Abstract
The excavation of Ghar-e Boof, a cave in the Zagros Mountains, places the site not only at the center of discussion on the transition to and development of its regional lithic tradition: the Rostamian (37,000–31,000 BP), but also in differentiating between plants used by humans or mere traces of the surrounding vegetation. The large pulses of Lathyrus or Vicia sp. recovered from this shallow cave in the southwest of Iran may, for instance, represent food collected from wild stands already in the early Upper Palaeolithic. The seeds of barley (Hordeum sp.), although not all clearly domesticated, are without doubt signs of disturbance or bioturbation since the historic era. Analysis of cave deposits over 30,000 years old raise a number of methodological and interpretive challenges. Human, taphonomic, or biomechanical disturbances impact the deposition of plant remains, as well as affect the composition of the assemblages, undermining spatial and ecological examination of the data set. Comprehension of provenance of the samples, site genesis, and matrix development, through detailed micromorphological and stratigraphical studies, is thus suggested in conjunction with the archaeobotanical analyses, to identify disturbances, define their cause, and treat them appropriately. Numerical studies and ecological interpretations of climate, vegetation composition, or indications of human activity therefore follow specific criteria discussed here. Despite signs of disturbance in the plant assemblages, archaeobotanic research can lead to recognition of environmental conditions, plausible human subsistence, site use and seasonality, and sound vegetation description.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
A methodology for archaeobotanical analysis of Upper Palaeolithic cave sediments is discussed through a case study of material from Ghar-e Boof in the Iranian Zagros Mountains. From this cave site, carbonized seed and fruit remains from seven horizons dating to the Rostamian (from 36,000 to 31,000 uncalibrated years BP) were examined. This methodology integrates approaches from various fields in archaeological sciences: botanic (Bouby and Marnival 2004; Borojevic 2011; van der Veen 2007), taphonomic (Johnson 2002), archaeological (Lange 1990) and statistical (Hubbard and Clapham 1992; Jones 1992; Mitka and Wasylikowa 1995; Popper 1988). It first tackles the issues of site taphonomy and formation, alongside intrusions in the plant assemblage of Ghar-e Boof. A secondary focus lies in the description of the extant vegetation and the identification of possible past vegetative food (like the collection of pulses of the Lathyrus and Vicia genera). Understanding the deposition history, chronological patterns and formation of a site, as mentioned in earlier papers (e.g., Allison and Briggs 1991; Behrensmeyer et al. 2000; Miksicek 1987; Schiffer 1983; Spicer 1980, 1991) is essential in reconstructing past diets and plant environment and to separate dubious from reliable evidence. In following these previous researches, which highlighted the issues of disturbance and site taphonomy in interpreting archaeobotanic assemblages from all types of sites and eras, four steps are put forward to address these challenges.
First, identified plant remains must be cataloged for ecological and archaeological temporal and spatial analysis. The second and third steps in the research are micromorphological and topographical analyses. They contribute to detecting and interpreting sedimentation processes and disturbances. The fourth step is in three parts: first, to integrate local animal and human sustenance, then to differentiate between intrusive or anachronistic plants, and finally, to describe the biotic environment. This facilitates recognizing animal and human agency in the deposition of plant remains, on one hand, and characterizing their usage and occupation of the site on the other.
Background
Study area
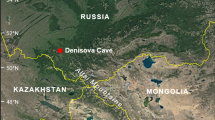
Ghar-e Boof cave was excavated in 2007 as part of Tübingen University’s TISAR Project. It lies in the Dasht-e Rostam valley in the southern Zagros Mountains of Iran, see Fig. 1 (Conard et al. 2006). During the Upper Palaeolithic this range of mountains formed a natural obstacle between the Levant and South West Asia. Dictated by an irregular, obstructive, and varied topography, today’s vegetation in the region is made up of diverse small patches. It is a mosaic of landscape structures and climatic conditions where much biological interchange occurs between the different ecotones. Due to the orientation of the intermountain valleys and their interconnections, the common direction of travel is to follow the range rather than to traverse it (Heydari-Guran 2007; Weeks 2006). Because people had to avoid depleting the wealth of their environment in search of nutrition, traveling across the entire breadth and height of the mountains was the best way of safeguarding prey and edible plants. Sites, though predominantly found nearer the valley floor, have therefore been identified in most parts of the Zagros relief, spreading from 700 to about 2,500 m.a.s.l. (Zeidi et al. 2009). The change in vegetation from the Pleistocene to the Holocene; as identified from lake pollen cores retrieved in the central Zagros Mountains, is characterized by a shift from an upland steppe vegetation to a river valley one (Bottema 1993; Fazeli 2008). Climate changed overall from drier and cooler to slightly damper and warmer conditions (Djamali et al. 2008; Stevens et al. 2001; Wasylikowa 2005).
The sediment was deposited during two broad time periods (see Fig. 2). First, overlying the limestone bedrock, an Upper Palaeolithic layer ~ 1-m-thick dates from around 36 to 31 kyears BP {horizons IV, IVa, IVb, III, IIIa, IIIb, and IIb.1}; see Table 1 for radiocarbon dates. Micromorphological analysis suggests that this layer was little altered or impacted over the following 25,000 years (Schilt 2011). Some remains from the Upper Palaeolithic occupations were also found in horizon IIb, but their deposition is likely the result of human and bio-taphonomic disturbances such as worm activity and root canals. This horizon, together with the surface horizon I and the intermediate layers II and IIa, form the second period of Ghar-e Boof. These horizons consist of modern rubbish, organic debris, some post-Islamic pottery and vast amounts of dung and ash from recent animal penning activities. The matrix itself was mostly comprised of this material as well as a little aeolian sand, mixed in with rockfall from the shelter’s wall and roof.
The Upper Palaeolithic
A review of the archaeology of the Zagros Mountains as presented by Hole and Woosley (1978), Conard and Ghasidian (2011), and Heydari-Guran (2014) reveals that hunter gatherers who frequented these highlands left a rather homogenous material record. Culturally, however, Ghar-e Boof is significant as the type site for the Rostamian lithic tradition. This early Upper Palaeolithic phase, comparable in radiocarbon dates with the Aurignacian in Europe, stands out from the Zagros’ other sites typified as the Baradostian (Ghasidian 2010). The main difference lies in the reduced size, fineness and lamellar appearance of the stone tools more reflective of an epi-Palaeolithic assemblage. Ethnographic accounts of hunter-gatherers provide no viable parallel for Palaeolithic transience and subsistence strategies in the West-Asian highlands. A tenuous assumption, however, that people then subsisted for about 60 % on vegetative foods may seem plausible in view of the diet maintained by the seasonally nomadic herdsman living there today (Gilbert 1983; Stauffer 1965). Landscape structure and seasonality, for example, in recognizing the maturity of edible/useful plants or the prime hunting time were therefore central in archaic migration and temporary residence cycles. Preference for camping or occupying caves in locations that optimized access to resources in relation to traveling time and energy/nutritional return finds support furthermore in Heydari-Guran’s surveys of Palaeolithic occupations in the Southern Zagros mountains (PhD in press).
Materials and methods
All the samples were obtained from a single 2 × 9 m trench. In this article, each square meter is referred to by two digits. The first digit is its Easting (row 6 or 7) and the second its Northing (running from squares 2 to 10). No samples are available from squares 4, 7, and 10. Each sample is the flotation residue of a 20 l bucket of excavated sediment. The samples were then dry-sieved through three mesh sizes: 1, 0.63, and 0.18 mm. These sizes were chosen to ease the sorting process. The finest sieve was chosen to optimize the chances of catching the very smallest seeds. Tübingen University’s comparative collection and reference books were consulted for verifying the identifications with a binocular microscope (Berggren 1981; van Zeist and Bakker-Heeres 1984; Nesbitt 2006; Bojňansý and Fargaŝová 2007). All available materials from horizons IV, IVa, IVb, III, IIIa, IIIb, IIb, and IIb.1 were examined and are presented in Appendix.
Plant remains do not fall into orderly or roughly regular numbers of specimens per square meter of a site. The distribution of seed and fruit may be influenced by human or animal movements, by postdepositional modification or even inadvertent introduction of specimens from the surrounding environment. For these reasons, it was considered appropriate to establish one parameter that is valid for all recovered specimens, and thus we decided to take only an equal volume of sample material. This would allow the analysis of distributions or other spatial factors to be made under standard conditions. About 10 % of the samples came from buckets with a different volume and so were excluded from analysis. A few samples from horizons I, II, and IIa were studied, but were likewise excluded as they were not strictly Upper Palaeolithic. Only carbonized specimens were studied and included in the analyses for this article, because desiccated material was often of a dubious age.
Birks, Cappers, and others have explained that several levels of detail are required in archaeobotanical research (Birks and Birks 2006; Cappers and Neef 2012). We suggest six levels for this site. The left side of the diagram (see Fig. 3) lists them spatially according to the area, location, and amount of material discovered. This from the largest parameter (a regional climate) to the smallest (an identified specimen). The left arrow indicates the decreasing amount of elapsed time covered, or needed for the formation of the material. Hence, we suggest to proceed from the geological timescale of the eons involved in the genesis of landscapes and their climate, to the passing of years for the formation of a site or a number of events for archaeological horizons and assemblages, and finally to a single event for the deposition of an identified specimen. The right-hand side of the diagram lists them qualitatively. The arrow there indicates decreasing complexity, amount of details involved and interpretability of evidence. For this site, the principal reason for the reduced interpretability of the evidence for the three lower levels of the diagram is postdepositional movement. Other causes affecting them, as well as the higher three levels on a longer timescale, are human activities and environmental change.
Following the above perspective, we decided the data set must be treated according to two separate methodologies; one for each aim of this paper (Mitka and Wasylikowa 1995). The first aim is to distinguish between cases of disturbance associated with formation and taphonomic processes, on the one hand, and human activities (past or recent) on the other. This first methodological approach involves analysis of details from the last four levels, to secure a comparative picture. The second aim is to describe the extant vegetation at the site. To achieve this, a list is compiled of the taxa identified from the last level of detail alone. For the first methodology, let us presume that strictly site taphonomic processes affect taxa equally, whereas human activities affect particular taxa. Thus the number of specimens per taxon is significant to distinguish between both cases, because human activities lead to concentrations or anomalies in the plant assemblage, while other disturbances alter the assemblage more homogeneously (Cappers and Neef 2012). For example, the burning of dung raises the volume of small Fabaceae; the laying down of bedding leads to higher Cyperaceae counts (van Zeist and Bakker-Heeres 1984). Similarly, the remains of human subsistence may be recognized in a dominance of edible seed and fruit. For the second aim, however, each identified taxon (either to a family, genus, or species) is counted, regardless of how many specimens were found, because it represents a trace of the surrounding vegetation.
Results
In order to compare the data set spatially and temporally, it was ordered by excavation square and horizon. Due to the volume of material studied (see Table 2); a brief overview of Ghar-e Boof’s plant remains is presented here, with reference to the whole data set in the Appendix. This assemblage of 352 samples, of which 118 had no finds, had 2,876 specimens identified to a plant family.
The plants found may be divided into three groups: those collected as food, those collected for a particular use, and those with no obvious relation to the human occupants of the cave. The first are, for example; small Poaceae seeds and Lathyrus and Vicia pulses (Lev et al. 2005). Gathering of sedges for bedding could be an example from the second group (Sievers and Muasya 2011). The third group are those plants unaffected by the cave occupants and are here referred to as the vegetation background. Because the taxa from the third group usually have a small specimen count, and an unclear status with regard to human activities, they are often discarded from numerical analyses as noise (Jones 1992). As an illustration of how much material remains if taxa are discounted as noise, because they do not reach a certain specimen count, Table 3 illustrates numerically what was found per square only in horizon III. A minimum of seven specimens was chosen as a threshold to separate this noise from the usable data. The count of seven was chosen as it represents the rough average of what is suggested by a few authors, namely, Jones (1992), Lange (1990), Mitka and Wasylikowa (1995), Baxter (1994), Hubbard and Clapham (1992), and Popper (1988).
We decided not to apply criteria for discounting noise because otherwise most of the plant diversity would be discarded in this paper. As may be seen from Appendix, the other horizons scored even poorer than horizon III. Contrasting the botanic data with the number of lithic artefacts found in horizon III puts the paucity in perspective with the level of intensity in occupation of the cave. Totalling 406 cores, 51 complete blades, 286 retouched bladelets, 82 end scrapers, and 92 retouched flakes, these stone artefacts establish Ghar-e Boof as an intensely used “base camp that could have simultaneously served as a workshop” (Ghasidian 2010, p. 161). Even without disregarding noise in the plant assemblage, the overall record remains low in specimens and identified taxa. Nonetheless, three broad interpretations could be made from the results and are presented here with their evidence in separate sections.
Disturbance or human activity
Sieving every bucket of excavated sediment means that localized analyses are possible as shown in Table 4. In some cases, a peculiar variety, quantity of specimens, or specific plants may indicate the results of geogenic or anthropogenic disturbance. In Table 4, the plant assemblages from the four quarters of a square, all recovered from the same level in horizon III, are compared. Identification of Oryza sp. (rice), Triticum sp. (wheat), and Hordeum sp. (barley) in only one quarter, but not the other three subsquares, indicates an anomaly. Moreover, rice and wheat were not found in any of the neighboring squares. The identification of rice, normally not encountered in the region before the Parthian Age, and the barley and wheat with distinctively domesticated morphologies, suggest bioturbation, rather than human activity transported material downwards from the higher levels (Miller 1981).
As mentioned in Fig. 3, identification of crop plants in any one sample does not mean the other specimens in the same sample are also intrusive. While the intrusive specimens reflect deposition with manipulation at a later moment, the other plant remains in the sample can reflect an unaffected in situ deposition, or a series of depositional events. Two observations may be drawn from these results. First, comparisons within or between plant assemblages may, in those cases where movement by disturbances are possible, be invalidated due to the uncertainty of the original deposition of specimens. Second, if discrepancies occur as illustrated in Table 4, they could be examined further to distinguish between anthropogenic or natural disturbances. Following Johnson’s (2002) natural disturbances in a studied matrix may be biomechanical, (e.g., through burrowing animals, worms, or plant roots), or geological like erosion, the movement of water, or temperature changes. The anthropogenic disturbances include pastoral and agrarian activities and the digging of pits and hearths. These actions lead to a recognizable dominance of particular taxa in a plant assemblage, like an increase in legume remains (see Fig. 4) resulting from dung burning (van Zeist and Bakker-Heeres 1984), weed and crop remains from winnowing and threshing (Cappers and Neef 2012), or the downward movement of intrusive material originating in those activities.
Locating intrusive material in the matrix can highlight those areas of a site that were disturbed. For this purpose, Table 5 records where the anachronistic barley grains were found at Ghar-e Boof. Besides their conspicuous seed morphology and rachides, one grain was radiocarbon dated to about 845 BP ± 25. Were it not for these indicators to the contrary, identification of barley grains in a Rostamian context could have furthered debate on inclusion of this cereal in the Upper Palaeolithic diet as grain collected from wild stands (Hillman et al. 1997; Piperno et al. 2004). Some of the small Poaceae seeds, on the other hand, like those of the Panicum, Setaria, or Tetrapogon genera (see Fig. 5), may have been eaten (Weiss et al. 2004). Although only 80 % of the samples with barley grains had intrusive chaff, the other grains are unlikely to be Palaeolithic in date. Before the results of Table 5 are interpreted, further information on site genesis and development must be given. A patch of larger ceiling collapse in the middle of the excavation trench, for instance, protected an underlying Palaeolithic horizon designated IIb.1. Later, this area around the boulders became a focus of dung sweeping and burning. Pits were dug disturbing the matrix up to horizon IIIa. The two modern radiocarbon dates (see Table 1) obtained above and below horizon IIb.1 further highlight the effects of this event. Many samples taken in the vicinity from horizon II, IIa, IIb, and III, but not from the relatively protected IIb.1 area, were disturbed with uncarbonized seed and fruit from the dung and anachronistic Triticum, Hordeum (see Fig. 6) and Oryza which had moved down from the ash lenses. It is noteworthy that samples taken nearer the edge of the excavation reflected more uncarbonized seed and fruit as well as proportionately more intrusive taxa. The likelihood of encountering disturbance could thus be said to increase toward the trench edges compared with the trench middle.
Table 5 shows that barley was recovered down to and within horizon III, an otherwise securely-dated Rostamian horizon, but not in horizon IIb.1. These results confirm Johnson’s opinion that contamination reaches far below the original deposition; in this case, 1.7 m lower down (Johnson 2002). The absence of barley in horizon IIIb, IV, Iva, and IVb is either because a depth limit was reached or because the biomechanical or taphonomic influences affecting the matrix had waned. Indeed micromorphological investigations identified extensive evidence for bioturbation, such as root canals and worm tunnels, in horizon III and the higher horizons, but less in the lower horizons (Schilt 2011). Comparing the occurrence of barley across the excavation, the middle of the cave appears no more affected than the entrance and rear. One could conclude therefore that the dung burning and animal penning activity that was focused in the cave middle was itself less a cause of contamination than the bioturbation and taphonomic disturbances which moved seed and fruit remains downwards.
An indication of how human activity alone affected, but did not disturb, the plant assemblage are the Lathyrus/Vicia remains found at Ghar-e Boof (see Fig. 7). These pulses were found in 21 samples across horizons III, IIIa, and IIIb, but could never be identified more precisely than to this cumulative type. The concentration of Lathyrus (vetchling) or Vicia (vetch) in square 6/3 of horizon III, in particular, point to these legumes being collected as food. The two dates obtained from the Lathyrus/Vicia remains recovered from horizon IIIb: 33,850 BP ± 650 and 34,900 BP ± 600, firmly secure these pulses as Upper Palaeolithic, rather than as modern intrusions. Though van Zeist and Bakker-Heeres (1984) suggests legumes of these genera were used as kindling in the Levantine Neolithic, such an interpretation makes little sense for Ghar-e Boof, where over 75 % of the 352 samples studied revealed wood charcoal. We prefer to interpret the pulses as food, rather than fuel remains (Kislev et al. 1992; Miller 1996; Hillman et al. 1997; Lev et al. 2005).
Distribution of plant remains and site genesis
One way of examining distribution or other spatial analysis under equal conditions is to study the number of specimens per sample volume (Jones 1992). Seed and fruit do not order into a roughly regular number of specimens recovered per square meter excavated (Schiffer 1983). This is because their location may be influenced by human or animal movements, postdepositional disturbance or inadvertent introduction of specimens from the surrounding environment. In sum, plant remains are not deposited evenly about the surface of an archaeological site. Thus, it is not surprising that the number of specimens and taxa recovered at Ghar-e Boof varies horizontally, i.e., among subsquares, and vertically, i.e., between horizons, or even between arbitrarily chosen depths. A first observation made from the complete data set (see Appendix) is that the cave rear and entrance did not preserve different plant assemblages. One might presume that the cave rear would preserve a more homogenous assemblage, compared with the other areas, because of its more enclosed nature, and the cave entrance a more varied assemblage because of its greater exposure to the outside, but this was not the case. The site middle preserved a different overall plant record, however. It is likely that the burning of dung and animal penning activities, which was focused there, increased the number of identified herbaceous plants. In particular. the Chenopodium, Echium, Medicago, and Scirpus genera, which are commonly grazed by ungulates in the Southern Zagros Mountains, were more widespread (Rechinger 1968). Perhaps the wealth in Caryophyllaceae (carnations) and Poaceae (grasses), compared to the overall paucity and variation in taxa in horizons II, IIb, and III, is a further sign that some mixing occurred.
To verify whether postdepositional impacts affect the matrix stratigraphically, arbitrary depths were compared in Table 6. A sample for the cave rear, middle, and entrance were examined at three different levels in horizon III. Because of the significant intrusion of recent material above and into horizon III, making vegetation reconstruction and both horizontal and vertical assemblage comparisons problematic, a depth near the horizon’s base was chosen for this table. The measured depths differ a little between squares due to the gradient, or slope of the cave sediments, but fit as a former level or surface of the cave. Note that repositioning of macro fossils may be affected and increase along a sites’ gradient (Spicer 1991).
The contrasts observable in Table 6 could be due to the dynamism of bioturbation, the effects of gravity or other taphonomic processes (Behrensmeyer et al. 2000). This is not only recognizable at Palaeolithic sites, for example, at Kebara cave, Israel (Bar-Yosef et al. 1992), but also at prehistoric and historic sites, where macrofossils moved downwards through the matrix from their initial position (Johnson 2002; Borojevic 2011). The prevalence of Boraginaceae specimens in the Lappula and Echium genera in both figures is a result of their preponderance in the vegetation and their better preservation potential due their hard calcitic coat (Rechinger 1968; Pustovoytov et al. 2004). Like the smaller Chenopodiaceae, these plants most likely grew along the cave edge in the Upper Palaeolithic, but were of no direct significance to the human occupiers.
Using the same divisions of cave rear, middle, and entrance at arbitrary depths, the occurrences of indicator taxa for damp—Portulaca oleraceae (common purslane) and Helianthemum salicifolium (willowleaf frostweed)—and dry conditions—Salsola laricina—were compared to investigate which plant ecology in the Dasht-e Rostam was predominantly favorable (Rechinger 1968). S. laricina, though absent in the cave rear, was found, associated with perennial plants that tolerate diverse circumstances, in the entrance and middle zone. It is unlikely therefore that its identification means overall drier, more saline conditions. Retrieval from all the excavated squares and horizons of the damp indicator taxa and Malva, Alopecurus, and Plantago, which also prefer moist ground, rather suggest an environment with a tendency toward wetter conditions.
Palaeovegetation description
For future work, we suggest that a description of the palaeovegetation would benefit from sampling together with charcoal and pollen analysis, which should transect the landscape. By including all of the area’s topographic features; valley floors, slopes, ridges, and gullies, and more botanic proxies, coverage of both the variety in elevation and available ecotopes in the area would be enlarged for analyses and discussion. Still, the record from Ghar-e Boof alone already informs what plants grew in its vicinity between 31,000 and 36,000 BP. All the identified taxa were categorized into six groups dependent on their life cycle or physiognomy—wild legumes: Fabaceae, grasses: Poaceae, shrubs and perennial herbs: Cistaceae and Boraginaceae, rushes and sedges: Cyperaceae, and mixed lifecycle herbaceous: Malvaceae, Scrophulariaceae, and Brassicaceae (Bakels 1999). This paper discourages associating plants for ecological interpretations in this way in the distant past, because such associations are prone to oversimplify, to making unsound clusters, and to ignoring possible changes in the plants’ or community’s life (Ellenberg 1979). We made these categorizations, however, to observe the composition of the vegetation in each horizon and to see if any changes occurred during the few thousand years of deposition at Ghar-e Boof. Though it is not debated here that the Hordeum, Triticum, Linum (Flax), and Papaver (poppy) found are not native to the Zagros Mountains, it would be erroneous to include anachronistic plants in these groupings. Thus the Hordeum, Oryza, and Triticum specimens were excluded from this examination. From Table 7, it is clear that the numerical discrepancy between the horizons is due to the different amount of samples studied in each horizon. Note that this table presents how often the taxa were found (the amount of records) in each horizon, not how many specimens in each taxa, following the methodology outlined in Section 3.
While depth affects the amount of contamination numerically; that is, the lower down a sample lies from the disturbance, the less greatly it is affected (see Table 5 on the barley contamination), Table 7 suggests it does not influence the vegetation composition. No specific indications of season of occupation were found because plant maturity, seed dispersal of the vegetation types identified, and the natural preservation thereof extended over too much time. The sole exception to this: S. laricina, a fragile seed indicative of the middle summer (Novikova et al. 2011) does not change this scenario. In sum, Dasht-e Rostam’s palaeovegetation was rich in legumes, particularly those no larger than 2 mm, and had a balance of rocky, wet, and steppe plants.
Conclusions
This paper explored the use of contextual information like site genesis and taphonomy, bioturbation, human activity, and nativity/coevality for evaluating the fidelity of plant assemblages. Leaning on theoretical frameworks and observations made by other authors, a dual methodology is described to tackle the paper's two aims. Through division of the data set in stratigraphic and spatial analytical units, cases of disturbance and irregularities in an assemblage were interpreted as either due to an agency external to the site (biological, human, or climatic) or due to its formation. These can alter the composition of an assemblage, introduce anachronistic or alien taxa, or displace plant remains from their original deposition. Thus archaeobotanical research should incorporate methods for identifying and treating disturbed samples since the source of the intrusion or disturbance may highlight human activities of archaeological interest, reflect diet or changes in the local vegetation. A broad description of the palaeovegetation, including a variety of small pulses, grasses, and perennials that charred accidentally during the cave occupation, was feasible. Identification of Lathyrus/Vicia-type pulses across Ghar-e Boof’s Upper Palaeolithic horizons was interpreted as a sign that the Rostamian occupants of the area collected the legumes as food. Summing up, it was found that disturbed samples, either through natural or anthropogenic agency in an area of the site, render a different plant record compared to unmodified samples. Examined and contrasted together, they can still provide valid windows to describe the past vegetation, environmental conditions, the route taken by plant remains up to deposition and possible human activities.
References
Allison PA, Briggs DEC (eds) (1991) Taphonomy, releasing the data locked in the fossil record
Bakels C (1999) Archaeobotanical investigations in the Aisne Valley, northern France, from the Neolithic up to the early Middle Ages. Veg Hist Archaeobot 8:71–77
Bar-Yosef O et al (1992) The excavations in Kebara Cave, Mount Carmel. Curr Anthropol 33:497–550
Baxter MJ (1994) Exploratory multivariate analysis in archaeology. Edinburgh Univ. Press, Edinburgh
Behrensmeyer AK, Kidwell SM, Gastaldo RA (2000) Taphonomy and paleobiology. Paleobiology 26(4):103–147
Berggren G (1981) Atlas of seeds from Northern Europe and Scandinavia
Birks HH, Birks HJB (2006) Multi-proxy studies in palaeolimnology. Veg Hist Archaeobot 15:235–251
Bojňansý V, Fargaŝová A (2007) Atlas of seeds and fruits of central and eastern Europe
Borojevic K (2011) Interpreting, dating and reevaluation the botanical assemblage from tell Kedesh: a case study of historical contamination. J Archaeol Sci 38:829–842
Bottema S (1993) The Palaeoenvironment of prehistoric man in the Near East: some aspects of palynological research. Jpn Rev 4:129–1490
Bouby L, Marinval P (2004) Fruits and seeds from Roman cremations in Limagne (Massif Central) and the spatial variability of plant offerings in France. J Archaeol Sci 31:77–86
Cappers RTJ, Neef R (2012) Handbook of plant palaeoecology
Conard N, Ghasidian E (2011) The Rostamian cultural group and the taxonomy of the Iranian Upper Paleolithic. In: Conard N, Drechsler P, andMorales A (eds) Festschrift in honour of Hans- Peter Uerpmann Between Sand and Sea 33–52
Conard N et al (2006) Report on the 2005 survey of the Tübingen Iranian Stone Age research project in the Provinces of Esfahan, Fars and Kohgiluyeh-Boyerahmad. In: Azarnoush M (ed) Archaeological reports, vol 5. ICAR 9–34
Djamali M et al (2008) A late Pleistocene long pollen record from Lake Urmia, NW Iran. Quat Res 69:413–420
Ellenberg H (1979) Zeigerwerte der Gefäßpflanzen Mitteleuropas
Fazeli H (2008) Vegetation history of the SE section of the Zagros Mountains during the last five millennia; a pollen record from the Maharlou Lake, Fars Province, Iran. Veg Hist Archaeobot 18:123–136
Ghasidian E (2010) Early Upper Palaeolithic occupation at Ghar-e Boof cave; a reconstruction of Cultural tradition in Southern Zagros Mountains of Iran
Gilbert AS (1983) On the origins of specialised nomadic pastoralism in Western Iran. World Archaeol 15(1):105–119
Heydari-Guran S (2007) The impact of geology and geomorphology on cave and rockshelter archaeological site formation, preservation and distribution in the Zagros Mountains of Iran. Geoarchaeol Int J 22(6):653–669
Heydari-Guran S (2014) Palaeolithic landscapes of Iran. Tübingen publications in prehistory. Kerns Verlag
Hillman G, Legge A, Rowley-Conwy P (1997) On the charred seeds from Epipalaeolithic Abu Hureyra: food or fuel? Curr Anthropol 38:651–659
Hole F, Woosley A (1978) Pollen evidence of subsistence and environment in ancient Iran. Paléorient 4:59–70
Hubbard R, Clapham A (1992) Quantifying macroscopic plant remains. Rev Palaeobot Palynol 73:117–132
Johnson D (2002) Darwin would be proud: bioturbation, dynamic denudation, and the power of theory in science. Geoarchaeology 17(1):7–40
Jones G (1992) Numerical analysis in archaeobotany. In: Van Zeist, Wasylikowa, Behre (eds) Progress in old world palaeoethnobotany 63–80
Kislev M, Nadel D, Carmi I (1992) Epiplaeolithic (19 000 BP) cereal and fruit diet at Ohalo II, Sea of Galilee. Rev Palaeobot Palynol 73:161–166
Lange A (1990) De Horden near Wijk bij Duurstede
Lev E, Kislev ME, Bar-Yosef O (2005) Mousterian vegetal food in Kebara cave, Mt. Carmel. J Archaeol Sci 33(3):475–484
Miksicek CH (1987) Formation processes of the Archaeobotanical record. Adv Archaeol Method Theory 10:211–247
Miller NF (1981) Plant remains from Ville Royale II, Susa. Cahiers de la Délégation Archéologique Française en Iran (DAFI) 12:137–142
Miller N (1996) Seed eaters of the ancient Near East: human or herbivore? Curr Anthropol 37:521–528
Mitka J, Wasylikowa K (1995) Numerical analysis of charred seeds and fruits from an 8000 year old site at Nabta Playa, Western desert, south Egypt. Acta Palaeobotanica 35(1):175–184
Nesbitt A (2006) Identification guide for Near Eastern grass seeds
Novikova N et al (2011) Ecosystem responses to hydrological regime changes in the steppe zone. Arid Ecosyst 1(3):142–148
Piperno D et al (2004) Processing of wild cereal grains in the Upper Palaeolithic revealed by starch grain analysis. Nature 430:670–673
Popper V (1988) Selecting quantitative measurements in paleoethnobotany
Pustovoytov K, Riehl S, Mittmann S (2004) Radiocarbon age of carbonate in fruits of Lithospermum from the early Bronze Age settlement of Hirbet ez-Zeraqōn. Veg Hist Archaeobot 13:207–212
Rechinger K (1968) Flora Iranica; flora des Iranischen Hochlandes und der Umrahmenden gebirge; Persien, Afganistan, teile von west-Pakistan, nord Iraq, Azerbaidjan und Turkmenistan 173 Cyperaceae and 48 Boraginacea. Akad. Verl. Anst. Graz
Schiffer MB (1983) Toward the identification of formation processes. Am Antiq 48(4):675–706
Schilt F (2011) Micromorphology of Upper Palaeolithic and historic sediments from Boof Cave, Iran. Tübingen University, unpublished Master’s thesis
Sievers C, Muasya AM (2011) Identification of the sedge Cladium mariscus subsp. jamaicense and its possible use in the Middle Stone Age at Sibudu, KwaZulu-Natal. South Afr Humanit 23:77–86
Spicer RA (1980) The importance of depositional sorting to the biostratigraphy of plant megafossils. In: Dilcher DL, Taylor TN (eds) Biostratigraphy of fossil plants - successional and paleoecological analyses. Dowden, Hutchinson and Ross Inc., Stroudsberg, pp 171–183
Spicer RA (1991) Plant taphonomic processes. In: Briggs DEG, Allison PA (eds) Taphonomy: releasing the data locked in the fossil record. Plenum Press, New York, pp 71–113
Stauffer T (1965) The economics of nomadism in Iran. Middle East J 19(3):284–302
Stevens L et al (2001) Proposed changes in seasonality of climate during the Lateglacial and Holocene at Lake Zeribar, Iran. The Holocene 11(6):747–755
van der Veen M (2007) Formation processes of desiccated and carbonized plant remains—the identification of routine practice. J Archaeol Sci 34:968–990
van Zeist W, Bakker-Heeres J (1984) Archaeobotanical studies in the Levant 3 Late-Paleolithic Mureybit. Palaeohistoria 26:171–199
Wasylikowa K (2005) Palaeoecology of Lake Zeribar, Iran, in the Pleniglacial, Lateglacial and Holocene, reconstructed from plant macrofossils. The Holocene 15:720–735
Weeks L (2006) The Neolithic settlement of highland SW IRan: new evidence from the Mamasani District. Iran 44:1–31
Weiss E, Wetterstrom W, Nadel D, Bar-Yosef O (2004) The broad spectrum revisited: evidence from plant remains. Proc Natl Acad Sci 101(26):9551–9555
Zeidi M et al (2009) Chapter 6. Survey of Dasht e Rostam e Yek and Dasht e Rostam e Do. In: Potts D et al (eds) The Mamasani Archaeological Project stage one 147–168
Acknowledgements
Unerring gratitude to Harald Hirschprung for his grand medical research. Further thanks to my parents, Saman Heydari-Guran, Katleen Deckers, Paul Goldberg, and Flora Schilt for their support and inspiration.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
Number of specimens | Number of records | |||||||||||
IIb | IIb.1 | III | IIIa | IIIb | IV | IIb | IIb.1 | III | IIIa | IIIb | IV | |
Alopecurus sp. | 16 | – | 8 | 1 | 1 | – | 1 | – | 7 | 1 | 1 | – |
Althaea officinalis | – | – | 1 | – | – | – | – | – | 1 | – | – | – |
Asteraceae indet. | – | – | 2 | – | – | – | – | – | 2 | – | – | – |
Astragalus sp. | – | 1 | 1 | – | – | – | – | 1 | 1 | – | – | – |
Atriplex sp. | – | 2 | 13 | 1 | – | – | – | 1 | 8 | 1 | – | – |
Brassicaceae indet. | – | – | 2 | – | – | – | – | – | 2 | – | – | – |
Bromium sp. | 1 | – | – | – | – | – | 1 | – | – | – | – | – |
Camelina sp. | – | – | 1 | – | – | – | – | – | 1 | – | – | – |
Carex sp. | – | 1 | 6 | – | 1 | – | – | 1 | 3 | – | 1 | – |
Centaurea sp. | – | 1 | – | – | – | – | – | 1 | – | – | – | – |
Cerastium sp. | – | – | 7 | – | – | – | – | – | 4 | – | – | – |
Chenopodiaceae indet. | 100 | 44 | 221 | 25 | 18 | 4 | 4 | 8 | 61 | 13 | 9 | 1 |
Chenopodium foliosum | – | – | 2 | – | – | – | – | – | 2 | – | – | – |
Chenopodium murale | 14 | 28 | 22 | 1 | – | – | 3 | 5 | 8 | 1 | – | – |
Chenopodium sp. | 21 | 9 | 139 | 14 | 12 | – | 2 | 2 | 48 | 5 | 4 | – |
Chenopodium urbicum | – | – | 1 | – | – | – | – | – | 1 | – | – | – |
Cyperaceae indet. | – | 2 | 5 | – | – | – | – | 2 | 5 | – | – | – |
Echium cf. vulgare | 1 | – | 157 | 20 | 3 | – | 1 | – | 58 | 15 | 3 | – |
Eleusine sp. | – | – | – | – | 1 | – | – | – | – | – | 1 | – |
Epilobium cf. palustre | – | – | – | 2 | – | – | – | – | – | 2 | – | – |
Fabaceae indet. | 17 | 4 | 74 | 9 | 1 | 1 | 2 | 3 | 33 | 5 | 1 | 1 |
Ficus sp. | – | 1 | 1 | – | – | – | – | 1 | 1 | – | – | – |
Galium sp. | – | – | 6 | – | 3 | – | – | – | 6 | 2 | – | – |
Genista sp. | – | – | 1 | – | 1 | – | – | – | 1 | – | 1 | – |
Geranium sp. | – | – | – | – | 1 | – | – | – | – | – | 1 | – |
Glaucium sp. | – | – | 2 | – | – | – | – | – | 2 | – | – | – |
Helianthemum salicifolium | – | 3 | 3 | – | – | – | – | 3 | 3 | – | – | – |
Helianthemum sp. | – | 1 | 8 | 1 | – | – | – | 1 | 4 | 1 | – | – |
Heliotropium europeum | – | 1 | 4 | – | – | 2 | – | 1 | 3 | – | – | 1 |
Hippocrepis sp. | 2 | 3 | 16 | 2 | – | – | 1 | 3 | 12 | 1 | – | – |
Hordeum sp. | 45 | – | 18 | 1 | – | – | 11 | – | 13 | 1 | – | – |
Hypericum sp. | 1 | – | – | – | – | – | 1 | – | – | – | – | – |
Lallemantia cf. peltata | – | – | – | 1 | – | – | – | – | – | 1 | – | – |
Lapulla sp. | – | 2 | 52 | 36 | 94 | – | – | 1 | 28 | 9 | 8 | – |
Lathyrus/Vicia sp. | 16 | 2 | 80 | 2 | 14 | 2 | 3 | 1 | 21 | 1 | 3 | 1 |
Lepidium sp. | – | – | 2 | – | 1 | – | – | – | 2 | – | 1 | – |
Linum sp. | 1 | – | – | – | – | – | 1 | – | – | – | – | – |
Lithospermae | 10 | 15 | 34 | 1 | – | – | 1 | 5 | 19 | 1 | – | – |
Lolium perenne | 1 | – | – | – | – | – | 1 | – | – | – | – | – |
Lolium sp. | 3 | 1 | 12 | 1 | – | – | 2 | 1 | 4 | 1 | – | – |
Lotus/Melilotus sp. | – | – | 2 | – | – | – | – | – | 2 | – | – | – |
Malva sp. | 3 | – | 24 | 2 | – | – | 3 | – | 21 | 1 | – | – |
Medicago sp. | 91 | 80 | 251 | 23 | 6 | – | 11 | 9 | 66 | 10 | 6 | – |
Melica sp. | – | – | 1 | – | – | – | – | – | 1 | – | – | – |
Minuartia sp. | – | – | 4 | 2 | – | – | – | – | 1 | 2 | – | – |
Onobrychis sp. | 1 | – | – | – | – | – | 1 | – | – | – | – | – |
Onosma cf. tauricum | – | – | 16 | 5 | 1 | – | – | – | 9 | 4 | 1 | – |
Oryza sp. | – | – | 2 | – | – | – | – | – | 2 | – | – | – |
Panicum sp. | – | – | – | 1 | 1 | – | – | – | – | 1 | 1 | – |
Panicum/Setaria–Tetrapogon | – | 2 | 9 | 4 | 2 | – | – | 2 | 7 | 4 | 2 | – |
Papaver sp. | – | – | 1 | – | – | – | – | – | 1 | – | – | – |
Persicaria hydropiper | – | – | 3 | – | 1 | – | – | – | 1 | – | – | – |
Phalaris sp. | 1 | – | 4 | – | – | – | 1 | – | 4 | – | – | – |
Plantago cf. lagopus | – | 2 | 3 | – | – | – | – | 1 | 3 | – | – | – |
Plantago sp. | – | 1 | 11 | 1 | 3 | – | – | 1 | 11 | 1 | 3 | – |
Poaceae indet. | 6 | 2 | 58 | 6 | 8 | – | 2 | 2 | 29 | 5 | 6 | – |
Polygonaceae indet. | – | – | 2 | – | – | – | – | – | 3 | – | 1 | – |
Polygonum aviculare | – | – | 2 | – | – | – | – | – | 1 | – | – | – |
Polygonum sp. | – | – | 1 | – | – | – | – | – | 1 | – | – | – |
Portulaca oleracea | – | – | 1 | – | – | – | – | – | 8 | 1 | 1 | – |
Portulacaceae indet. | – | – | 11 | 1 | 5 | – | – | 1 | 2 | – | – | – |
Prunus sp. | – | – | 6 | 2 | – | – | – | – | – | – | – | – |
Ranunculus sp. | – | 4 | 2 | – | – | – | – | – | 2 | – | – | – |
Reseda cf. lutea | – | – | 2 | – | – | – | – | – | 15 | – | 3 | – |
Reseda sp. | – | – | 23 | – | 3 | – | – | – | 6 | – | 1 | – |
Rumex sp. | – | – | 4 | – | – | – | – | – | 4 | – | – | – |
Salsola cf. laricina | – | – | 2 | – | 2 | – | – | – | 2 | – | 1 | – |
Scirpus sp. | 68 | 75 | 282 | 61 | 20 | 1 | 10 | 5 | 64 | 7 | 6 | 1 |
Scorpiurus sp. | 2 | 1 | 6 | – | – | – | 2 | 1 | 5 | – | – | – |
Scrophularia sp. | – | 1 | 2 | 1 | – | – | – | 1 | – | – | – | – |
Silene cf. otitis | 1 | – | 3 | – | – | – | 1 | – | 3 | – | – | – |
Silene colorata | – | – | 1 | – | – | – | – | – | 1 | – | – | – |
Silene linicola | – | – | 1 | – | – | – | – | – | 1 | – | – | – |
Sisymbrium sp. | – | – | 2 | – | 1 | – | – | – | 2 | – | 1 | – |
Solanum sp. | – | – | 1 | – | – | – | – | 1 | – | – | – | – |
Stellaria sp. | – | – | 3 | 1 | – | – | – | – | 3 | 1 | – | – |
Stipa sp. | – | – | 3 | – | – | – | – | – | 3 | – | – | – |
Trifolium sp. | 2 | 16 | 31 | 1 | – | – | 2 | 4 | 18 | 1 | – | – |
Trigonella sp. | 1 | – | 3 | – | – | – | 1 | – | 2 | – | – | – |
Triticum sp. | 11 | – | 2 | – | – | – | 3 | – | 2 | – | – | – |
Urtica urens | – | 1 | – | – | – | – | – | – | 1 | – | – | – |
Valerianella cf. dentata | 1 | – | 3 | 2 | – | – | 1 | – | 3 | 1 | – | – |
Verbascum sp. | – | 1 | – | – | – | – | – | – | 1 | – | – | – |
Veronica sp. | – | – | – | – | – | – | – | 1 | 2 | 1 | – | – |
Number of samples | 17 | 12 | 205 | 35 | 33 | 50 | ||||||
Number of specimens | 437 | 307 | 1,687 | 231 | 204 | 10 | ||||||
The amount of records means how often the taxa were found in each horizon
Rights and permissions
About this article
Cite this article
Baines, J.A., Riehl, S., Conard, N. et al. Upper Palaeolithic archaeobotany of Ghar-e Boof cave, Iran: a case study in site disturbance and methodology. Archaeol Anthropol Sci 7, 245–256 (2015). https://doi.org/10.1007/s12520-014-0191-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12520-014-0191-6