Abstract
Control of the invasive vine mealybug Planococcus ficus (Signoret) can be achieved through integration of different management strategies such as the application of chemical treatments and biological control. However, compatibility between these control methods is considered crucial when adopting integrated pest management (IPM) programs against this insect pest. The recently used insecticides Prev-Am®, a contact biopesticide, and spirotetramat, a systemic tetramic acid insecticide, were assessed in laboratory conditions for their side effects on the vine mealybug parasitoid Anagyrus sp. near pseudococci with reference to chlorpyriphos-methyl, an organophosphate insecticide conventionally used for mealybug control in vineyards. Regarding contact toxicity, chlorpyriphos-methyl caused 100% parasitoid mortality 24 h after treatment, but parasitoid mortality from Prev-Am® or spirotetramat was almost absent. Neither the total parasitoid offspring/female nor the sex ratio of the progeny was negatively affected by Prev-Am® or spirotetramat relative to the untreated control. According to the IOBC classification for laboratory trials, both Prev-Am® and spirotetramat were rated harmless (IOBC category 1), whereas chlorpyriphos-methyl was categorized as harmful (IOBC category 4) to A. sp. near pseudococci. None of the insecticides adversely affected the development of the parasitoid pupal stage inside mealybug mummies or the survival of the emerged parasitoids. The practical implications of the results for implementing mealybug IPM programs in vineyards are emphasized and discussed.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In several agricultural areas around the world, chemical treatments are extensively applied to cope with insect problems and are considered a key component of programs of Integrated Pest Management (IPM). However, several studies have shown that pesticides can negatively affect non-target species occurring in agro-ecosystems, including vertebrate fauna (Banks and Stark 1998; Carlile 2006; Pimentel 2009). Negative side effects of pesticides on arthropod natural enemies can often be observed, and toxicity of the applied chemical compounds varies from one insecticide to another (Carmo et al. 2010; Moens et al. 2011; Theiling and Croft 1988) and is dependent on the targeted insect species (Stark et al. 2003). Such circumstances are favorable for pesticide-induced disruption of natural enemies and consequently for the occurrence of further increases in outbreaks of insect pests, destabilizing IPM objectives. The compatibility of the natural enemies of the insects with pesticides should be seriously considered for successful programs of IPM (Stark et al. 2007). The tools of biological control could thus be strengthened through pesticide selectivity (Hautier et al. 2006; Johnson and Tabashnik 1999).
These practical considerations are true in the case of mealybug (Hemiptera: Pseudococcidae) pests (Franco et al. 2004; Mansour et al. 2010b; Suma et al. 2009; Walton and Pringle 1999) for which the most common control tactic is the application of insecticide treatments (Franco et al. 2009). Among these pseudococcids, the vine mealybug (VM) Planococcus ficus (Signoret) is considered the predominant species occurring in vineyards throughout the world (Daane et al. 2004a; Dalla Montà et al. 2001; Trjapitzin and Trjapitzin 1999; Walton et al. 2009). It causes serious damage to grapevines by transmitting viral diseases (Bertin et al. 2010; Mahfoudhi et al. 2009; Tsai et al. 2010). Control of P. ficus in vineyards currently is by applying new synthetic or natural compounds that are very useful in limiting outbreaks of this insect. For instance, the systemic tetramic acid insecticide spirotetramat, applied as foliar sprays, proved to be highly effective in decreasing densities of P. ficus occurring in vineyards and has a long residual activity against this insect pest (Brück et al. 2009; Mansour et al. 2010a). The systemic nicotinoid insecticide imidacloprid applied through drip irrigation supplied an outstanding efficacy in limiting mealybug numbers on grapevines (Daane et al. 2006; Mansour et al. 2010c). The new contact biopesticide Prev-Am® also provided promising results in reducing P. ficus densities on grapevines (Mansour et al. 2010a). However, the side effects of some of these insecticides on parasitoids of P. ficus have not yet been described, especially on the wasp Anagyrus pseudococci s.l. (Hymenoptera: Encyrtidae), the most common parasitoid of this mealybug species in several grape-growing areas (Dalla Montà et al. 2001; Franco et al. 2009; Gutierrez et al. 2008; Trjapitzin and Trjapitzin 1999). Increasing evidence suggests that the assessment of the influence of newly applied insecticides on this encyrtid would prove useful for enhancing IPM programs against P. ficus.
The present study aimed to evaluate, under laboratory conditions, the side effects of the two newer insecticides spirotetramat and Prev-Am® on the parasitoid A. sp. near pseudococci sensu Triapitsyn et al. (2007), compared to chlorpyriphos-methyl, which is considered the universal reference insecticide for mealybug control in vineyards. The practical implications for refining IPM-compatible programs for P. ficus in vineyards are emphasized.
Materials and methods
Insect colonies and rearing conditions
Mass culture of P. ficus was conducted on pumpkin squash (Cucurbita moschata (Duchesne) Poir) and sprouted potatoes (Solanum tuberosum L., var. ‘Spunta’) maintained in plastic boxes held under controlled laboratory conditions (25° ± 1°C; 60% r.h.; 12 h:12 h L:D photoperiod). Each fresh squash and/or sprouted potato was inoculated with suitable mealybug egg masses. P. ficus crawlers were allowed to develop until 3rd instar/adult female stages prior to their exposure to mature females of A. sp. near pseudococci. Mass rearing of A. sp. near pseudococci was carried out, in a separate room, on P. ficus colonies fed on squash and/or sprouted potatoes and was maintained under the above conditions. Prior to their introduction into cages with mealybug cultures, newly emerged female parasitoids were isolated and stored in small plastic vials for 24 h with an appropriate number of 1- or 2-day-old male parasitoids (1 male for 2–3 females) for mating.
Laboratory colonies of both P. ficus and A. sp. near pseudococci originated from intensive collections performed in July–August 2010 from grapevines at a vineyard in southern Sicily. Mealybug populations were sampled as mature females with ovisacs, while parasitoids were collected at the pupal stage residing inside mummified mealybugs. Representative specimens from the collected mealybugs and emerged parasitoids were identified prior to their introduction into the laboratory.
Chemicals
Two new insecticides and one conventional insecticide for control of P. ficus were tested for their side effects on A. sp. near pseudococci: spirotetramat (Movento® 150 OD, Bayer CropScience), a systemic tetramic acid insecticide; Prev-Am® (ORO AGRI International Ltd.), a contact biopesticide containing orange oil, borax and organic surfactants; and chlorpyriphos-methyl (Reldan® 22, Dow AgroSciences), an organophosphate insecticide, which was treated as the reference insecticide. All insecticides were tested at doses corresponding to their maximum recommended field rates of 120 ml per hectoliter, 300 ml hl−1 and 250 ml hl−1, respectively. The dose used for the reference insecticide is that registered for mealybug control in Italian vineyards. The applied doses of both spirotetramat and Prev-Am® were those registered for mealybug control in Tunisian vineyards, which are cultivated under climatic and agro-ecological conditions similar to those in Sicilian grape-growing areas. Spirotetramat and Prev-Am® are not yet officially registered for mealybug control in Italian vineyards.
Trials
Laboratory trials were performed according to the standard principles adopted by the IOBC/WPRS Working group ‘Pesticides and Beneficial Organisms’ (Hassan et al. 1994; Sterk et al. 1999). The tests were carried out using a Potter Precision Spray Tower (Burkard Manufacturing Co. Ltd., Rickmansworth, Herts., UK) with constant pressure, operated by compressed air. The purpose was to evaluate the effects on fertility and sex ratio of the progeny from contact toxicity 24, 48 and 72 h after treatment in adult A. sp. near pseudococci.
Direct toxicity was assessed after exposure of the mature female parasitoids to a fresh dry pesticide film applied to the internal surface of cubic glass cages (578 cm3) made of six plates joined externally with transparent adhesive tape (Viggiani and Tranfaglia 1978). Three of the four lateral glass plates had a central hole (0.5 cm diam) to allow air-flow and to provide the specimens with food by a cotton dispenser imbibed with a solution of sugar and protein (Protonectar, Lega Italy, Faega, Italy).
Insecticide distribution was uniform (1.5–1.8 mg cm−2) on the treated glass plates. The cages were assembled after the glass plates were completely dried. The untreated control was sprayed with tap water. Each single newly emerged A. sp. near pseudococci female was allowed to mate after having been isolated with one male parasitoid, 1–2 days old. Five 1- or 2-day-old mature A. sp. near pseudococci females were introduced into each cage, and five replicates were performed per treatment. The parasitoids used in the experiments derived from the F8 laboratory parasitoid generation. The cages were held under controlled laboratory conditions (25 ± 1°C; 60% r.h.; 12 h:12 h L:D photoperiod).
Mortality of parasitoid adult females 24, 48 and 72 h after treatment was recorded. Surviving females observed after 24 h of exposure were moved to clean cylindrical transparent containers (10 cm diam, 14 cm height). Each female parasitoid was offered one potato sprout infested with at least 50 P. ficus 3rd instar nymphs and adult females in order to evaluate the effect of the applied insecticides on both wasp fertility and sex ratio of the progeny. All introduced parasitoids were left with the host P. ficus until death.
To assess the influence of insecticide applications on the development of the pupal stage of A. sp. near pseudococci, P. ficus mummies were sprayed with the insecticides, under the Potter Precision Spray Tower, at the same dose used for the toxicity test on adults. The untreated control was sprayed with tap water. Prior to insecticide spraying, each mummy was immobilized by gently pressing it onto a strip of adhesive tape placed on one of the previously used glass plates. Immobilization also ensured uniform deposit of the insecticide on the surface of the mummy’s body. All P. ficus mummies were randomly selected from the mass culture 10–11 days after parasitoid introduction and ca 7–9 days before eventual parasitoid emergence. Five replicates were performed per treatment, with 15 sprayed mummies per replicate. The sprayed mummies were placed in petri dishes and were stored under the climatic conditions described above. Parasitoid emergence was checked twice a day, and both the number of emerged parasitoids and the number of days needed for emergence were recorded for each replicate. Subsequently, all emerged male and female parasitoids were placed in small vials and were provided with a solution of sugar and protein (Protonectar). Eventual mortality (until 96 h after emergence) from insecticide ingestion by A. sp. near pseudococci when chewing an exit hole through the host puparium was recorded.
Data analysis
The levels of parasitoid mortality related to contact toxicity were adjusted for control mortality using Abbott’s formula (Abbott 1925). The reduction in parasitism compared to the control (RP%) was calculated. The pesticides were classified into four categories (IOBC classes of toxicity) according to the levels of mortality and/or to the reduction in beneficial (parasitism) capacity: 1, harmless (<30%); 2, slightly harmful (30–79%); 3, moderately harmful (80–99%); and 4, harmful (>99%) (Hassan et al. 1994; Sterk et al. 1999).
To normalize the data, the percentages of mortality were transformed [arcsin (√x)], while the other data were subjected to [log (x + 1)] transformation. The transformed data were analyzed by a one-way analysis of variance (ANOVA), and the means were separated using Tukey's HSD test at P = 0.05. All statistical analyses were performed using Statistica 6.0 (StatSoft Inc. 2003).
Results
Contact toxicity
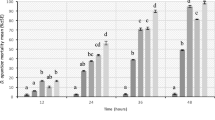
Adult parasitoids showed significantly lower (F3,16 = 0.132; P < 0.0001) overall levels of mortality (24, 48 and 72 h after treatment) when exposed to either spirotetramat or Prev-Am® treatments compared to chlorpyriphos-methyl exposure, which caused 100% parasitoid mortality 24 h after treatment (Table 1). None of the exposed parasitoid individuals died in the control treatment. Such a result was statistically similar to that recorded in either spirotetramat or Prev-Am® treatments. Indeed, mortality of parasitoids was recorded once on both Prev-Am® and spirotetramat-treated glasses, namely 24 and 48 h after treatment, respectively. The overall level of parasitoid mortality for the latter two insecticides was estimated at 4%.
Effects on fertility and sex ratio
Neither spirotetramat nor Prev-Am® significantly affected (F2,12 = 1.431; P = 0.27) the total progeny produced per single female (Table 1). The total number of offspring ranged from 22.68 ± 3 and 25.66 ± 3.36 parasitoids/female for spirotetramat and Prev-Am®, respectively, to 29.28 ± 2.02 parasitoids/female for the untreated control. Insecticide treatments did not significantly influence (F2,12 = 0.863; P = 0.44) the sex ratio of the progeny, which was male-biased for the untreated control (2.53:1 M:F) and the spirotetramat and Prev-Am® treatments (Table 1).
The RP% was 12.36% for Prev-Am®, 22.54% for spirotetramat and 100% for chlorpyriphos-methyl. Considering both the mortality% and RP% and based on the classes of toxicity adopted by the IOBC, the insecticides spirotetramat and Prev-Am® are rated as harmless (class 1: both percentages were <30%). Chlorpyriphos-methyl is categorized as harmful (class 4: both percentages were >99%) towards A. sp. near pseudococci (Table 1).
Effect on pupae and parasitoid survival
Statistical analysis did not reveal any significant impact (F3,16 = 0.0883; P = 0.47) of treatments on the percentage of emergence of A. sp. near pseudococci from P. ficus mummies (Table 2). The overall rate of parasitoid emergence exceeded 92% for all treatments, with a rate >98% for the untreated control. No significant difference (F3,16 = 2.45; P = 0.1) was found among treatments based on the number of days (after insecticide application) needed for parasitoid emergence, confirming the absence of any adverse insecticidal effect on pupal stage development (Table 2). The survival of the emerged parasitoids (male and female) was not negatively affected by the tested insecticides, since no mortality was observed up to 96 h after parasitoid emergence.
Discussion
The systemic tetramic acid insecticide spirotetramat and the new contact biopesticide Prev-Am® were proven to be highly selective in contact with A. sp. near pseudococci. The lack of toxicity of spirotetramat towards A. sp. near pseudococci could be linked to its limited contact efficacy. Spirotetramat is especially active against juvenile stages of a broad spectrum of sucking pests including, in addition to mealybugs, soft and armored scales, aphids, psyllids, and whiteflies (Nauen et al. 2008). Nauen et al. (2008) stated that spirotetramat exhibits an excellent systemic and translaminar efficacy against targeted insect pests, whereas its contact efficacy is rather limited. However, the application of the organophosphate insecticide chlorpyriphos-methyl resulted in the mortality of all tested parasitoids 24 h after tarsal contact. Organophosphate insecticides have previously been reported as having a high toxicity to parasitoids (Hassan 1997; Jacas Miret and Garcia-Marí 2001; Suma and Mazzeo 2008; Suma et al. 2009).
Neither spirotetramat nor Prev-Am® adversely affected the total parasitoid offspring/female or sex ratios of the progeny. These results further support the findings related to contact toxicity that these two insecticides are selective towards A. sp. near pseudococci.
Similar results were obtained by Suma and Mazzeo (2008) for the insecticides buprofezin and pyriproxyfen used in controlling P. citri in citrus orchards. These insecticides did not negatively influence fertility and sex ratio in A. sp. near pseudococci. In our laboratory conditions, the total parasitoid offspring for untreated controls was estimated at 29.28 ± 2.02 parasitoids/female. Güleç et al. (2007) found that under controlled laboratory conditions (28 ± 1°C, 65 ± 10% r.h. and 16 h:8 h L:D photoperiod), the average number of offspring per A. pseudococci female was 22.35 ± 1.68 in 15-day-old and 34.8 ± 2.56 in 21-day-old P. ficus.
The sex ratios recorded for the untreated control and the other treatments were male-biased, perhaps because the majority of the exposed mealybugs were 3rd instar nymphs (in addition to adult females). Previous laboratory studies indicated that the sex ratio of A. pseudococci was male-biased in 15-day-old but was equal in 21-day-old P. ficus (Güleç et al. 2007), was strongly male-biased in 3rd instar nymphs, but was female-biased in adult P. ficus females (Daane et al. 2004b), indicating that most parasitoid females were obtained from larger host mealybugs.
Based on the percentage mortality and on the reduction in parasitism, the newer insecticides spirotetramat and Prev-Am® are rated harmless to A. sp. near pseudococci, making both suitable for IPM systems against P. ficus in vineyards. The conventionally used insecticide chlorpyriphos-methyl was categorized as harmful towards A. sp. near pseudococci. Such a result would prevent the application of chlorpyriphos-methyl when adult parasitoids are the most abundant developmental stage of A. sp. near pseudococci in vineyards. In a previous study, spirotetramat was similarly categorized as harmless to other natural enemies of P. ficus, such as the predatory green lacewing Chrysoperla carnea (Stephens) (Neuroptera: Chrysopidae) and the parasitoid Coccidoxenoides perminutus Girault (Hymenoptera: Encyrtidae) (Schnorbach et al. 2008). Campos et al. (2008) found that chlorpyriphos on leaves of mandarin potted plants under extended laboratory conditions had low toxicity towards A. pseudococci s.l. reared from P. citri.
No significant differences were found among all treatments based on the number of parasitoids emerging from host mummies. The number of days needed for parasitoid emergence was statistically similar in all treatments. The adult stage of A. sp. near pseudococci was clearly more susceptible to chlorpyriphos-methyl than the juvenile stage residing inside P. ficus mummies. Chemical treatments against P. ficus in vineyards would thus prove to be compatible with biological control by A. sp. near pseudococci with the prerequisite that timing of insecticide treatments should coincide with the presence of the parasitoid pupal stage in the vineyard. Differences between laboratory and field conditions should therefore be considered when adopting this management tactic.
None of the emerged individual parasitoids, regardless of sex, died until 96 h after emergence. This result is a strong indication that, especially regarding chlorpyriphos-methyl-treated mummies, ingestion by the parasitoid of the insecticide present on the dorsal part of the host puparium did not constitute a risk of toxicity for emerged A. sp. near pseudococci. In contrast, other laboratories have observed that A. sp. near pseudococci died as they gnawed an exit hole with their mandibles through the dorsal portions of P. ficus mummies treated with fipronil or α-cypermethrin, pesticides used for the control of ants in South African vineyards (Mgocheki and Addison 2009).
The results obtained in the present study could enhance IPM programs against P. ficus, an economic pest attacking grapevines, by combining insecticide application with biological control using the encyrtid A. sp. near pseudococci. Spirotetramat and Prev-Am® could be applied without risk of toxicity to A. sp. near pseudococci. In contrast, the timing of application of chlorpyriphos-methyl should depend on the timing of parasitoid release to avoid parasitoid mortality due to tarsal contact with this insecticide. Since spirotetramat is a systemic insecticide, evaluating the suitability of A. sp. near pseudococci development inside spirotetramat-contamined mealybugs could prove useful. A follow-up study could attempt to understand whether or not a systemic toxicity of spirotetramat towards the encyrtid could occur.
References
Abbott, W. S. (1925). A method of computing the effectiveness of an insecticide. Journal of Economic Entomology, 18, 265–267.
Banks, J., & Stark, J. D. (1998). What is ecotoxicology? An ad-hoc grab bag or an interdisciplinary science? Integrative Biology, 5, 195–204.
Bertin, S., Cavalieri, V., Graziano, C., & Bosco, D. (2010). Survey of mealybug (Hemiptera: Pseudococcidae) vectors of Ampelovirus and Vitivirus in vineyards of northwestern Italy. Phytoparasitica, 38, 401–409.
Brück, E., Elbert, A., Fischer, R., Krueger, S., Kühnhold, J., Klueken, A. M., et al. (2009). Movento®, an innovative ambimobile insecticide for sucking insect pest control in agriculture: Biological profile and field performance. Crop Protection, 28, 838–844.
Campos, J. M., Martínez-Ferrer, M. T., & Forés, V. (2008). Secondary effects of seven pesticides on Anagyrus pseudococci (Girault) and Leptomastix dactylopii Howard (Hymenoptera: Encyrtidae), parasitoids of Planococcus citri (Risso) (Hemiptera: Pseudococcidae). IOBC/WPRS Bulletin, 38, 111–116.
Carlile, B. (2006). Pesticide selectivity, health and the environment. Cambridge, UK: Cambridge University Press.
Carmo, E., Bueno, A., & Bueno, R. (2010). Pesticide selectivity for the insect egg parasitoid Telenomus remus. Biocontrol, 55, 455–464.
Daane, K. M., Bentley, W. J., Walton, V. M., Malakar-Kuenen, R., Yokota, G. Y., Millar, J. G., et al. (2006). New controls investigated for vine mealybug. California Agriculture, 60, 31–38.
Daane, K. M., Bentley, W. J., & Weber, E. A. (2004). Vine mealybug: a formidable pest spreads throughout California vineyards. Practical Winery and Vineyard Journal, 3, 35–40.
Daane, K. M., Malakar-Kuenen, R. D., & Walton, V. M. (2004). Temperature-dependent development of Anagyrus pseudococci (Hymenoptera: Encyrtidae) as a parasitoid of the vine mealybug, Planococcus ficus (Homoptera: Pseudococcidae). Biological Control, 31, 123–132.
Dalla Montà, L., Duso, C., & Malagnini, V. (2001). Current status of scale insects (Hemiptera: Coccoidea) in the Italian vineyards. Bollettino di Zoologia Agraria e Bachicoltura, II.3, 343–350.
Franco, J. C., Suma, P., Silva, E. B., Blumberg, D., & Mendel, Z. (2004). Management strategies of mealybug pests of citrus in Mediterranean countries. Phytoparasitica, 32, 507–522.
Franco, J. C., Zada, A., & Mendel, Z. (2009). Novel approaches for the management of mealybug pests. In I. Ishaaya & A. R. Horowitz (Eds.), Biorational control of arthropod pests—application and resistance management (pp. 233–278). Dordrecht, the Netherlands: Springer.
Gutierrez, A. P., Daane, K. M., Ponti, L., Walton, V. M., & Ellis, C. K. (2008). Prospective evaluation of the biological control of vine mealybug: refuge effects and climate. Journal of Applied Ecology, 45, 524–536.
Güleç, G., Kilinçer, A. N., Kaydan, M. B., & Ülgentürk, S. (2007). Some biological interactions between the parasitoid Anagyrus pseudococci (Girault) (Hymenoptera: Encyrtidae) and its host Planococcus ficus (Signoret) (Hemiptera: Coccoidea: Pseudococcidae). Journal of Pest Science, 80, 43–49.
Hassan, E. (1997). Chlorpyrifos toxicity to Aphytis lingnanensis Compere (Hymenoptera: Aphelinidae) a parasitoid of California red scale, Aonidiella aurantii (Mask.) in citrus. Zeitschrift für Pflanzenkrankheiten und Pflanzenschutz, 104, 102–104.
Hassan, S. A., Bigler, F., Bogenschütz, H., Boller, E., Brun, J., Calis, J. N. M., et al. (1994). Results of the sixth joint pesticide testing programme of the IOBC/WPRS-working group “Pesticides and beneficial organisms”. Entomophaga, 39, 107–119.
Hautier, L., Jansen, J.-P., Mabon, N., & Schiffers, B. (2006). Building a selectivity list of plant protection products on beneficial arthropods in open field: a clear example with potato crop. IOBC/WPRS Bulletin, 29, 21–32.
Jacas Miret, J. A., & Garcia-Marí, F. (2001). Side-effects of pesticides on selected natural enemies occurring in citrus in Spain. IOBC/WPRS Bulletin, 24, 103–112.
Johnson, M. W., & Tabashnik, B. E. (1999). Enhanced biological control through pesticide selectivity. In T. Fisher, T. S. Bellows, L. E. Caltagirone, D. L. Dahlsten, C. Huffaker, & G. Gordh (Eds.), Handbook of biological control (pp. 297–317). San Diego, CA, USA: Academic.
Mahfoudhi, N., Digiaro, M., & Dhouibi, M. H. (2009). Transmission of grapevine leafroll viruses by Planococcus ficus (Hemiptera: Pseudococcidae) and Ceroplastes rusci (Hemiptera: Coccidae). Plant Disease, 93, 999–1002.
Mansour, R., Grissa Lebdi, K., & Rezgui, S. (2010). Assessment of the performance of some new insecticides for the control of the vine mealybug Planococcus ficus (Signoret) in a Tunisian vineyard. Entomologia Hellenica, 19, 21–33.
Mansour, R., Suma, P., Mazzeo, G., Buonocore, E., Grissa Lebdi, K., & Russo, A. (2010). Using a kairomone-based attracting system to enhance biological control of mealybugs (Hemiptera: Pseudococcidae) by Anagyrus sp. near pseudococci (Hymenoptera: Encyrtidae) in Sicilian vineyards. Journal of Entomological and Acarological Research, 42, 161–170.
Mansour, R., Youssfi, F. E., Grissa Lebdi, K., & Rezgui, S. (2010). Imidacloprid applied through drip irrigation as a new promising alternative to control mealybugs in Tunisian vineyards. Journal of Plant Protection Research, 50, 314–319.
Mgocheki, N., & Addison, P. (2009). Effect of contact pesticides on vine mealybug parasitoids, Anagyrus sp. near pseudococci (Girault) and Coccidoxenoides perminutus (Timberlake) (Hymenoptera: Encyrtidae). South African Journal of Enology and Viticulture, 30, 110–116.
Moens, J., De Clercq, P., & Tirry, L. (2011). Side effects of pesticides on the larvae of the hoverfly Episyrphus balteatus in the laboratory. Phytoparasitica, 39, 1–9.
Nauen, R., Reckmann, U., Thomzik, J., & Thielert, W. (2008). Biological profile of spirotetramat (Movento®)—a new two-way systemic (ambimobile) insecticide against sucking pest species. Bayer CropScience Journal, 61, 245–278.
Pimentel, D. (2009). Environmental and economic costs of the application of pesticides primarily in the United States. In R. Peshin & A. K. Dhawan (Eds.), Integrated pest management: innovation-development process, Vol. 1 (pp. 89–111). Dordrecht, the Netherlands: Springer Science and Business Media B.V.
Schnorbach, J., Elbert, A., Laborie, B., Navacerrada, J., Bangels, E., & Gobin, B. (2008). Movento®, an ideal tool for integrated pest management in pomefruit, citrus and vegetables. Bayer CropScience Journal, 61, 377–402.
Stark, J. D., Banks, J. E., & Acheamponga, S. (2003). Estimating susceptibility of biological control agents to pesticides: influence of life history strategies and population structure. Biological Control, 29, 392–398.
Stark, J. D., Vargas, R., & Banks, J. E. (2007). Incorporating ecologically relevant measures of pesticide effect for estimating the compatibility of pesticides and biocontrol agents. Journal of Economic Entomology, 100, 1027–1032.
Statsoft Inc. (2003). Statistica (data analysis software system) version 6. StatSoft, Inc., Tulsa, OK, USA. http://www.statsoft.com.
Sterk, G., Hasssan, S. A., Baillod, M., Bakker, F., Bigler, F., Blümel, S., et al. (1999). Results of the seventh joint pesticide testing programme carried out by the IOBC/WPRS-working group “Pesticides and beneficial organisms”. Biocontrol, 44, 99–117.
Suma, P., & Mazzeo, G. (2008). Laboratory evaluation of pesticide secondary effects on Anagyrus sp. nov. near pseudococci, parasitoid of the citrus mealybug Planococcus citri. IOBC/WPRS Bulletin, 38, 99–103.
Suma, P., Zappalà, L., Mazzeo, G., & Siscaro, G. (2009). Lethal and sub-lethal effects of insecticides on natural enemies of citrus scale pests. Biocontrol, 54, 651–661.
Theiling, K. M., & Croft, B. A. (1988). Pesticide side-effects on arthropod natural enemies: A database summary. Agriculture, Ecosystems & Environment, 21, 191–218.
Triapitsyn, S. V., González, D., Vickerman, D. B., Noyes, J. S., & White, E. B. (2007). Morphological, biological, and molecular comparisons among the different geographical populations of Anagyrus pseudococci (Hymenoptera: Encyrtidae), parasitoids of Planococcus spp. (Hemiptera: Pseudococcidae), with notes on Anagyrus dactylopii. Biological Control, 41, 14–24.
Trjapitzin, S. V., & Trjapitzin, V. A. (1999). Parasites of mealybugs (Homoptera, Pseudococcidae) on cultivated grapes in Argentina, with description of a new species of the genus Aenasius Walker (Hymenoptera, Encyrtidae). Entomological Review, 79, 386–390.
Tsai, C. W., Rowhani, A., Golino, D. A., Daane, K. M., & Almeida, R. P. P. (2010). Mealybug transmission of grapevine leafroll viruses: an analysis of virus–vector specificity. Phytopathology, 100, 830–834.
Viggiani, G., & Tranfaglia, A. (1978). A method for laboratory test of side effects of pesticides on Leptomastix dactylopii (How.) (Hym. Encyrtidae). Bollettino del Laboratorio di Entomologia Agraria Filippo Silvestri, 35, 8–15.
Walton, V. M., Krüger, K., Saccaggi, D. L., & Millar, I. M. (2009). A survey of scale insects (Sternorryncha: Coccoidea) occurring on table grapes in South Africa. Journal of Insect Science, 9, 47.
Walton, V. M., & Pringle, K. L. (1999). Effects of pesticides used on table grapes on the mealybug parasitoid Coccidoxenoides peregrinus (Timberlake) (Hymenoptera: Encyrtidae). South African Journal of Enology and Viticulture, 20, 31–34.
Acknowledgments
We are deeply grateful to Mr. Walid Salhi (Elmoussem Agricole company, Tunis, Tunisia) for providing us with a sample of the biopesticide Prev-Am®. The first author received a grant of study from the AVERROES program (Erasmus Mundus—European Commission).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mansour, R., Suma, P., Mazzeo, G. et al. Evaluating side effects of newer insecticides on the vine mealybug parasitoid Anagyrus sp. near pseudococci, with implications for integrated pest management in vineyards. Phytoparasitica 39, 369–376 (2011). https://doi.org/10.1007/s12600-011-0170-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12600-011-0170-8