provided | ||
Free of selective reporting | Low Risk | Protocol not available, but the outcomes in the methods match those in the results | ||
Free of other bias | Unclear Risk | Baseline characteristics are balanced, but there is no source of funding declared | ||
Henderson 2008 | Adequate sequence generation | Unclear Risk | Reported as a randomized trial without any further details | |
Allocation concealment | Unclear Risk | No description of allocation concealment reported | ||
Blinding | Unclear Risk | Not reported | ||
Incomplete outcome data addressed | Low Risk | All patients completed the study and were included in the analyses (intention-to-treat) | ||
Free of selective reporting | Low Risk | Protocol not available, but the outcomes in the methods match those in the results | ||
Free of other bias | Unclear Risk | No information on baseline characteristics and no information on financial support. | ||
Hood 1991 | Adequate sequence generation | Unclear Risk | Reported the use of unmarked envelopes with no further details | |
Allocation concealment | Unclear Risk | Reported the use of a sealed envelope technique with no further details | ||
Blinding | Low Risk | Reported that all patients had their skin prepared and an elastoplast placed over the possible injection site to minimize bias, while staff providing rescue analgesia administration and assessing the quality of analgesia after the operation were blinded to the patients’ allocation | ||
Incomplete outcome data addressed | Low Risk | All patients completed the study and were included in the analyses (intention-to-treat) | ||
Free of selective reporting | Low Risk | Protocol not available, but the outcomes in the methods match those in the results | ||
Free of other bias | Unclear Risk | Baseline characteristics are balanced, but there is no source of funding declared | ||
Monzon 2010 | Adequate sequence generation | Low Risk | computer-generated | |
Allocation concealment | Low Risk | The randomization list was kept by one of the authors who did not interact with the patients. He gave instructions to the patient’s emergency department nurse about which treatment should be administered. | ||
Blinding | Unclear Risk | Not reported | ||
Incomplete outcome data addressed | Unclear Risk | No intention-to-treat and 13.6% of patients excluded. | ||
Free of selective reporting | Low Risk | Protocol not available, but the outcomes in the methods match those in the results. | ||
Free of other bias | Low Risk | Baseline characteristics are balanced; no funding | ||
Mouzopoulos 2009 | Adequate sequence generation | Low Risk | Reported that randomization was performed using a computer-generated randomization code | |
Allocation concealment | Unclear Risk | No description of allocation concealment reported | ||
Blinding | Low Risk | Reported that patients were blinded to the treatment using a placebo with identical appearance and route of administration to the study medication | ||
Incomplete outcome data addressed | Low Risk | Intention-to-treat principle was not used in the analyses with 12/219 (5.48%) of randomized patients not included in the analyses | ||
Free of selective reporting | Low Risk | Protocol not available, but the outcomes in the methods match those in the results | ||
Free of other bias | Unclear Risk | Baseline characteristics are balanced, but there is no source of funding declared | ||
Murgue 2006 | Adequate sequence generation | Unclear Risk | Reported as a randomized trial without any further details | |
Allocation concealment | Unclear Risk | Not reported | ||
Blinding | Unclear Risk | Not reported | ||
Incomplete outcome data addressed | Low Risk | All patients completed the study and were included in the analyses (intention-to-treat) | ||
Free of selective reporting | Low Risk | Protocol not available, but the outcomes in the methods match those in the results | ||
Free of other bias | Unclear Risk | Baseline characteristics are balanced, but there is no source of funding declared | ||
Turker 2003 | Adequate sequence generation | Unclear Risk | Reported as a randomized trial without any further details | |
Allocation concealment | Unclear Risk | No description of allocation concealment reported | ||
Blinding | Unclear Risk | Reported that the outcomes assessment was blinded (single-blind) | ||
Incomplete outcome data addressed | Low Risk | All patients completed the study and were included in the analyses (intention-to-treat) | ||
Free of selective reporting | Low Risk | Protocol not available, but the outcomes in the methods match those in the results | ||
Free of other bias | Unclear Risk | Baseline characteristics are balanced but there is no source of funding declared |
Rights and permissions
About this article
Cite this article
Rashiq, S., Vandermeer, B., Abou-Setta, A.M. et al. Efficacy of supplemental peripheral nerve blockade for hip fracture surgery: multiple treatment comparison. Can J Anesth/J Can Anesth 60, 230–243 (2013). https://doi.org/10.1007/s12630-012-9880-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12630-012-9880-8