Abstract
Objectives
To investigate the effect of a medical food (Souvenaid) on body mass index (BMI) and functional abilities in patients with mild Alzheimer’s disease (AD).
Design/setting/participants/intervention /measurements
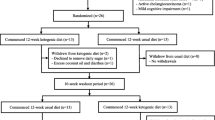
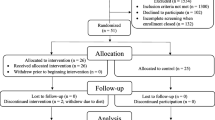
These analyses were performed on data from a 12-week, double-blind, randomized, controlled, multicenter, proof-of-concept study with a similarly designed and exploratory 12-week extension period. Patients with mild AD (Mini-Mental State Examination score of 20–26) were randomized to receive either the active product or an iso-caloric control product While primary outcomes included measures of cognition, the 23-item Alzheimer’s Disease Cooperative Study-Activities of Daily Living (ADCS-ADL) scale was included as a secondary outcome. Both ADCS-ADL and BMI were assessed at baseline and Weeks 6, 12 and 24. Data were analyzed using a repeated-measures mixed model.
Results
Overall, data suggested an increased BMI in the active versus the control group at Week 24 (ITT: p = 0.07; PP: p = 0.03), but no treatment effect on ADCS-ADL was observed. However, baseline BMI was found to be a significant treatment effect modifier (ITT: p = 0.04; PP: p = 0.05), and an increase in ADCS-ADL was observed at Week 12 in patients with a ‘low’ baseline BMI (ITT: p = 0.02; PP: p = 0.04).
Conclusions
These data indicate that baseline BMI significantly impacts the effect of Souvenaid on functional abilities. In addition, there was a suggestion that Souvenaid increased BMI.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Rosenstein LD. Differential diagnosis of the major progressive dementias and depression in middle and late adulthood a summary of the literature of the early 1990s. Neuropsychol Rev. 1998 Sep;8(3): 109–167.
Selkoe DJ. Alzheimer’s disease is asynaptic failure. Science 2002;298:789–791.
Terry RD. Alzheimer’s disease and the aging brain. J Geriatr Psychiatry Neurol. 2006 Sep;19(3): 125–128.
Wurtman RJ, UIus EH, Cansev M, et al. Synaptic proteins and phospholipids are increased in gerbil brain by administering uridine plus docosahexaenoic acid orally. Brain Res. 2006 May 9;1088(1):83–92.
Cansev M, Wurtman RJ, Sakamoto T, et al. Oral administration of circulating precursors for membrane phosphatides can promote the synthesis of new brain synapses. Alzheimers Dement. 2008 Jan;4(1 Suppl 1):S153–S168.
Cansev M, Wurtman RJ. Chronic administration of docosahexaenoic acid or eicosapentaenoic acid, but not arachidonic acid, alone or in combination with uridine, increases brain phosphatide and synaptic protein levels in gerbils. Neuroscience. 2007 Aug 24;148(2):421–431.
Van Wijk NW, Böhlke M, Maher TT, et al. Combined dietary vitamins B6, B12, and folate increase plasma choline levels in rats. Abstract. 2011;10th International Conference of Alzheimer’s and Parkinson’s Diseases (AD/PD), Spain, Barcelona Program number 194.
Broersen LM, De Wilde M, I. L, et al. Reduced beta-amyloid plaque burden and neurodegeneration in APP/PS1 transgenic mice following multi-nutrient dietary intervention Society for Neuroscience (San Diego, CA). 2007;Programme number 157. 116/U127.
Barberger-Gateau P, Raffaitin C, Letenneur L, et al. Dietary patterns and risk of dementia the Three-City cohort study. Neurology. 2007 Nov 13;69(20):1921–1930.
Luchsinger JA, Tang MX, Miller J, et al. Relation of higher folate intake to lower risk of Alzheimer disease in the elderly. Arch Neurol. 2007 Jan;64(1):86–92.
Shatenstein B, Kergoat MJ, Reid I. Poor nutrient intakes during 1-year follow-up with community-dwelling older adults with early-stage Alzheimer dementia compared to cognitively intact matched controls. J Am Diet Assoc. 2007 Dec;107(12):2091–2099.
Scheltens P, Kamphuis PJ, Verhey FR, et al. Efficacy of a medical food in mild Alzheimer’s disease: A randomized, controlled trial. Alzheimers Dement. 2010 Jan;6(1):1–10e1.
Barberger-Gateau P, Commenges D, Gagnon M, et al. Instrumental activities of daily living as a screening tool for cognitive impairment and dementia in elderly community dwellers. J Am Geriatr Soc. 1992 Nov;40(11):1129–1134.
White H, Pieper C, Schmader K. The association of weight change in Alzheimer’s disease with severity of disease and mortality: a longitudinal analysis. J Am Geriatr Soc. 1998 Oct;46(10):1223–1227.
Guerin O, Andrieu S, Schneider SM, et al. Different modes of weight loss in Alzheimer disease: a prospective study of 395 patients. Am J Clin Nutr. 2005 Aug;82(2):435–441.
Faxen-Irving G, Basun H, Cederholm T. Nutritional and cognitive relationships and long-term mortality in patients with various dementia disorders. Age and Ageing. 2005 Mar;34(2):13641.
Spaccavento S, Del Prete M, Craca A, et al. Influence of nutritional status on cognitive, functional and neuropsychiatrie deficits in Alzheimer’s disease. Arch Gerontol Geriatr. 2009 May–Jun;48(3):356–360.
Ousset PJ, Nourhashemi F, Reynish E, et al. Nutritional status is associated with disease progression in very mild Alzheimer disease. Alzheimer Dis Assoc Disord. 2008 Jan–Mar;22(1):66–71.
Cook Z, Kirk S, Lawrenson S, et al. Use of BMI in the assessment of undernutrition in older subjects: reflecting on practice. Proc Nutr Soc. 2005 Aug;64(3):313–317.
(CHMP) CfMPfHU. Guideline on Medicinal Products for the Treatment of Alzheimer’s Disease and Other Dementias (Document CPMP/EWP/553/95 Rev 1). London, UK: European Medicines Agency; 2009.
Mohs RC, Knopman D, Petersen RC, et al. Development of cognitive instruments for use in clinical trials of antidementia drugs: additions to the Alzheimer’s Disease Assessment Scale that broaden its scope. The Alzheimer’s Disease Cooperative Study. Alzheimer Dis Assoc Disord. 1997;11Suppl 2: S13–21.
Galasko D, Bennett D, Sano M, et al. An inventory to assess activities of daily living for clinical trials in Alzheimer’s disease. The Alzheimer’s Disease Cooperative Study. Alzheimer Dis Assoc Disord. 1997; 11Suppl 2:833–839.
Irving GF, Freund-Levi Y, Eriksdotter-Jonhagen M, et al. Omega-3 fatty acid supplementation effects on weight and appetite in patients with Alzheimer’s disease: the omega-3 Alzheimer’s disease study. J Am Geriatr Soc. 2009 Jan;57(1):11–17.
Gauthier S, Lopez OL, Waldemar G, et al. Effects of donepezil on activities of daily living: integrated analysis of patient data from studies in mild, moderate and severe Alzheimer’s disease. IntPsychogeriatr. 2010 Sep;22(6):973–983.
Galanos AN, Pieper CF, Comoni-Huntley JC, et al. Nutrition and function is there a relationship between body mass index and the functional capabilities of community-dwelling elderly? J Am Geriatr Soc. 1994 Apr;42(4):368–373.
Guerin O, Soto ME, Brocker P, et al. Nutritional status assessment during Alzheimer’s disease: results after one year (the REAL French Study Group). J Nutr Health Aging. 2005;9(2):81–84.
de Jong N, Chin APMJ, de Groot LC, et al. Functional biochemical and nutrient indices in frail elderly people are partly affected by dietary supplements but not by exercise. J Nutr. 1999 Nov;129(11):2028–2036.
Faxen-Irving G, Basun H, Cederholm T. Nutritional and cognitive relationships and long-term mortality in patients with various dementia disorders. Age Ageing. 2005 Mar;34(2):136–141.
Chin A Paw MJ, Dekker JM, Feskens EJ, et al. How to select a frail elderly population? A comparison of three working definitions. J Clin Epidemiol. 1999 Nov;52(11): 1015–1021.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kamphuis, P.J.G.H., Verhey, F.R.J., Olde Rikkert, M.G.M. et al. Effect of a medical food on body mass index and activities of daily living in patients with Alzheimer’s disease: Secondary analyses from a randomized, controlled trial. J Nutr Health Aging 15, 672–676 (2011). https://doi.org/10.1007/s12603-011-0339-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12603-011-0339-3