Abstract
The publication of a well preserved Eocene primate, Darwinius masillae (Cercamoniinae, Notharctidae), has revived the debate on the phylogenetic relationships of Adapiformes and extant primates (Franzen et al., PLos ONE 4(5):e5723, 2009). Recently, Lebrun et al. (J Anat 216:368–380, 2010) showed that the morphology of the bony labyrinth of strepsirrhine primates conveys a strong phylogenetic signal. The study of labyrinthine morphology may thus bring a new piece of evidence to resolve phylogenetic relationships within a group. The investigation of the labyrinthine morphology of another Cercamoniinae, Pronycticebus gaudryi, reveals no synapomorphy with the labyrinths of modern anthropoids. On the contrary, Pronycticebus is closer in labyrinthine shape to extant strepsirrhines, which supports the hypothesis that the Cercamoniinae and other Adapiformes are the sister group of toothcombed primates.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The recent description of Darwinius masillae (Cercamoniinae, Notharctidae), a well-preserved Eocene primate, has revived the debate on the phylogenetic relationships of Adapiformes and anthropoid primates (Franzen et al. 2009). Several decades ago, a few researchers had argued that extant anthropoids share an adapiform ancestor (Franzen 1994; Gingerich 1973, 1975, 1981; Gingerich and Schoeninger 1977; Rasmussen 1986, 1990). Since then, phylogenetic analyses of primate relationships have favoured the hypothesis that Adapiformes are stem strepsirrhines (see, for instance, Kay et al. 1997; Marivaux et al. 2005; Ni et al. 2004; Ross et al. 1998; Seiffert et al. 2005). However, Franzen et al. (2009) have revived the hypothesis of a special link between Adapiformes and anthropoids, claiming that Darwinius masillae was part of a group “representative of the early haplorhine diversification”. This view was criticised by Williams et al. (2010), in a review of the anatomical features differentiating haplorhine and strepsirrhine primates. Williams et al. (2010) argued that Darwinius is certainly not a haplorhine, and gave further support to the hypothesis that Adapiformes are basal strepsirrhines (but see Gingerich et al. 2010).
In this paper, we analyse the phylogenetic relationships of Adapiformes with other primates from the perspective of the morphology of the inner ear. Morphological variation of the bony labyrinth across taxa reflects differences in locomotor behaviour (Silcox et al. 2009; Spoor and Zonneveld 1998; Spoor et al. 2007; Walker et al. 2008) and hearing performance (Coleman and Boyer 2012; Coleman and Colbert 2010; Coleman et al. 2010; Echteler et al. 1994; Gleich et al. 2005; Kirk and Gosselin-Ildari 2009; West 1985). This structure is thus highly functionally constrained. Nevertheless, a part of labyrinthine morphological variation may still convey phylogenetic information. Basilar membrane length, the number of spiral turns and cochlear volume relate to low frequency and high frequency sensitivity (Coleman and Boyer 2012; Echteler et al. 1994; Kirk and Gosselin-Ildari 2009; West 1985). However, other aspects of the morphology of the cochlea, such as its general geometry and orientation relative to the semi-circular canal system, are less likely to reflect hearing performance. Concerning the semi-circular canal system, even though canal radius tends to increase with agility, semi-circular canal morphology conveys a strong phylogenetic signal (Spoor et al. 2007). As large species tend to be less agile than small ones, a possible way to maximise the phylogenetic information conveyed by this structure is to remove the allometric component of labyrinthine morphological variation.
Building on these hypotheses, Lebrun et al. (2010) proposed a protocol analysis designed to capture the allometry-free component of the geometry of the semi-circular canal system, and the general orientation and geometry of the cochlea within the labyrinth. These authors showed that in strepsirrhine primates the geometry of the bony labyrinth conveys a strong phylogenetic signal. Also, they showed that change in labyrinthine morphology is adequately described with an evolutionary random walk model, i.e. random phenotypic dispersal in morphospace. Under this hypothesis, average shapes calculated for each node of a phylogenetic tree give an estimate of the inner ear morphology of the respective last common ancestors (LCAs). Lebrun et al. (2010) showed that the morphology of Eocene Adapinae is close to the inferred state of the ancestral toothcombed primate labyrinthine morphology. In addition, adapine labyrinthine morphology is close to that of Malagasy primates, which supports the hypothesis that Adapiformes are the sister group of toothcombed primates.
The morphology of the inner ear of Darwinius masillae is not preserved (Hurum 2011). However, a more complete and undistorted cranium of another member of Cercamoniinae, Pronycticebus gaudryi, is available for study. The ear region of Pronycticebus is well preserved. The bullae were intact when the specimen was discovered (Grandidier 1904), and were subsequently partially prepared. A small segment of a free ectotympanic ring was exposed and described by Simons (1962) as Loris-like. Other students instead described the ear region of Pronycticebus as Lemur-like, a free-floating ectotympanic ring actually found in Malagasy primates (Couette et al. 2011; Le Gros Clark 1934; Saban 1963; Szalay 1971), giving support to the strepsirrhine status of Pronycticebus. Because the bullae of the type specimen are filled with a hard calcitic matrix, the specimen was never completely dissected. However, CT scan techniques allow for virtual and non-invasive dissection of that region. Investigation of the morphology of the labyrinth of Pronycticebus was carried out, and study of its morphological affinities with that of extant primates and other Eocene primate taxa will provide an independent piece of evidence to test hypotheses about the phylogenetic relationships of Adapiformes with other primates (Lebrun et al. 2011). Our earlier analyses showed that primate labyrinthine morphology is consistent at the family level, and exhibits an even higher degree of consistency at the subfamily level (Lebrun et al. 2010). Building upon these findings, we make the assumption that the labyrinths of Darwinius masillae and of Pronycticebus gaudryi share strong morphological affinities. Also, the teeth of Darwinius show derived similarities with those of Agerinia (Herbomel and Godinot 2011), and Agerinia and Pronycticebus have been considered as closely related (Godinot 1998; Szalay 1971). Hence, Darwinius is probably quite closely related to Pronycticebus gaudryi, and the labyrinth of the latter is likely a good proxy for that of Darwinius masillae.
Materials and methods
Comparative sample
We compare the labyrinth of Pronycticebus with a broad sample of extant and extinct primates, in order to have a good representation of present and past primate labyrinthine morphological diversity. The comparative dataset consists of 93 inner ears of primates (see Table 1). Thirty-nine inner ears belong to modern strepsirrhine taxa, representing 14 lemuroid and 9 lorisoid genera. Forty-two modern haplorhine specimens were digitised, representing 16 genera of platyrrhines, 20 genera of catarrhines and the genus Tarsius. Concerning the fossil sample, 11 inner ears of Adapiformes were included, representing 3 adapine genera and the cercamoniine Pronycticebus. Finally, 2 inner ears of fossil Omomyiformes belonging to the Microchoerinae subfamily were analysed (see Table 1). Only left inner ears were integrated in the sample when preserved. For the four adapine specimens which had only their right inner ear preserved, virtual mirror images of their right labyrinth were produced and used for analysis.
Data acquisition
X-ray micro-computed tomography (μCT) and synchrotron X-ray microtomography (SR-μCT) were used to acquire 3D data. Most fossil specimens were scanned at the European Synchrotron Radiation Facility (ESRF) on beam lines ID17 and ID19 (see Table 1). Synchrotron tomography results in high contrast and spatial resolutions data (Tafforeau et al. 2006), which greatly facilitate segmentation of the bony labyrinth cavities filled by dense sediment when fossils are highly mineralised (Lebrun et al. 2010). Digital volume data of Pronycticebus gaudryi were obtained via high resolution micro-CT on a SkyScan 1076 scanner. Three-dimensional surfaces representing the bony labyrinths were produced with Amira 3.1.1 (Visage Imaging) and Avizo 6.3.1 (Visualization Sciences Group) via thresholding using the half maximum height technique (Spoor et al. 1993) and manual segmentation. The choice of the threshold value could affect to some extent the reconstruction of the semicircular canals and of the cochlea (Coleman and Colbert 2007). Fortunately, thresholding effects do not affect the location of the lumen centre of the semicircular canals and the cochlea (Gunz et al. 2012; Lebrun et al. 2010). As such, labyrinthine shape was quantified with 22 landmarks, located at the centres of the lumina of the semicircular canals, of the ampullae, and of the cochlear helix (see Supporting Information Figure S1), following the protocol of Lebrun et al. (2010).
Data analysis
Using generalised least-squares fitting (Rohlf 1990) and principal components analysis (PCA) of shape (Dryden and Mardia 1998), the form of each specimen’s landmark configuration was represented by its centroid size S, and by its multidimensional shape vector v in linearised Procrustes shape space. In order to take into account potential confounding effects of size allometry on primate labyrinthine shape (Lebrun et al. 2010), size-corrected shapes were obtained as follows. Regression of Procrustes coordinates by the logarithm of centroid size were computed for Lorisoidea, Lemuroidea, Catarrhines, Platyrrhines, Adapiformes, Omomyiformes, and Tarsiiformes, yielding group-specific allometric shape vectors (ASVg). The ASVg represent directions in shape space which characterise group-specific allometric patterns of labyrinthine shape variation. A common allometric shape vector (ASVc), obtained as the mean of all the ASVg, provided a direction in shape space that minimises potential divergence in labyrinthine allometric patterns across taxonomic groups. All labyrinths were then projected on ASVc, the residuals representing the size-independent component of labyrinthine shape. Size-independent shape variation was analysed by PCA of shape using the interactive software package MORPHOTOOLS (Lebrun 2008; Specht 2007; Specht et al. 2007).
In order to assess the morphological affinities of Pronycticebus with extant primates, Adapinae and Microchoerinae, phenetic trees were produced as follows. For each group (Adapinae, Microchoerinae, Lemuroidea, Lorisoidea, Tarsius, Catarrhines, Platyrrhines), an average size-corrected labyrinthine shape was produced. Taxa were then clustered using the NJ (neighbour joining) procedure. A landmark-based random sampling procedure, as described in Lockwood et al. (2004), was executed 1,000 times. The associated consensus NJ tree was computed using PHYLIP (Felsenstein 1989).
Results
Visualising patterns of size-corrected labyrinthine shape variation in morphospace and in physical space permits characterisation of high-level taxon-specific morphologies (Fig. 1): lorisoids, lemuroids, anthropoids, Tarsius, adapiforms and omomyiforms are well discriminated in PC1–PC2 space (32.05 % of total shape variation), without almost any overlap. The labyrinth of Pronycticebus is closest in morphology to that of Adapinae and of Lemuroidea families such as Lepilemuridae, Lemuridae and Cheirogaleidae, and differs substantially from that of anthropoids (see Table 2). Anthropoid labyrinths show extension in the anteromedial to posterolateral direction, whereas the labyrinths of extant strepsirrhines and Tarsius tend to be compressed in this direction. The lateral canal of anthropoids is extended in the anteromedial to posterolateral direction while that of prosimians shows extension in the anterolateral to posteromedial direction (see Fig. 1b, deformations along PC1). In Pronycticebus, Adapinae and Microchoerinae, the posterior semi-circular canal assumes a high position relative to the lateral canal (see Figs. 2 and 3), which results in partial fusion of the lateral and posterior semi-circular canals. Fossil specimens are well discriminated from extant strepsirrhines, anthropoids and Tarsius in PC1–PC2 space. Furthermore, the semi-circular canals of these Eocene primates are round, and their three semi-circular canals are of approximately similar size. This condition differs from that found in Tarsius and small-bodied anthropoids: their anterior canal is relatively smaller (see also Fig. 1b). The common crus of Pronycticebus is orthogonal to the plane of the lateral semi-circular canal, a condition that can also be observed in small-bodied Adapinae, like Adapis, and in some lemurs, such as Cheirogaleus, and in Lorisidae. The common crura of microchoerine primates, of Tarsius and Anthropoidea tend to point posteriorly (see Fig. 3).
Principal components analysis (PCA) of labyrinthine size-corrected shape variation. a Graphing the first two components of shape space, PC1 and PC2, shows differences in labyrinthine morphology across anthropoids, extant strepsirrhines, Eocene primates and Tarsius. Black symbols strepsirrhines, grey symbols haplorhines, triangles Lorisoidea, squares Lemuroidea, Ooen circles Adapinae filled circle Pronycticebus, stars Platyrrhines, X Catarrhines, diamonds Tarsius; + Microchoerinae. Symbol size variation represents labyrinthine centroid size variation. b Patterns of labyrinthine shape variation associated with PC1 and PC2, respectively. Grey arrows anteromedial-to-posterolateral and anterolateral-to-posteromedial directions
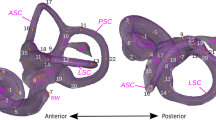
Left bony labyrinth of Pronycticebus gaudryi. The labyrinth is positioned in superior (a) and lateral (b) views (by convention, the lateral semicircular canal is positioned horizontally). Specimen: QU 11056. Scale bar 5 mm. Dashed arrows give the orientation of the common crus and of the cochlea. Note that the common crus is orthogonal to the plane of the lateral semi-circular canal
Left bony labyrinths of primates of a Cheirogaleus major, b Galago moholi, c Adapis sp., d Microchoerus erinaceus, e Tarsius spectrum, f Callithrix jacchus. For each specimen, the labyrinth is positioned in superior (left) and lateral (right) views. Specimens: a MNHN MO 2002-87, b MNHN MO 1885-196, c MUNCH XV-1869-1530, d MONTP PR-1771, e AIM-ZU AS1821, f AIM-ZU 10168. Scale bar 5 mm. Taxa of similar labyrinthine size to that of Pronycticebus were chosen. Dashed arrows give the orientation of the common crus and of the cochlea
Pronycticebus, Adapis and Lemuroidea exhibit similarities in cochlear shape, orientation and number of turns, measured following West (1985); their cochleae exhibit between 2 and 2.5 turns. In Lemuroidea, Lorisoidea and Adapiformes, the turns of the cochlear spiral are in broad and close contact, which gives this structure a relatively more globose and flattened aspect. In contrast, the cochlear orientation and aspect of Tarsius and Microchoerinae resembles that of small anthropoids such as Callithrix (see Fig. 3): Microchoerinae, Tarsius and anthropoids exhibit cochleae oriented more anteriorly than those of Adapiformes and Lemuroidea (see Figs. 2 and 3). Also, the second turn of their cochleae tends to stay further away from the plane of the first turn.
The phenetic similarity tree based on inner ear morphology is illustrated in Fig. 4. This tree gives indications about the affinities of taxon-specific labyrinthine shapes, and does not reflect the current view of primate phylogeny. This phenetic tree groups together extant platyrrhines and catarrhines, while Adapinea and Pronycticebus branch close to each other, and are distant from extant anthropoids. Microchoerinae and Tarsius branch together, but appear also distant from anthropoids.
Phenetic neighbour joining tree based on inner ear morphology (average labyrinthine shape of taxa) reflecting bony labyrinth morphological affinities (size-corrected shape distances) between Adapinae, Pronycticebus, Lemuroidea, Lorisoidea, Microchoerinae, Tarsius, Platyrrhines, Catarrhines. Bootstrap values for 1,000 resamplings are given at each node
Discussion
Our results show that inner ear morphology is a useful taxonomic marker, supporting the results of Lebrun et al. (2010). Our data show that the inner ear of Pronycticebus is morphologically closest to that of adapine Adapiformes, which indicates that inner ear morphological variation of Adapiformes is small. This result confirms that the inner ear of Pronycticebus can reasonably be used as a proxy for that of other putative Cercamoniinae primates like Darwinius.
Primate inner ear symplesiomorphies
The Eocene primates analysed in this study share similarities in the semi-circular canal system: their semi-circular canals are round and are of largely similar size, their posterior canal assumes a relatively high position, a consequence of which is its partial fusion with the lateral canal. We propose that these shared Adapiformes/Omomyiformes features represent symplesiomorphies of primate labyrinthine morphology.
Labyrinthine shape variation and cranial evolution
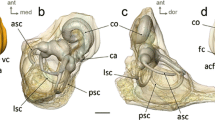
Our results suggest that, despite being a functionally constrained structure, a substantial residual part of primate labyrinthine morphology conveys non-functional information. This residual morphological variation may be related to specific cranial morphological characters. We observed in fossil Adapinae and Microchoerinae that the bony channels of the posterior limb of the lateral canal are merged with the inferior part of the posterior canal, forming a second common crus. The presence of a second common crus is found in a variety of extant and extinct placental and non-placental mammals (see, for instance, Benoit et al. 2012; Ekdale 2009; Hyrtl 1845; Schmelzle et al. 2007). In all extant specimens observed in this study, the posterior canal assumes a lower position relatively to the lateral canal, the consequence of which is the absence of fusion of the inferior part of the posterior canal with the posterior limb of the lateral canal. It may be asked how the presence of a second common crus in Eocene Adapinae and Microchoerinae and its absence in all extant primate specimens observed here relate to primate cranial evolution. Primates, and in particular anthropoids, exhibit a well-documented general evolutionary trend towards increased encephalisation during the Cenozoic (Jerison 1973, 1979; Radinski 1977), that is toward increasing their relative brain size. Evolving larger brain size implies modifications of the morphology of the braincase, and has a potential impact on the otic capsules and the surrounding petrous bone (Jeffery and Spoor 2004). The coronal orientation of the petrous bone has been shown to correlate with relative brain size in primates (Spoor 1997), which may influence the geometry of the labyrinth, and in particular the relative position of the semi-circular canals and the orientation of the cochlea. Also, in modern humans, the petrous bone is wedged between the cerebral temporal lobe and the cerebellum (see, for instance, Jeffery and Spoor 2004). This configuration is found in other extant primate species, such as Otolemur garnetti, Tarsius syrichta and Callithrix jacchus (see Fig. S2). In these species, the regions of the petrous bone holding the anterior and posterior canals are in close contact with the cerebral temporal lobes and the cerebellum. In Adapis parisiensis, the petrous bone being more laterally positioned relative to the brain, the position and shape of the posterior and anterior canals are less likely to be influenced by brain structures (see Fig. S2). The lower position of the posterior canal relative to the lateral canal in modern forms may be the result of a “packing” issue subsequent to the increasing volume of the brain during evolution. The orientation of the cochlea and the shape of the semi-circular canals may also covary with other aspects of cranial morphology, such as basicranial flexion. In order to better understand the non-functional component of primate labyrinthine morphological variation, a comprehensive covariation analysis between cranial and inner ear morphology is required.
Morphological differences between the two primate suborders
Researchers working on the dentitions of the earliest Omomyiforms and Adapiformes note only little difference (Gingerich 1986; Godinot 1978; Simons 1962; Szalay 1976). Hopefully, other cranial structures are useful to discriminate among primate groups. The middle ear region has long been studied (e.g. Gregory 1915, 1920; MacPhee and Cartmill 1986; Saban 1963), and differences in vascularisation and pneumatisation are found between both suborders. Our results suggest that the inner ear is another structure that differs in morphology between both suborders. Despite the morphological similarities cited above, the inner ear of Eocene microchoerines on the one hand, and that of adapine Adapiformes and Pronycticebus, on the other, can be well distinguished, in particular in their cochlear and common crus orientation and in their cochlear aspect.
Tarsius and Omomyiformes on the one hand and extant anthropoids on the other differ in overall labyrinthine shape (see Figs. 1, 4). However, several inner ear characters link these extant and extinct haplorhines. Omomyiformes and Tarsius share similarities in the orientation of the common crus and shape of the canals (see Fig. 3). And even though Tarsiers have more spiral turns and have longer cochleae than Omomyids (Coleman and Boyer 2012), they share with small anthropoids some other aspects of morphology of the cochlea. We found that Tarsius, microchoerines and small anthropoids have anteriorly oriented cochleae exhibiting a second turn staying away from the plane of the first turn, and posteriorly oriented common crura. These three characters are candidate synapomorphies for the inner ears of haplorhines. On the other hand, extant and extinct strepsirrhine inner ears share morphological similarities. The inner ears of Adapinae and Pronycticebus are close in morphology to those of Malagasy lemurs. Lebrun et al. (2010) suggested that, within extant strepsirrhines, the inner ear condition of Lemuroidea is primitive, while that of Lorisoidea is derived. Furthermore, Lebrun et al. (2010) found morphological affinities between the inner ears of Lemuroidea and Adapiformes and that of the demopteran Cynocephalus, the three semi-circular canals of which are straight, round and approximately of similar size. However, as the inner ears of the dermopteran Galeopterus and of the scandantian Tupaia differ widely from those of primates, it cannot be ruled out that the similarities observed between Cynocephalus on the one hand, and Adapiformes and Lemuroidea on the other, represent morphological convergences. As such, it cannot yet be assessed whether the labyrinthine similarities observed in Adapidae and Lemuroidea represent shared derived features within strepsirrhine primates rather than primate inner ear symplesiomorphies.
Is there evidence for a link between Adapiformes and Haplorhini?
There is virtually no similarity between the inner ears of Adapiformes and those of extant anthropoids, which gives no tangible arguments that would support the hypothesis of Franzen et al. (2009) that Darwinius, and other notharctid Adapiformes represent a group of primates which gave rise to anthropoids. Our results, on the other hand, do not contradict the classical hypothesis linking Adapiformes and toothcombed strepsirrhines, based on wrist and ankle synapomorphies (Beard et al. 1988).
The sample of fossil primates analysed here does not allow one to securely define synapomorphies of strepsirrhine inner ears. Such inferences would require the inclusion of other Adapiformes and earlier Omomyiformes. Also, extant anthropoids labyrinthine morphology may be well derived, as illustrated by the clear division of Adapiform–Omomyiform inner ear in PC1–PC2 space on the one hand and of those of modern anthropoids on the other. Though shared characters exist between the inner ears of Tarsius, Omomyidae and small bodied anthropoids, further research is needed to assess whether they represent haplorhine synapomorphies.
Comparisons with inner ears of Eocene eosimiid (Beard et al. 1996; Jaeger et al. 1999) and of late Eocene African anthropoids (Seiffert et al. 2005) are thus required to understand the morphological evolution of this structure within haplorhines.
Conclusion
No synapomorphy was found between the labyrinths of Pronycticebus and those of modern anthropoids. On the contrary, Pronycticebus is closer in labyrinthine shape to extant strepsirrhines, which better supports the hypothesis that Cercamoniinae and other Adapiformes are the sister group of toothcombed primates. Also, candidate synapomorphies of haplorrhine inner ear have been proposed, which are absent in Adapiformes primates. Our results call for further comparative analyses including the inner ear of early Eocene Adapiformes for which the cranium is preserved, such as Cantius, and of early Eocene Omomyiformes such as Teilhardina and of fossil anthropoids.
References
Beard KC, Dagosto M, Gebo DL, Godinot M (1988) Interrelationships among primate higher taxa. Nature 331:712–714
Beard KC, Wang B, Dawson MR, Huang X, Tong Y (1996) Earliest complete dentition of an anthropoid primate from the late middle Eocene of Shanxi Province, China. Science 272:82–85
Benoit J, Orliac M, Tabuce R (2012) The petrosal of Chambius (Macroscelidea, Afrotheria) from the Eocene of Djebel Chambi (Tunisia). J Syst Palaeontology (in press)
Coleman MN, Boyer DM (2012) Inner ear evolution in Primates through the Cenozoic. Implications for the evolution of hearing. Anat Rec 295:615–631
Coleman MN, Colbert MW (2007) Technical note: CT Thresholding protocols for taking measurements on three-dimensional models. Am J Phys Anthropol 133:723–725
Coleman MN, Colbert MW (2010) Correlations between auditory structures and hearing sensitivity in non-human primates. J Morphol 271:511–532
Coleman MN, Kay RF, Colbert MW (2010) Auditory morphology and hearing sensitivity in fossil New World monkeys. Anat Rec 293:1711–1721
Couette S, Lebrun R, Godinot M (2011) The haplorhine/strepsirhine position of Darwinius masillae: new insights based on the CT analysis of Pronycticebus gaudryi middle ear morphology. In: Lehmann T, Schaal SFK (eds) The world at the time of Messel: Puzzles in the palaeobiology, palaeoenvironment, and the history of the early primates. (22nd Int Senckenberg, conference volume). Senckenberg Gesellschaft für Naturforschung, Frankfurt am Main, pp 44–45
Dryden IL, Mardia KV (1998) Statistical shape analysis. Wiley, Chichester
Echteler SM, Fay RR, Popper AN (1994) Structure of the mammalian cochlea. In: Fay RR, Popper AN (eds) Comparative hearing: Mammals. Springer, New York, pp 134–171
Ekdale EG (2009) Variation within the bony labyrinth of mammals. PhD dissertation, University of Texas, Austin
Felsenstein J (1989) PHYLIP—Phylogeny Inference Package (Version 3.2). Cladistics 5:164–166
Franzen JL (1994) The Messel primates and anthropoid origins. In: Fleagle JG, Kay RF (eds) Anthropoid origins. Plenum Press, New York, pp 99–122
Franzen JL, Gingerich PD, Habersetzer J, Hurum JH, von Koenigswald W, Smith BH (2009) Complete primate skeleton from the middle Eocene of Messel in Germany: morphology and paleobiology. PLoS One 4(5):e5723
Gingerich PD (1973) Anatomy of the temporal bone in the Oligocene anthropoid Apidium and the origin of Anthropoidea. Folia Primatol 19(5):329–337
Gingerich PD (1975) A new genus of Adapidae (Mammalia, Primates) from the late Eocene of Southern France, and its significance for the origin of higher primates. Contributions from the Museum of Paleontology, University of Michigan 24:163–170
Gingerich PD (1981) Eocene Adapidae, paleobiogeography, and the origin of South American Platyrrhini. In: Ciochon RL, Chiarelli AB (eds) Evolutionary biology of the new world monkeys and continental drift. Plenum Press, New York, pp 123–138
Gingerich PD (1986) Early Eocene Cantius torresi—oldest primate of modern aspect from North America. Nature 320:319–321
Gingerich PD, Schoeninger M (1977) The fossil record and primate phylogeny. J Hum Evol 6:483–505
Gingerich PD, Franzen JL, Habersetzer J, Hurum JH, Smith BH (2010) Darwinius masillae is a Haplorhine—Reply to Williams et al. (2010). J Hum Evol 59(5):574–579
Gleich O, Dooling RJ, Manley GA (2005) Audiogram, body mass, and basilar papilla length: correlations in birds and predictions for extinct archosaurs. Naturwissenschaften 92:595–598
Godinot M (1978) Un nouvel Adapidé (Primate) de l’Eocène inférieur de Provence. CR Acad Sci Paris, ser D 286:1869–1872
Godinot M (1998) A summary of adapiform systematics and phylogeny. Folia Primatol 69(suppl 1):218–249
Grandidier G (1904) Un nouveau lémurien fossile de France, le Pronycticebus gaudryi. Bull Mus Natl Hist Nat Paris 10:9–13
Gregory WK (1915) I: On the relationship of the Eocene Lemur Notharctus to the Adapidae and the other primates. II: on the classification and phylogeny of the Lemuroidea. Bull Geol Soc Am 26:419–446
Gregory WK (1920) On the structure and relations of Notharctus, an American Eocene primate. Bull Am Mus Nat Hist 42:95–263
Gunz P, Ramsier M, Kuhrig M, Hublin J-J, Spoor F (2012) The mammalian bony labyrinth reconsidered, introducing a comprehensive geometric morphometric approach. J Anat 6:529–543
Herbomel E, Godinot M (2011) A new species of Agerinia (Primates, Adapiformes) and its bearing on the phylogenetic affinities of Darwinius masillae. In: Lehmann T, Schaal SFK (eds) The world at the time of Messel: Puzzles in the palaeobiology, palaeoenvironment, and the history of the early primates. (22nd Int Senckenberg, conference volume). Senckenberg Gesellschaft für Naturforschung, Frankfurt am Main, pp 80–81
Hurum JH (2011) Cranial morphology of the Eocene primate Darwinius masillae. In: Lehmann T, Schaal SFK (eds) The world at the time of Messel: Puzzles in the palaeobiology, palaeoenvironment, and the history of the early primates. (22nd Int Senckenberg, conference volume). Senckenberg Gesellschaft für Naturforschung, Frankfurt am Main, p 84
Hyrtl J (1845) Vergleichend-anatomische Untersuchungen über das innere Gehörorgan des Menschen und der Säugethiere. Friedrich Ehrlich, Prague
Jaeger J-J, Thein T, Benammi M, Chaimanee Y, Soe AN, Thit L, Than T, San W, Ducrocq S (1999) A new primate from the middle Eocene of Myanmar and Asian early origin of anthropoids. Science 286:528–530
Jeffery N, Spoor F (2004) Prenatal growth and development of the modern human labyrinth. J Anat 204:71–92
Jerison HJ (1973) Evolution of the brain and intelligence. Academic Press, New York
Jerison HJ (1979) Brain, body and Encephalization in early primates. J Hum Evol 8:615–635
Kay RF, Ross C, Williams BA (1997) Anthropoid origin. Science 275:797–803
Kirk EC, Gosselin-Ildari AD (2009) Cochlear labyrinth volume and hearing abilities in primates. Anat Rec 292:765–776
Le Gros Clark WF (1934) On the skull structure of Pronycticebus gaudryi. Proc Zool Soc Lond 1:19–27
Lebrun R (2008) Evolution and development of the Strepsirrhine Primate Skull. PhD thesis, Université Montpellier II, University Zürich Irchel
Lebrun R, Ponce de León MS, Tafforeau P, Zollikofer CPE (2010) Deep evolutionary roots of strepsirrhine primate labyrinthine morphology. J Anat 216:368–380
Lebrun R, Couette S, Godinot M (2011) The labyrinthine morphology of Pronycticebus gaudryi (Primates, Adapiformes). In: Lehmann T, Schaal SFK (eds) The world at the time of Messel: puzzles in the palaeobiology, palaeoenvironment, and the history of the early primates. (22nd Int Senckenberg, conference volume). Senckenberg Gesellschaft für Naturforschung, Frankfurt am Main, pp 98–99
Lockwood C, Kimbel WH, Lynch JM (2004) Morphometrics and hominoid phylogeny: support for a chimpanzee—human clade and differentiation among great ape subspecies. Proc Natl Acad Sci USA 101:4356–4360
MacPhee RDE, Cartmill M (1986) Basicranial structures and primate systematics. In: Swindler DR (ed) Comparative primate biology, vol I, Systematics, evolution, and anatomy. Liss, New York, pp 219–275
Marivaux L, Antoine P-O, Baqui SRH, Benammi M, Chaimanee Y, Crochet J-Y, de Franceschi D, Iqbal N, Jaeger J-J, Metais G, Roohi G, Welcomme J-L (2005) Anthropoid primates from the Oligocene of Pakistan (Bugti Hills): data on early anthropoid evolution and biogeography. Proc Natl Acad Sci USA 102:8436–8441
Ni X, Wang Y, Hu Y, Li C (2004) A euprimate skull from the early Eocene of China. Nature 427:65–68
Radinski LB (1977) Early primate brains: facts and fiction. J Hum Evol 6:79–86
Rasmussen DT (1986) Anthropoid origins: a possible solution to the Adapidae-Omomyidae paradox. J Hum Evol 15:1–12
Rasmussen DT (1990) The phylogenetic position of Mahgarita stevensi: Protoanthropoid or lemuroid? Int J Primatol 11:439–469
Rohlf FJ (1990) Rotational fit (Procrustes) Method. In: Rohlf FJ, Bookstein FL (eds) Proceedings of the Michigan morphometrics Workshop. The University of Michigan Museum of Zoology, pp 227–236
Ross CF, Williams BA, Kay RF (1998) Phylogenetic analysis of anthropoid relationships. J Hum Evol 35:221–306
Saban R (1963) Contribution a l’étude de l’os temporal des primates. Description chez l’homme et les prosimiens. Anatomie comparée et phylogénie. Mém Mus Natl Hist Nat Paris, sér A 29:1–378
Schmelzle T, Sánchez-Villagra MR, Maier W (2007) Vestibular labyrinth diversity in diprotodontian marsupial mammals. Mamm Stud 32:83–97
Seiffert ER, Simons CVM, Clyde WC, Rossie JB, Attia Y, Bown TM, Chatrath PS, Mathison ME (2005) Basal anthropoids from Egypt and the antiquity of Africa’s higher primate radiation. Science 310:300–304
Silcox MT, Bloch JI, Boyer DM, Godinot M, Ryan TM, Spoor F, Walker A (2009) The semicircular canal system in early primates and euprimates. J Hum Evol 56:315–327
Simons EL (1962) A new Eocene primate genus, Cantius, and a revision of some allied European lemuroids. Bulletin of the British Museum (Natural History). Geology 7:1–36
Specht M (2007) Spherical surface parameterization and its application to geometric morphometric analysis of the braincase. PhD dissertation, University of Zürich Irchel, Zürich
Specht M, Lebrun R, Zollikofer CPE (2007) Visualizing shape transformation between Chimpanzee and human braincases. Visual Comput 23(9–11):743–751
Spoor F (1997) Basicranial architecture and relative brain size of Sts 5. (Australopithecus africanus) and other Plio-Pleistocene hominids. S Afr J Sci 93:182–187
Spoor C, Zonneveld F (1998) Comparative review of the human bony labyrinth. Am J Phys Anthropol 27:211–251
Spoor F, Zonneveld F, Macho G (1993) Linear measurements of cortical bone and dental enamel by computed tomography: applications and problems. Am J Phys Anthropol 91(4):469–484
Spoor F, Garland TJR, Krovitz G, Ryan TM, Silcox MT, Walker A (2007) The primate semicircular canal system and locomotion. Proc Natl Acad Sci USA 104(26):10808–10812
Szalay FS (1971) The European adapid primates Agerina and Pronycticebus. Am Mus Novit 2466:1–18
Szalay FS (1976) Systematics of the Omomyidae (Tarsiiformes, Primates). Taxonomy, phylogeny, and adaptations. Bull Am Mus Nat Hist 156:157–450
Tafforeau P, Boistel R, Boller E, Bravin A, Brunet M, Chaimanee Y, Cloetens P, Feist M, Hoszowska J, Jaeger J-J, Kay RF, Lazzari V, Marivaux L, Nel A, Nemoz C, Thibault X, Vignaud P, Zabler S (2006) Applications of X-ray synchrotron microtomography for non-destructive 3D studies of paleontological specimens. Appl Phys Mater 83(2):195–202
Walker A, Ryan TM, Silcox MT, Simons EL, Spoor F (2008) The semicircular canal system and locomotion: the case of extinct lemuroids and lorisoids. Evol Anthropol 17:135–145
West CD (1985) The relationship of the spiral turns of the cochlea and the length of the basilar membrane to the range of audible frequencies in ground dwelling mammals. J Acoust Soc Am 77(3):1091–1100
Williams BA, Kay RF, Kirk EC, Ross CF (2010) Darwinius masillae is a strepsirrhine—a reply to Franzen et al. (2009). J Hum Evol 59(5):574–579
Acknowledgments
We thank Pascal Tassy and Christine Argot for giving access to the cranium of Pronycticebus gaudryi. We are grateful to Hugo Dutel and Olivier Lambert, who helped to perform the scan of Pronycticebus, and to the “ATM forms possibles”, for financing that scan. We express our gratitude to Marcia Ponce de León (Anthropological Institute and Museum Zürich) for giving access to extant specimens and to scanning facilities. We thank the staff of beamlines ID19 and ID17 (European Synchrotron Radiation Facility), and the Montpellier RIO Imaging (MRI) platform. We are grateful to Lionel Hautier, Matt Low and Alan Heaver from the University of Cambridge for giving access to the Cambridge specimen of Adapis parisiensis and for performing the CT-Scan. We also are grateful to the following curators: Edmée Ladier (Musée d’Histoire Naturelle de Montauban), Nathalie Mémoire (Musée d’Histoire Naturelle de Bordeaux), Kurt Heissig (Museum und Institut für Paläontologie, München), Burkart Engesser and Arne Ziems (Naturhistorisches Museum Basel), Jacques Cuisin (Collection des Mammifères et des Oiseaux, Muséum d’Histoire Naturelle de Paris), Monique Vianey-Liaud, Bernard Marandat and Suzanne Jiquel (Institut des Sciences de l’Evolution de Montpellier). This research was supported by the French ANR-ERC PALASIAFRICA Program (ANR-08-JCJC-0017). We also thank Mark N. Coleman and Timothy M. Ryan for their helpful comments and suggestions.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is a contribution to the special issue “Messel and the terrestrial Eocene—Proceedings of the 22nd Senckenberg Conference”
Electronic supplementary material
Below is the link to the electronic supplementary material.
Figure S1
Landmarks used for geometric morphometric analysis of the bony labyrinth (specimen: Lepilemur ruficaudatus AIM-11054). Grey arrows anteromedial-to-posterolateral and anterolateral-to-posteromedial directions. (PNG 703 kb)
Figure S2
Position within the skull of the left labyrinth of a Adapis parisiensis b Otolemur crassicaudatus, c Tarsius syrichta and d Callithrix jacchus. Left: superior view of the skull and left labyrinth, the superior part of the calvaria being virtually removed. Right stereoscopic lateral view of the left labyrinth within the braincase. In Adapis parisiensis, the labyrinth is positioned in a more lateral position relatively to the brain structures. Arrows in Otolemur, Tarsius and Callithrix, the posterior canal assumes a lower position relatively to the lateral canal. Scale bars 1 cm. Specimens: a Cambridge M 538, b AIM-1841, c AIM-1732; d AIM-10168. (PNG 2862 kb)
Rights and permissions
About this article
Cite this article
Lebrun, R., Godinot, M., Couette, S. et al. The labyrinthine morphology of Pronycticebus gaudryi (Primates, Adapiformes). Palaeobio Palaeoenv 92, 527–537 (2012). https://doi.org/10.1007/s12549-012-0099-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12549-012-0099-z