Abstract
Polyurethanes foaming is considered as one of key technologies in refrigerator manufacturing to ensure insulation performance. We propose a method for the prediction of refrigerator panel deformation with considering full processes such as filling of polyurethane, chemical reactions, and thermal deformation of solidified polyurethane during foaming. The important influencing factors of the after-foaming deformation are studied and defined using three physical properties: temperature, density, and pressure in order to predict the panel deformation precisely. Analysis of the factors affecting the final deformation resulted in greater than 80% accuracy for each factor compared with those of experiment. Based on the fundamental research results, we tried to predict the deformation of a rear panel after foaming process. We evaluated the usefulness of the proposed analytical approach by experiments, and good agreements between two results obtained. So, we believe that this approach can be used to predict the deformation of a refrigerator panel and some side effects on it.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Recently, the energy consumption of household appliances has been considered as one of important issues to be settled down. Generally, the performance of a refrigerator can be divided into two factors, cooling ability to adjust the internal temperature quickly to a target temperature, and heat insulating performance to minimize heat transfer to the outside environment [1,2,3,4]. Polyurethane (PU) foams are widely used in industries such as construction, automotive, and home appliances, because of their high value characteristics like lightweight, adiabatic effect, somewhat high strength, and high productivity [5,6,7].
Insulation, which is known as a key technology for energy saving, is maintained through the insulator placed between the outer and inner walls of the refrigerator. To provide insulation ability in a refrigerator, the insulation material being used in the door and cabinet is PU foams. Polyol and isocyanate are mixed with water to produce rigid PU following a chemical reaction. As the process is accompanied by a rapid volume expansion and curing, an air layer known as the internal cell is formed. This internal cell in the solidified PU foam is responsible for refrigerator insulation [8, 9]. A filling process for the production of rigid PU insulation is an essential refrigerator manufacturing process. In the process, sol–gel state urethane is infilled into between the steel plate (the outside of a refrigerator) and inner case (inner wall). As mentioned, the PU foam has good heat insulation performance, but there are quality problems related to the deformation of the cabinet of a refrigerator after the filling and curing of PU foams. The outer shape of a product is an important factor to buy it. Therefore, the deformation of the cabinet should be solved to manufacture a higher valued refrigerator. Up to now, research on simulating the internal foaming process is intensively on going, however, unfortunately, clear concepts and theories have not been established and reported. Especially, the prediction of the post-deformation of structures with thermal deformation after the foaming process is a marginal step.
In this study, in order to solve the deformation of a refrigerator cabinet considering full processes such as filling, curing, and thermal deformation of PU foam. For this, basic experiments like the temperature, density, and pressure measurement were conducted during the filling process of PU foam. Then, the filling behavior of the refrigerator was analyzed using a commercial analysis code of Anyfoam [10]. In this connection, the final deformation was predicted through the thermal transformation analysis code of ABAQUS [11] by conducting a material coupling derived from the fill analysis.
2 Theoretical Study on Foaming Reaction
2.1 Theory
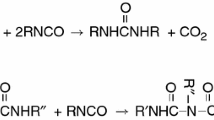
Several chemical reactions occur during the foaming process along with various phenomena; however, there are a significant lack of basic research and test values related to the process. In order to interpret and implement the series of phenomena occurring during the process, the numerical analyses are conducted using some basic chemical and undiluted properties of the process, although they are limited. For the simplicity and generalization of interpretation, the chemical reaction in the process assumed three main responses: ① gelling reaction: fluid solidification reaction, ② chemical blowing reaction: the reaction whereby the molecular structure is changed by chemical reaction, and ③ physical blowing reaction: the molecular structure expanded on absorbing gas.
The allophanate reaction and biuret reaction, which appear incidentally, assumed to be included in the basic reactions, and the chemical reaction rates of the gelling and blowing reaction are independent of each other. The PU foams are produced in a single phase, and the vaporization of the physical foams occurs because the temperature is raised by the exothermal reaction. It is assumed that the production of carbon dioxide is controlled by the chemical blowing reaction, which is the reaction of water with isocyanate [12]. Through the assumption that the internal cells appear regularly after the hard PU filling process, the density distribution and temperature change over time can be determined by the energy and mass conservation laws with the reaction rate and thermodynamic formula of the polyol and isocyanate. The energy conservation equation of the foam expansion that is followed by the reaction with water can be expressed as Eq. (1) [13]. The notations in equations are listed in the section of nomenclature. The PU formation is a nonreversible reaction in which heat is generated by the chemical vaporization of the PU and the heat generated by the vaporization of the foam is not transferred back to the solid. Thus, a general equation can be expressed in terms of the Arrhenius equation.
However, according to Tesser et al. [14], a large amount of heat is generated during the PU chemical reaction in the closed mold, which is not smoothly transferred to the outside, and the heat is applied to the foamed raw material once again. When the temperature dependence of the liquid phase and solid phase is regarded as a reversible reaction, the equation is as follows.
2.2 Physical Properties
The reaction rate, internal heat, foam strength, and others are determined according to the contents of each composition in the liquid isocyanate and resin blend [15]. In order to predict the filling behavior of the foam, it is necessary to measure the properties of the forming solution according to mixing conditions.
A cup test is a widely used method for determining internal reactional relationships in chemical combinations. The reaction rate and rising temperature depend on the major PU component and additives. Therefore, in order to measure the chemical reaction rate of the premixed foam liquid injected into a refrigerator, a data on the variation of foam height (h) during chemical reaction should be obtained through the cup test. Also, a numerical simulation was carried out simultaneously through a three-dimensional computer aided design method. The test results yielded a maximum height of about 200 mm after 100 s. In Fig. 1, the simulation and experimental results (rise profile) on the cup test are compared and summarized. The Anyfoam was used to analyze the profile over time based on the governing equations mentioned in Eqs. (1) and (2). The results show that the behavior of the test and simulation is in good agreement.
After the liquid-state PU solution is injected into the refrigerator mold, the vertical and horizontal foaming rates should be measured according to the injection rate for analysis. The initial position of the liquid-state PU before the foaming reaction occurred was used to determine the foaming behavior after the placement of liquid-state PU. As shown in Fig. 2a, the horizontal position test was performed by placing the nozzle at a height of 300 mm from the ground. After the foaming reaction was completed, the Δt in Fig. 2 showed 98% accuracy when compared to that obtained from the simulation result (∆t) (see Fig. 2b, c).
3 Foam Filling Behavior Inside a Mold
In order to approximate the actual foaming process of a refrigerator experimentally, a mold with 2 m long was utilized. The PU is injected, causing a chemical reaction, and the temperature and pressure change with time in the mold. The density of the final foamed product after the foaming reaction can be measured. The shape of the mold and sensing points are depicted in detail in Fig. 3.
3.1 Temperature Variation
To measure the inner temperature of the test mold over time, the thermocouples were set at 10 points in the center of the thickness direction of the PU foam (see Fig. 4). As the undiluted solution is injected at the lower end of the mold and filled into the upper part, symmetric conditions are set in the width direction. And five points along the center line, other five points in the middle line of the side wall and the center line at equal intervals are arranged in the longitudinal direction based on the center line (‘A’ in Fig. 4). The initial conditions were set at a mold temperature of 45 °C and a foaming stock solution temperature of 26 °C, which were the conditions used during actual refrigerator manufacturing. The foam was separated from the mold after 255 s. The initial temperature rise curve showed that the start time progressively delayed because the foaming reaction occurred firstly near the inlet caused by the positional difference. The center and middle lines showed a maximum temperature rise of 154 °C regardless of their positions, and they slowly cooled down after reaching the maximum temperature.
Simulations have conducted after imposing the same conditions as the experiment. The temperature distribution of the center and middle line at time is shown in Fig. 5. The solid lines denote the experimental test results and the dotted line is the simulation results. The accuracy of numerical prediction with respect to the test is about 88% of the mean value, and the root mean square value is about 98%. The temperature at the 1770 mm point in the center line is cooled relatively fast compared with the other points. The reason is that the 1770 mm point in the centerline locates far from the injection port. The effects of location of mold length and gravity were not considered in the simulation, however it may be necessary to be improved in the further study.
3.2 Density Distribution After Foaming Process
After the PU reaction, the internal density of the fully solidified foam was measured. The 2 m long mold foam was cut into some pieces of 90 × 90 × 20 mm except for the part in contact with the mold, and the density of each piece was measured and compared with the simulation. As shown in Fig. 6, the density of the foam in the width direction is symmetrical, and the density decreases as the progress of foaming in the longitudinal direction. We believe that the reason is that the foamed portion has a relatively large cell volume because the foamed portion at the initial stage has higher pressure after foaming, and it requires longer final foaming time than at the final stage. And, the reason why the lower right portion has higher density is that irregular initial seizure occurs because the injection nozzle is located at the lower left portion [16].
3.3 Pressure in Foaming Process
During the foaming process, the physical foaming reaction generates pressure on the outer mold that has great influence on the deformation after the foam component is taken out of the mold. In this study, pressure gauges were installed on the mold surface to measure the pressure generated in the mold. The positions for the pressure measurement were 170, 570, 970, 1370, and 1770 mm from the injection nozzle of foam. The sensor used for pressure measurement was VPRF-5 k from Valcom Co., Ltd. As the amount of the foaming agent influences the foaming pressure [17], the pressure at each position was measured while changing the injection amount.
Figure 7 shows the pressure measurement results under various conditions. The pressure due to the foaming reaction increases as the foaming liquid increases, and decreases as the distance from the injection port increases. Just packing is a theoretical method that injects a foam solution to match the entire volume exactly. The just packing point of 1770 mm point showed a contraction phenomenon where minus (−) pressure occurred when the low pressure occurred at 200 s in the mold due to a small amount of bubbling liquid. Through the experimental data, we have derived the equation for predicting the pressure obtained based on density and temperature.
4 Prediction of Deformation of Refrigerator Panel
4.1 Prediction of Density During Foam Filling
Based on the accuracy of the material properties and mold state described above, a refrigerator cabinet model that was actually mass-produced was subjected to the foam filling analysis and then the internal densities were compared. In the actual manufacturing process of a refrigerator, after removing the panel on the exterior surface and the innercase, we measured the foam density profiles as shown in the Fig. 8 (top side: one point, middle barrier: one point, bottom side: one point, right side: three points, left side: three points, and center: three points). The measurement period was a box plot of 17 sample measurements through random sampling of refrigerators, and the measurements had been performed over a long-term period (9 months) to reflect the sampling scatter. Based on the actual process conditions and density profiles, the numerical analyses were conducted and the results were summarized as shown in Fig. 9. The internal filling starts from four injection nozzles and the final state of density and temperature were calculated. The simulation results show that the accuracy of mean value is near 99% (max. error 2.3% at point rear #4), the overall results are quiet similar to those of experiment on the angle point of density distribution (see Fig. 10). We think that it is necessary to consider the real ambient temperature where the foaming takes place for high precise prediction.
4.2 Behavior of Polyurethane Foam
Generally, during the foaming process, a volume expansion accompanied by the generation of CO2 gas occurs in the P chemical reaction, thereby causing pressure (expansion effect) [18, 19]. By the pressure in the mold, deformation occurs when a solidified PU structure is demolded. Also, thermal deformation occurs due to thermal expansion coefficient until the inner temperature goes down to room temperature (cooling shrinkage effect). In order to consider these phenomena, the deformation of the PU structure after foaming is assumed as the constitutive Eq. (3).
where εelast is the elastic strain due to internal pressure during foaming reaction and εtherm is strain due to thermal expansion and contraction.
As shown in Fig. 11a, the modulus of elasticity (E) of the internal foam due to the solidification of the PU is affected by the final density of PU; higher density, higher elastic modulus. In the numerical simulation model, the elastic modulus were reflected to every finite elements based on the predicted results of density that were obtained by interpolation and extrapolation of measured data. The analysis was performed through ABAQUS user’s MAT subroutine (UMAT). Deformation due to the relative shrinkage difference among the local positions in the foam is reflected in the numerical analysis when the thermally expanded high temperature foam structure is cooled down to room temperature assuming that the PU foam is completely solidified immediately after the demolding.
Figure 11b shows the thermal expansion coefficient of PU foam according to density variation. As the density increases, the coefficient of thermal expansion decreases linearly. The initial temperature was applied to each element in the finite element model, and the mold and room temperature were assumed as 25 °C.
4.3 Prediction of Thermal Deformation
In the refrigerator foaming process, about 2 h after the demolding process, the chemical reaction inside the refrigerator is mostly stabilized and the deformation does not progress further. After the filling of the PU liquid, structure analysis was done for the prediction of shrinkage that occurred in the cooling process. As mentioned above, the PU foam is directly affected by density variation. So, we configured an additional program using the ABAQUS UMAT based on test data. The relation between the modulus of elasticity and coefficient of thermal expansion according to density variation was established in the analysis.
In the structure analysis, temperature, density, pressure, and chemical reaction, which were obtained from Anyfoam filling analysis results, were considered. The initial stress values of initial foam in the model were coupled with those of Anyfoam results. The pressure distribution was derived by regression analysis of the function on temperature and density. Tables 1 and 2 show the thickness and physical properties of the structures reflected in the thermal deformation analysis.
In order to compare the analysis results with experimental ones, the deformation of the outer rear panel of the refrigerator was measured using three-dimensional scanning after 2 h later from demolding. The scanner (GOM’s ATOS3) has a resolution of about 5 μm. Three samples were measured, and the deformation patterns and average amounts were obtained. The measurement results are shown in Fig. 12a. The direction of the view is the backside of the refrigerator. The maximum deformation occurs at the bottom of the center and protrudes outwards. The averaged value of deformation was 3.35 mm.
Figure 12b shows the analysis result of deformation at the same condition of experiment. The error amount between the analytical value and test value is approximately 0.07 mm, and the accuracy estimates as 81.8%. The deformation mode and maximum value matched well compared with experiment and analysis results. Therefore, we believe that the proposed approach can be utilized for prediction of deformation of a refrigerator cabinet with considering full processes including filling, chemical reaction and thermal deformation.
5 Conclusion
The prediction of deformation of a refrigerator cabinet after foaming process is important to manufacture high quality products. However, unfortunately, there is no report on this issue for precise prediction of deformation with considering the full foaming processes. To solve this problem, we developed an analytical approach by coding in-house codes and evaluated the usefulness of the proposed method. In this work, the deformation of a refrigerator panel after the foaming process was predicted by full conditions like filling of PU, chemical reaction, and thermal–mechanical deformation. The density of PU foam was predicted through the pre-foam filling analysis and the final filling results were calculated by coupling the physical test data obtained an actual foaming process to the finite element model. Through this work, it was possible to reduce the insulation of the refrigerator cabinet and to minimize deformation of the cabinet by predicting the unfilled or weak density areas through the foam filling analysis.
Abbreviations
- \(C\) :
-
Specific heat (J/kg K)
- \(CO_{2}\) :
-
Mass of carbon dioxide per unit polymer mass
- \(G\) :
-
Mass of physical blowing agent in the gas phase per unit polymer mass
- \(L\) :
-
Mass of physical blowing agent in the liquid phase per unit polymer mass
- BG:
-
Physical blowing agent in gas phase
- BL:
-
Physical blowing agent in liquid phase
- \(( - H)_{OH}\) :
-
Heat of gelling reaction (J/g-equiv)
- \(\left( { - H} \right)_{W}\) :
-
Heat of blowing reaction (J/g-mol)
- \(\left[ {OH} \right]\) :
-
Concentration of OH end groups (g-equiv/m3)
- OH:
-
Polyol hydroxyl
- o:
-
Initial value
- \(\rho_{P}\) :
-
Density of polymer (kg/m3)
- \(X\) :
-
Fractional conversion
- x:
-
Mole fraction in the polymer
- \(\lambda\) :
-
Heat of vaporization of blowing agent per unit mass (J/kg)
- R:
-
Gas law constant (J/g-mol K)
- T:
-
Temperature (K)
References
Lee, G. H., Park, B. K., & Lee, W. I. (2017). Microstructure and property characterization of flexible syntactic foam for insulation material via mold casting. International Journal of Precision Engineering and Manufacturing,4(2), 169–176.
Członka, S., Bertino, M. F., Strzelec, K., Strąkowska, A., & Masłowski, M. (2018). Rigid polyurethane foams reinforced with solid waste generated in leather industry. Polymer Tesing,69, 225–237.
Tan, S., Abraham, T., Ference, D., & MacOsko, C. W. (2011). Rigid polyurethane foams from a soybean oil-based Polyol. Polymery,52, 2840–2846.
Lee, D. G., Park, J. H., Park, K. H., Ha, B. K., Kim, H. S., & Park, S. H. (2013). Investigation of the frictional impulse induced abnormal noise in a refrigerator. International Journal of Precision Engineering and Manufacturing,14(4), 559–566.
Gwon, J. G., Sung, G., & Kim, J. H. (2015). Modulation of cavities and interconnecting pores in manufacturing water blown flexible poly(urethane urea) foams. International Journal of Precision Engineering and Manufacturing,16(11), 2299–2307.
Lee, L. J., Zeng, C., Cao, X., Han, X., Shen, J., & Xu, G. (2005). Polymer nanocomposite foams. Composite Science and Technology,65, 2344–2363.
Gwon, J. G., Kim, S. K., & Kim, J. H. (2016). Sound absorption behavior of flexible polyurethane foams with distinct cellular structures. Materials and Design,89, 448–454.
Sung, G., Kim, J. W., & Kim, J. H. (2016). Fabrication of polyurethane composite foams with magnesium hydroxide filler for improved sound absorption. Journal of Industrial and Engineering Chemistry,44, 99–104.
Thiessen, S., Knabben, F. T., Melo, C., & Gonçalves, J. M. (2018). A study on the effectiveness of applying vacuum insulation panels in domestic refrigerators. International Journal of Refrigeration,96, 10–16.
Trias, F. X., Oliet, C., Rigola, J., & Pérez-Segarra, C. D. (2018). A simple optimization approach for the insulation thickness distribution in house hold refrigerators. International Journal of Refrigeration,93, 169–175.
Anyfoam User’s Manual, Released 2.3.0.11 SNWISE Coporation.
Abaqus, 2017. Analysis User’s Manual, Version 6.12. Dassault Systemes.
Lapprand, A., Boisson, F., Delolme, F., Méchin, F., & Pascault, J. P. (2005). Reactivity of isocyanates with urethanes: Conditions for allophanate formation. Polymer Degradation and Stability,90, 363–373.
Baser, S. A., & Khakhar, D. V. (1994). Modeling of the dynamics of water and R-11 blown polyurethane foam formation. Polymer Engineering & Science,34(8), 642–649.
Tesser, R., Serio, M. D., Sclafani, A., & Santacesaria, E. (2004). Modeling of polyurethane foam formation. Journal of Applied Polymer Science,92, 1875–1886.
Mashhadi, M. M., Lopesa, C. S., & LLorca, J. (2018). Effect of anisotropy on the mechanical properties of polyurethane foams: An experimental and numerical study. Mechanics of Materials,124, 143–154.
Chen, Y., Das, R., & Battley, M. (2017). Effects of cell size and cell wall thickness variations on the strength of closed-cell foams. International Journal of Engineering Science,120, 220–240.
Peter, B., van Herwijnen, W. G. H., Martin, R., & Johannes, K. (2018). Investigation of important influencing factors on the tensile shear strength of two component polyurethane with distinct foaming behavior. International Journal of Adhesion and Adhesives,84, 343–349.
Morris, D. B., & Fogg, B. (1979). Rigid polyurethane foam: Refrigerator cabinet design and construction. International Journal of Refrigeration,2, 105–112.
Acknowledgements
This research was supported by the Technology Innovation Program (20000201) funded by the Ministry of Trade, Industry & Energy (MOTIE, Korea) and also was supported by the Korea institute of Energy Technology Evaluation and Planning (KETEP) and the Ministry of Trade, Industry & Energy (MOTIE) of the Republic of Korea (No. 20173030041340).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yang, WJ., Lee, GY. & Park, SH. Analysis on Chemical and Physical Behaviors of Polyurethane Foam for Prediction of Deformation of Refrigerator Panels. Int. J. Precis. Eng. Manuf. 20, 2041–2049 (2019). https://doi.org/10.1007/s12541-019-00159-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12541-019-00159-0