Abstract
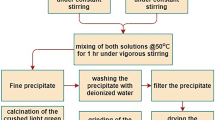
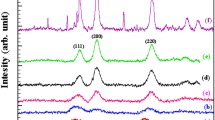
A mechanical activation process was introduced as a facile method for producing nickel oxide nanopowders. The precursor compound Ni(OH)2·NiCO3·4H2O was synthesized by chemical precipitation. The precursor was milled with NaCl diluent. A high-energy ball milling process led to decomposition of the precursor and subsequent dispersal in NaCl media. Nickel oxide nanocrystalline powders were produced by subsequent heat treatment and water washing. Milling rotation speed, milling time, ball-to-powder ratio (BPR), and nickel chloride-to-precursor ratio were introduced as influential parameters on the wavelength of maximum absorption (λ max). The effects of these parameters were investigated by the Taguchi method. The optimum conditions for this study were a milling rotation speed of 150 r/min, a milling time of 20 h, a BPR of 15/1, and a NaCl-to-powder weight ratio (NPR) of 6/1. In these conditions, λ max was predicted to be 292 nm. The structural properties of the samples were determined by field emission scanning electron microscopy, X-ray diffraction, and energy dispersive spectrometry.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
J. Moghaddam, S. Kolahgar-Azari, and S. Karimi, Determination of optimum conditions for nano-silver preparation from AgCl based on the taguchi design by the use of optical properties of silver, Ind. Eng. Chem. Res., 51(2012), No. 8, p. 3224.
Y.H. He, K. Vinodgopal, M. Ashokkumar, and F. Grieser, Sonochemical synthesis of ruthenium nanoparticles, Res. Chem. Intermed., 32(2006), No. 8, p. 709.
R.M. Kassab, K.T. Jackson, O.M. El-Kadri, and H.M. El-Kaderi, Nickel-catalyzed synthesis of nanoporous organic frameworks and their potential use in gas storage applications, Res. Chem. Intermed., 37(2011), No. 7, p. 747.
D. Adler and J.J. Feinleib, Electrical and optical properties of narrow-band materials, Phys. Rev. B, 2(1970), No. 8, p. 3112.
I. Hotovy, J. Huran, L. Spiess, S. Hascik, and V. Rehacek, Preparation of nickel oxide thin films for gas sensors applications, Sens. Actuators B, 57(1999), No. 1–3, p. 147.
E.L. Miller and R.E. Rocheleau, Electrochemical behavior of reactively sputtered iron-doped nickel oxide, J. Electrochem. Soc., 144(1997), No. 9, p. 3072.
Y.P. Wang, J.W. Zhu, X.J. Yang, L.D. Lu, and X. Wang, Preparation of NiO nanoparticles and their catalytic activity in the thermal decomposition of ammonium perchlorate, Thermochim. Acta, 437(2005), No. 1–2, p. 106.
R.C. Makkus, K. Hemmes, and J.H.W. de Wit, A Comparative study of NiO(Li), LiFeO2, and LiCoO2 porous cathodes for molten carbonate fuel cells, J. Electrochem. Soc., 141(1994), No. 12, p. 3429.
M. Ghosh, K. Biswas, A. Sundaresan, and C.N.R. Rao, MnO and NiO nanoparticles: synthesis and magnetic properties, J. Mater. Chem., 16(2006), No. 1, p. 106.
X. Wang, L.J. Ye, P. Hu, and F.L. Yuan, Synthesis of single-crystalline hollow octahedral NiO, Cryst. Growth Des., 7(2007), No. 12, p. 2415.
C.N. Huang, S.Y. Chen, and P. Shen, Condensation and decomposition of NiO-dissolved rutile nanospheres, J. Phys. Chem. C, 111(2007), No. 8, p. 3322.
B. Zhao, X.K. Ke, J.H. Bao, C.L. Wang, L. Dong, Y.W. Chen, and H.L. Chen, Synthesis of flower-like NiO and effects of morphology on its catalytic properties, J. Phys. Chem. C, 113(2009), No. 32, p. 14440.
M.S. Wu and H.H. Hsieh, Nickel oxide/hydroxide nanoplatelets synthesized by chemical precipitation for electrochemical capacitors, Electrochim. Acta, 53(2008), No. 8, p. 3427.
Z.Q. Wei, H.X. Qiao, H. Yang, C.R. Zhang, and X.Y. Yan, Characterization of NiO nanoparticles by anodic arc plasma method, J. Alloys Compd., 479(2009), No. 1–2, p. 855.
J.R.A. Sietsma, J.D. Meeldijk, J.P. den Breejen, M.V. Helder, A.J. van Dillen, P.E. de Jongh, and K.P. de Jong, The Preparation of supported NiO and Co3O4 nanoparticles by the nitric oxide controlled thermal decomposition of nitrates, Angew. Chem. Int. Ed., 119(2007), No. 24, p. 4631.
L.X. Yang, Y.J. Zhu, H. Tong, Z.H. Liang, L. Li, and L.J. Zhang, Hydrothermal synthesis of nickel hydroxide nanostructures in mixed solvents of water and alcohol, J. Solid State Chem., 180(2007), No. 7, p. 2095.
C.K. Xu, K.Q. Hong, S. Liu, G.H. Wang, and X.N. Zhao, A novel wet chemical route to NiO nanowires, J. Cryst. Growth, 255(2003), No. 3–4, p. 308.
L.L. Wu, Y.S. Wu, H.Y. Wei, Y.C. Shi, and C.X. Hu, Synthesis and characteristics of NiO nanowire by a solution method, Mater. Lett., 58(2004), No. 21, p. 2700.
M.B. Zheng, J.M. Cao, Y.P. Chen, X.J. Ma, S.G. Deng, and J. Tao, Facile fabrication of nickel oxide hollow spheres and amorphous carbon/nickel nanoparticles composites using colloidal carbonaceous microspheres as template, Chem. Lett., 34(2005), No. 8, p. 1174.
W. Xing, F. Li, Z.F. Yan, H.M. Cheng, and G.Q. Lu, Synthesis of wormlike nanoporous nickel oxide with nanocrystalline framework for electrochemical energy storage, Int. J. Nanosci., 3(2004), No. 3, p. 321.
X.M. Liu, X.G. Zhang, and S.Y. Fu, Preparation of urchinlike NiO nanostructures and their electrochemical capacitive behaviors, Mater. Res. Bull., 41(2006), No. 3, p. 620.
L.Y. Bai, F.L. Yuan, P. Hu, S.K. Yan, X. Wang, and S.H. Li, A facile route to sea urchin-like NiO architectures, Mater. Lett., 61(2007), No. 8–9, p. 1698.
X.M. Ni, Y.F. Zhang, D.Y. Tian, H.G. Zheng, and X.W. Wang, Synthesis and characterization of hierarchical NiO nanoflowers with porous structure, J. Cryst. Growth, 306(2007), No. 2, p. 418.
A. Al-Hajry, A. Umar, M. Vaseem, M.S. Al-Assiri, F. El-Tantawy, M. Bououdina, S. Al-Heniti, and Y.B. Hahn, Low-temperature growth and properties of flower-shaped β-Ni(OH)2 and NiO structures composed of thin nanosheets networks, Superlattices Microstruct., 44(2008), No. 2, p. 216.
L.P. Zhu, G.H. Liao, Y. Yang, H.M. Zhao, J.F. Wang, and S.Y. Fu, Self-assembled 3D flower-like hierarchical β-Ni(OH)2 hollow architectures and their in-situ thermal conversion to NiO, Nanoscale Res. Lett., 4(2009), No. 6, p. 550.
H.Z. Wang and Y.T. Qian, Malic acid assisted precursor route to hierarchical structured nickel oxide, Cryst. Res. Technol., 45(2010), No. 5, p. 545.
I. Hotovy, V. Rehacek, P. Siciliano, S. Capone, and L. Spiess, Sensing characteristics of NiO thin films as NO2 gas sensor, Thin Solid Films, 418(2002), No. 1, p. 9.
T.Y. Kim, J.Y. Kim, S.H. Lee, H.W. Shim, S.H. Lee, E.K. Suh, and K.S. Nahm, Characterization of ZnO needle-shaped nanostructures grown on NiO catalyst-coated Si substrates, Synth. Met., 144(2004), No. 1, p. 61.
F. Li, H.Y. Chen, C.M. Wang, and K.A. Hu, A novel modified NiO cathode for molten carbonate fuel cells, J. Electroanal. Chem., 531(2002), No. 1, p. 53.
M. Salavati-Niasari, N. Mir, and F. Davar, A novel precursor in preparation and characterization of nickel oxide nanoparticles via thermal decomposition approach, J. Alloys Compd., 493(2010), No. 1–2, p. 163.
S.A. Needham, G.X. Wang, and H.K. Liu, Synthesis of NiO nanotubes for use as negative electrodes in lithium ion batteries, J. Power Sources, 159(2006), No. 1, p. 254.
T. Tsuzuki and P.G. McCormick, Mechanochemical synthesis of nanoparticles, J. Mater. Sci., 39(2004), No. 16–17, p. 5143.
T. Tsuzuki, J.S. Robinson, and P.G. McCormick, UV-Shielding ceramic nanoparticles synthesized by mechanochemical processing, J. Aust. Ceram. Soc., 38(2002), No. 1, p. 15.
R. Aghababazadeh, B. Mazinani, A. Mirhabibi, and M. Tamizifar, ZnO nanoparticles synthesised by mechanochemical processing, J. Phys. Conf. Ser., 26(2006), No. 1, p. 312.
B. Fotoohi, A Study of Mechanochemical Activation in Solid-State Synthesis of Advanced Ceramic Composites [Dissertation], University of Birmingham, Birmingham, 2010.
G.J.C. Carpenter and Z.S. Wronski, Nanocrystalline NiO and NiO-Ni(OH)2 composite powders prepared by thermal and mechanical dehydroxylation of nickel hydroxide, Nanostruct. Mater., 11(1999), No. 1, p. 67.
B.D. Cullity, Elements of X-ray Diffraction, Second edition, Addison-Wesley, Reading, Massachusetts, 1978, p. 37–68.
C. Suryanarayana, Mechanical alloying and milling, Prog. Mater. Sci., 46(2001), No. 1–2, p. 1.
M.A. Gondal, T.A. Saleh, and Q.A. Drmosh, Synthesis of nickel oxide nanoparticles using pulsed laser ablation in liquids and their optical characterization, Appl. Surf. Sci., 258(2012), No. 18, p. 6982.
K. Anandan and V. Rajendran, Morphological and size effects of NiO nanoparticles via solvothermal process and their optical properties, Mater. Sci. Semicond. Process., 14(2011), No. 1, p. 43.
Y.G. Liu, Z.Y. Tang, Q. Xu, X.Y. Zhang, and Y. Liu, Ni(OH)2 particles synthesized by high energy ball milling, Trans. Nonferrous Met. Soc. China, 16(2006), No. 5, p. 1218.
A.D. Paola, E. García-López, G. Marcì, and L. Palmisano, A survey of photocatalytic materials for environmental remediation, J. Hazard. Mater., 211–212(2012), No. 11, p. 3.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ahmadisoltansaraei, K., Moghaddam, J. Preparation of NiO nanoparticles from Ni(OH)2·NiCO3·4H2O precursor by mechanical activation. Int J Miner Metall Mater 21, 726–735 (2014). https://doi.org/10.1007/s12613-014-0964-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12613-014-0964-z