Abstract
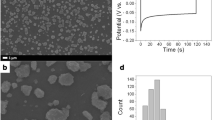
This study experimentally analyzed the influence of the diffusion rate of Cu ions on the growth of microstrucutres during the coelectrodeposition of copper and nickel. To change the diffusion rate of the Cu ions resolved in the electrodeposition solution, electrodeposition was performed under three different stirring conditions (0, 150, 300 rpm). The microstructure grew in an orientation vertical to the microstructure, similar to a probe without stirring, while a popcorn-shaped cluster with electrodeposition particles at the upper part of the microstructure was found under the stirring condition. Forced circulation of the electrodeposition solution by stirring drastically enhanced the movement of the Cu ions, and popcorn shaped clusters are formed. The average height and width of the structure electrodeposited for 20 minutes through stirring at 300 rpm were 1.5 and 4.3 times greater, respectively, than those of the structure electrodeposited without stirring. The substrate surfaces covered with electrodeposited microstructures were modified to be superhydrophobic by coating them with hydrophobic plasma polymerized fluorocarbon (PPFC).
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Shin, H. C., Dong, J., and Liu, M. L., “Nanoporous structures prepared by an electrochemical deposition process,” Advanced Materials, Vol. 15, No. 19, pp. 1610–1614, 2003.
Lee, S., Oh, D., Jung, I., Bae, K., Jung, P., and Chung, K. et al., “Fabrication of nickel micromesh sheets and evaluation of their water-repellent and water-proof abilities,” Int. J. Precis. Eng. Manuf., Vol. 10, No. 3, pp. 161–166, 2009.
Li, G. Y., Li, X. P., Wang, H., Yang, Z. Q., Yao, J. Y., and Ding, G. F., “Fabrication and characterization of superhydrophobic surface by electroplating regular rough micro-structures of metal nickel,” Microelectronic Engineering, Vol. 95, pp. 130–134, 2012.
Michaelis, S., Timme, H. J., Wycisk, M., and Binder, J., “Additive electroplating technology as a post-cmos process for the production of mems acceleration-threshold switches for transportation applications,” Journal of Micromechanics and Microengineering, Vol. 10, No. 2, pp. 120–123, 2000.
Ke, F. S., Huang, L., Cai, J. S., and Sun, S. G., “Electroplating synthesis and electrochemical properties of macroporous Sn-Cu alloy electrode for lithium-ion batteries,” Electrochimica Acta, Vol. 52, No. 24, pp. 6741–6747, 2007.
Bicelli, L. P., Bozzini, B., Mele, C., and D’Urzo, L., “A review of nanostructural aspects of metal electrodeposition,” International Journal of Electrochemical Science, Vol. 3, No. 4, pp. 356–208, 2008.
Qiu, R., Zhang, X. L., Qiao, R., Li, Y., Kim, Y. I., and Kang, Y. S., “CuNi dendritic material: Synthesis, mechanism discussion, and application as glucose sensor,” Chemistry of Materials, Vol. 19, No. 17, pp. 4174–4180, 2007.
Gu, C. D. and Zhang, T. Y., “Electrochemical synthesis of silver polyhedrons and dendritic films with superhydrophobic surfaces,” Langmuir, Vol. 24, No. 20, pp. 12010–12016, 2008.
Zhou, Q., Wang, S., Jia, N., Liu, L., Yang, J., and Jiang, Z., “Synthesis of highly crystalline silver dendrites microscale nanostructures by electrodeposition,” Materials Letters, Vol. 60, No. 29–30, pp. 3789–3792, 2006.
Nikolic, N. D., Popov, K. I., Pavlovic, L. J., and Pavlovic, M. G., “Morphologies of copper deposits obtained by the electrodeposition at high overpotentials,” Surface and Coatings Technology, Vol. 201, No. 3–4, pp. 560–566, 2006.
Nikolic, N. D., Brankovic, G., Maksimovic, V. M., Pavlovic, M. G., and Popov, K. I., “Application of pulsating overpotential regime on the formation of copper deposits in the range of hydrogen codeposition,” Journal of Solid State Electrochemistry, Vol. 14, No. 2, pp. 331–338, 2010.
Hang, T., Li, M., Fei, Q., and Mao, D., “Characterization of nickel nanocones routed by electrodeposition without any template,” Nanotechnology, Vol. 19, No. 3, Paper No. 035201, 2008.
Shao, W. B. and Zangari, G., “Dendritic growth and morphology selection in copper electrodeposition from acidic sulfate solutions containing chlorides,” The Journal of Physical Chemistry C, Vol. 113, No. 23, pp. 10097–10102, 2009.
Deng, M. J., Huang, F. L., Sun, I. W., Tsai, W. T., and Chang, J. K., “An entirely electrochemical preparation of a nano-structured cobalt oxide electrode with superior redox activity,” Nanotechnology, Vol. 20, No. 17, Paper No. 175602, 2009.
Chang, J. K., Hsu, S. H., Sun, I. W., and Tsai, W. T., “Formation of nanoporous nickel by selective anodic etching of the nobler copper component from electrodeposited nickel-copper alloys,” The Journal of Physical Chemistry C, Vol. 112, No. 5, pp. 1371–1376, 2008.
Liu, Z., Xia, G., Zhu, F., Kim, S., Markovic, N., Chien, C. L., and Searson, P.C., “Exploiting finite size effects in a novel core/shell microstructure,” Journal of Applied Physics, Vol. 103, No. 6, Paper No. 064313, 2008.
Wang, Q. T., Wang, G. Z., Han, X. H., Wang, X. P., and Hou, J. G., “Controllable template synthesis of Ni/Cu nanocable and Ni nanotube arrays: A one-step coelectrodeposition and electrochemical etching method,” The Journal of Physical Chemistry B, Vol. 109, No. 49, pp. 23326–23329, 2005.
Choi, W. S., Jung, H. R., Kwon, S. H., Lee, J. W., Liu, M. L., and Shin, H. C., “Nanostructured metallic foam electrodeposits on a nonconductive substrate,” Journal of Materials Chemistry, Vol. 22, No. 3, pp. 1028–1032, 2012.
Ollivier, A., Muhr, L., Delbos, S., Grand, P. P., Matlosz, M., and Chassaing, E., “Copper-nickel codeposition as a model for masstransfer characterization in copper-indium-selenium thin-film production,” Journal of Applied Electrochemistry, Vol. 39, No. 12, pp. 2337–2344, 2009.
Witten, T. A. and Sander, L. M., “Diffusion-limited aggregation, a kinetic critical phenomenon,” Physical Review Letters, Vol. 47, No. 19, pp. 1400–1403, 1981.
Lee, S. M., Park, C. Y., Bae, S. I., Go, J. S., Shin, B., and Ko, J. S., “Surface wettability in terms of the height and depression of diverse microstructures and their sizes,” Japanese Journal of Applied Physics, Vol. 48, No. 9, Paper No. 095504, 2009.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, J.M., Lee, S.H., Kim, Y.J. et al. Effect of the diffusion rate of the copper ions on the co-electrodeposition of copper and nickel. Int. J. Precis. Eng. Manuf. 14, 2009–2014 (2013). https://doi.org/10.1007/s12541-013-0273-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12541-013-0273-x