Abstract
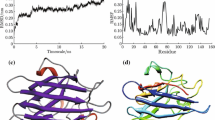
The enzyme catalase breaks down H2O2, a potentially harmful oxidant, to H2O and O2. Besides oxidase activity, the enzyme also exhibits peroxidase activity. Therefore, it plays an important role in maintaining health and regulating pathophysiology of the organisms. However, 3D structure of this important enzyme in invertebrates particularly in crabs is not yet available. Therefore, an attempt has been made to predict the structure of the crab catalase and to envisage its catalytic interaction with H2O2. A three dimensional model of crab catalase was constructed using the NADPH binding site on Beef Liver catalase from Bos taurus (PDBID: 7CAT) as template by comparative modeling approach. Backbone conformation of the modeled structure by PROCHECK revealed that more than 98% of the residues fell in the allowed regions, ERRAT results confirmed good quality of modeled structure and VERIFY3D profile was satisfying. Molecular docking has been used to know the binding modes of hydrogen peroxide with the crab catalase protein. The receptor structures used for docking were derived from molecular dynamics (MD) simulations of homology modeled structure. The docking results showed that the three important determinant residues Arg68, Val70 and Arg108 in catalase were binding with H2O2 as they had strong hydrogen bonding contacts with the substrate. Our analysis provides insight into the structural properties of crab catalase and defines its active sites for binding with substrate. These data are important for further studies of catalase of invertebrates in general and that of crabs in particular.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Altshul, S.F., Gish, W., Miller, W., Myers, E.W., Lipman, D.J. 1990. Basic local alignment search tool. J Mol Biol 215, 403–410.
Bailey, T.L., Elkan, C. 1994. Fitting a mixture model by expectation maximization to discover motifs in biopolymers. In: Proceedings of the 2nd International Conference on Intelligent Systems for Molecular Biology, Menlo Park, California, 28–36.
Berendsen, H.J.C., Postma, J.P.M., Gunsteren, W.F., DiNola, A., Haak, J.R. 1984. Molecular dynamics with coupling to an external bath. J Chem Phys 81, 368–3690.
Bermen, H.M., Westbrook, J., Feng, Z., Gilliland, G., Bhat, T.N., Weissig, H., Shindyalov, I.N., Bourne, P.E. 2000. The protein data bank. Nucleic Acids Res 28, 235–242.
Bohacova, V., Zamocky, M., Godocikova, J., Buckova, M., Polek, B. 2006. The Expression and diversity of catalases in isolates of genus comamonas in response to the oxidative stress of a polluted environment. Curr Microbiol 53, 430–434.
Boon, E.M., Downs, A., Marcey, D. 2010. Catalase: H2O2: H2O2 Oxidoreductase. http://www.callutheran.edu/BioDev/omm/catalase/cat1.htm.
Brooks, B.R., Bruccoleri, R.E., Olafson, B.D., States, D.J., Swaminathan, S., Karplus, M. 1993. CHARMm: A program for macromolecular energy minimization and dynamics calculations. J Comp Chem 4, 187–217.
Chris, O., Alessandra, V., Alan, E., Wilfred, F.V.G. 2004. A biomolecular force field based on the free enthalpy of hydration and solvation: The GROMOS force-field parameter sets 53A5 and 53A6. J Comput Chem 25, 1656–1676.
Colovos, C., Yeates, T.O. 1993. Verification of protein structures: Patterns of nonbonded atomic interactions. Protein Sci 2, 1511–1519.
Eisenberg, D., Luthy, R., Bowie, J.U. 1997. VERIFY3D: Assessment of protein models with three-dimensional profiles. Methods Enzymol 277, 396–404.
Essmann, U., Perera, L., Berkowitz, M.L., Darden, T., Lee, H., Pedersen, L.G. 1995. A smooth particle mesh ewald method. J Chem Phys 103, 8577–8593.
Ewing, T.J., Makino, S., Skillman, A.G., Kuntz, I.D. 2001. DOCK 4.0: Search strategies for automated molecular docking of flexible molecule database. J Comput Aided Mol Des 15, 411–428.
Fita, I., Rossmann, M.G. 1985. The NADPH binding site on beef liver catalase. Proc Natl Acad Sci USA 82, 1604–1608.
Frishman, D., Argos, P. 1995. Knowledge-based protein secondary structure assignment. Proteins 23, 566–579.
Guex, N., Peitsch, M.C. 1997. SWISS-MODEl and the Swiss-PdbViewer: An environment for comparative protein modeling. Electrophoresis 18, 2714–2723.
Hess, B., Bekker, H., Berendsen, H.J.C., Fraaije, J.G.E.M. 1997. LINCS: A linear constraint solver for molecular simulations. J Comput Chem 18, 1463–1472.
Hess, B., Kutzner, C., Spoel, D., Lindahl, E. 2008. GROMACS 4: Algorithms for Highly Efficient, Load-Balanced, and Scalable Molecular Simulation. J Chem Theory Comput 4, 435–447.
Ishida, T., Kinoshita, K. 2007. PrDOS: Prediction of disordered protein regions from amino acid sequence. Nucleic Acids Res 35, W460–W464.
Jones, G., Willett, P., Glen, R.C., Leach, A.R., Taylor, R. 1997. Development and validation of a genetic algorithm for flexible docking. J Mol Biol 267, 727–748.
Laskoswki, R.A., MacArthur, M.W., Moss, D.S., Thornton, J.M. 1993. PROCHECK: A program to check the sterochemical quality of protein structures. J Appl Cryst 26, 283–291.
Laurie, A.T.R., Jackson, R.M. 2005. Q-SiteFinder: an energy-based method for the prediction of protein-ligand binding sites. Bioinformatics 21, 1908–1916.
Linding, R., Jensen, L.J., Diella, F., Bork, P., Gibson, T.J., Russell, R.B. 2003a. Protein disorder prediction: implications for structural proteomics. Structure 11, 1453–1459.
Linding, R., Russell, R.B., Neduva, V., Gibson, T.J. 2003b. GlobPlot: Exploring protein sequences for globularity and disorder. Nucleic Acids Res 31, 3701–3708.
Mates, J.M., Sanchez-Jimenez, F. 1999. Antioxidant enzymes and their implications in pathophysiologic process. Front Biosci 4, 339–345.
McDowall, J. 2010. Catalase. http://www.ebi.ac.uk/interpro/potm/20049/Page1.htm).
Mutsuda, M., Ishikawa, T., Takeda, T., Shigeoka, S. 1996. The catalase-peroxidase of Synechococcus PCC 7942 purification, nucleotide sequence analysis and expression in Escherichia coli. Biochem J 15, 251–257.
Paital, B., Chainy, G.B.N. 2010. Antioxidant defence and oxidative stress parameters in tissues of mud crab (Scylla serrata) with reference to changing salinity. Comp Biochem Physiol C 151, 142–151.
Sali, A., Blundell, T.L. 1993. Comparative protein modeling by satisfaction of spatial restraints. J Mol Biol 234, 779–815.
Sali, A., Overington, J.P. 1994. Derivation of rules for comparative protein modeling from a database of protein structure alignments. Protein Sci 31, 1582–1596.
Sali, A., Matsumoto, R., McNeil, H.P., Karplus, M., Stevens, R.L. 1993. Three-dimensional models of four mouse mast cell chymases, identification of proteoglycan-binding regions and protease-specific antigenic epitops. J Biol Chem 268, 9023–9034.
Sali, A., Pottertone, L., Yuan, F., Vlijmen, V.H., Karplus, M. 1995. Evaluation of comparative protein modeling by MODELLER. Proteins 23, 318–326.
Singh, B.K., Dubey, V.K. 2009. In Silico studies on tryparedoxin peroxidase of Leishmania infantum: Structural aspects. Curr Pharma Biotech 10, 626–630.
Singh B.K., Nandini, S., Jagannadham, M.V., Dubey, V.K. 2008a. Modeled structure of trypanothione reductase of Leishmania infantum. BMB Reports 41, 444–447.
Singh B.K., Nandini, S., Dubey, V.K. 2008b. Modeled structure of trypanothione synthetase of Leishmania infantum for development of novel therapeutics for leishmaniasis. Curr Trend Biotech & Pharma 2, 390–395.
Subbarao, N., Haneef, I. 1991. Defining topological equivalences in macromolecules. Protein Eng 4, 877–884.
Switala, J., Loewen, P.C. 2002. Diversity properties among catalase. Arch Biocehm Biophys 401, 145–154.
Tetko, I.V., Gasteiger, J., Todeschini, R., Mauri, A., Livingstone, D., Ertl, P., Palyulin, V.A., Radchenko, E.V., Zefirov, N.S., Makarenko, A.S., Tanchuk, V.Y., Prokopenko, V.V. 2005. Virtual computational chemistry laboratory — design and description. J Comput Aided Mol Des 19, 453–463.
Thompson, J.D., Higgins, D.G., Gibson, T.J. 1994. CLUSTAL W: Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22, 4673–4680.
Vriend, G. 1990. WHAT IF: A molecular modeling and drug design program. J Mol Graph 8, 52–56.
Wallace, A.C., Laskowski, R.A., Thornton, J.M. 1995. LIGPLOT: A program to generate schematic diagrams of protein-ligand interaction. Protein Eng 8, 127–134.
Wang, R., Lu, Y., Wang, S. 2003. Comparative evaluation of 11 scoring functions for molecular docking. J Med Chem 46, 2287.
Winston, G.W., Giulio, R.T.D. 1991. Prooxidant and antioxidant mechanisms in aquatic organisms. Aquat Txicol 19, 137–161.
Wood, J.M., Decker, H., Hartmann, H., Chavan, B., Rokos, H., Spencer, J.D., Hasse, S., Thornton, M.J., Shalbaf, M., Paus, R., Schallreuter, K.U. 2009. Senile hair graying: H2O2-mediated oxidative stress affects human hair color by blunting methionine sulfoxide repair. Faseb J 23, 2065–2075.
Yang, Z.R., Thomson, R., McNeil, P., Esnouf, R.M. 2005. RONN: The bio-basis function neural network technique applied to the detection of natively disordered regions in proteins. Bioinformatics 21, 3369–3376.
Zdobnov, E.M., Apweiler, R. 2001. InterProScan-an integration platform for the signature-recognition methods in InterPro. Bioinformatics 17, 847–848.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Paital, B., Kumar, S., Farmer, R. et al. In silico prediction and characterization of 3D structure and binding properties of catalase from the commercially important crab, Scylla serrata. Interdiscip Sci Comput Life Sci 3, 110–120 (2011). https://doi.org/10.1007/s12539-011-0071-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12539-011-0071-z