Abstract
During an environmental survey, living (stained) benthic foraminiferal faunas were investigated at 14 stations within the Cassidaigne Canyon (NW Mediterranean Sea) and the surrounding area. For many decades, industrial bauxite residues (namely red mud) have drained into this canyon via a submarine pipe. Stations investigated in this paper are located between water depths of 288 and 2,432 m from the shelf break to the deeper basin and at a distance ranging between ~5 and ~70 km from the pipe outlet. At almost every site, surface sediment is characterized by red mud deposits and their geochemical imprints. Our ecological observations show that foraminiferal standing stocks and simple diversity (S) decrease across the margin in response to the decreasing food input to the seafloor with increasing water depth. The foraminiferal composition echoes the overall meso-oligotrophic patterns of our study area. The contribution of opportunistic and stress-tolerant species, commonly identified as recolonizers of freshly disturbed areas, is minor at the 14 stations. This clearly shows that red mud dispersal in the Cassidaigne Canyon and the surrounding area has no major ecological impact on foraminiferal diversity.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Deep-sea foraminifera (Eukaryota, Rhizaria) are considered a major ecological group of benthic meiofauna (see the review by Gooday 2003). In natural settings, their faunal characteristics (standing stocks, diversity, microhabitat) are controlled by many physico-chemical parameters. Amongst these, organic matter flux reaching the sea floor is the most important ecological constraint in many settings (Gooday 2003). The organic matter flux acts indirectly as an ecological limiting factor when it induces either temporary or long-term hypoxia, either in the sediment or in the bottom water (Fontanier et al. 2014; Gooday et al. 2000; Kurbjeweit et al. 2000; Schumacher et al. 2007). Sediment gravity flows that pass down active submarine canyons can supply organic detritus and inorganic particles to the deep ocean. Foraminiferal faunas living in these disturbed habitats are characterized by either more or less advanced colonization stages occurring after physical disturbance (e.g., turbidity flows), or by equilibrium phases related to the gradual accumulation of organic matter (e.g., eutrophication) (Duros et al. 2011, 2013; Fontanier et al. 2008a, 2008b; Hess and Jorissen 2009; Hess et al. 2005; Koho et al. 2007, 2008). Due to their relatively short life cycle, environmental change can be quickly recorded via changes in the foraminiferal assemblage structure. Therefore, foraminifera make ideal candidates for the monitoring of environmental stress related to human activities (Schönfeld et al. 2012).
Since 1967, the Gardanne alumina refinery (France) has been disposing of bauxite residues (namely red mud) in the Cassidaigne Canyon (see review by Dauvin 2010). Bauxite red mud is drained away by a submarine pipe and discharged at a water depth of 320 m. This sedimentary material spreads along the axis of the Cassidaigne Canyon and on its lateral flanks at great depths (> 2,000 m) (Dauvin 2010; Fabri et al. 2014). Environmental studies have been carried out for the last five decades in order to elucidate the impact of red mud on the deep-sea metazoan benthos (Bourcier 1969; Bourcier et al. 1993; Bourcier and Zibrowius 1973; Fabri et al. 2014; Vitiello and Vivier 1974; Vivier 1978a, 1978b). Close to the pipe outlet, hydro-sedimentary constraints related to the flooding of red muddy material (i.e., high sedimentation rate) preclude benthic meiofauna and macrofauna settlement along the canyon axis. In the surrounding areas, normal macrofauna with suspension and deposit feeders are able to thrive, despite the presence of in a thin layer of red mud.
Fontanier et al. (2012) conducted an ecological investigation of foraminiferal faunas from two stations located at water depths of 725 m and 1,528 m along the axis of the Cassidaigne Canyon (NW Mediterranean Sea) (Fig. 1). At both study sites, sediments were highly contaminated by iron, titanium, vanadium and chromium compared to normal hemipelagic sediments. At the shallower station located close to the pipe outlet, the living benthic foraminiferal community was characterized by very low diversity (only three species) and by an unusual dominance of Gyroidina umbonata (Silvestri, 1898) and Bulimina marginata d’Orbigny, 1826. The physical disturbance related to red mud deposition is likely the major hydro-sedimentary parameter precluding the settlement of diverse fauna. Conversely, the living foraminiferal fauna from the deeper site was typical of oligo-mesotrophic conditions prevailing in natural environments. There, bauxite residues have no obvious environmental impact on foraminiferal faunas.
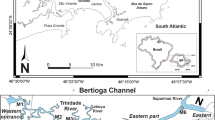
Bathymetry, study area and location of the 14 stations sampled during the ALPEJAN 01 cruise. Triangles correspond to stations also investigated by Fontanier et al. (2012)
Based on more extensive core collections and area coverage, here we investigate the potential impact of red mud dispersal on deep-sea foraminiferal faunas living in the Cassidaigne Canyon and its surroundings (Western Mediterranean Sea). In the framework of an environmental survey, foraminiferal communities were sampled at 14 stations located between water depths of 288 m and 2,432 m, with varying proximities to the pipe outlet (Fig. 1; Table 1). Most of these sites are located in a geographical zone where bauxite residues have been previously detected (Dauvin 2010). The main objective of our study was to assess the environmental impact of dumped red mud on benthic foraminiferal assemblage characteristics and composition during the sampling period (September 2012).
Study area
The Cassidaigne Canyon (also called Cassis Canyon) is situated at the frontier between the eastern Gulf of Lions and the Ligurian Sea (NW Mediterranean). The canyon head, located at a water depth of 200 m, borders the Cassis Bay at a distance of only 7 km away from the coast. The Cassidaigne Canyon is characterized by a narrow canyon axis (1 km in width).
The Northern Current (NC), which forms the northern branch of the cyclonic Liguro-Provençal Current (LPC), follows the continental margin from the Provence coast (France) to the coast of Catalonia (Spain) (Béthoux and Prieur 1983; Millot 1990). The NC determines the general patterns of surface water circulation. Below the surface waters (> 200 m), spreads the modified Levantine Intermediate Water (LIW), which is characterized by a salinity maximum (~38.5) and a relative temperature maximum (> 13 °C). The Western Mediterranean Deep Water (WMDW) occurs below the LIW with a diffusive boundary at 500 to 800 m (Béthoux et al. 2002; Béthoux and Prieur 1983). It is generally characterized by a rather homogeneous temperature (~13 °C) and salinity (38.40–38.45) (Béthoux et al. 2002; Béthoux and Prieur 1983).
Our present study is based on sediment cores collected during the monitoring cruise “ALPEJAN 01”, which took place in September 2012. We sampled 14 stations in the surroundings of the Cassidaigne Canyon (Table 1; Fig. 1). Stations U03 (288 m) and U05 (750 m) are located at the head and on the eastern flank of the Cassidaigne Canyon. Stations U06, U07, U08 and U09 are situated along the Planier Canyon between water depths of 600 and 2,000 m. Both stations U02 and U10 are located along the deep valley where both the Marseille and Planier tributary canyons converge (> 2,100 m). U11 and U12 (> 2,200 m) are under the influence of the Marseille/Planier/Cassidaigne Canyon system. U28, U27 and U26 represent a bathymetric transect from the interfluve between the Petit Rhône and Grand Rhône Canyons (~1,750 m) to the deep basin (~2,400 m). Station U13 is located in the western branch of the Cap-Sicié Canyon (France), less than 7 km from the coast and around 25 km south-east of the pipe outlet.
Material and methods
Samplings
Sediment samples were collected with a box corer (surface area of 2,500 cm2). Two deployments were conducted at almost all sites (Fig. 1; Table 1). In the first box core, two sectors with equal surfaces (1,250 cm2) were defined with a plastic plate (Fig. 2). One sector was subsampled with a Plexiglas tube (internal diameter 9.3 cm, surface area of 68 cm2). The uppermost 2 cm of this sediment core was sliced in half-centimeter intervals and dedicated to foraminiferal analyses.
Benthic foraminiferal analysis
On board, sediment samples dedicated for foraminiferal study were transferred into 250 cm3 bottles, which were filled with 95 % ethanol containing 2 g/L Rose Bengal stain, commonly used to identify live foraminifera (Murray and Bowser 2000; Walton 1952). All samples were gently shaken for several minutes to obtain a homogeneous mixture. Fourteen days after the cruise, they were sieved through a 125-μm screen and the sieve residues were stored in 95 % ethanol. Well-stained foraminifera (all chambers except the last one stained bright pink) were sorted in wet samples and stored in Plummer slides. Strict staining criteria were applied and doubtful individuals without perfectly stained tests were not included. Non-transparent agglutinated and miliolid taxa were broken on many occasions for inspection of the interior of the test. The highest number of stained specimens (285 individuals) was recorded in the 0–0.5 cm interval at station U03 (288 m, Fig. 1). The absence of stained individuals was noted only once, in the 1.5–2 cm sample from station U26 (2,432 m, Fig. 1). Most live foraminifera were identified at the species level (see Appendix A). At each station, we calculated diversity indices, including simple diversity S (representing the number of species), Shannon index H’ (log base e), Rarefied Species Richness E(S35) and Dominance index D (Hayek and Buzas 1997; Murray 2006). These indices were based on counts of stained specimens from the four depth horizons that were analyzed in each core. In order to investigate the spatial variability of live (stained) foraminiferal faunas in our study area, cluster analysis (Ward’s method) was applied for the 14 cores. We used arcsin values for the square root of “pi” for all taxa recorded in each core. The “pi” value is the relative abundance (%) of one species divided by 100. A cluster diagram was constructed (Ward’s method) based on the squared Euclidean distances between all cores.
Results
Visual observations of surface sediments
The surface sediment collected at 12 of the 14 stations included reddish brown deposits (Fig. 2; Table 1). Along the Planier, the Marseille and the Cassidaigne Canyons, this material formed either a centimeter-thick surface layer or patches at the sediment–water interface (Fig. 2). No evidence was found for the deposition of abnormal (bauxite-derived) sediment at stations U05 (745 m water depth) and U13 (958 m water depth). Sieve residues (> 125 μm) were characterized by biogenic particles (mainly dead foraminifera and pteropods) and terrigeneous minerals (mainly quartz and micas).
Foraminiferal faunas
Standing stocks and diversity
Foraminiferal standing stocks ranged between ~50 (U02, 2,104 m) and ~950 (U03, 288 m) individuals/100 cm2 (Fig. 3). Values were lower (< 100 individuals/100 cm2) at depths greater than 2,000 m compared to shallower stations. Simple diversity (S) varied between eight (U26, 2,432 m) and 68 (U03, 288 m) species (Fig. 4). Diversity decreased with increasing water depth, with S values lower than 20 species below 2,000 m. The only exception was recorded at station U13 (958 m), where only 19 taxa were identified. Shannon index H’ and Rarefied Species Richness E(S35) followed the same trend (Fig. 4), as values were low at stations deeper than 2,000 m compared to more shallow sites. Once again, station U13 was an exception, with low H’ (~1.8) and E(S35) (~11) values corresponding to the very low simple diversity and a dominance of Globobulimina spp. Dominance index D and Shannon index values were inversely related.
Faunal composition
At the head of the Cassidaigne Canyon (station U03, 288 m), Melonis barleeanus (Williamson, 1858) (29 %), Reophax scorpiurus de Montfort, 1808 (23 %) and Valvulineria bradyana (Fornasini, 1900) (8 %) dominated the living fauna (Fig. 5). In the Sicié Canyon, on the eastern flank of the Cassidaigne Canyon and along the Planier Canyon axis (< 2,000 m water depth), M. barleeanus was still dominant. Its relative contribution is 10 % and 30 % at stations U13 (958 m) and U05 (745 m), respectively. Globobulimina spp. [mainly Globobulimina affinis (d’Orbigny, 1839)] were particularly dominant (~55 %) at station U13. Uvigerina mediterranea Hofker, 1932 and Uvigerina peregrina Cushman, 1923 were both important contributors (> 10 %) at stations U05, U13, U06, U07 and U08 (ranging between water depths of 745 and 1,547 m). Deeper than ~2,000 m, Nodellum membranaceum (Brady, 1879) was the dominant species, with percentages between 11 % (U02, 2,104 m) and 60 % (U26, 2,432 m) (Fig. 5). Porcellaneous taxa [Pyrgo depressa (d’Orbigny, 1826), Pyrgo anomala (Schlumberger, 1891), Biloculinella labiata (Schlumberger, 1891)], monothalamous agglutinated Lagenammina spp., and epilithic species attached on pteropod fragments (Ammolagena tubulata Rhumbler, 1931, Crithionina mamilla Goës, 1894, Hemisphaerammina bradyi Loeblich and Tappan, 1957) were secondary taxa (Appendix A). Cluster analysis (Ward’s method) of the foraminiferal faunas recorded at our 14 stations is illustrated by a cluster diagram (Fig. 6), which clearly shows that stations belonging to major bathymetric zones are grouped in rather well-constrained sub-clusters (see the sub-clusters 1, 2 and 3 in Fig. 6). The only outlier is station U13.
Discussion
Evidence of red mud dispersal
Reddish brown surface sediment observed at almost every station provides qualitative evidence regarding the geographical dispersal of bauxite residues (Table 1). Only stations U05 (725 m) and U13 (958 m) are not contaminated by red mud deposits. In fact, the titanium (Ti) content of surface sediments at these sites matches quite well with lithological observations (Table 1; Fig. 7). Titanium is considered a geochemical proxy of red mud dispersal (Dauvin 2010; Fontanier et al. 2012). Fairly high Ti values were recorded at most stations in our study area (> 4.000 μg/g), even at great depths (10.500 μg/g at station U11, 2,222 m). In contrast, samples from station U05 and U13 yielded relatively low Ti content (< 2.000 μg/g) close to natural sedimentary signatures (Fig. 7) (Fontanier et al. 2012). It should be kept in mind that Ti content recorded at the 14 stations is much lower compared to those recorded by Fontanier et al. (2012) in the axis of the Cassidaigne Canyon (> 18.000 μg/g). Unsurprisingly, it seems that bauxite residues focus preferentially in the canyon axis, close to the pipe outlet.
Titanium (Ti) and organic carbon (OC) content in surface sediments collected at 14 stations (ALPEJAN 01 cruise, September 2012). The Ti content is indicated using a three-class color code. Organic carbon is indicated by numerical values (in % dry weight). This data was taken from the survey report by CREOCEAN (unpublished data). A portion of this data is available on the ALTEO website (http://www.alteo-environnement-gardanne.fr/-Publications-du-Comite-). Titanium and OC values are also illustrated for both stations located in the axis of the Cassidaigne Canyon (Fontanier et al. 2012). Both stations are identified by triangles
Microscope observations of sieved sediment (> 125 μm) collected at all sites do not reveal any large-sized oxi-hydroxide minerals (boehmite, goethite) that characterize red mud layers sampled in the vicinity of the pipe (Fontanier et al. 2012). It is likely that only fine bauxite-derived particles rich in kaolinite compose the reddish brown layers and patches observed at our sites. Nepheloid layers and sediment gravity flows are considered major hydro-sedimentary processes involved in the transfer of bauxite-derived material from the head of the Cassidaigne Canyon into adjacent canyons and deep basins (Dauvin 2010; Fabri et al. 2014; Fontanier et al. 2012). Furthermore, the episodically strong up-welling and down-welling currents coupled with efficient sediment transfer by both gravity and suspension flows could trigger the complete spatial coverage of the natural seafloor by anthropogenic discharges (Fabri et al. 2014). At a water depth of around 1,200 m, the narrowing of the canyon axis leads to the partitioning of the red mud flux, as a substantial amount of bauxite residue persists along the axis of the Cassidaigne Canyon while the remainder is diverted into the Planier Canyon (Ti frequently > 4.000 μg/g, Fig. 7). Finally, these two distinct pathways seem to converge at a water depth of around 2,000 m, where high Ti concentration is measured (10.500 μg/g at station U11, Fig. 7). From these observations, the total coverage of discharged red mud is estimated to be more than 900 km2. In contrast, stations U05 (725 m) and U13 (958 m) are located in areas not accessible by the bauxite residue (Ti < 2.000 μg/g, Fig. 7). Station U13, located 25 km from the pipe outlet, is sheltered within the neighboring Cap-Sicié Canyon and sits beyond the natural dispersal range of the red mud. Even though station U05 is located proximal to the pipe outlet, its elevated position on a terrace 1,000 m above the canyon floor prevents deposition of bauxite material by sediment gravity flows.
Using caution with foraminifera in biomonitoring studies
This ecological study represents a snapshot of environmental conditions prevailing in September 2012 in the Cassidaigne Canyon and surrounding area. It does not follow a conventional ecological survey based on foraminiferal baselines. Fortunately, many works on living foraminiferal faunas were performed in the Gulf of Lions and in the Ligurian Sea before our present study (Bizon and Bizon 1984; Contreras-Rosales et al. 2012; De Rijk et al. 2000; Fontanier et al. 2008a, 2008b; Goineau et al. 2011, 2012; Schmiedl et al. 2000). They deal with various environmental settings (shelf, open-slope, active and passive canyons), and for this reason, provide useful information concerning what we might expect in terms of natural foraminiferal abundance and distribution in our study area. Furthermore, a foraminiferal response to red mud pollution has already been documented by Fontanier et al. (2012). This later work and other recent papers dealing with foraminiferal recolonization in canyon settings (Duros et al. 2011, 2013; Hess and Jorissen 2009; Hess et al. 2005) provide a reliable basis on which to assess the potential impact of red mud dispersal on foraminiferal biodiversity. To understand the overall variability of an ecosystem, it is recommended to study three replicates at each sampling site (Schönfeld et al. 2012). Therefore, the ecological interpretation of our foraminiferal data set may be problematic since only one core per station is described. However, in most ecological papers dedicated to living (stained) foraminiferal communities from deep-sea ecosystems, only one core per site is commonly investigated. Nevertheless, readers should consider our observations on living faunas with care and consider that our interpretations may be biased by potential spatial (decimetric to metric scale) variability that we unfortunately cannot appreciate with our data sets.
A concern with the use of coring devices for ecological research is the integrity of the retrieved sample. For this study, we used a box corer, which obtains representative samples without the loss of the surface veneer of sediment (Murray 2006). Weather and sea conditions were mild and favorable during the sampling period, which permitted optimal handling of the sampler and minimized sediment disturbance when the corer came back on deck. For instance, we observed well-preserved ripple marks on the sediment surface at the deeper stations (see photographs related to stations U27 and U26 in Fig. 2). For some shallower stations, the sediment surface was chaotic (see photographs related to stations U05, U13, U06, U07 and U10 in Fig. 2). This disturbance of surficial sediments may be related to bow-wave effects during corer penetration. In addition, some clear signs of disturbance of the surface sediments by contact with the box corer were evident at station U10 (Fig. 2). For all these reasons, we retrieved sediment subsamples in the least disturbed and the flattest areas of the box core.
The meso-oligotrophic setting of the Cassidaigne Canyon
Our foraminiferal data show a decrease of both standing stock and diversity (S) with water depth. Low-diversity foraminiferal faunas are recorded in the deeper basin, compared to more diverse communities from bathyal stations. Such an ecological trend was previously documented in slope and canyon environments from the Western Mediterranean Sea (Contreras-Rosales et al. 2012; Fontanier et al. 2008b). Along this margin, this trend is likely related to the scarcity of food at varying depths, which is fairly well illustrated by a decrease of organic carbon content in surface sediments (Fig. 6). This reduction of organic detritus with depth may be related to a diminishing of exported primary productivity. Alternatively, lateral input of altered organic compounds and inorganic particles by nepheloid layers and/or sediment gravity flows could dominate down-slope sedimentary fluxes. This may generate a complex interplay between the concentration and dilution of organic load within anthropogenic (red mud) and hemipelagic deposits. This scenario, based on a dominance of lateral sedimentary transport, can explain the high organic content (~1.10 % DW) recorded at the very deep station U11 (2,222 m), where both the Marseille/Planier and the Cassidaigne Canyons converge (Fig. 1).
That being said, the faunal composition of foraminiferal communities brings complementary and crucial information about the overall environmental conditions prevailing in the Cassidaigne Canyon and its surroundings. Melonis barleeanus is the major species at stations between ~290 and ~2,000 m where organic content is relatively high (> 1.0 % DW). Accordingly, this taxon is abundant in mesotrophic and well-oxygenated ecosystems (Caralp 1989a, 1989b; Duros et al. 2011, 2013; Fontanier et al. 2003, 2005, 2008a, 2008b; Koho et al. 2007; Kurbjeweit et al. 2000; Licari et al. 2003; Schmiedl et al. 2000). In both canyon and open slope settings, M. barleeanus thrives in deep microhabitats below the sediment–water interface where it feeds on degraded organic matter (Duros et al. 2011, 2013; Fontanier et al. 2003, 2005, 2008a, 2008b; Koho et al. 2007; Licari et al. 2003; Schmiedl et al. 2000). This supports the idea that most of the bathyal stations (except U13) are characterized by the input of low-quality organic compounds, likely transported by along-slope currents. Furthermore, the co-occurrence of U. mediterranea and U. peregrina is in agreement with bathyal faunas described in the Western Mediterranean Sea (Bizon and Bizon 1984; Contreras-Rosales et al. 2012; De Rijk et al. 2000; Fontanier et al. 2008a, 2008b; Schmiedl et al. 2000). These uvigerinids are generally described as shallow infaunal species able to feed on relatively fresh organic detritus in mesotrophic environments (Contreras-Rosales et al. 2012; De Rijk et al. 2000; Duros et al. 2011, 2013; Eberwein and Mackensen 2006; Fontanier et al. 2002, 2003, 2006, 2008b; Koho et al. 2007, 2008; Schmiedl et al. 2000). Uvigerina peregrina can behave as an opportunistic taxon feeding on freshly exported phytodetritus in canyon and slope environments, whereas U. mediterranea is a reactive species able to grow and reproduce when food is available in the surface sediment (Duros et al. 2011, 2013; Fontanier et al. 2003). The faunal association between M. barleeanus and uvigerinids shows that mesotrophic conditions prevail naturally in canyon systems, independent of the presence of bauxite residues.
Valvulineria bradyana and R. scorpiurus are noticeably dominant (with M. barleeanus) at the shallowest station U03 (288 m). Valvulineria bradyana commonly thrives in outer-shelf and upper bathyal environments enriched in organic detritus (Fontanier et al. 2002; Goineau et al. 2011; Hess and Jorissen 2009; Langezaal et al. 2006). This species, which is potentially opportunistic, may rely on different types of marine phytodetritus produced throughout the year (Goineau et al. 2011; Langezaal et al. 2006). Moreover, V. bradyana is a major contributor in rich and diverse faunas encountered in canyon-head environments where no redepositional processes occur (Hess and Jorissen 2009). Reophax scorpiurus is well documented from upper to lower bathyal environments (Duros et al. 2011, 2013; Fontanier et al. 2002, 2005, 2013; Kurbjeweit et al. 2000). In some studies, it is reported to occupy a deep infaunal microhabitat, suggesting a preference for degraded organic detritus or bacterial biomass related to organic matter degradation (Gooday 1986). Alternatively, this taxon behaves like a recolonizer, flourishing in areas after physical disturbances such as enhanced bioturbation, turbidites, ash falls, nepheloid layers and deep-sea storms (Fontanier et al. 2013; Hess and Kuhnt 1996; Kaminski 1985). Although the co-occurrence of V. bradyana and M. barleeanus suggests that meso-eutrophic conditions prevail at the shallowest station, the strong contribution of R. scorpiurus (~23 %) may be related to an opportunistic response to physical disturbance driven by hydro-sedimentary instability at this site.
Below 2,000 m, living foraminifera are dominated by a faunal association including N. membranaceum, P. depressa, Thurammina albicans Brady, 1879, Pullenia bulloides (d’Orbigny, 1846) and Gyroidina orbicularis d’Orbigny, 1826. They are all typical of oligotrophic basins from the Western Mediterranean Sea (Bizon and Bizon 1984; De Rijk et al. 2000; Fontanier et al. 2008b, 2012). The presence of epilithic species and suspension feeders (e.g., H. bradyi, C. mamilla, A. tubulata, Ammobaculites agglutinans (d’Orbigny, 1846) and Lagenammina spp.) suggests that bottom currents sweep across canyon and deep basin floors (Contreras-Rosales et al. 2012; Fontanier et al. 2008b, 2013; Nigam et al. 2004; Schönfeld 2002). Such currents, when channeled by the local bathymetry, may contribute to the dispersal of bauxite residues from the upper canyons to the lower margin.
Station U13 (958 m) possesses a large proportion of Globobulimina spp. (54 %). This site is located in the western branch of Cap-Sicié Canyon, 25 km away from the pipe outlet and only 7 km from the coast. As explained above, this sample site is sheltered from the red mud dispersal area. There, sediment contains a high organic content (~1.4 % DW) thattriggers the dominance of Globobulimina spp.. These highly specialized taxa are able to rely on low-quality organic detritus that collects within canyon depressions (Fontanier et al. 2005, 2008a).
Assessing environmental stress in relation to red mud dispersal
In Fig. 8, we illustrate the proportion of opportunistic, stress-tolerant foraminiferal taxa that were documented as potential recolonizers of freshly disturbed areas [Psammosphaera spp., Saccammina spp., Technitella spp., R. scorpiurus, Quinqueloculina seminula (Linneaus, 1758), G. umbonata, B. marginata] (Fontanier et al. 2012, 2013; Hess and Jorissen 2009; Hess et al. 2005; Hess and Kuhnt 1996; Kaminski 1985). At most stations, they account for less than 10 % of the living faunas. At station U03, where opportunistic species account for 30 %, R. scorpiurus may be a reliable proxy of sedimentary instability related to red mud deposition. Despite this, the dominance of M. barleeanus in the highly diverse faunas observed at this station supports the idea of a relatively stable environment. Our faunal observations differ strongly from those found previously in the axis of Cassidaigne Canyon where opportunistic species (G. umbonata and B. marginata) constituted almost all living faunas (Fontanier et al. 2012). Therefore, in light of our foraminiferal data, it appears that the dispersal of bauxite residues in the surrounding Cassidaigne Canyon area has no major ecological impact on foraminiferal diversity.
Conclusions
A previous study performed at the head of the Cassidaigne Canyon (Western Mediterranean Sea) highlighted the impact of mechanical stress related to downslope transport of bauxite deposits on benthic biodiversity (Fontanier et al. 2012). Due to more extensive coring, we can evaluate the impact of red mud dispersal in the Cassidaigne Canyon and its surrounding area. Foraminiferal communities were sampled at 14 stations located between water depths of 288 m and 2,432 m at varying proximities to the pipe outlet (Fig. 1; Table 1). Most of these sites are located in a geographical zone where bauxite residues have been previously detected (Dauvin 2010). Deposits of red mud are observed in the Cassidaigne and Planier Canyons down to ~2,000 m (coverage area ~900 km2). Our study shows that the diversity, composition and standing stock patterns for foraminiferal faunas in this area represent assemblages predominantly constrained by overall meso-oligotrophic conditions. The reduction of sedimentary organic detritus with varying water depths and the ecological constraint determined by bottom currents generate gradual changes in foraminiferal communities, regardless of red mud presence. Compared to the canyon axis, there was no obvious environmental impact of dispersed bauxite residues on benthic biodiversity during the sampling period (September 2012). Future foraminiferal studies focusing strictly on the Cassidaigne Canyon axis, close to the pipeline outlet, are required to fully appreciate the geographical extent of the aforementioned hydro-sedimentary pollution.
References
Béthoux J-P, Prieur L (1983) Hydrologie et circulation en Méditerranée Nord-Occidentale Pétrole et techniques 229:25–34
Béthoux J-P, Durrieu de Madron X, Nyffeler F, Tailliez D (2002) Deep water in the western Mediterranean: peculiar 1999 and 2000 characteristics, shelf formation hypothesis, variability since 1970 and geochemical inferences. J Mar Syst 33–34:117–131
Bizon G, Bizon JJ (1984) Les foraminifères des sédiments profonds. In: Bizon JJ, Burollet PF (eds) Ecologie des microorganismes en Méditerranée occidentale ‘ECOMED’: Ecologie des foraminifères en Méditerranée nord-occidentale. Association Française des Techniciens du Pétrole, Paris, pp 103–139
Bourcier M (1969) Ecoulement des boues rouges dans le canyon de la Cassidaigne (Décembre 1968). Tethys 1:779–782
Bourcier M, Zibrowius H (1973) Les boues rouges déversées dans le canyon de la Cassidaigne (région de Marseille). Observations en soucoupe plongeante SP350 (Juin 1971) et résultats de dragages. Tethys 4:811–842
Bourcier M, Stora G, Gerino M (1993) Réponse du macrobenthos d’un canyon sous-marin méditerranéen à des apports particulaires telluriques et anthropiques. C R Acad Sci Paris III 316:191–196
Caralp HM (1989a) Abundance of Bulimina exilis and Melonis barleeanum: relationship to the quality of marine organic matter. Geo-Mar Lett 9:37–43
Caralp HM (1989b) Size and morphology of benthic foraminifer Melonis barleeanum: relationships with marine organic matter. J Foraminifer Res 19:235–245
Contreras-Rosales LA, Kohoa KA, Duijnsteea IAP, de Stigter HC, García R, Koning E, Epping E (2012) Living deep-sea benthic foraminifera from the Cap de Creus Canyon (western Mediterranean): faunal–geochemical interactions. Deep-Sea Res I 64:22–42
Dauvin JC (2010) Towards an impact assessment of bauxite red mud waste on the knowledge of the structure and functions of bathyal ecosystems: the example of the Cassidaigne Canyon (north-western Mediterreanean Sea). Mar Pollut Bull 60:197–206
De Rijk S, Jorissen FJ, Rohling EJ, Troelstra SR (2000) Organic flux control on bathymetric zonation of Mediterranean benthic foraminifera. Mar Micropaleontol 40:151–166
Duros P, Fontanier C, Metzger E, Pusceddu A, Cesbron F, de Stigter HC, Bianchelli S, Danovaro R, Jorissen FJ (2011) Live (stained) benthic foraminifera in the Whittard Canyon, Celtic margin (NE Atlantic). Deep-Sea Res I 58:128–146
Duros P, Fontanier C, Metzger E, Cesbron F, Deflandre B, Schmidt S, Buscail R, Zaragosi S, Kerhervé P, Rigaud S, Jorissen FJ (2013) Live (stained) benthic foraminifera from the Cap-Ferret Canyon (Bay of Biscay, NE Atlantic): a comparison between the canyon axis and the surrounding areas. Deep-Sea Res I 74:98–114
Eberwein A, Mackensen A (2006) Regional primary productivity differences off Morocco (NW-Africa) recorded by modern benthic foraminifera and their stable carbon isotopic composition. Deep-Sea Res I 53:1379–1405
Fabri M-C, Pedel L, Beuck L, Galgani F, Hebbeln D, Freiwald A (2014) Megafauna of vulnerable marine ecosystems in French mediterranean submarine canyons: Spatial distribution and anthropogenic impacts Deep-Sea Res II
Fontanier C, Jorissen FJ, Licari L, Alexandre A, Anschutz P, Carbonel P (2002) Live benthic foraminiferal faunas from the Bay of Biscay: faunal density, composition, and microhabitats. Deep-Sea Res I 49:751–785
Fontanier C, Jorissen FJ, Chaillou G, David C, Anschutz P, Lafon V (2003) Seasonal and interannual variability of benthic foraminiferal faunas at 550 m depth in the Bay of Biscay. Deep-Sea Res I 50:457–494
Fontanier C, Jorissen FJ, Chaillou G, Anschutz P, Griveaud C (2005) Live foraminiferal faunas from a 2,800 m deep lower canyon station from the Bay of Biscay: Faunal response to focusing of refractory organic matter. Deep-Sea Res I 52:1189–1227
Fontanier C, Jorissen FJ, Anschutz P, Chaillou G (2006) Seasonal variability of foraminiferal faunas at 1,000 m depth in the Bay of Biscay. J Foraminifer Res 36:61–76
Fontanier C, Jorissen FJ, Geslin E, Zaragosi S, Duchemin S, Laversin M, Gaultier M (2008a) Live and dead foraminiferal faunas from the Saint-Tropez Canyon (Bay of Fréjus): observations based on in situ and incubated cores. J Foraminifer Res 38:137–156
Fontanier C, Jorissen FJ, Lansard B, Mouret A, Buscail R, Schmidt S, Kerhervé P, Buron F, Zaragosi S, Hunault G, Ernoult E, Artero C, Anschutz P, Rabouille C (2008b) Live (stained) foraminiferal faunas from open slope environments separating submarine canyons in the Gulf of Lions (NW Mediterranean): diversity, density and microhabitats. Deep-Sea Res I 55:1532–1553
Fontanier C, Fabri M-C, Buscail R, Biscara L, Koho K, Reichart GJ, Cossa D, Galaup S, Chabaud G, Pigot L (2012) Deep-sea foraminifera from the Cassidaigne Canyon (NW Mediterranean): assessing the environmental impact of bauxite red mud disposal. Mar Pollut Bull 64:1895–1910
Fontanier C, Metzger E, Deflandre B, Waelbroeck C, Jouffreau M, LeFlock N, Jorissen FJ, Etcheber H, Bichon S, Chabaud G, Poirier D, Grémare A (2013) Live (stained) benthic foraminifera off Walvis Bay (Namibia): a deep-sea ecosystem under the influence of benthic nepheloid layer. J Foraminifer Res 43:50–66
Fontanier C, Duros P, Toyofuku T, Oguri K, Koho KA, Buscail R, Grémare A, Radakovitch O, Deflandre B, De Nooijer LJ, Bichon S, Goubet S, Ivanovsky I, Chabaud G, Menniti C, Reichart GJ, Kitazato H (2014) Living (stained) deep-sea foraminifera off Hachinohe (NE Japan, Western Pacific): Environmental interplay in oxygen-depleted ecosystems J Foraminifer Res
Goineau A, Fontanier C, Jorissen FJ, Lansard B, Buscail R, Mouret A, Kerhervé P, Zaragosi S, Ernoult E, Artéro C, Anschutz P, Metzger E, Rabouille C (2011) Live (stained) benthic foraminifera from the Rhône prodelta (Gulf of Lion, NW Mediterrenean): environmental controls on a river-dominated shelf. J Sea Res 65:58–75
Goineau A, Fontanier C, Jorissen F, Buscail R, Kerhervé P, Cathalot C, Pruski AM, Lantoine F, Bourgeois S, Metzger E, Legrand ECR (2012) Temporal variability of live (stained) benthic foraminiferal faunas in a river-dominated shelf—faunal response to rapid changes of the river influence (Rhône prodelta, NW Mediterranean). Biogeosci Discuss 8:9033–9086
Gooday A (1986) Meiofaunal foraminiferans from the bathyal Porcupine Seabight (northeast Atlantic): size structure, standing stock, taxonomic composition, species diversity and vertical distribution in the sediment. Deep Sea Res Oceanogr Abstr 33:1340–1373
Gooday A (2003) Benthic Foraminifera (Protista) as tools in deep-water palaeoceanography: environmental influences on faunal characteristics. Adv Mar Biol 46:1–90
Gooday AJ, Bernhard JM, Levin LA, Suhr SB (2000) Foraminifera in the Arabian Sea oxygen minimum zone and other oxygen-deficient settings: taxonomic composition, diversity, and relation to metazoan faunas. Deep-Sea Res II 47:24–54
Hayek LEC, Buzas MA (1997) Surveying natural populations. Columbia University Press, New York
Hess S, Jorissen FJ (2009) Distribution patterns of living benthic foraminifera from Cap Breton canyon, Bay of Biscay: faunal response to sediment instability. Deep-Sea Res I 56:1555–1578
Hess S, Kuhnt W (1996) Deep-sea benthic foraminiferal recolonization of the 1991 Mt. Pinatubo ash layer in the South China Sea. Mar Micropaleontol 28:171–197
Hess S, Jorissen FJ, Venet V, Abu-Zied R (2005) Benthic foraminiferal recovery after recent turbidite deposition in Cap Breton Canyon (Bay of Biscay). J Foraminifer Res 35:114–129
Kaminski MA (1985) Evidence for control of abyssal agglutinated foraminiferal community structure by substrate disturbance. Mar Geol 66:113–131
Koho KA, Kouwenhoven TJ, de Stigter HC, van der Zwaan GJ (2007) Benthic foraminifera in the Nazaré canyon, Portuguese continental margin: sedimentary environments and disturbance. Mar Micropaleontol 66:27–51
Koho KA, García R, de Stigter HC, Epping E, Koning E, Kouwenhoven TJ, van der Zwaan GJ (2008) Sedimentary labile organic carbon and pore water redox control on species distribution of benthic foraminifera: a case study from Lisbon-Setúbal canyon (southern Portugal). Prog Oceanogr 79:55–82
Kurbjeweit F, Schmiedl G, Schiebel R, Hemleben C, Pfannkuche O, Wallman K, Schäfer P (2000) Distribution biomass and diversity of benthic foraminifera in relation to sediment geochemistry in the Arabian Sea. Deep-Sea Res II 47:2913–2955
Langezaal AM, Jorissen FJ, Braun B, Chaillou G, Fontanier C, Anschutz P, van der Zwaan GJ (2006) The influence of seasonal processes on geochemical profiles and foraminiferal assemblages on the outer shelf of the Bay of Biscay. Cont Shelf Res 26:1730–1755
Licari LN, Schumacher S, Wenzhöfer F, Zabel M, Mackensen A (2003) Communities and microhabitats of living benthic foraminifera from the tropical east Atlantic: impact of different productivity regimes. J Foraminifer Res 33:10–31
Millot C (1990) The Gulf of Lions’ hydrodynamics. Cont Shelf Res 10:884–895
Murray JW (2006) Ecology and applications of benthic foraminifera. Cambridge University Press, New York
Murray JW, Bowser SS (2000) Mortality, protoplasm decay rate, and reliability of staining techniques to recognise ‘living’ foraminifera: a review. J Foraminifer Res 30:66–70
Nigam R, Mazumder A, Saraswat R (2004) Ammolagena clavata (Jones and Parker, 1860), an agglutinated benthic foraminiferal species-first report from the recent sediments, Arabian Sea, Indian Ocean Region. J Foraminifer Res 34:74–78
Schmiedl G, de Bovée F, Buscail R, Charrière B, Hemleben C, Medernach L, Picon P (2000) Trophic control of benthic foraminiferal abundance and microhabitat in the bathyal Gulf of Lions, Western Mediterranean Sea. Mar Micropaleontol 40:167–188
Schönfeld J (2002) Recent benthic foraminiferal assemblages in deep high-energy environments from the Gulf of Cadiz (Spain). Mar Micropaleontol 44:141–162
Schönfeld J, Alve E, Geslin E, Jorissen F, Korsun S, Spezzaferri S, group motF (2012) The FOBIMO (FOraminiferal BIo-MOnitoring) initiative—Towards a standardised protocol for soft-bottom benthic foraminiferal monitoring studies. Mar Micropaleontol 94–95:1–13
Schumacher S, Jorissen FJ, Dissard D, Larkin KE, Gooday AJ (2007) Live (Rose Bengal stained) and dead benthic foraminifera from the oxygen minimum zone of the Pakistan continental margin (Arabian Sea). Mar Micropaleontol 62:45–73
Vitiello P, Vivier MH (1974) Données quantitatives sur la méiofaune d’une zone profonde de déversements industriels. Bull Union Océanographes Fr 6:13–16
Vivier MH (1978a) Conséquences d’un déversement de boue rouge d’alumine sur le méiobenthos profond (canyon de Cassidaigne, Méditerranée). Tethys 8:249–262
Vivier MH (1978b) Influence d’un déversement industriel profond sur la nématofaune (canyon de Cassidaigne, Méditerranée). Tethys 8:307–321
Walton WR (1952) Techniques for recognition of living foraminifera. Contr Cushman Lab Forum Res 3:56–60
Acknowledgments
We thank the crew members of R/V “JANUS II” (COMEX) (M. Puggioni, E. Dombrowski, M. Compère, E. Tanazacq, G. Willem, C. Terzian, J. Morvan, E. Seguin) and all scientific participants (M. Avon – AMC; D. Ribéra, D. Bravalt - BIO-TOX; E. Delord, C. Reveret –CREOCEAN; P. Thibault, B. Langlois, A. Fédérighi - ALTEO) on the ALPEJAN 01 Cruise. We thank members of the Scientific Committee (Comité Scientifique de Suivi) in charge of the biomonitoring assessment. We extend special thanks to Michel Avon (AMC) who provided useful pictures taken during the cruise. C.F. (first author of this paper) performed foraminiferal analyses in autumn 2012, in the framework of an industrial contract financially linking the Université Nantes Angers Le Mans (UMR CNRS 6112 LPG, LUNAM) to ALTEO. In spring 2013, ALTEO allowed C.F. to use foraminiferal data for this publication.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM Appendix A
Census data for live (stained) benthic foraminifera in the > 125-μm size fraction at sampled stations (XLSX 53.7 kb).
Rights and permissions
About this article
Cite this article
Fontanier, C., Biscara, L., Mamo, B. et al. Deep-sea benthic foraminifera in an area around the Cassidaigne Canyon (NW Mediterranean) affected by bauxite discharges. Mar Biodiv 45, 371–382 (2015). https://doi.org/10.1007/s12526-014-0281-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12526-014-0281-9