Abstract
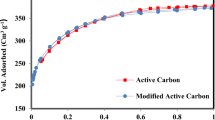
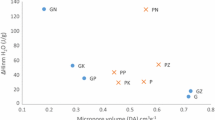
The activated carbon with high surface area was prepared by KOH activation. It was further modified by H2SO4 and HNO3 to introduce more surface functional groups. The pore structure of the activated carbons before and after modification was analyzed based on the nitrogen adsorption isotherms. The morphology of those activated carbons was characterized using scanning electronic microscopy (SEM). The surface functional groups were determined by Fourier transform infrared spectroscopy (FTIR). The quantity of those groups was measured by the Boehm titration method. Cr(VI) removal by the activated carbons from aqueous solution was investigated at different pH values. The results show that compared with H2SO4, HNO3 destructs the original pore of the activated carbon more seriously and induces more acidic surface functional groups on the activated carbon. The pH value of the solution plays a key role in the Cr(VI) removal. The ability of reducing Cr(VI) to Cr(III) by the activated carbons is relative to the acidic surface functional groups. At higher pH values, the Cr(VI) removal ratio is improved by increasing the acidic surface functional groups of the activated carbons. At lower pH values, however, the acidic surface functional groups almost have no effect on the Cr(VI) removal by the activated carbon from aqueous solution.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Barton S.S., Evans M.J.B., Halliop E., and MacDonald J.A.F., Acidic and basic sites on the surface of porous carbon, Carbon, 1997, 35: 1361.
Toles C.A., Marshall W.E., and Johns M.M., Surface functional groups on acid-activated nutshell carbons, Carbon, 1999, 37: 1207.
Chingombe P., Saha B., and Wakeman R.J., Effect of surface modification of an engineered activated carbon on the sorption of 2,4-dichlorophenoxy acetic acid and benazolin from water, J. Colloid Interface Sci., 2006, 297(2): 434.
Zhou H.L., Li D.Y., Gong G.Z., Tian Y.J., and Chen Y.F., Adsorption of soluble metal ions from red mud by modified activated carbon, Key Eng. Mater., 2008, 368–372: 1541.
Zhou H.L., Li D.Y., Tian Y.J., and Chen Y.F., Extraction of scandium from red mud by modified activated carbon and kinetics study, Rare Met., 2008, 27(3): 223.
Raji C. and Anirudhan T.S., Batch cr(vi) removal by polyacrylamide-grafted sawdust: Kinetics and thermodynamics, Water Res., 1998, 32: 3772.
Selvi K., Pattabhi S., and Kadirvelu K., Removal of Cr(VI) from aqueous solution by adsorption onto activated carbon, Bioresour. Technol., 2001, 80(1): 87.
Uysal M. and Ar I., Removal of Cr(VI) from industrial wastewaters by adsorption. Part I: Determination of optimum conditions, J. Hazard. Mater., 2007, 149: 482.
Khezami L. and Capart R., Removal of chromium(VI) from aqueous solution by activated carbons: Kinetic and equilibrium studies, J. Hazard. Mater., 2005, 123(1–3): 223.
Zhao N.Q., Wei N., Li J.J., Qiao Z.J., Cui J., and He F., Surface properties of chemically modified activated carbons for adsorption rate of Cr(VI), Chem. Eng. J., 2005, 115(1–2): 133.
Park S.J. and Jang Y.S., Pore structure and surface properties of chemically modified activated carbons for adsorption mechanism and rate of Cr(VI), J. Colloid Interface Sci., 2002, 249(2): 458.
Aggarwal D., Goyal M., and Bansal R.C., Adsorption of chromium by activated carbon from aqueous solution, Carbon, 1999, 37: 1989.
Zhou H.L., Preparation of activated carbon with high specific surface area and its application [Dissertation], Institute of Process Engineering, Chinese Academy of Sciences, Beijing, 2008: 40.
Boehm H.P., Surface oxides on carbon and their analysis: A critical assessment, Carbon, 2002, 40: 145.
Tamai H., Shiraki K., Shiono T., and Yasuda H., Surface functionalization of mesoporous and microporous activated carbons by immobilization of diamine, J. Colloid Interface Sci., 2006, 295(1): 299.
Abdel-Nasser A. and EI-Hendawy, Variation in the ftir spectra of a biomass under impregnation, carbonization and oxidation conditions, J. Anal. Pyrol., 2006, 75: 159.
Fang J., Gu Z.M., Gang D.C., Liu C.X., Ilton E.S., and Deng B.L., Cr(VI) removal from aqueous solution by activated carbon coated with quaternized poly(4-vinylpyridine), Environ. Sci. Technol., 2007, 41(13): 4748.
Imai A. and Gloyna E.F., Effects of ph and oxidation state of chromium on the behavior of chromium in the activated sludge, Water Res., 1990, 24(9): 1143.
Dionex, Determination of Cr(VI) in Water, Wastewater and Solid Waste Extracts, Dionex Technical Note TN26, 1998, http://www.dionex.com/en-us/documents/technical-notes/ic-hplc/lp-72156.html
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhou, H., Chen, Y. Effect of acidic surface functional groups on Cr(VI) removal by activated carbon from aqueous solution. Rare Metals 29, 333–338 (2010). https://doi.org/10.1007/s12598-010-0059-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12598-010-0059-6