Abstract
Noroviruses (NoVs) are a leading cause of epidemic and sporadic acute gastrointestinal illness globally. These viruses can potentially contaminate rural private wells and non-community drinking water systems, and cause waterborne disease outbreaks related to consumption of contaminated ground water. Detection of NoVs in water samples can be challenging because they are genetically and antigenically diverse, and noncultivable. In the present study, the detection limits of a novel broadly reactive GI assay and an existing GII NoV real-time TaqMan reverse transcriptase-polymerase chain reaction (RT-qPCR) assay in ground water concentrates was determined. Ground water samples (50 l) from two sources (Lawrenceville, GA and Gainesville, FL, USA) were seeded with electron microscopy-enumerated and RT-qPCR quantified NoV and concentrated using hollow-fiber ultrafiltration (UF) followed by either polyethylene glycol (PEG) precipitation or microconcentrators. Detection limits for GI NoV ranged from 1 × 104 (GA source) to 2 × 105 (FL source) virus particles in 50 l water samples (corresponding to 200–3,000 particles/l) and 5 × 104 (GA source) to 5 × 105 (FL source) virus particles (corresponding to 1,000–10,000 particles/l) for GII NoV. The reported UF method, sample processing procedures, and RT-qPCR assays should be effective tools for sensitive detection of NoVs in large-volume water samples.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Noroviruses (NoVs) are a leading cause of epidemic and sporadic acute gastrointestinal illness (AGI) globally. The virus can be transmitted through several different transmission routes, including person-to-person, food, water, and contaminated environments (Glass et al. 2009). In 2006, NoVs were identified as the etiologic agent in three of the six AGI outbreaks associated with drinking water that were reported to the US Centers for Disease Control and Prevention (Yoder et al. 2008). NoVs are especially of concern for rural private wells and non-community drinking water systems, which have frequently been associated with waterborne disease outbreaks (WBDOs) related to consumption of contaminated ground water (Liang et al. 2006; O’Reilly et al. 2007; Yoder et al. 2008). Detection of NoVs in environmental samples can be challenging because NoVs are a genetically diverse group of viruses that can be divided into at least five genogroups (GI-V) of which GI, II, and IV have been shown to infect humans whereas GIII and GV have been detected in ruminants and mice, respectively.
NoVs have been suspected to be a major cause of waterborne AGI illness in the US for many years, including suspicion that they have represented a substantial proportion of the “unidentified AGI” WBDO reports since the 1970s (Kaplan et al. 1982; Huffman et al. 2003). Since the first report of a NoV TaqMan realtime RT-PCR assay in 2003 (Kageyama et al. 2003), numerous research groups have reported similar assays targeting the ORF1–ORF2 junction region (Cannon and Vinje 2008). This assay is now being employed in most clinical and environmental virology laboratories (Kageyama et al. 2003; Jothikumar et al. 2005; Wolf et al. 2007) and has also helped in identifying NoV as the etiologic agent in waterborne outbreaks (Jothikumar et al. 2005; Rutjes et al. 2006; Wolf et al. 2007).
Our group previously reported a reverse transcriptase-real-time polymerase chain reaction (RT-qPCR) assay that has been used widely for detection of NoVs in water and food samples by other researchers (Baert et al. 2008; Ngazoa et al. 2008; Park et al. 2008). However, the GI assay was found to be less sensitive than another RT-qPCR assay for detecting GI.3 NoV (Gentry et al. 2009b). In the present study, we validated a novel GI NoV real-time RT-qPCR assay on a panel of all NoV GI genotypes and on 50 l ground water samples processed using hollow-fiber ultrafiltration (UF).
Methods and Materials
Virus Strains and RNA Transcripts
A panel of 33 archived fecal samples including strains from 8 NoV GI genotypes (GI.1-8) were used to evaluate the novel GI NoV RT-qPCR assay developed in this study. The samples were collected during outbreaks of acute gastroenteritis from 1998 to 2008 and have been stored as whole stool at 4 or −70°C. In addition, 17 samples containing other enteric viruses [Coxsackievirus A8, hepatitis A virus (strain HM-175), sapovirus (genogroup I), hepatitis E virus (genotype 2), 12 GII NoV specimens, and murine norovirus] were included. To determine the detection limit of the novel GI RT-qPCR assay, a 10-fold serial dilution of a quantified GI.3b RNA transcript (Gentry et al. 2009a) was included in all experiments.
For use in seeding ground water samples, two clarified stool extracts (GI.4 and GII.4) were enumerated by electron microscopy. Briefly, 20% fecal suspensions in PBS were clarified by centrifugation at 2,500×g for 30 min. Viruses were concentrated by ultracentrifugation at 100,000×g for 90 min, resuspended in PBS and a 400 mesh formvar and carbon-coated grid was floated on 25 μl of virus for 15–60 min at room temperature. Any excess fluid was blotted and 25 μl of 2% (w/v) phosphotungstic acid (pH 6.5) was added. The grid was air dried and analyzed under the electron microscope (50,000× magnification). NoV particles were estimated based on counts made from three images per sample.
Development of Novel GI Norovirus RT-qPCR Assay
The previous reported GI assay had a relative low sensitivity for GI.1, GI.3b, and GI.6 viruses (Jothikumar et al. 2005). Therefore, the forward primer was modified (Table 1) which increased the GC percentage of the seven bases at the 3′ end from 43 to 57%, corresponding to an increase of the dG value from −9.5 to −12.2 kCal/mol. The novel GI TaqMan probe [RING1(c)] was designed based on combining both RING1(a) and RING1(b) probes (Kageyama et al. 2003; Rolfe et al. 2007) (Table 1). For NoV GII detection we used the oligonucleotide primers and TaqMan probe described previously (Jothikumar et al. 2005).
TaqMan Real-Time RT-PCR Conditions
All samples and dilutions were tested in duplicate on an iCycler iQ5 Real-time PCR Detection System (Bio-Rad, Hercules, CA). Reactions were carried out in a 20 μl final reaction mixture using the QuantiTect probe RT-PCR kit (Qiagen, Carlsbad, CA), 2 μl of template RNA, 400 nM (GI assay) or 250 nM (GII assay) of forward and reverse oligonucleotide primers, 200 nM of FAM-labeled RING1(c) probe (or 100 nM of FAM-labeled JJV1P probe), 0.4 μl of 50× non-acetylated BSA (Sigma-Aldrich, St. Louis, MO), and 1.0 μl of 20× gp32 (New England Biolabs, Ipswitch, MA). In each experiment, 10-fold serially diluted GI.3b or GII.4 RNA transcripts were included as positive controls and water as a negative control. Cycling conditions included: (i) reverse transcription for 30 min at 50°C, (ii) 15 min at 95°C to activate Taq polymerase, and (iii) 45 cycles of 10 s at 95°C, 30 s at 55°C, and 15 s at 72°C.
Water Samples
Eighteen 50 l untreated ground water samples were obtained in cubitainers from two different locations between July and September 2008. The ground water samples were collected from a fractured crystalline rock aquifer (Source 1) used as source water by the City of Lawrenceville, GA (USA) water treatment plant and the Floridan aquifer system (Source 2) used as source water by the Gainesville, FL (USA) water utility plant. All water samples were stored at 4°C and used in NoV recovery experiments within 2 weeks of collection.
The physico-chemical quality of the water samples was characterized within 2 days of sampling using the following water quality parameters: temperature, pH, turbidity, specific conductance (SC), alkalinity, total hardness, total organic carbon (TOC). Temperature, pH, turbidity, SC, alkalinity, and TOC testing was performed as described previously (Hill et al. 2007). Total hardness was measured using the Hach total hardness test method 8213 and AL-DT digital titrator. In addition, Escherichia coli were enumerated in water samples by membrane filtration and modified mTEC agar according to EPA method 1603.
Sample Processing for Ultrafiltration Experiments
The filtration procedure was performed as described previously (Hill et al. 2007). Briefly, F200NR ultrafilters (Fresenius Medical Care, Lexington, MA, USA) were incubated overnight with 5% calf serum (Invitrogen, Carlsbad, CA, USA) to prevent nonspecific binding. Prior to beginning the UF procedure, sodium polyphosphate (NaPP, catalog number 305553; Sigma-Aldrich, St. Louis, MO, USA) was added to the water sample to a final concentration of 0.01% (w/v). After UF was completed and the retentate collected, virus was eluted with 500 ml elution solution consisting of 0.01% Tween 80, 0.01% NaPP, and 0.001% Antifoam Y-30 emulsion (Sigma-Aldrich, St. Louis, MO, USA). The average final volume of the combined retentate and eluate (i.e., UF concentrate) was 406 ± 54 ml.
Two approaches were investigated for secondary processing of the UF concentrates: polyethylene glycol (PEG) precipitation and centrifugal ultrafilters (i.e., microconcentrators). Two 30-kDa Centricon Plus-70 units (Millipore, Billerica, MA, USA) were used according to the manufacturer’s instructions to concentrate half of the UF concentrate for each experiment. The average final Centricon concentrate volume achieved was 2.8 ± 1.9 ml. The other half of the UF concentrate from each experiment was concentrated by PEG precipitation (12% PEG 8000, 0.9 M NaCl, and 1% bovine serum albumin) for 2 h at 4°C (Polaczyk et al. 2008). Pelleted material was resuspended with phosphate buffered saline (PBS) containing 0.01% Tween 80 and 0.001% Antifoam Y-30 emulsion, resulting in average PEG concentrates of 3.1 ± 1.0 ml.
Three replicate experiments were performed at each of three seed levels (103–3 × 105 GI.4 virus particles and 104–106 GII.4 virus particles) added to 50 l water samples to determine RT-qPCR detection rates associated with UF and each of the secondary concentration techniques.
Nucleic Acid Extraction
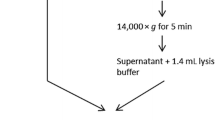
Nucleic acid extraction was performed using a previously reported non-commercial lysis buffer containing 4.5 M guanidinium isothiocyanate (Hill et al. 2007). Briefly, 500 μl of freshly prepared lysis buffer was added to 500 μl of concentrated (either PEG precipitated or Centricon) water sample in a 2 ml screw-cap polypropylene tubes (“BioStor Vials,” National Scientific Supply, Claremont, CA, USA) containing 100 mg of 0.2- and 0.5-mm high-purity, yttrium (III) oxide-stabilized zirconium oxide (ZrO x ) beads (Union Process, Akron, OH, USA). Prior to use, the ZrO x beads were acid washed with 0.1 N HCl for 10 min, rinsed with DI water five times, and then baked in a laboratory oven at 200°C for 30 min, then allowed to cool. The 2 ml tube containing the lysis buffer and sample was shaken for 2 min at high speed setting in a Mini-Bead-Beater-8 (Biospec, Bartlesville, OK, USA). After bead beating, the sample was centrifuged for 30 s at 10,000×g and the supernatant was transferred to a silica spin column (Omega Biotek, Norcross, GA, USA), and passed through the column by centrifugation at 10,000×g for 1 min. After two washes (100% ethanol and 75% ethanol), nucleic acid was eluted in 80 μl of TE buffer. A final purification step was performed on a polyvinylpolypyrrolidone (PVPP) spin column (Spin-IV-HRC columns, Zymo Research Corporation, Orange, CA).
RT-qPCR Analysis of Ground Water Samples
To determine the detection sensitivity of the RT-qPCR assays for ground water samples, different amounts of virus particles (1 × 103–3 × 105 of GI.4 and 104–106 virus particles of GII.4) were seeded into 50 l ground water samples. After concentration and RNA extraction as described above, eight replicates of each RNA extract were tested for GI.4 and GII.4 NoV by RT-qPCR. A sample was defined positive when at least two of the eight replicate reactions tested positive. To evaluate RT-qPCR inhibition associated with ground water concentrates, GI.4 or GII.4 NoV RNA was added to RNA extracts and to nuclease-free water as an inhibition control. The samples were assayed in the same thermal cycler run, and the CT values compared. The difference in CT values between the nuclease-free control and UF-concentrated water samples was used as a measure of RT-qPCR inhibition associated with the UF-concentrated water sample, as described previously (Hill et al. 2007).
Results
GI Norovirus TaqMan RT-qPCR Assay Development
Our novel GI NoV RT-qPCR assay detected strains from all eight tested genotypes, including GI.1 and GI-3b viruses and other GI genotypes with the same sensitivity compared to our previously reported assay (Jothikumar et al., 2005). Gradient PCR results demonstrated that the novel assay provided robust results for a genetically diverse panel of GI NoVs at annealing temperatures of 55°C and lower (Fig. 1) and standard curves (Fig. 2) showed that the assay was efficient. Further, the new GI NoV RT-qPCR assay did not cross-react with GII Nov, GIV NoV, (GV) murine norovirus, GI sapovirus, coxsackievirus A8, hepatitis A virus (strain HM-175), and hepatitis E virus (genotype 2) (data not shown).
Detection Sensitivity for GI and GII Noroviruses in Ground Water
A total of 18, 50 l samples were collected from the two ground water locations. The pH of the ground water samples ranged from pH 7.07 for Source 1 water to pH 7.87 for Source 2 water (Table 2). The turbidity of the two water sources differed greatly, with Source 2 averaging 0.17 nephelometric turbidity units (NTU) and Source 1 water averaging 9.7 NTU. Alkalinity and total hardness results indicated that both water types had moderate buffering capacities and mineral content. TOC results also indicated that Source 1 (18.6 mg/l) and Source 2 (19.5 mg/l) water had levels of organic matter typical for crystalline rock and limestone aquifers. E. coli was not detected in any of the water samples (100 ml tested).
GI NoV was detected in two of the three samples from Source 1 that were seeded with 2 × 104 virus particles and concentrated using UF and Centricon Plus-70 microconcentrators, but a lower detection limit (1 × 104 virus particles) was achieved when using PEG precipitation instead of microconcentrators (Table 3). In Source 2 water, GI NoV was detected at a seed level of 3 × 105 virus particles in three of three experiments when either secondary concentration method was performed. However, a lower detection limit (2 × 105 virus particles) was achieved when using Centricon Plus-70 microconcentrators instead of PEG precipitation. For both secondary concentration methods, inhibition of RT-qPCR was estimated to be 1.7 and a 2.8 CT values for the GI NoV assays performed with RNA extracts from Source 1 and Source 2 water concentrates, respectively.
GII NoV was detected in three of three Source 1 water samples at seed levels of 1 × 105 and 5 × 104 virus particles when either secondary concentration method was used (Table 4). However, when the seed level was reduced to 1 × 104 virus particles, no virus was detected following either secondary concentration method. In Source 2 water, GII NoV was detected at a seed level of 1 × 106 virus particles in three of three experiments when either secondary concentration method was performed, but a lower detection limit (5 × 105 virus particles) was achieved when using microconcentrators. At a seed level of 1 × 105 virus particles, GII NoV was detected in one out of three Source 2 water experiments when PEG precipitation was performed, but was not detected in Centricon concentrates. For both secondary concentration methods, inhibition of RT-qPCR was estimated to be 2.0 and a 3.3 CT values for the GII NoV assays performed with RNA extracts from Source 1 and Source 2 water concentrates, respectively.
Discussion
We developed a novel broadly reactive RT-qPCR assay for the detection of GI NoV. Combined with a previous reported GII NoV RT-qPCR assay, as few as 1 × 104 NoV particles could be detected in 50 l seeded ground water samples. Compared with previous reported detection limits (Jothikumar et al. 2005; Gentry et al. 2009a), the novel GI assay detected as few as 1.6 copies of GI.3 NoV. A recent study showed that the RNA copy numbers per EM-enumerated virus particles ranged from 1:1 to 60:1 for GI and GII NoV (Costantini et al. 2010). The detection limit in ground water samples was 1 × 104 to 2 × 105 virus particles in 50 l water samples (corresponding to 200–3,000 particles/l) for GI NoV and 5 × 104 to 5 × 105 virus particles (corresponding to 1,000–10,000 particles/l) for GII NoV. RT-qPCR inhibitors were detected in RNA extracts of water samples from both sources and therefore improved nucleic acid extraction methods should be explored to enable detection of NoVs at lower levels. NoV contamination levels between 110 and 1.4 × 109 RNA copies/l have been reported in environmental water samples (Gentry et al. 2009a).
Recovery of NoVs from environmental water samples typically involves methods that rely on adsorption of viruses to negatively or positively charged filters (Hunt et al. 2010; Gabrieli et al. 2009; Gentry et al. 2009a; Hamza et al. 2009). While the virus adsorption–elution approach can be effective for recovering viruses from water samples, this method has been shown to be affected by water quality conditions such as pH, ionic strength, and organic content (Sobsey and Glass 1984; Guttman-Bass and Catalano-Sherman 1986; Lukasik et al. 2000; Lambertini et al. 2008). UF is an increasingly accepted alternative for virus recovery from water samples, and has been utilized for detection of NoV in water samples (Hewitt et al. 2007; Hernandez-Morga et al. 2009). While no differences between the two alternative secondary concentration techniques for GII.4 NoV were observed, PEG precipitation resulted in lower method detection limits than the Centricon procedure for GI.4 NoV. In Source 2 water, however, the Centricon procedure resulted in slightly lower detection limits than the PEG precipitation method. Thus, the data from this study did not indicate a consistent difference in NoV recovery performance between the Centricon and PEG precipitation methods. To our knowledge, this is the first study to report sensitivity data for detection of GI and GII NoV in seeded ground water samples.
However, our study did have several limitations. The number of ground water sampling sites investigated in this study was modest. While these ground water samples were substantially different in certain important characteristics (e.g., aquifer type and turbidity), we caution for extrapolating the results from this study to all ground water samples. In addition, the RT-qPCR method detection limit was affected by co-extraction of inhibitors in the water samples. Use of a clean water control could have provided optimal detection sensitivity data as a reference to data obtained with environmental water samples.
The results from this study show that UF followed by the use of either microconcentrators or PEG precipitation can be effective in detecting NoV in 50 l ground water samples. These sampling and analytical methods should be useful tools for monitoring water resources for NoVs and responding to outbreaks of disease associated with contaminated water. Future applications of the use of UF and NoV RT-qPCR include monitoring for NoVs in drinking water, surface water, and reclaimed water. The broad reactivity and sensitivity of the novel GI NoV RT-qPCR assay also suggest that this assay can be effective for detecting GI NoVs in other environmental samples, including food.
References
Baert, L., Uyttendaele, M., & Debevere, J. (2008). Evaluation of viral extraction methods on a broad range of ready-to-eat foods with conventional and real-time RT-PCR for Norovirus GII detection. International Journal of Food Microbiology, 123, 101–108.
Cannon, J., & Vinje, J. (2008). Foodborne viruses. In E. Palombo & C. Kirkwood (Eds.), Viruses in the environment (1st ed., pp. 45–76). Ames: Iowa State Press.
Costantini, V., Grenz, L., Fritzinger, A., Lewis, D., Biggs, C., Hale, A., et al. (2010). Diagnostic accuracy and analytical sensitivity of IDEIA norovirus assay for routine screening of human norovirus. Journal of Clinical Microbiology, 48, 2770–2778.
Gabrieli, R., Maccari, F., Ruta, A., Paná, A., & Divizia, M. (2009). Norovirus detection in groundwater. Food and Environmental Virology, 1, 92–96.
Gentry, J., Vinje, J., Guadagnoli, D., & Lipp, E. K. (2009a). Norovirus distribution within an estuarine environment. Applied and Environmental Microbiology, 75, 5474–5480.
Gentry, J., Vinje, J., & Lipp, E. K. (2009b). A rapid and efficient method for quantitation of genogroups I and II norovirus from oysters and application in other complex environmental samples. Journal of Virological Methods, 156, 59–65.
Glass, R. I., Parashar, U. D., & Estes, M. K. (2009). Norovirus gastroenteritis. New England Journal of Medicine, 361, 1776–1785.
Guttman-Bass, N., & Catalano-Sherman, J. (1986). Humic-acid interference with virus recovery by electropositive microporous filters. Applied and Environmental Microbiology, 52, 556–561.
Hamza, I. A., Jurzik, L., Stang, A., Sure, K., Uberla, K., & Wilhelm, M. (2009). Detection of human viruses in rivers of a densly-populated area in Germany using a virus adsorption elution method optimized for PCR analyses. Water Research, 43, 2657–2668.
Hernandez-Morga, J., Leon-Felix, J., Peraza-Garay, F., Gil-Salas, B. G., & Chaidez, C. (2009). Detection and characterization of hepatitis A virus and norovirus in estuarine water samples using ultrafiltration—RT-PCR integrated methods. Journal of Applied Microbiology, 106, 1579–1590.
Hewitt, J., Bell, D., Simmons, G. C., Rivera-Aban, M., Wolf, S., & Greening, G. E. (2007). Gastroenteritis outbreak caused by waterborne norovirus at a New Zealand ski resort. Applied and Environmental Microbiology, 73, 7853–7857.
Hill, V. R., Kahler, A. M., Jothikumar, N., Johnson, T. B., Hahn, D., & Cromeans, T. L. (2007). Multistate evaluation of an ultrafiltration-based procedure for simultaneous recovery of enteric microbes in 100-liter tap water samples. Applied and Environmental Microbiology, 73, 4218–4225.
Huffman, D. E., Nelson, K. L., & Rose, J. B. (2003). Calicivirus—an emerging contaminant in water: state of the art. Environmental Engineering Science, 20, 503–515.
Hunt, R. J., Borchardt, M. A., Richards, K. D., & Spencer, S. K. (2010). Assessment of sewer source contamination of drinking water wells using tracers and human enteric viruses. Environmental Science and Technology, 44, 7956–7963.
Jothikumar, N., Lowther, J. A., Henshilwood, K., Lees, D. N., Hill, V. R., & Vinje, J. (2005). Rapid and sensitive detection of noroviruses by using TaqMan-based one-step reverse transcription-PCR assays and application to naturally contaminated shellfish samples. Applied and Environmental Microbiology, 71, 1870–1875.
Kageyama, T., Kojima, S., Shinohara, M., Uchida, K., Fukushi, S., Hoshino, F. B., et al. (2003). Broadly reactive and highly sensitive assay for Norwalk-like viruses based on real-time quantitative reverse transcription-PCR. Journal of Clinical Microbiology, 41, 1548–1557.
Kaplan, J. E., Feldman, R., Campbell, D. S., Lookabaugh, C., & Gary, G. W. (1982). The frequency of a Norwalk-like pattern of illness in outbreaks of acute gastroenteritis. American Journal of Public Health, 72, 1329–1332.
Lambertini, E., Spencer, S. K., Bertz, P. D., Loge, F. J., Kieke, B. A., & Borchardt, M. A. (2008). Concentration of enteroviruses, adenoviruses, and noroviruses from drinking water by use of glass wool filters. Applied and Environmental Microbiology, 74, 2990–2996.
Liang, J. L., Dziuban, E. J., Craun, G. F., Hill, V., Moore, M. R., Gelting, R. J., et al. (2006). Surveillance for waterborne disease and outbreaks associated with drinking water and water not intended for drinking—United States, 2003–2004. Morbidity and Mortality Weekly Report, 55, 31–65.
Lukasik, J., Scott, T. M., Andryshak, D., & Farrah, S. R. (2000). Influence of salts on virus adsorption to microporous filters. Applied and Environmental Microbiology, 66, 2914–2920.
Ngazoa, E. S., Fliss, I., & Jean, J. (2008). Quantitative study of persistence of human norovirus genome in water using TaqMan real-time RT-PCR. Journal of Applied Microbiology, 104, 707–715.
O’Reilly, C. E., Bowen, A. B., Perez, N. E., Sarisky, J. P., Miller, M. D., Hubbard, B. C., et al. (2007). A waterborne outbreak of gastroenteritis with multiple etiologies among resort island visitors and residents: Ohio, 2004. Clinical Infectious Diseases, 44, 506–512.
Park, Y., Cho, Y. H., Jee, Y., & Ko, G. (2008). Immunomagnetic separation combined with real-time reverse transcriptase PCR assays for detection of norovirus in contaminated food. Applied and Environmental Microbiology, 74, 4226–4230.
Polaczyk, A. L., Jothikumar, N., Cromeans, T. L., Hahn, D., Roberts, J. M., Amburgey, J. E., et al. (2008). Ultrafiltration-based techniques for rapid and simultaneous concentration of multiple microbe classes from 100-L tap water samples. Journal of Microbiological Methods, 73, 92–99.
Rolfe, K. J., Parmar, S., Mururi, D., Wreghitt, T. G., Jalal, H., Zhang, H., et al. (2007). An internally controlled, one-step, real-time RT-PCR assay for norovirus detection and genogrouping. Journal of Clinical Virology, 39, 318–321.
Rutjes, S. A., van den Berg, H., Lodder, W. J., & Husman, A. M. D. (2006). Real-time detection of noroviruses in surface water by use of a broadly reactive nucleic acid sequence-based amplification assay. Applied and Environmental Microbiology, 72, 5349–5358.
Sobsey, M. D., & Glass, J. S. (1984). Influence of water quality on enteric virus concentration by microporous filter methods. Applied and Environmental Microbiology, 47, 956–960.
Wolf, S., Williamson, W. M., Hewitt, J., Rivera-Aban, M., Lin, S., Ball, A., et al. (2007). Sensitive multiplex real-time reverse transcription-PCR assay for the detection of human and animal noroviruses in clinical and environmental samples. Applied and Environmental Microbiology, 73, 5464–5470.
Yoder, J., Roberts, V., Craun, G. F., Hill, V., Hicks, L. A., Alexander, N. T., et al. (2008). Surveillance for waterborne disease and outbreaks associated with drinking water and water not intended for drinking—United States, 2005–2006. MMWR Surveillance Summaries, 57, 39–62.
Acknowledgments
We thank Charles Humphrey (CDC) for performing electron microscopy. This publication was supported in part by funds made available through CDCs Coordinating Office for Terrorism Preparedness and Emergency Response. The use of trade names and names of commercial sources is for identification only and does not imply endorsement by the CDC or the U.S. Department of Health and Human Services. The findings and conclusions in this presentation are those of the authors and do not necessarily represent those of the CDC. This article received clearance through the appropriate channels at the CDC prior to submission.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hill, V.R., Mull, B., Jothikumar, N. et al. Detection of GI and GII Noroviruses in Ground Water Using Ultrafiltration and TaqMan Real-time RT-PCR. Food Environ Virol 2, 218–224 (2010). https://doi.org/10.1007/s12560-010-9049-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12560-010-9049-y