Abstract
A 9-week feeding trial was conducted to evaluate the optimum dietary level of PROTIDE (PRO), a single-cell protein obtained from the bacteria Corynebacterium ammoniagenes, as a substitute for fish meal in the diet of whiteleg shrimp Litopenaeus vannamei. Five diets were formulated that replaced fish meal at 0% (PRO0), 10% (PRO2), 20% (PRO4), 30% (PRO6) and 40% (PRO8). Fifty shrimp averaging 0.15 ± 0.02 g (mean ± SD) body weight were randomly distributed between 20 experimental tanks and fed one of the five experimental diets. At the end of the experiment, final weight, weight gain, specific growth rate and feed conversion ratio of shrimp fed PRO0 and PRO2 diets were significantly improved compared to those fed PRO6 and PRO8 diets (P < 0.05). The proximate composition of muscle and the whole body indicated an increase in crude protein content with an increase in dietary PRO level. These results suggest that, for whiteleg shrimp, the optimum dietary level of PRO when used as a replacement for fish meal should be greater than 10% (PRO2) but less than 20% (PRO4) without any additional dietary amino acid supplementation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The whiteleg shrimp Litopenaeus vannamei is the most important cultivated shrimp species in the world and has the highest value of all traded crustacean products. The global production of whiteleg shrimp increased from 154,515 tons in 2000 to 4,155,827 tons in 2016 with more than an 18 billion US dollars (USD) increase in its aquacultural value during that time (FAO 2018). This species has several merits that make it more suitable for aquaculture than other penaeid shrimps, such as a relatively low dietary protein requirement, high density tolerance, and adaptability to wide ranges of salinity and temperature (Argue et al. 2002; Lightner et al. 2009; Moss et al. 2001, 2007, 2010).
The significant growth in shrimp aquaculture has led to a corresponding expansion of feed production for shrimp. Commercial shrimp feed formulations commonly include between 25 and 50% fish meal (FM), representing the primary and most expensive protein ingredient of these products (Gonzalez-Rodriguez and Abdo de la Parra 2004). In the mid-1990s, FM cost ca. 500 USD per million tons, but as of May 2018 the average cost had risen to 1,510 USD per million tons (World Bank Commodity 2018). The economic success of the aquaculture industry depends in part on a reduction in the use of FM in fish feed (Choi et al. 2004). Consequently, fish nutritionists have conducted several studies to evaluate the efficiency of various conventional and unconventional protein sources when used as substitutes for FM in fish feed. Some of these studies evaluated the substitution of FM by single-cell protein (SCP) in fish and shrimp diets (Beck et al. 1979; Davies and Wareham 1988; Lara-Flores et al. 2003; Glencross et al. 2014; Vizcaíno et al. 2014) and found that SCPs have significant potential for use in aquafeed formulations.
SCPs are produced from single-cell microorganisms (e.g. bacteria, yeast, fungi, and algae) that utilize different carbon sources for growth (Ravinda 2000). SCPs are commonly sourced from the wastes produced by other agricultural industries including cereal husks, crop peel, cocoa processing wastes, sugar cane (bagasse), citrus, mango and coconut waste, etc. Some of the wastes comprise lignocelluloses, recalcitrant compounds that can resist enzymatic and microbial treatments (Karimi and Taherzadeh 2016). These wastes are of public and environmental concern due to the large quantities in which they are produced and the difficulty of their disposal. Converting these wastes into SCPs serves the dual purposes of mitigating degradation of the environment from waste accumulation and providing a good protein source for fish diets (Palmegiano et al. 2005). Bacteria present a promising source of SCP due to their rapid growth in various substrates, high protein value, and cellular vitamins and photopigments (Kantachote et al. 2005; Kornochalert et al. 2014; Chumpol et al. 2018). PROTIDE (PRO) is a new SCP concentrate obtained from the bacteria Corynebacterium ammoniagenes by a specific fermentation process (patented by CJ Cheiljedang, Republic of Korea). The nutrient profile of PRO closely resembles that of Peruvian and menhaden FM with a crude protein content of 64.7% on a dry matter basis. Moreover, the production of PRO does not rely on finite marine resources, which is a significant hurdle to sustainable aquaculture.
Given its good nutritional properties, PRO is of particular research interest for use as a FM substitute. However, it has yet to be evaluated for its potential in whiteleg shrimp diets. Therefore, the present study was conducted to evaluate the effects of a dietary SCP (PRO) as a substitute for FM, and its effects on the growth, whole-body proximate composition and amino acid content of whiteleg shrimp L. vannamei.
Materials and methods
Experimental design and diet preparation
Commercially available PRO was provided by CJ Cheiljedang (http://www.cjbio.net/html/product/application.asp). The Gram-positive aerobic bacteria C. ammoniagenes undergoes several fermentation processes with inosine-5′-monophosphate until the final product, PRO, is achieved (Fig. 1). The proximate composition and formulation of the diets are presented in Table 1. Five isonitrogenous (45% crude protein) and isocaloric (16.4 kJ gross energy/g diet) experimental diets were formulated to contain 0 (PRO0), 2 (PRO2), 4 (PRO4), 6 (PRO6) and 8 % (PRO8) PRO to replace 0, 10, 20, 30 and 40% FM, respectively. These amounts were based on the results of a previous study (Chumpol et al. 2018). The proximate composition analysis and the amino acid profile of dietary PRO and FM are summarized in Tables 2 and 3, respectively. FM and dehulled soybean meal were included as the major protein sources, fish and soybean oil were the main lipid sources, while carbohydrates derived from wheat meal and corn starch were used to adjust the energy content of the diet.
Diet preparation and storage followed Bai and Kim (1997). First, dry ingredients were mixed and then soybean oil, fish oil, and water (30% of diet) were added to the mixture to form a paste. A laboratory mincing machine (Shinsung, Seoul) was used to obtain uniform pellets of 0.6-mm and 1.5-mm diameter. The feed pellets were dried and stored at − 20 °C until use.
Experimental shrimp and feeding trial
Post-larval whiteleg shrimp were transported from the Tae-an hatchery of the National Fisheries Research and Development Institute to the Feeds and Foods Nutrition Research Centre, Pukyong National University, Busan, Republic of Korea for the feeding trial. All the shrimp were fed a commercial shrimp diet (DongA One, Seoul) for 1 week for acclimation to the experimental conditions. Fifty shrimp with an initial average weight of 0.15 ± 0.02 g (mean ± SD) were randomly distributed between 20 tanks. Each tank was then randomly assigned to one of the four replicated treatments for five experimental diets. Shrimp were fed four times per day (at 0900, 1200, 1500 and 1800 hours) for 9 weeks at a fixed rate of 7% of body weight per day. Total shrimp weight in each experimental tank was determined every 3 weeks and the feeding rate was adjusted accordingly. This feeding trial was conducted using a semi-recirculating system, supplied with filtered seawater at the rate of 0.8 l min−1 from a reservoir tank (8 tons). Aeration was provided to maintain dissolved oxygen at 6.0 ± 0.5 mg l−1. Water temperature, salinity and pH during the experiment were maintained at 27 ± 0.5 °C, 32 ± 1 practical salinity units and 7.5 ± 0.3, respectively. Each rearing tank was siphoned every day in the morning, and dead shrimp removed, weighed, and recorded daily.
Sample collection and analyses
When the feeding trial was completed, all the shrimp from the experimental tanks were weighed and counted in order to calculate weight gain (WG), specific growth rate (SGR), feed conversion ratio (FCR), protein efficiency ratio (PER) and survival.
After the final weighing, six shrimp were randomly collected from each tank and frozen at − 20 °C for whole-body and muscle proximate analysis. Muscle samples were collected by removing the exoskeleton, head, telson and other internal organs. Proximate composition analyses of experimental diets and shrimp body were performed using the standard methods of the Association of Official Analytical Chemists (AOAC) (2005). Samples of diets and shrimp were dried at 105 °C to a constant weight to determine their moisture content. Ash content was determined by incineration at 550 °C, crude protein by the Kjeldahl method (nitrogen × 6.25) after acid digestion and crude lipid by Soxhlet extraction using the Soxhlet system 1046 (Tacator, Hoganas, Sweden). Amino acid analyses for PRO, fish meal and shrimp muscle were performed according to Macchi et al. (2000). Freeze-dried samples (0.02 g) were hydrolysed with 15 ml of 6 N HCl at 110 °C for 24 h. Hydrolysed samples were evaporated, recovered in sodium citrate buffer (0.2 N, pH 2.2) and filtered (0.2 μm). Following ninhydrin reaction, sample absorbance was measured at 570 nm and 440 nm using a S433 amino acid analyser (Sykam, Eresing, Germany). Samples were hydrolysed using performic acid for methionine and cystine analysis.
Sample collection and analyses
Assumptions for statistical tests such as normality and homogeneity of variances were checked. Data were analysed according to one-way ANOVA to test the effects of the dietary treatments. When significant differences were observed in the treatments, a least significant difference test for multiple comparisons was performed. Treatment effects were considered significant at the P < 0.05 level. All statistical analyses were carried out by SAS version 9.0 software (SAS Institute, Cary, NC).
Results
Growth performance
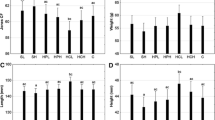
The growth performance and survival of whiteleg shrimp fed the five experimental diets for 9 weeks are shown in Table 4. At the end of the feeding trial, the weights of shrimp fed the PRO2 diet were significantly higher than those fed the PRO6 and PRO8 diets (P < 0.05). However, there were no significant differences in the final weight among shrimp fed the PRO0, PRO2 and PRO4 diets, among shrimp fed the PRO0, PRO4 and PRO6 diets, or among those fed the PRO4, PRO6 and PRO8 diets (P > 0.05). WG and SGR of shrimp fed the PRO0 and PRO2 diets were significantly higher than those fed PRO6 and PRO8 diets. There were no significant differences in WG and SGR among shrimp fed the PRO0, PRO2 and PRO4 diets, or among those fed the PRO4, PRO6 and PRO8 diets. The FCR was significantly higher for shrimp fed the PRO4, PRO6 and PRO8 diets compared to those fed the PRO0 and PRO2 diets, but the differences in FCR were not significant between shrimp fed the PRO0 and PRO2 diets. PER was significantly higher for shrimp fed the PRO0 and PRO2 diets than for those fed the PRO4, PRO6, and PRO8 diets. However, there were no significant differences in PER among shrimp fed the PRO4, PRO6 and PRO8 diets. The survival of shrimp fed the PRO6 and PRO8 diets was significantly higher than that of shrimp fed the PRO0 and PRO2 diets. However, there were no significant differences in survival among shrimp fed the PRO0, PRO2 and PRO4 diets or among those fed the PRO4, PRO6 and PRO8 diets.
Whole-body and muscle proximate composition
Table 5 shows the proximate composition of whiteleg shrimp fed the five experimental diets for 9 weeks. Whole-body crude protein contents of shrimp fed PRO4, PRO6 and PRO8 diets were significantly higher than those of shrimp fed the control diet. However, there were no significant differences among shrimp fed PRO4, PRO6 and PRO8 diets, among shrimp fed PRO2, PRO4 and PRO8 diets, or among those fed PRO0 and PRO2 diets. Furthermore, the muscle protein contents of PRO0 and PRO2 groups were significantly lower than those of all other groups.
Whole-body crude lipid contents of shrimp fed the PRO8 diet were significantly higher than those of shrimp in all the other dietary treatments. The control group showed the lowest whole-body lipid content, although this was not significantly different from the content in the PRO4 group. However, the muscle lipid contents of shrimp fed the PRO0 and PRO2 diets were significantly higher than those of shrimp fed the PRO2, PRO6 and PRO8 diets.
The whole-body crude ash contents of shrimp fed the PRO6 and PRO8 diets were significantly higher than those fed the PRO2 and PRO4 diets. However, there were no significant differences among shrimp fed the PRO0, PRO6 and PRO8 diets, among shrimp fed the PRO0 and PRO4 diets, or among those fed the PRO2 and PRO4 diets. Furthermore, the muscle ash contents of shrimp fed the PRO2 and PRO4 diets were significantly higher than those fed the PRO6 and PRO8 diets. Whole-body and muscle moisture contents did not show any significant differences among shrimp fed any of the experimental diets.
Muscle amino acid composition
The muscle amino acid compositions of whiteleg shrimp are presented in Table 6. Although significant differences were found for some of the amino acids, no clear trends were observed. Serine, glycine and alanine levels of shrimp fed the PRO8 diet were significantly higher than levels of those fed the PRO0, PRO2, PRO4 and PRO6 diets. The levels of valine, isoleucine, leucine, phenylalanine and lysine of shrimp fed the PRO0, PRO2, PRO4 and PRO6 diets were significantly higher than those of shrimp fed the PRO8 diet. Also, the glutamine and proline levels of shrimp fed the PRO4 and PRO6 diets were significantly higher than those of shrimp fed the PRO0, PRO2 and PRO8 diets. The asparagine level of shrimp fed the PRO4 diet was significantly higher than that of shrimp fed the PRO8 diet. Furthermore, the cysteine level of shrimp fed the PRO6 diet was significantly higher than that of shrimp fed the PRO0 diet. However, there were no significant differences in threonine, tyrosine and methionine levels among shrimp fed the different experimental diets.
Discussion
In the present study, the formulation and proximate composition of the diets are comparable to those of commercial diets used for whiteleg shrimp. Also, the proximate and amino acid compositions of dietary PRO closely resemble those of dietary fish meal (Tables 2, 3), as indicated by the results obtained for growth and survival with no visibly deleterious effects on shrimp health. During the experiment, shrimp readily consumed all the feed, indicating that the experimental diets were palatable to them.
Research on the potential use of SCP in fish diets started in the late 1970s (Beck et al. 1979; Bergstrom 1979; Dabrowski et al. 1980; Davies and Wareham 1988); because of the encouraging results obtained, research is still ongoing in this field. In Mozambique tilapia Oreochromis mossambicus and sea bass Dicentrarchus labrax up to 40% and 50% FM was replaced with an industrial SCP and brewer’s yeast, respectively (Davies and Wareham 1988; Oliva-Teles and Goncalves 2001). Gamboa-Delgado et al. (2016) used torula yeast Candida utilis as a protein source in the diet of whiteleg shrimp and succeeded in replacing 60% of the FM with this. Replacement of up to 45% FM with yeast extract in the diet of L. vannamei had no adverse effects on the digestibility of the feed or the growth of the shrimp (Zhao et al. 2015). Although few studies have investigated bacteria as a SCP in the diet of shrimp, it has been well proven that other types of SPC can have a positive effect on their immune system and growth (Biswas et al. 2012; Deng et al. 2013). Viola and Zohar (1984) found that a bacterial SCP, Pruteen, which was used to replace 50% of the dietary FM protein for hybrid tilapia Oreochromis niloticus × Oreochromis aureus, was beneficial to its immune system and growth. Other studies have used a SCP to replace a smaller percentage of FM in feed. In a recent study, Rosales et al. (2017) reported that SCP (dried yeast) can be used to replace up to 50% of the crude protein (40%) provided by half FM and half soybean meal in the diet of red drum Sciaenops ocellatus; in effect, SCP replaced approximately 10% of the FM in this experiment. Our experiment showed that up to 20% of the fish meal in a practical diet for whiteleg shrimp could be effectively replaced by PRO, a SCP derived from the bacteria Corynebacterium ammoniagenes, without any significant reduction in WG. In accordance with our results, Lunger et al. (2006) concluded that 25% of feed FM could be replaced with SCP without growth reduction in cobia Rachycentron canadum. However, 15% of yeast-based SCPs in the diet of Red-Stirling tilapia Oreochromis niloticus (Ribeiro et al. 2014) and gray mullet Mugil cephalus (Luzzana et al. 2005) resulted in growth reduction. The adverse effects on growth in our and several other experiments that used SCPs as FM substitutes could be related to differences in target species, sources of SCP, feed formulations, and physicochemical conditions of the experiments. A more accurate comparison could be made by knowing the cost of each diet before and after FM substitution.
In the present study, an increasing trend of survival was observed with increasing PRO in the diet. To the best of our knowledge, only a few studies have observed significant relationships between bacterial SCPs and fish/shrimp survival. Christensen et al. (2003) found that application of whole-cell bacterial SPC had a statistically significant impact on a specific systemic immune response in mice. Chumpol et al. (2018) also showed that administration of bacterial SPC in the diet of L. vannamei increases its innate immunity, as evidenced by elevated superoxide dismutase and phenoloxidase activities. Therefore, in the present study, the observed trend of increased survival may have been due to the amplification of non-specific immune responses by PRO. According to the results of our study, up to 20% of FM can be substituted by PRO without a significant drop in growth factors such as WG, SGR and FW. However, substitution of more than 10% FM caused a reduction in PER and increased the FCR. Similar effects have been observed in other FM replacements studies (e.g. El-Sayed 1998) when replacing FM with shrimp meal, blood meal, meat and bone meal and poultry by-product meal, as the PER and FCR were significantly slowed down. PER is calculated from fish body mass gain and intake of protein, and defines the quality of protein in feed. In contrast, FCR defines the efficiency of converting feed into fish body mass (NRC 2011). Both of these values are closely related to the quality, digestibility and utilisation of feed. It is likely that there are multiple factors that influence these parameters in diets with a higher SCP content. For example, it was previously suggested that the cell walls of SCP are the main cause of low nitrogen digestibility in feed (Yamada and Sgarbieri 2005). Tlusty et al. (2017) replaced 55% of FM with a specific bacterial SCP from Methylobacterium extorquens in the diet of Atlantic salmon Salmo salar and reported lower digestibility of the SCP-containing diet.
The whole-body and muscle proximate composition of shrimp in this study did not seem to show a specific trend in response to different levels of dietary PRO, although the whole-body lipid content of shrimp fed 8.0% PRO was higher than that of shrimp fed all the other diets. Similar results were reported for red drum (Rosalesa et al. 2017), hybrid striped bass (Gause and Trushenski 2011) and rainbow trout (Hauptman et al. 2014) when FM was replaced with SCPs. This could have been related to the retention of lipids, and lipid storage, in the body of shrimp due to an imbalance in the protein composition of the PRO. In the present study, the muscle amino acid composition of whiteleg shrimp corresponded to the amino acid composition of PRO and FM based on dietary inclusion levels. In contrast, significantly lower levels of essential amino acids such as arginine, histidine, isoleucine, lysine, leucine, phenylalanine and valine were retained in the muscle of whiteleg shrimp fed higher amounts of PRO. This could indicate that the amino acid requirements of shrimp were not met by the higher level of PRO in the diet, as evidenced by shrimp growth reduction. Methionine and cysteine contents in the whole body of finfish and shellfish have been reported to be adversely affected in various fish meal replacement studies (Xie et al. 2016). Therefore, additional dietary supplementation of these amino acids in fish meal-replacement feed has been recommended as the justified approach to maintain their profile in shrimp. However, in the present investigation, none of the experimental diets were formulated with additional supplementation of methionine or cysteine. In order to achieve a higher percentage substitution of FM, future research should focus on the efficacy of methods that can be used to increase the digestibility of SCP (cell wall disruption) and provide essential amino acids in the diet at higher replacement levels. Most of the works reviewed here have evaluated FM replacements from a biological or nutritional point of view, whereas economic analyses of these protein sources have scarcely been undertaken. Therefore, it is highly recommended that future experiments should also focus on the economic consequences of FM replacement by SPCs.
In conclusion, the results of the present experiment indicate that the optimum level of PRO, as a replacement for fish meal, should be greater than 10% but less than 20% for whiteleg shrimp culture, without any additional dietary amino acid supplementation. PRO, derived from the bacteria C. ammoniagenes, can be recommended as a SCP for shrimp feed, providing farmers with an under-utilized and cost-effective protein source.
Change history
22 April 2024
A Correction to this paper has been published: https://doi.org/10.1007/s12562-024-01783-0
References
AOAC (2005) Official methods of analysis, 18th edn. Association of Official Analytical Chemists, Gaithersburg
Argue BJ, Arce SM, Lotz JM, Moss SM (2002) Selective breeding of Pacific white shrimp (Litopenaeus vannamei) for growth and resistance to Taura syndrome virus. Aquaculture 204:447–460
Bai SC, Kim KW (1997) Effects of dietary animal protein sources on growth and body composition in Korean rockfish, Sebastes schlegeli. J Aquac 10:77–85 (In Korean with English abstract)
Beck H, Gropp J, Koops H, Tiews K (1979) Single cell protein in trout diets. In: Halver JE, Tiews K (eds) Finfish nutrition and fish feed technology, 2nd edn. Heenemann, Berlin, pp 269–280
Bergstrom E (1979) Experiments on the use of single cell proteins in Atlantic salmon diets. In: Halver JF, Tiews K (eds) Finfish nutrition and fish feed technology. Heenemann, Berlin, pp 105–116
Biswas G, Korenaga H, Nagamine R, Kono T, Shimokawa H, Itami T, Sakai M (2012) Immune stimulant effects of a nucleotide-rich baker’s yeast extract in the kuruma shrimp, Marsupenaeus japonicus. Aquaculture 366:40–45
Choi SM, Wang XJ, Park GJ, Lim SR, Kim KW, Bai SC, Shin IS (2004) Dietary dehulled soybean meal as a replacement for fish meal in fingerling and growing olive flounder, Paralichthys olivaceus. Aquac Res 35:410–418
Christensen H, Larsen L, Frøkiær H (2003) The oral immunogenicity of BioProtein, a bacterial single-cell protein, is affected by its particulate nature. Br J Nutr 90:169–178
Chumpol S, Kantachote D, Nitoda T, Kanzaki H (2018) Administration of purple nonsulfur bacteria as single cell protein by mixing with shrimp feed to enhance growth, immune response and survival in white shrimp (Litopenaeus vannamei) cultivation. Aquaculture 489:85–95
Dabrowski K, Hassard S, Quinn J, Pitcher TJ, Flinn AM (1980) Effect of Geotrichum candidum protein substitution in pelleted fish feed on the growth of rainbow trout (Salmo gairdneri Rich.) and on utilization of the diet. Aquaculture 21:213–232
Davies SJ, Wareham H (1988) A preliminary evaluation of an industrial single cell protein in practical diets for Mozambique tilapia (Oreochromis mossambicus, Peters). Aquaculture 73:189–199
Deng D, Mei C, Mai K, Tan BP, Ai Q, Ma H (2013) Effects of a yeast-based additive on growth and immune responses of white shrimp, Litopenaeus vannamei (Boone, 1931), and aquaculture environment. Aquac Res 44:1348–1357
El-Sayed A-FM (1998) Total replacement of fish meal with animal protein sources in Nile tilapia, Oreochromis niloticus (L.), feeds. Aquac Res 29:275–280
FAO (2018) FISHSTAT Plus, universal software for fishery statistical time series. Food and Agriculture Organization, United Nations, Rome (Electronic webpage)
Gamboa-Delgado J, Fernández-Díaz B, Nieto-López M, Cruz-Suárez LE (2016) Nutritional contribution of torula yeast and fish meal to the growth of shrimp Litopenaeus vannamei as indicated by natural nitrogen stable isotopes. Aquaculture 453:116–121
Gause B, Trushenski J (2011) Replacement of fish meal with ethanol yeast in the diets of sunshine bass. N Am J Aquac 73:97–103
Glencross B, Irvin S, Arnold S, Blyth D, Bourne N, Preston N (2014) Effective use of microbial biomass products to facilitate the complete replacement of fishery resources in diets for the black tiger shrimp, Penaeus monodon. Aquaculture 431:12–19
Gonzalez-Rodriguez B, Abdo de la Parra I (2004) Replacement of fish meal with co-extruded wet tuna viscera and corn meal in diets for white shrimp (Litopenaeus vannamei Boone). Aquac Res 35:1153–1157
Hauptman BS, Barrows FT, Block SS, Gaylord TG, Paterson JA, Rawles SD, Sealey WM (2014) Evaluation of grain distillers dried yeast as a fish meal substitute in practical-type diets of juvenile rainbow trout, Oncorhynchus mykiss. Aquaculture 432:7–14
Kantachote D, Torpee S, Umsakul K (2005) The potential use of anoxygenic phototrophic bacteria for treating latex rubber sheet wastewater. Electron J Biotechnol 8:314–323
Karimi K, Taherzadeh MJ (2016) A critical review on analysis in pretreatment of lignocelluloses: degree of polymerization, adsorption/desorption, and accessibility. Bioresour Technol 203:348–356
Kornochalert N, Kantachote D, Chaiprapat S, Techkarnjanaruk S (2014) Use of Rhodopseudomonas parustris P1 stimulated growth by fermented pineapple extract to treat latex rubber sheet wastewater to obtain single cell protein. Ann Microbiol 64:1021–1032
Lara-Flores M, Olvera-Novoa MA, Guzman-Menedez RE, Lopez-Madrid W (2003) Use of the bacteria Streptococcus faecium and Lactobacillus acidophilus, and the yeast Saccharomyces cerevisiae as growth promoters in Nile tilapia (Oreochromis niloticus). Aquaculture 216:193–201
Lightner DV, Redman RM, Arce SM, Moss MS (2009) Specific pathogen free shrimp stocks in shrimp farming facilities as a novel method for disease control in crustaceans. In: Shumway SE, Rodrick GE (eds) Shellfish safety and quality. Woodhead, Cambridge, pp 384–424
Lunger AN, Craig SR, Mclean E (2006) Replacement of fish meal in cobia (Rachycentron canadum) diets using an organically certified protein. Aquaculture 257:393–399
Luzzana U, Valfrè F, Mangiarotti M, Domeneghini C, Radaelli G, Moretti VM, Scolari M (2005) Evaluation of different protein sources in fingerling grey mullet Mugil cephalus practical diets. Aquac Int 13:291–303
Macchi FD, Shen FJ, Keck RG, Harris RJ (2000) Amino acid analysis, using postcolumn ninhydrin detection, in a biotechnology laboratory. Methods Mol Biol 159:9–30
Moss SM, Arce SM, Argue BJ, Otoshi CA, Calderon FRO, Tacon AGJ (2001) Greening of the blue revolution: efforts toward environmentally responsible shrimp culture. In: Browdy CL, DE Jory (eds) The new wave: proceedings of the special session on sustainable shrimp farming. World Aquaculture Society, Baton Rouge, pp 1–19
Moss DR, Arce SA, Otoshi CA, Doyle RW, Moss SM (2007) Effects of inbreeding on survival and growth of Pacific white shrimp (Litopenaeus vannamei). Aquaculture 27S1:S30–S37
Moss SM, Moss DR, Otoshi CA, Arce SM (2010) An integrated approach to sustainable shrimp farming. Asian Fish Sci 23:591–605
NRC (National Research Council) (2011) Nutrient requirements of fish and shrimp. National Academies, Washington
Oliva-Teles A, Goncalves P (2001) Partial replacement of fishmeal by brewer’s yeast (Saccaromyces cerevisae) in diets for sea bass (Dicentrarchus labrax) juveniles. Aquaculture 202:269–278
Palmegiano GB, Agradi E, Forneris G, Gai F, Gasco L, Rigamonti E, Sicuro B, Zoccarato I (2005) Spirulina as a nutrient source in diets for growing sturgeon (Acipenser baeri). Aquac Res 36:188–195
Ravinda AP (2000) Value-added food: single cell protein. Biotechnol Adv 18:459–479
Ribeiro CS, Moreira RG, Cantelmo OA, Esposito E (2014) The use of Kluyveromyces marxianus in the diet of red-stirling tilapia (Oreochromis niloticus, Linnaeus) exposed to natural climatic variation: effects on growth performance, fatty acids, and protein deposition. Aquac Res 45:812–827
Rosalesa M, Castilloa S, Pohlenz C, Gatlin DM III (2017) Evaluation of dried yeast and threonine fermentation biomass as partial fish meal replacements in the diet of red drum Sciaenops ocellatus. Anim Feed Sci Technol 232:190–197
Tlusty M, Rhyne A, Szczebak JT, Bourque B, Bowen JL, Burr G, Marx CJ, Feinberg L (2017) A transdisciplinary approach to the initial validation of a single cell protein as an alternative protein source for use in aquafeeds. PeerJ 3170:1–19
Viola S, Zohar G (1984) Nutrition studies with market size hybrids of tilapia (Oreochromis niloticus) in intensive culture. Bamidgeh 36:3–15
Vizcaíno AJ, López G, Sáez MI, Jiménez JA, Barros A, Hidalgo L, Camacho-Rodríguez J, Martínez TF, Cerón-García MC, Alarcón FJ (2014) Effects of the microalga Scenedesmus almeriensis as fishmeal alternative in diets for gilthead sea bream, Sparus aurata, juveniles. Aquaculture 431:34–43
World Bank Commodity (2018) Commodity price data of the World Bank. Washington DC. http://www.worldbank.org/en/research/commodity-markets. Accessed 2018
Xie S, Liu Y, Zeng S, Niu J, Tian L (2016) Partial replacement of fish-meal by soy protein concentrate and soybean meal based protein blend for juvenile Pacific white shrimp, Litopenaeus vannamei. Aquaculture 464:296–302
Yamada E, Sgarbieri AVC (2005) Yeast (Saccharomyces cerevisiae) protein concentrate: preparation, chemical composition, and nutritional and functional properties. J Agric Food Chem 53:3931–3936
Zhao L, Wang W, Huang X, Guo T, Wen W, Feng L, Wei L (2015) The effect of replacement of fish meal by yeast extract on the digestibility, growth and muscle composition of the shrimp Litopenaeus vannamei. Aquac Res 48:311–320
Acknowledgements
This study was supported by the Environmental-friendly Aquaculture Technology using Biofloc project (R20160218) of the National Institute of Fisheries Science, the Animal Biosciences Research Institute, CJ CheilJedang and the Feeds and Foods Nutrition Research Center, Pukyong National University.
Author information
Authors and Affiliations
Corresponding author
Additional information
The original online version of this article was revised for retrospective open access order.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hamidoghli, A., Yun, H., Won, S. et al. Evaluation of a single-cell protein as a dietary fish meal substitute for whiteleg shrimp Litopenaeus vannamei. Fish Sci 85, 147–155 (2019). https://doi.org/10.1007/s12562-018-1275-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12562-018-1275-5